Abstract
Background:
Previous studies have suggested that geldanamycin (GA) inhibits the replication of several viruses in vitro. Here, we aimed to synthesize and evaluate the antiviral activity of 17-amino-17-demethoxygeldanamycin derivatives.
Methods:
A series of 17-substituted and 17-,19-disubstituted GA derivatives were screened for antiviral activities against eight different viral strains, including herpesvirus, hepatitis virus, retrovirus and picornavirus.
Results:
Most of the tested compounds showed inhibitory activity against the viruses and showed reduced cytotoxicity in vitro as compared with the parent compound GA. In vivo efficacy evaluation results showed that compound 6 noticeably inhibited duckling hepatitis B virus DNA replication in duckling serum after oral administration. Viral rebound did not occur after termination of the treatment. The modified GA derivatives also showed median lethal dose values that were higher than that of the parent GA in mice intraperitoneally treated with the study compounds.
Conclusions:
Targeting heat-shock protein 90 could be a new antiviral approach that is not prone to the development of drug resistance. The 17-amino-17-demethoxygeldanamycin derivatives could be novel agents with potential antiviral activity.
Introduction
Viruses with high infection rates and rapid propagation can cause worldwide human and animal disease pandemics. With the emergence of previously unknown viruses and new virus variants in recent years, viral infection remains a major cause of human health problems. Moreover, the existing antiviral agents mainly target the key viral enzymes that are involved in the process of replication. At the same time, many viral enzyme mutations result in drug resistance. Some antiviral agents are also of limited use because of their existing toxic side effects. There is a crucial need for developing new agents with novel antiviral mechanisms and broad antiviral activities.
Heat-shock protein 90 (HSP90) is a cellular molecular chaperone that binds to multiple proteins and facilitates the proper refolding of these proteins. It increases the stability of the bound proteins and prevents their degradation through the ubiquitin–proteasome pathway. Previous studies have focused primarily on the stabilization of tumour cell oncogenic proteins by HSP90 [1,2]. Recent studies have found that some viral proteins can also directly or indirectly form chaperone complexes with HSP90 to assure stabilization and correct folding; therefore, HSP90 has been identified as an essential host factor required for stabilization of viral proteins. Moreover, attempts to introduce mutations into HSP90 have been difficult because HSP90 is a conserved cellular protein; thus, HSP90 has the potential to be developed as a novel target for broad-spectrum antiviral agents. Optimized HSP90 inhibitors should have broad-spectrum antiviral activity and should be able to reduce the possibility of developing drug resistance.
Geldanamycin (GA), an HSP90 inhibitor, is a naturally occurring benzoquinone ansamycin antibiotic that is isolated from the fermentation of Streptomyces hygroscopicus, and has been widely recognized for its potent activity against tumours [3–5]. GA has also been frequently used to investigate the relationship between HSP90 activity and viral replication. Results from these studies have suggested that GA inhibits the replication of multiple viruses by abolishing the activity of HSP90. For example, by inhibiting HSP90 activity, GA could suppress hepatitis B virus (HBV) DNA replication and RNA packaging because of its specific inhibition of the interaction between the HBV reverse transcriptase and ɛ RNA [6,7]. GA also suppresses normal functions of viral proteins by preventing the formation of the complex comprising HSP90 and viral proteins; such complexes play an important role in, for example, hepatitis C virus (HCV) RNA replication and in the HIV type-1 (HIV-1) transcription process [8–11]. Inhibition of HSP90 activity also causes inhibition of viral protein synthesis (herpesvirus and some negative-stranded RNA viruses) because it facilitates the degradation of viral RNA polymerases [12–14]. These studies have also indicated that GA could inhibit viral replication by interfering with the chaperone-mediated process in viral protein stabilization and folding. Li et al. [15] and Geller et al. [16] have identified GA as a novel parent compound that exhibits broad-spectrum activities against DNA or RNA viruses in cell cultures and in animals. However, high toxicity and poor oral bioavailability are major drawbacks that limit its use in clinical settings. As previous studies have verified that derivatization of GA by substituting the 17-position of GA with an amino group has greatly decreased the toxicity while retaining the anti-tumour activity [17,18], we synthesized GA derivatives substituted with small molecule alkylamines that consist of heterocycles or strong polar groups, such as phosphates or carbohydrates, in the 17-position to improve both the pharmacokinetic properties and the safety profile of GA.
