Abstract
Background:
Diarylheptanoids (AO-0002 [7-(4″-hydroxy-3″-methoxyphenyl)-1-phenyl-4E-hepten-3-one] and AO-0011 [(5S)-5-hydroxy-7-(4″-hydroxyphenyl)-1-phenyl-3-heptanone]) isolated from Alpinia officinarum have been reported to exhibit anti-influenza virus activity in vitro. Hence, efficacies against influenza virus infection and the mode of antiviral action were evaluated in vivo and in vitro, respectively.
Methods:
In a murine influenza virus infection model, diarylheptanoids were orally administered three times daily to mice infected with influenza A/PR/8/34 virus for 6 days after infection. AO-0002 was examined for its antiviral activity against the wild types of influenza viruses A/PR/8/34 (H1N1), oseltamivir-resistant A/PR/8/34 (H1N1), A/Bangkok/93/03 (H1N1), A/Ishikawa/7/82 (H3N2), A/Fukushima/13/43 (H3N2), B/Singapore/222/79 and B/Fukushima/15/93 in plaque reduction or yield reduction assays. The mode of anti-influenza virus action was assessed by a virus adsorption assay, immunofluorescence assay of viral antigens, and inhibition of viral messenger RNA synthesis using real-time reverse transcriptase PCR. Results: AO-0002 at 100 mg/kg was significantly effective in reducing the body weight loss and prolonging survival times of infected mice without toxicity, but AO-0011 was not. AO-0002 at 30 and 100 mg/kg significantly reduced virus titres in bronchoalveolar lavage fluids of the lungs on days 3 and 6 after infection. AO-0002 exhibited anti-influenza virus activity against all viruses used, including the oseltamivir-resistant strain in vitro. The compound had no effect on virus adsorption or invasion into cells, but dose-dependently suppressed the expression of viral messenger RNA and antigens.
Conclusions:
AO-0002 was suggested to have a different anti-influenza virus action to that of oseltamivir and was verified to show anti-influenza activity in vitro and in vivo.
Introduction
The H1N1 influenza virus has recently spread worldwide. The appearance of an influenza virus more virulent than the pandemic H1N1 is now predicted. Influenza viruses infect the respiratory tract in humans and cause a variety of symptoms, including fever, nasal secretions, cough, headache, muscle pain and pneumonia. These clinical symptoms often become severe, especially in high-risk groups such as the elderly and infants [1,2]. The amantadine and neuraminidase inhibitors zanamivir and oseltamivir have been used for the treatment and prevention of influenza virus infection [3–5], but the appearance of viruses resistant to them has also been reported [3,6–8]; thus, it is important to develop new types of anti-influenza virus agents with anti-influenza virus actions different from those of the known agents.
Traditional medicines, including medicinal plants, have been pursued as suitable alternative sources of antiviral agents [9,,–16]. An extract of Alpinia officinarum Hance was previously shown to exhibit therapeutic efficacy against herpes simplex virus type-1 infection in mice [17]. Some of the diarylheptanoids isolated from Alpinia officinarum Hance have been shown to exhibit biological activities, such as antioxidant activity [18,19], suppression of inducible nitric oxide synthase expression [20] and inhibition of biosynthesis of prostaglandins and leukotrienes [21,22]. Recently, we reported that 10 diarylheptanoids exhibit anti-influenza virus activity in vitro [23]. In the present study, we assessed the efficacies of 7-(4″-hydroxy-3″-methoxyphenyl)-1-phenyl-4E-hepten-3-one (AO-0002) and (5S)-5-hydroxy-7-(4″-hydroxyphenyl)-1-phenyl-3-heptanone (AO-0011) among the diarylheptanoids with anti-influenza virus activity on an intranasal influenza virus infection model in mice and examined the mode of anti-influenza virus action of AO-0002, which was effective in mice.
Methods
Cells and viruses
Madin–Darby canine kidney (MDCK) cells were grown and maintained in Eagle's minimum essential medium (Nissui Pharmaceutical Co. Ltd, Tokyo, Japan) supplemented with 8% and 2%, respectively, heat-inactivated fetal calf serum (Equitech-Bio, Inc., Kerrville, TX, USA) [24]. Influenza viruses were propagated in MDCK cells. Influenza viruses A/PR/8/34 (H1N1) and B/Singapore/222/79 were provided by H Ochiai (Toyama University, Toyama, Japan). Oseltamivir-resistant A/PR/8/34 (H1N1) was plaque-purified 10x from A/PR/8/34 (H1N1)-infected MDCK cell cultures in the presence of 60 μg/ml of oseltamivir phosphate, which was purchased from Roche Diagnostics KK (Tokyo, Japan), and dissolved in distilled water. Influenza viruses A/Bangkok/93/03 (H1N1), A/Ishikawa/7/82 (H3N2), A/Fukushima/13/43 (H3N2) and B/Fukushima/15/93 were provided by S Shigeta (Fukushima Medical University, Fukushima, Japan).
Compounds
AO-0002 (molecular weight [Mw] =310.4) and AO-0011 (Mw =298.4; Figure 1) were isolated from rhizomes of Alpinia officinarum Hance (Zingiberaceae) as described previously [25,26]. For in vivo assays, AO-0002 and AO-0011 were dissolved at various concentrations in 1% dimethyl sulfoxide (DMSO) and administered orally to mice. As a positive control, oseltamivir phosphate purchased from Roche Diagnostics KK, was dissolved in distilled water and administered to mice. For in vitro assays, AO-0002 was dissolved in DMSO to make a final concentration of 0.2% for use in plaque reduction and yield reduction assays using MDCK cells, and examined for its anti-influenza virus activity, as described previously [16,24]. Ribavirin was purchased from Wako Pure Chemical Industries Ltd (Osaka, Japan). Ribavirin and oseltamivir phosphate were dissolved in distilled water for plaque reduction and yield reduction assays [16,24].
Animals
Female BALB/c mice (6 weeks old, 20–22 g) were purchased from Kyudo Animal Laboratory (Kumamoto, Japan). The mice were housed in groups of five or six per cage with food and water ad libitum under a 12 h light/12 h dark diurnal cycle (light at 7:30 AM). The temperature in the room was kept at 24 ±2°C. The mice were acclimated for at least 5 days before starting experimental procedures. Animal studies followed the animal experimentation guidelines of Kyushu University of Health and Welfare (Nobeoka, Japan), and were carried out in an approved biosafety level facility there.