To our knowledge, this is the first report to describe the screening of GA derivatives as antiviral agents. The aim of the present study was to evaluate the antiviral activity of the synthesized 17-amino-17-demethoxygeldanamycin derivatives against a larger panel of viruses, including herpesvirus, hepatitis virus, retrovirus and picornavirus. On the basis of the antiviral activity in vitro, the selected compounds with good efficacy and their structural features were further investigated for their toxicity and antiviral activity in vivo.
Methods
Chemistry
Unless noted otherwise, all reagents and solvents were purchased from commercial sources and used without purification. GA was purchased from the Shanghai Institute of Pharmaceutical Industry (Shanghai, China) and the purity was >95% by HPLC analysis. 1H NMR spectra were recorded in CDCl3 or dimethylsulfoxide-d6 (DMSO-d6) solution with a Mercury-400 spectrometer (Varian, Inc., Palo Alto, CA, USA. High-resolution mass spectrometry (HRMS) data were obtained using an AutoSpec Ultima-Tof spectrometer (Waters, Milford, PA, USA) with an electrospray ionization (ESI) source. Melting points were determined using an X6 microscope melting point apparatus without correction. Column chromatography was performed on silica gel and eluted with a 1–20% methanol gradient in dichloromethane.
All 17-amino-17-demethoxygeldanamycin compounds were synthesized from GA and the corresponding amines (Figure 1). In our previous study, compounds
The purity of all GA derivatives in this study was >95%, which was achieved using the HLPC area normalization method with a C18 column (5 μm; 4.6×250 mm; Dikma Technologies, Inc., Lake Forest, CA, USA), eluted with water (containing 0.02 mol/l KH2PO4) and methanol at a flow rate of 1 ml/min, and the analyte was detected by ultraviolet absorption at 332 nm.