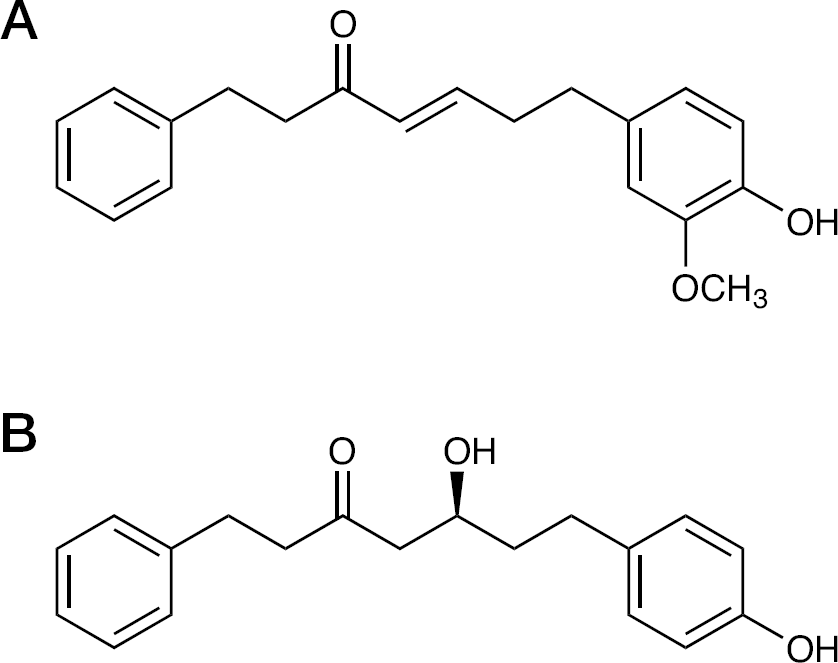
Chemical structures of compounds
Influenza virus infection in mice
Female BALB/c mice were intranasally infected or mock-infected with 500 plaque-forming units (PFU) of the A/PR/8/34 strain under anaesthesia with intramuscular ketamine and xylazine at 42 and 7.6 μg/g of body weight, respectively [16]. Diarylheptanoids dissolved in 1% DMSO were administered orally to mice by gavage at 0, 30 and 100 mg/kg of body weight once at 4 h prior to, and once at 1 h and once again at 6 h after virus infection on day 0, and three times daily from day 1 to day 5 after infection. A 1% DMSO solution was used as a control. Oseltamivir phosphate at 1 mg/kg was orally administered to infected mice, once at 4 h prior to infection and once after infection on day 0, and twice daily from day 1 to day 4 as described previously [16]. In total, five or six mice in infected groups or three mice in mock-infected groups were weighed daily from day 0 to day 7 after infection, and the weight changes were calculated based on the body weight of each mouse on day 0. The number of surviving mice in groups treated with or without diarylheptanoids or oseltamivir was observed daily from day 0 to day 14.
Determination of virus yields in bronchoalveolar lavage fluid
Virus yields in the bronchoalveolar lavage fluid (BALF) of influenza virus-infected mice have been widely used to evaluate the anti-influenza virus activity of compounds in intranasal influenza virus infection models in mice [13,14,16]. BALF was prepared from the lungs of four and three mice in infected groups administered AO-0002 at 0, 30 and 100 mg/kg on days 3 and 6, respectively, after infection, and the virus titres of BALF were determined by plaque assays [16].
Plaque reduction assays
MDCK cells grown in 60 mm plastic dishes were inoculated with 100 PFU/0.2 ml of influenza virus. After adsorption at room temperature for 1 h, the inoculum was removed and the cells were overlaid with 5 ml of a nutrient agarose (0.8%) medium containing various concentrations of AO-0002 or ribavirin, and maintained in a humidified atmosphere containing 5% CO2 for 3 days. The infected cells were fixed with 5% formalin solution and stained with 0.03% methylene blue (Nacalai Tesque, Kyoto, Japan). The number of plaques was counted under a dissecting microscope [16]. The effective concentrations for 50% plaque reduction (EC50) were determined from a curve relating the plaque number to the concentrations of AO-0002.
Effects of AO-0002 on adsorption and invasion of virus into MDCK cells were examined using plaque assays. MDCK cells grown in 60 mm plastic dishes were inoculated with 100 PFU/0.2 ml of influenza virus A/PR/8/34 (H1N1) containing various concentrations of AO-0002. After adsorption at room temperature for 1 h, the inoculum was removed and cells were washed 3x with phosphate-buffered saline (PBS). The cells were overlaid with 5 ml of a nutrient agarose medium, maintained for 3 days and stained as described earlier.
Cytotoxicity assays
Cytotoxicity of AO-0002 was examined by the growth inhibition of MDCK cells as described previously [16]. Briefly, MDCK cells were seeded at a concentration of 2.5×104 cells/well in 24-well plates and grown at 37°C for 2 days. The culture medium was replaced with fresh medium containing AO-0002 at various concentrations and then cells were further grown for 3 days. The cells were treated with trypsin, and the number of viable cells was determined by the trypan blue exclusion test. The 50% cytotoxic concentrations of AO-0002 reducing cell viability (CC50) were determined from a curve relating the percentage of viable cells to the concentrations of AO-0002.
Yield reduction assays
AO-0002 was examined for its anti-influenza virus activity in yield reduction assays. Monolayers of MDCK cells in 60 mm plastic dishes were infected with an A/PR/8/34 (H1N1) strain at 2 PFU/cell for 1 h. The cells were washed 3x with PBS and incubated in maintenance medium containing various concentrations of AO-0002 or ribavirin at 37°C for 18 h. The cultures were frozen and thawed, and then centrifuged at 3,000 rpm for 15 min. Virus titres in culture medium were determined using plaque assays on MDCK cells [16].