The general procedure for the synthesis of compounds
Compound
Compound
Compound
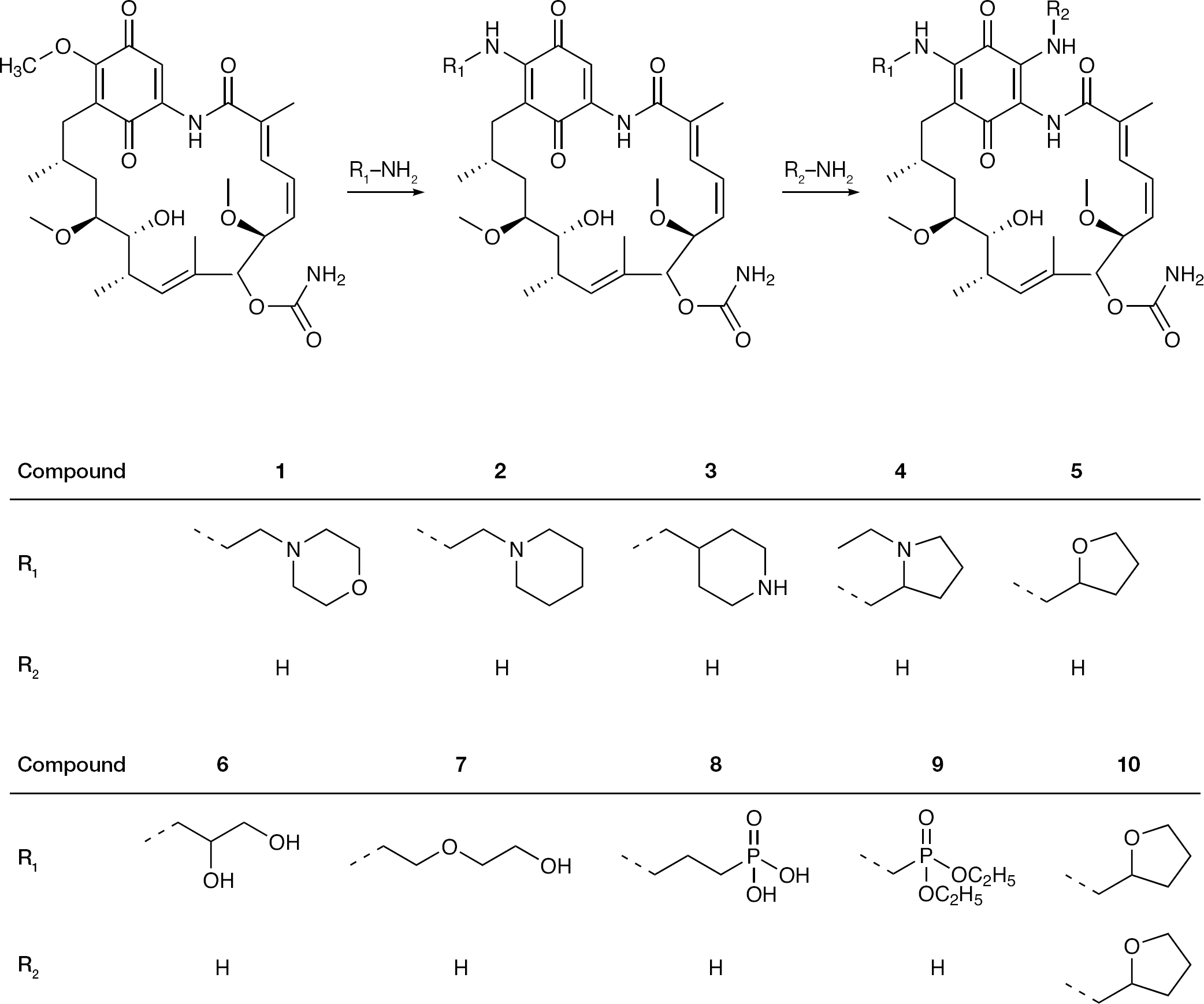
Synthesis and chemical structures of geldanamycin derivatives
Compound
Cells
African green monkey kidney (Vero) cells, human erythroleukaemia (HEL) cells and metallothionein T-4 (MT-4) cells were obtained from the Institute of Virology, Chinese Academy of Preventive Medicine (Beijing, China). Hepatocellular carcinoma (HepG2.2.15) cells were donated by Mount Sinai Medical Center (New York, NY, USA) and human liver cells (Huh7.5) were kindly provided by Vertex Pharmaceuticals, Inc., (Boston, MA, USA).
Viruses and plasmids
Human cytomegalovirus (HCMV; AD169 strain), herpes simplex virus type-1 (HSV-1; KOS strain), HSV type-2 (HSV-2; SAV strain), HIV-1 (strain IIIB) and Coxsackie viruses (Coxsackie virus B3 Nancy strain [Cox B3] and Coxsackie virus B6 Schmitt strain [Cox B6]) were all from the Institute of Virology, Chinese Academy of Preventive Medicine. The plasmid pFL-J6/JFH/JC1 with the complete chimeric HCV complementary DNA was provided by Vertex Pharmaceuticals, Inc. HCV virus stock was prepared as previously reported [19].
Reagents and animals
Lamivudine (3TC) was purchased from GlaxoSmithKline (Suzhou, China), zidovudine was purchased from Shanghai Desano Co. (Shanghai, China), aciclovir was purchased from Hubei Yitai Co. (Tianmen, China) and ribavirin was purchased from Xinxiang Chemial Pharmaceutical Co. (Xinxiang, China). Interferon-α2b was from Schering–Plough Co. (Brinny, Ireland). We synthesized valganciclovir from ganciclovir; the purity was >95% by HPLC analysis.