Immunofluorescence assays
Confluent monolayers of MDCK cells grown on cover glasses in 24-well plates were infected at 2 PFU/cell with A/PR/8/34 (H1N1) in the presence of AO-0002 at 0, 20, 40 or 60 μg/ml, as described previously. The cells were washed 3x with PBS at 0 or 7 h after infection and fixed in ice-cold acetone for 15 min. The cells were directly stained with fluorescein isothiocyanate-conjugated goat anti-influenza A virus antibody (ViroStat, Portland, ME, USA), which was prepared from the purified immunoglobulin G fraction of antiserum of goat immunized by the purified virions of USSR, H1N1 strain, diluted 1:25 in PBS at room temperature for 40 min, washed 3x with PBS and mounted on glass slides. The cells were observed using an immunofluorescence microscope (Olympus, Tokyo, Japan) at x400 magnification.
Real-time reverse transcritpase PCR
A reverse transcriptase (RT)-PCR assay that is targeted against the influenza A (H1N1) matrix (M) gene has been recommended by the World Health Organization for general influenza A virus screening, including avian influenza A virus (H5N1) [27]. Expression of the influenza virus A/PR/8/34 (H1N1) M gene in infected MDCK cells was measured by real-time RT-PCR, and the data were evaluated by normalizing results to those of the mouse β-actin gene, which is a housekeeping gene. Briefly, a confluent monolayer of MDCK cells in a 60 mm dish was mock-infected or infected at 2 PFU/cell and incubated in maintenance medium containing various concentrations of AO-0002 or ribavirin at 37°C, as described in the yield reduction assay. Because the viral messenger RNAs (mRNAs) and proteins of influenza virus PR8 strain have been reported to be sufficiently transcribed and translated, respectively, at 6 h after infection with >1 PFU/cell in MDCK cells in the yield reduction assay [24], RNA was isolated from the infected MDCK cells using Trizol reagent (Invitrogen Corp., Carlsbad, CA, USA) at 6 h after infection and the isolated RNA was transcribed into complementary DNA (cDNA) by ReverTra Ace α (Toyobo Co. Ltd, Osaka, Japan) using an oligo-deoxythymidine(20) primer, according to the manufacturer's instructions. The cDNAs of M gene mRNA and β-actin mRNA were amplified and analysed on a Roche LightCycler P2000 real-time PCR system using a LightCycler FastStart DNA Master SYBR Green I kit (Roche Diagnostics, Indianapolis, IN, USA) with specific primers according to the manufacturer's instructions. Pairs of the specific primers were as follows: M gene forward 5′-TAACTAGCCTGACTAGCAACCTC-3′ and M gene reverse 5′-CGCTCAGACATGAGAACAGAATGG-3′; β-actin forward 5′-TGGAATCCTGTGGCATCCATGAAAC-3′ and β-actin reverse 5′-TAAAACGCAGCTCAGTAACAGTCCG-3′. The number of copies of cDNAs of M and β-actin mRNAs were determined by comparing the crossing point values of the cDNA samples to those of the TA vectors harbouring parts of the influenza virus M gene (nucleotides 1–1027) and β-actin gene (nucleotides 728–1076), respectively.
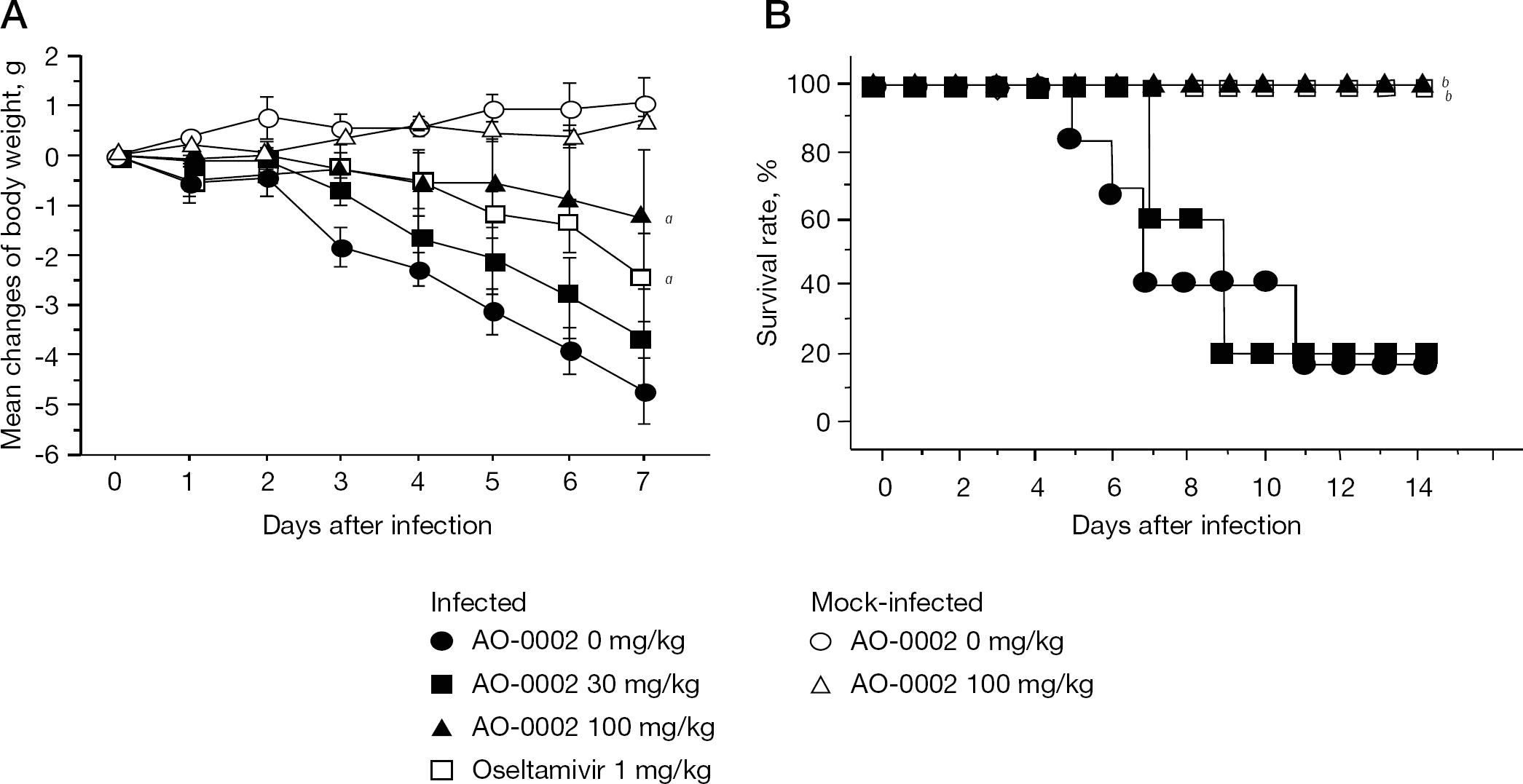
Effects of AO-0002 on body weight and survival rates of influenza virus-infected cells
Statistical analyses
Interactions between treated and untreated groups in the changes of net body weights of infected mice were analysed using a two-way repeated measures ANOVA, followed by Dunn's procedure as a multiple comparison procedure, for days 2–5 after infection. The Kaplan–Meier method, followed by a long-rank test was used to evaluate the statistical differences in mortality [28]. Statistical significances of differences between the EC50 and CC50 values were evaluated using Student's t-test. Interactions between treated and untreated groups in relative amounts of amplified DNA and virus yields were evaluated using Student's t-test. A P-value of ≤0.05 was considered to be statistically significant.
Results
Anti-influenza virus activity of diarylheptanoids in mice We previously showed that AO-0002 and AO-0011 (Figure 1) exhibited relatively stronger anti-influenza virus activity in vitro than other diarylheptanoids [23]. Their efficacy was examined in a murine influenza virus infection model. As shown in Figure 2A, the mean body weights of infected mice administered 1% DMSO (AO-0002 0 mg/kg) decreased noticeably from days 2 to 7 after infection. AO-0002 at 100 mg/kg significantly reduced the loss of mean body weight in infected mice (P<0.05 by the repeated measures of two-way ANOVA), but at 30 mg/kg it did not. AO-0011 at 30 and 100 mg/kg was not significantly effective in reducing the body weight loss (RS and TS, data not shown). Oseltamivir at 1 mg/kg significantly reduced the body weight loss of infected mice as did AO-0002 at 100 mg/kg. There was no significant difference between the mean ±SE changes of body weights of mock-infected mice administered AO-0002 at 0 and 100 mg/kg at 14 days after infection (1.5 ±0.3 and 1.7 ±0.2 g, respectively). In addition, AO-0002 at 100 mg/kg, as well as oseltamivir at 1 mg/kg, was significantly effective in prolonging the survival times of infected mice as compared with the control (Figure 2B; P<0.05 by the Kaplan–Meier method), but at 30 mg/kg it was not; thus, AO-0002 exhibited potential anti-influenza efficacy in mice without toxicity, but AO-0011 did not.
To assess the potential anti-influenza virus activity of AO-0002 in vivo, the effects of AO-0002 at 30 and 100 mg/kg on virus titres in the BALF of infected mice were examined on days 3 and 6 after infection, as shown in Figure 3. In this murine infection model, it has been reported that virus titre in BALF reached the maximum level on days 3–4 after infection and decreased after day 4 [28,29]; thus, the virus titres in the BALF on days 3 and 6 were evaluated as the periods in which viruses are multiplied maximally and eliminated, respectively. On day 3, AO-0002 at 100 mg/kg was significantly effective in reducing virus titres in the BALF of infected mice compared with the control, and both 30 and 100 mg/kg were also significantly effective on day 6 (P<0.05 by Student's t-test); however, no significant reduction of virus titres was observed in infected mice administered AO-0011 at 30 and 100 mg/kg (RS and TS, data not shown). Therefore, AO-0002 suppressed virus growth in the respiratory tract of mice and also exhibited potential anti-influenza virus activity in mice.
Anti-influenza virus activity of AO-0002 in vitro
AO-0002 was examined for its anti-influenza virus activity and cytotoxicity to allow characterization of its potential antiviral activity in vitro. As shown in Table 1, the mean ±SE EC50 values (5.1 ±0.03 to 21.6 ±1.0 μg/ml) of AO-0002 for wild-type strains A/PR/8/34 (H1N1), A/Bangkok/93/03 (H1N1), A/Ishikawa/7/82 (H3N2), A/Fukushima/13/43 (H3N2), B/Singapore/222/79 and B/Fukushima/15/93 were significantly lower than the CC50 values (61.9 ±3.1 μg/ml; P<0.05 by Student's t-test). The mean ±SE EC50 value (45.3 ±2.4 μg/ml) of oseltamivir for the oseltamivir-resistant A/PR/8/34 (H1N1) strain was approximately 189x higher than that for the wild type (0.24 ±0.01 μg/ml); however, similar to the wild-type strain, the oseltamivir-resistant strain was as susceptible to AO-0002. In this assay, the EC50 value of ribavirin as a positive control was similar to the results reported previously [16,30]. AO-0002 showed potential antiviral activity against, not only wild types of influenza virus, but also an oseltamivir-resistant strain in vitro.