Kunming mice were purchased from the Chinese Academy of Medical Sciences (Beijing, China). Beijing ducklings were purchased from Beijing Nanyan Fowl, Inc. (Beijing, China). The animals were housed in standard laboratories at 22 ±1°C and had free access to food and water except for a short fasting period before intraperitoneal or oral drug administration. These experiments were approved by our institution's animal Ethics Committee (Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China).
Cytotoxicity assays
The cytotoxicity of tested compounds in Vero, HEL and HepG2.2.15 cells were monitored using cytopathic effect (CPE) assays as described previously [20]. Vero and HEL cells (2.5×104 cells/well) were cultured in 96-well plates. After 24 h, the monolayers were incubated in the presence of various concentrations of the test compounds. After 3 days of culture at 37°C and 5% CO2, the cells were assayed by CPE. HepG2.2.15 cells (2.0×104 cells/well) were cultured in 96-well plates. After 24 h, the monolayer cells were incubated in the presence of various concentrations of the test compounds for 4 days, then washed and a fresh medium containing the test compounds was added. After 8 days of incubation at 37°C and 5% CO2, the cells were assayed by CPE. The 50% cytotoxic concentration (CC50) was defined as the compound concentration that reduced the viability of untreated cell cultures by 50%.
The cytotoxicity in MT-4 and Huh7.5 cells were monitored by tetrazolium (MTT) assays [15]. Huh7.5 cells (1×104 cells/well) were planted into 96-well plates. After 6 h, test compounds with various concentrations were added. Cell viability was detected using MTT assays at 96 h. MT-4 cells (2×104 cells/well) were plated into a 96-well plate and incubated in the presence of various concentrations of the test compounds. After 4 days of culture at 37°C and 5% CO2, the cell viability was quantified using MTT assays.
Anti-HBV activity assay in vitro
The anti-HBV activities of compounds together with a positive control drug were tested in HepG2.2.15 cells. The HepG2.2.15 cells were cultured in 96-well plates and treated with the test compounds at 37°C for 4 days, then washed and a fresh medium containing the test compounds was added. After 8 days of incubation at 37°C and 5% CO2, the cells were harvested and intracellular DNA was extracted. The inhibition of the viral DNA in treated cells versus untreated cells was determined by dot blot hybridization.
Anti-HCV activity assay in vitro
Huh7.5 cells were cultured in 96-well plates at a density of 1×104 cells/well. After 6 h of incubation, cells were infected with HCV virus stock (45 IU/cell) and treated concomitantly with tested compounds or interferon-α2b. At 96 h after inoculation in 5% CO2 at 37°C, cells were harvested and intracellular total RNA was extracted using an RNeasy mini kit (Qiagen, Hilden, Germany). The HCV RNA was quantified directly with a one-step real-time reverse transciptase PCR kit (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's protocol.
Anti-HIV-1 activity assay in vitro
The anti-HIV-1 activity of the test compounds on HIV-1 replication was measured by inhibition of HIV-1 p24 antigen in MT-4 cell cultures as described previously [21]. MT-4 cells (2×105 cells/ml) infected with HIV-1 at 100 median tissue culture infective dose (TCID50)/ml were cultured in 96-well plates and incubated in the presence of various concentrations of test compounds or a positive control drug. After 4 days of culture at 37°C and 5% CO2, the culture supernatants were collected and assayed for their p24 antigen levels by ELISA.
Anti-HSV, Coxsackie virus and HCMV activity assays in vitro
Confluent Vero cells grown in 96-well microplates were infected with 100 TCID50 HSV-1, HSV-2, Cox B3 or Cox B6 virus. HEL cells were infected with HCMV. After 1 h adsorption at 37°C, the monolayer cells were washed with phosphate-buffered saline and incubated at 37°C in the maintenance medium (minimum essential medium plus 2% fetal bovine serum) with or without different concentrations of the test compounds or a positive control drug. The CPE in cells was recorded when the CPE of the viral control group reached >75% and the antiviral activity of compound was determined using Reed–Muench analysis [22].