Anti-influenza virus activity and cytotoxicity of AO-0002
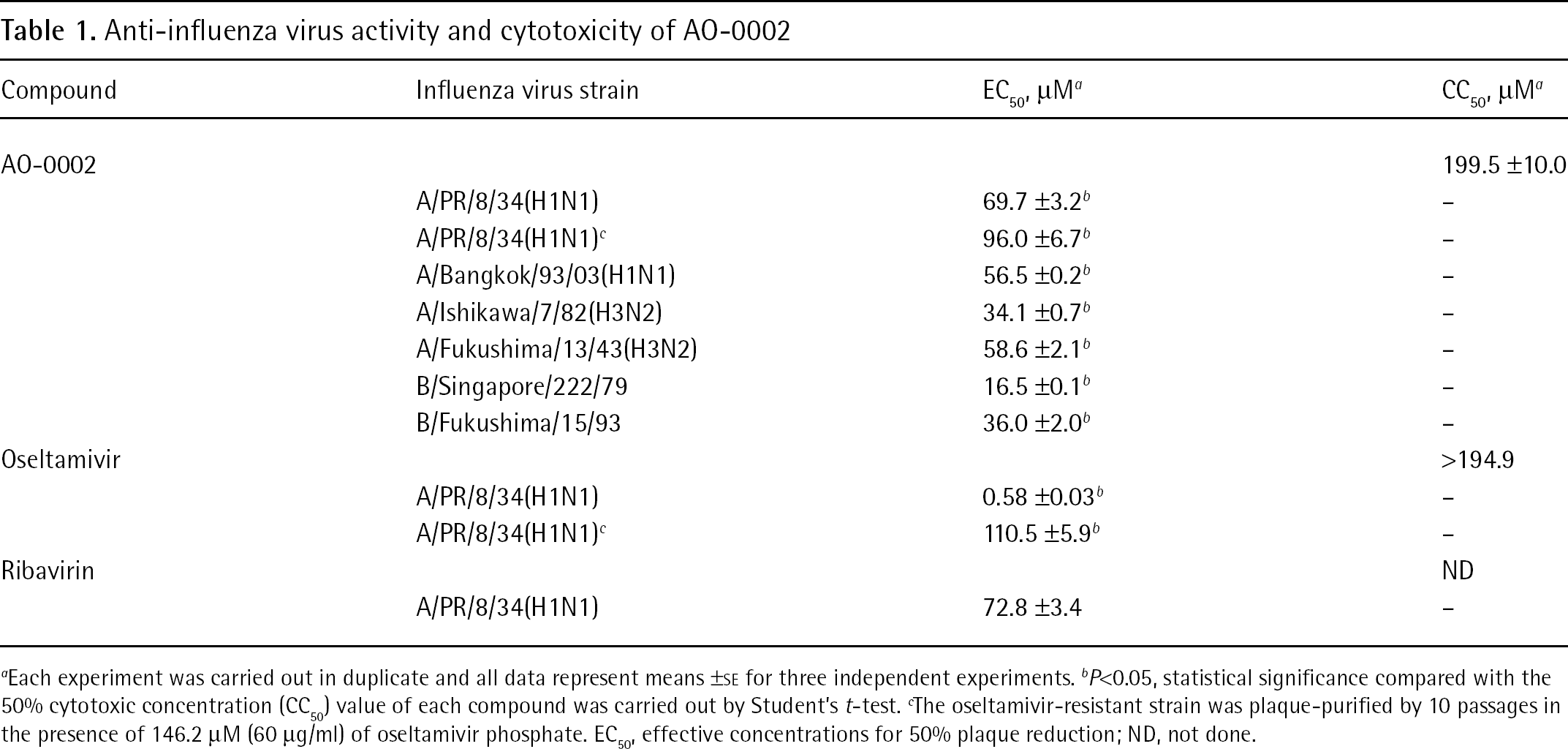
Each experiment was carried out in duplicate and all data represent means ±SE for three independent experiments.
P<0.05, statistical significance compared with the 50% cytotoxic concentration (CC50) value of each compound was carried out by Student's t-test.
The oseltamivir-resistant strain was plaque-purified by 10 passages in the presence of 146.2 μM (60 μg/ml) of oseltamivir phosphate. EC50, effective concentrations for 50% plaque reduction; ND, not done.
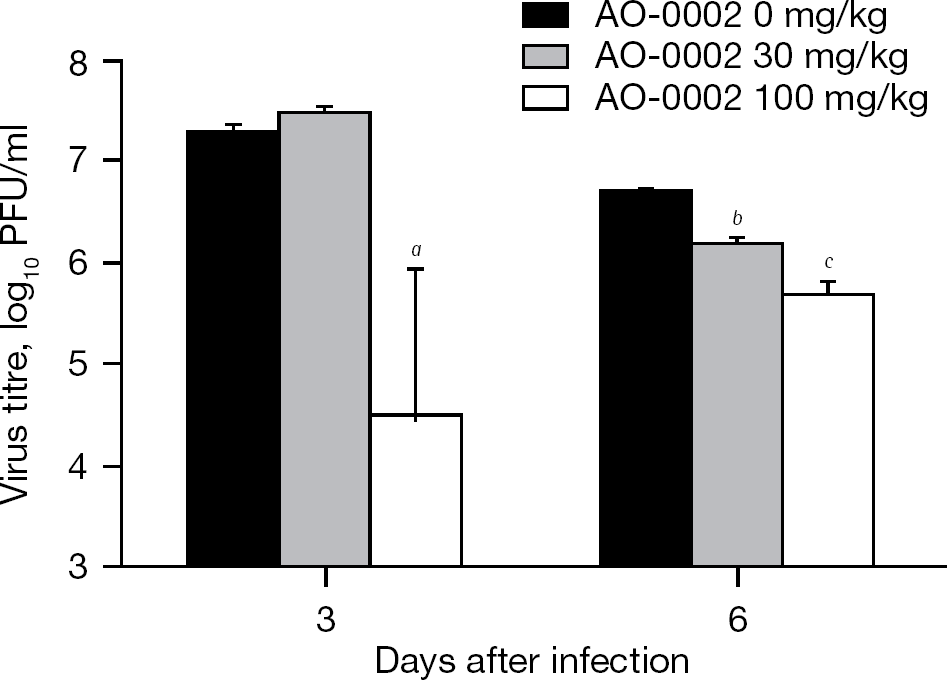
Effects of AO-0002 on influenza virus yields in bronchoalveolar lavage fluid of infected mice
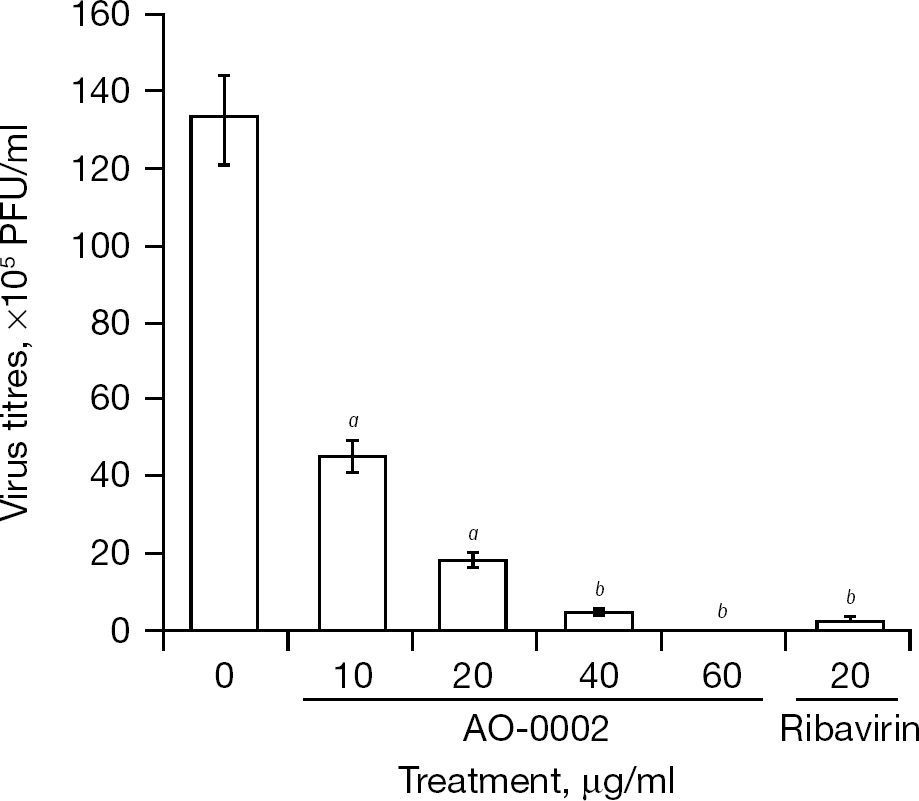
Effect of AO-0002 on growth of influenza virus
The anti-influenza virus activity of AO-0002 was confirmed against influenza virus A/PR/8/34 (H1N1) in a yield reduction assay (Figure 4). AO-0002 reduced virus yields in a dose-dependent manner, and virus yields were reduced by 1/7th, 1/27th and 1/1,331th in the presence of 20, 40 and 60 μg/ml, respectively, of AO-0002 compared with its absence. The reduction of virus yield at 40 μg/ml of AO-0002 was similar to that at 20 μg/ml of ribavirin; thus, the anti-influenza virus activity of AO-0002 was confirmed in vitro.
Effect of AO-0002 on virus adsorption to and/or invasion of Madin–Darby canine kidney cells
A plaque reduction assay was performed to evaluate the effect of AO-0002 at 0, 20, 40 and 60 μg/ml on virus adsorption to and/or invasion of MDCK cells. As shown in Figure 5, AO-0002 at 20, 40 and 60 μg/ml did not reduce plaque numbers significantly. No concentration of AO-0002 interfered with the adsorption and/or invasion of influenza virus to MDCK cells significantly.