Acute toxicity tests in mice
Kunming mice (20 ±2 g) were divided into groups with eight mice per group. The control group received DMSO only. On the basis of the pre-experiment results, each of the test compounds dissolved in DMSO was administrated with three doses to three groups of animals. Mice were placed unrestrained in their cages after treatment and observed for 5 days to monitor behavioural changes and death. The median lethal dose (LD50) was calculated using the Bliss method according the death number.
Anti-DHBV DNA activity in a DHBV infection model
One-day-old Beijing ducklings, inoculated intravenously with duckling hepatitis B virus (DHBV)-DNA-positive serum from Shanghai ducks (0.2 ml/duckling), were randomly divided into four groups (6 ducklings/group) for treatment. The virus control group was treated with 0.1% carboxymethylcellulose in water, the positive drug group was treated with 3TC (50 mg/kg) and the two test compound groups were treated with compound
The detection of DHBV DNA levels in the serum samples were done using the dot blot hybridization method as described previously [23,24]. Briefly, 50 μl of duckling serum was spotted directly onto the nitrocellulose filters. After denaturation and fixation, the filters were hybridized with a DHBV 32PDNA probe. Phosphorimaging dots were obtained on the films using autoradiography. Films were then scanned for the integrated optical density (IOD) of each dot and analysed using Gel-Pro Analyzer 3.1 (Media Cybernetics, Inc., Silver Spring, MD, USA). The IOD values of imaging dots were displayed as a marker of DHBV DNA levels.
In order to eliminate the background difference between groups, the relative ratio (IOD RR) between the post- and pretreatment result of each group was used to show the change in DHBV DNA levels. Lower IOD RR values represent a greater decrease in DHBV DNA levels.