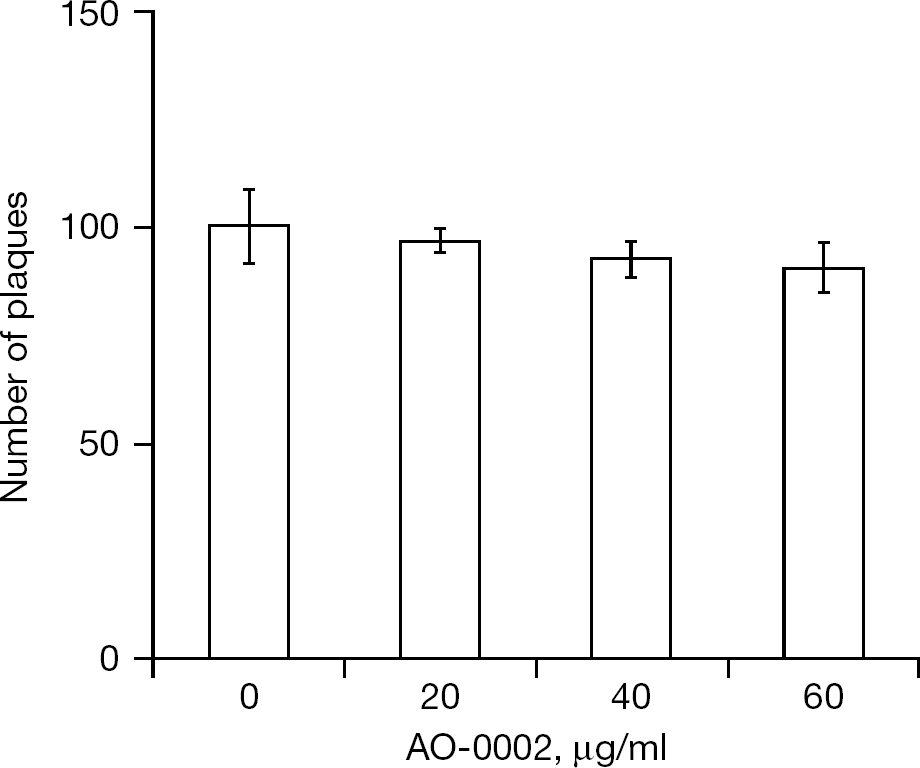
Effect of AO-0002 on influenza virus adsorption and/or invasion
Effect of AO-0002 on expression of viral antigens
Influenza virus antigens were detected in infected cells exposed to AO-0002 for 7 h after infection by a direct immunofluorescence assay (Figure 6). In most of the infected cells, viral antigens were strongly detected in the absence of AO-0002 (Figure 6C), as compared with the mock-infected cells and infected cells immediately after adsorption (Figure 6A and 6B, respectively); however, some viral antigens were detected in the presence of AO-0002 at 20 μg/ml (Figure 6D), and they were negligible at 40 μg/ml (Figure 6E). AO-0002 was suggested to suppress the expression of viral antigens in the infected cells.
Effect of AO-0002 on synthesis of viral messenger RNA
The effect of AO-0002 on the synthesis of influenza virus M gene mRNA was examined by real-time RT-PCR in influenza virus-infected cells exposed to AO-0002 at 0, 20 and 40 μg/ml for 6 h after infection. As shown in Figure 7A, AO-0002 at 20 and 40 μg/ml did not significantly affect the mRNA synthesis of the β-actin gene, which is a housekeeping gene in mock-infected cells; however, AO-0002 at 20 and 40 μg/ml significantly reduced mRNA synthesis from the M gene (Figure 7B; P<0.002 and P<0.003 by Student's t-test, respectively) as did ribavirin at 20 and 40 μg/ml (P<0.002 and P<0.003 by Student's t-test, respectively). Thus, AO-0002 was selectively effective in reducing transcription of the M gene in the infected cells.