Statistical analyses
The repeated measurement data from in vivo anti-DHBV DNA activity tests were analysed using the general linear models procedure in SPSS version 13.0 (SPSS, Inc., Chicago, IL, USA) for Windows. Multivariate analysis of variance was used to test the significance of the group effect. The criterion for significance was set at P<0.05.
Results
Eight viral strains, including herpesvirus (HCMV, HSV-1 and HSV-2), hepatitis virus (HBV and HCV), retrovirus (HIV-1) and picornavirus (Cox B3 and Cox B6), were used in the in vitro antiviral assay for the synthesized GA derivatives.
In vitro herpesvirus inhibitory activities
The antiviral activities of GA and its derivatives were tested in cultured HEL cells infected with HCMV and Vero cells infected with HSV. The activities of the GA derivatives against herpesvirus are shown in Table 1. All study compounds showed an inhibitory effect on the herpesvirus at test concentrations. Although the 50% inhibitory concentration (IC50) values of all derivatives were higher than that of the parent compound GA, the selectivity index (SI) of some derivatives were above that of GA because of their reduced cytotoxicity. The SI of compounds
Anti-herpesvirus activity and cytotoxicity of GA derivatives
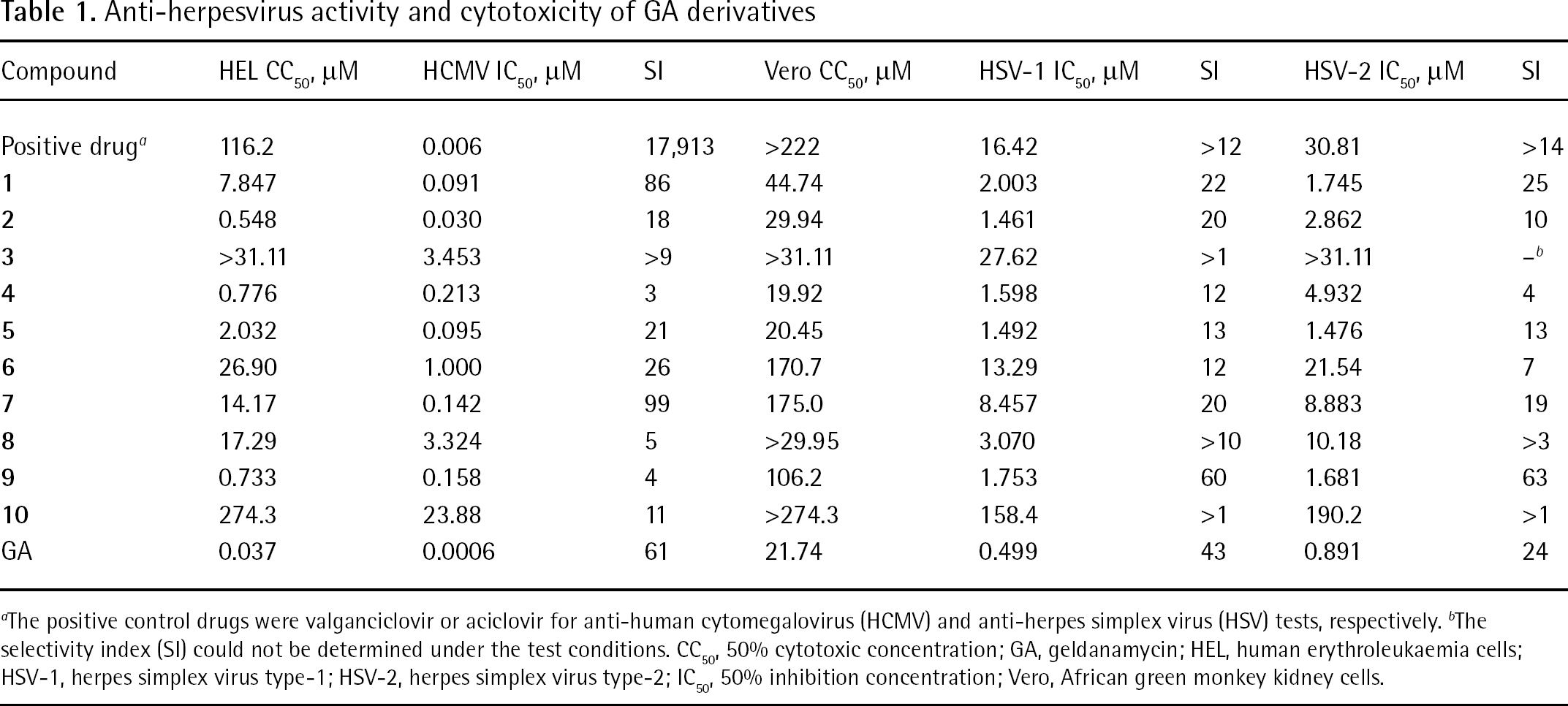
The positive control drugs were valganciclovir or aciclovir for anti-human cytomegalovirus (HCMV) and anti-herpes simplex virus (HSV) tests, respectively.
The selectivity index (SI) could not be determined under the test conditions. CC50, 50% cytotoxic concentration; GA, geldanamycin; HEL, human erythroleukaemia cells; HSV-1, herpes simplex virus type-1; HSV-2, herpes simplex virus type-2; IC50, 50% inhibition concentration; Vero, African green monkey kidney cells.