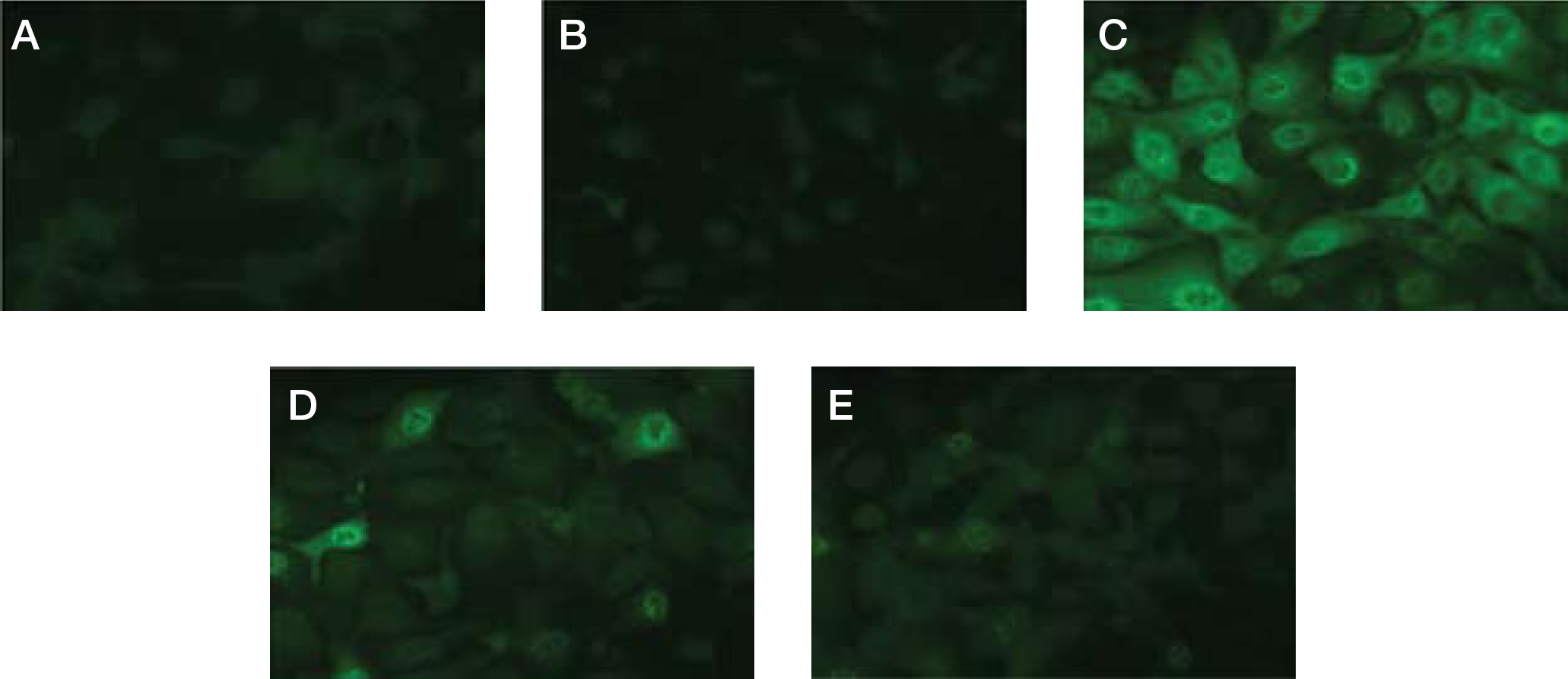
Effect of AO-0002 on expression of virus antigens
Discussion
Oral administration of AO-0002 exhibited efficacy against influenza infection in mice (Figure 2) and the efficacious dose (100 mg/kg) significantly reduced virus titres in BALF on days 3 and 6 (Figure 3); however, 30 mg/kg of AO-0002 produced a significant reduction of virus titre on day 6 but not on day 3 (Figure 3). In our infection model in mice, influenza viruses grow at a maximum level in the lungs on day 3 [28,29]; thus, the oral administration of AO-0002 at 30 mg/kg might not be significantly sufficient to reduce the virus multiplication at the maximum level on day 3. AO-0002 was potentially effective in producing anti-influenza virus activity in mice. It showed anti-influenza virus activity in vitro [23], and the activity was also confirmed in mice. The anti-influenza virus activity of AO-0002 was suggested to be maintained in mice after oral administration. AO-0002 has been reported to exhibit biological activities, such as antioxidant activity [18,19] and suppressive activity of inducible nitric oxide synthase expression [20]. In the lungs of influenza virus-infected mice, the reduction of the number of infiltrated cells and the levels of nitric oxide, its metabolites and oxygen radicals produced from them has been reported to contribute to the alleviation of influenza infection [31,32]; thus, the biological activities of AO-0002 reported might contribute to the alleviation of influenza infection and it would be interesting to evaluate the immunological effect of AO-0002 in mice infected with influenza virus. Although the effective dose of AO-0002 for mice was much higher than that of oseltamivir, it was suggested as a promising anti-influenza virus agent candidate for the treatment of influenza infection. This is the first study demonstrating the efficacy of a diarylheptanoid for influenza infection based on its anti-influenza virus activity in mice.
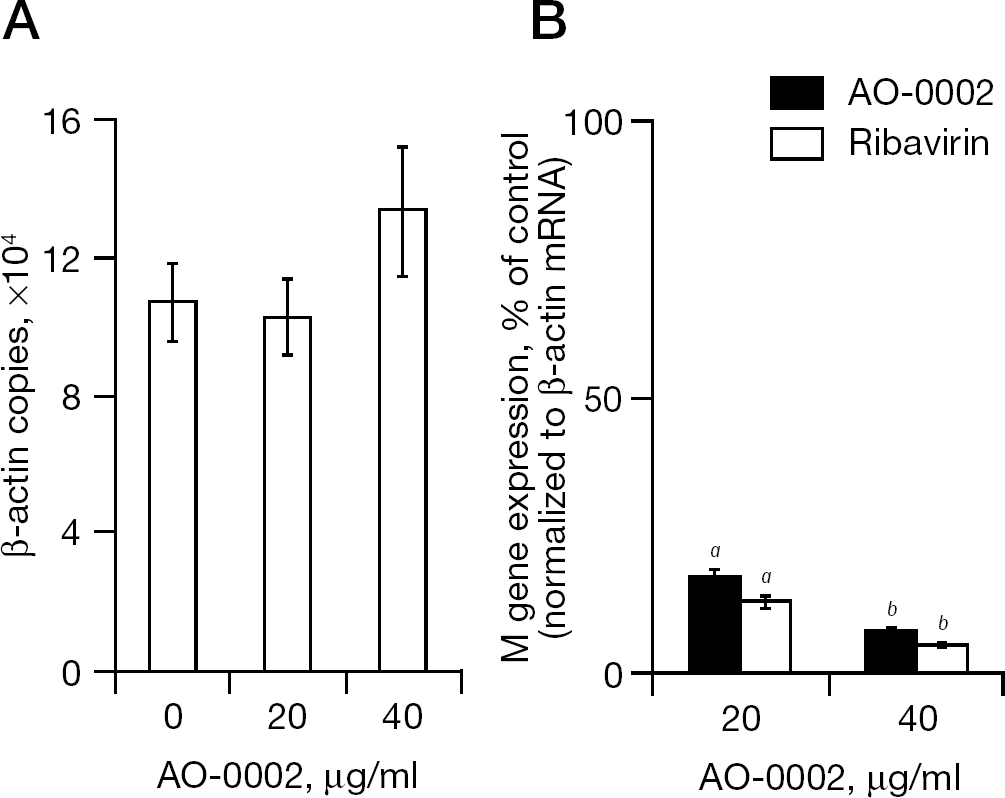
Effect of AO-0002 on expression of host β-actin and viral M genes
Various kinds of diarylheptanoids have been isolated from Alpinia officinarum [22,25,26,33,34] and shown to exhibit anti-influenza virus activity in vitro [23]. Two of these, AO-0002 and AO-0011, have been reported to exhibit significant anti-influenza virus activity in vitro, with AO-0011 being more effective than AO-0002 [23]; however, in this study, only AO-0002 was found to be effective against influenza infection in mice (Figure 2). AO-0011, which has a slightly different chemical structure from AO-0002 (Figure 1), was not effective against influenza infection in vivo. It has been indicated that the chemical structure of AO-0002 is much more favourable than that of AO-0011 with regard to anti-influenza virus activity in vivo; thus, the relationship between their structures and anti-influenza properties in vivo might be worth studying to obtain more effective anti-influenza virus diarylheptanoids.
Similar inhibitory effects of AO-0002 were observed for all influenza viruses examined in vitro (Table 1), including the wild types of influenza A/H1N1, A/H3N2 and B viruses that currently afflict the human population. Their EC50 values were significantly lower than the CC50 value of AO-0002, suggesting that AO-0002 has effective antiviral activity against different subtypes of influenza virus. The oseltamivir-resistant influenza A/H1N1 virus was as susceptible to AO-0002 as the wild types were. In our preliminary experiments, AO-0002 did not exhibit antiviral activity against herpes simplex virus type-1, poliovirus and respiratory syncytial virus in plaque reduction assays, but did against measles virus (RS and KK, data not shown). It is probable that AO-0002 possesses a different mode of anti-influenza virus action from oseltamivir, and this mode of antiviral action might also be effective against measles virus. The mean ±SE EC50 values of AO-0002 for the wild types of influenza A and B viruses examined (5.1 ±0.03 to 21.6 ±1.0 μg/ml) were similar to that of ribavirin (17.8 ±0.8 μg/ml) in a plaque reduction assay (Table 1). In a yield reduction assay (Figure 4), virus yields of the PR8 strain were reduced more than 1/127th and 1/1,331th times in the presence of 40 and 60 μg/ml AO-0002, respectively, compared with its absence. The reduction of virus yield at 40 μg/ml of AO-0002 was also similar to that of ribavirin at 20 μg/ml, suggesting that AO-0002 has an anti-influenza virus activity comparable to that of ribavirin.
The plaque numbers of A/PR/8/34(H1N1) strains were reduced to 50% at a mean ±SE EC50 of 21.6 ±1.0 μg/ml by AO-0002 in the plaque reduction assay (Table 1); however, if the virus was exposed to AO-0002 for only 1 h during the adsorption process at 40 and 60 μg/ml, which are higher than the EC50 value, there was no reduction in the plaque numbers (Figure 5). This indicated that AO-0002 did not interfere with the adsorption and invasion into MDCK cells of the influenza virus A/PR/8/34(H1N1) strain (Figure 5); thus, AO-0002 was suggested to suppress the stage of virus replication after adsorption and/or invasion.
When the expression of viral antigens was observed by an immunofluorescence assay, AO-0002 was shown to be effective in suppressing their expression in infected cells (Figure 6). The expression was probably suppressed in the stage before viral protein synthesis. Indeed, we found that transcription of the influenza virus M gene was inhibited by AO-0002 in infected cells, although transcription of the β-actin gene was not affected by the presence of AO-0002 in mock-infected cells (Figure 7). It is probable that AO-0002 selectively suppressed viral mRNA synthesis in infected cells without cytotoxicity. The mode of anti-influenza virus action of AO-0002 was suggested to be different from that of oseltamivir and requires further study.
Footnotes
Acknowledgements
We thank Akane Hino for her excellent technical assistance and Katherine Ono for her editorial assistance. This study was partly supported by Grants-in-Aid for Scientific Research (grant number 20590131) and by Young Scientists (grant number 20790430) from the Japan Society for the Promotion of Science and Health, and Labor Sciences Research Grants (Research on Risk of Chemical Substances) from the Ministry of Health, Labor and Welfare of Japan.
The authors declare no competing interests.