As summarized in Table 1, the CC50 values of the tested compounds were all significantly higher than that of GA, both in HEL and Vero cells: the CC50 values of all GA derivatives in the HEL cells were >10-fold of that of GA (0.037 μM). Among the compounds, the cytotoxicity of compounds
In vitro hepatitis virus inhibitory activities
The anti-HBV activity of GA and its derivatives was tested in HepG2.2.15 cells (Table 2). Compounds
Anti-HBV and anti-HCV activity and cytotoxicity of GA derivatives
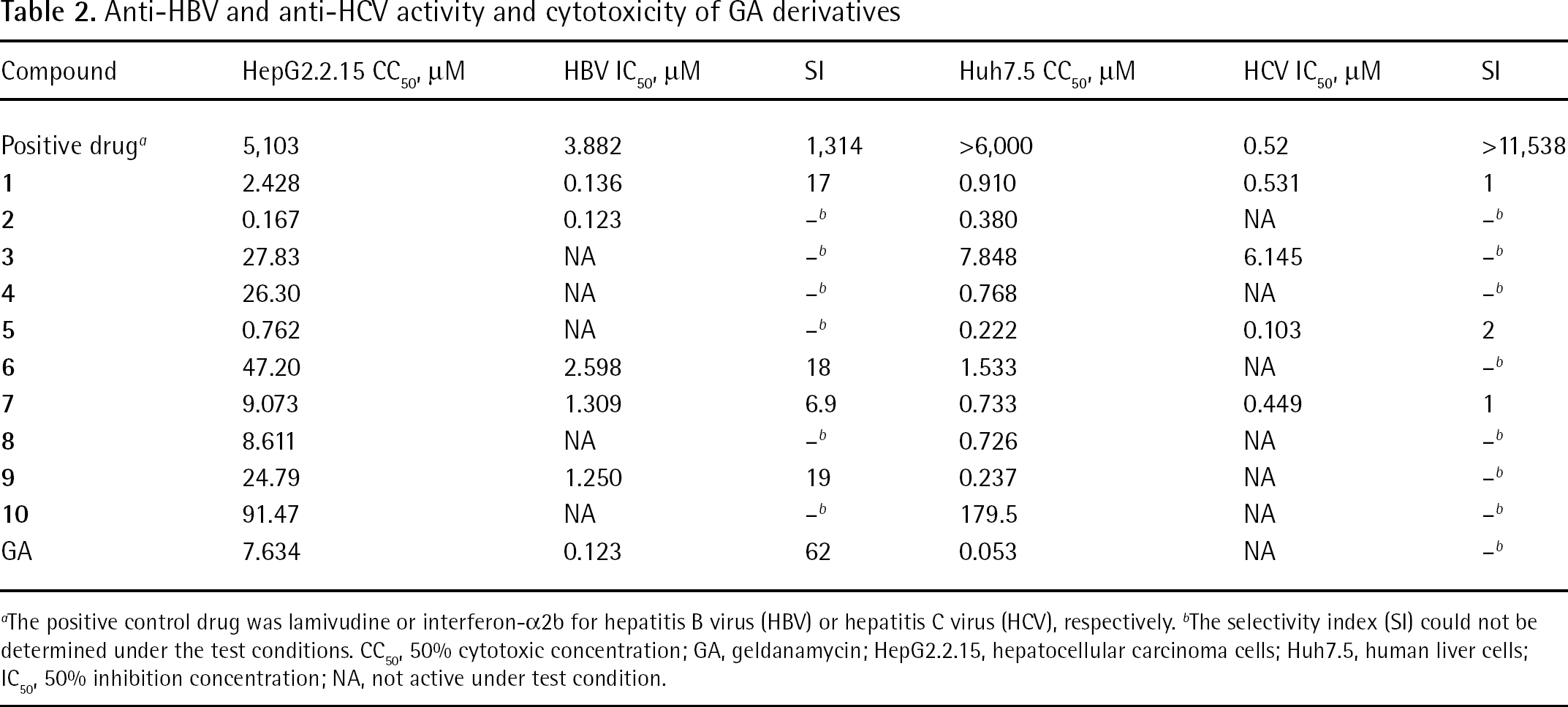
The positive control drug was lamivudine or interferon-α2b for hepatitis B virus (HBV) or hepatitis C virus (HCV), respectively.
The selectivity index (SI) could not be determined under the test conditions. CC50, 50% cytotoxic concentration; GA, geldanamycin; HepG2.2.15, hepatocellular carcinoma cells; Huh7.5, human liver cells; IC50, 50% inhibition concentration; NA, not active under test condition.
The anti-HCV activity of GA and its derivatives was tested in Huh7.5 cells. The CC50 values of all GA derivatives were higher than that of GA (Table 2), indicating that the cytotoxicity of the GA derivatives were lower than that of GA; however, the derivatives showed no significant anti-HCV activity under the test concentration.
In vitro HIV-1 virus inhibitory activities
Anti-HIV-1 activity of compounds
Anti-HIV-1 activity and cytotoxicity of GA derivatives
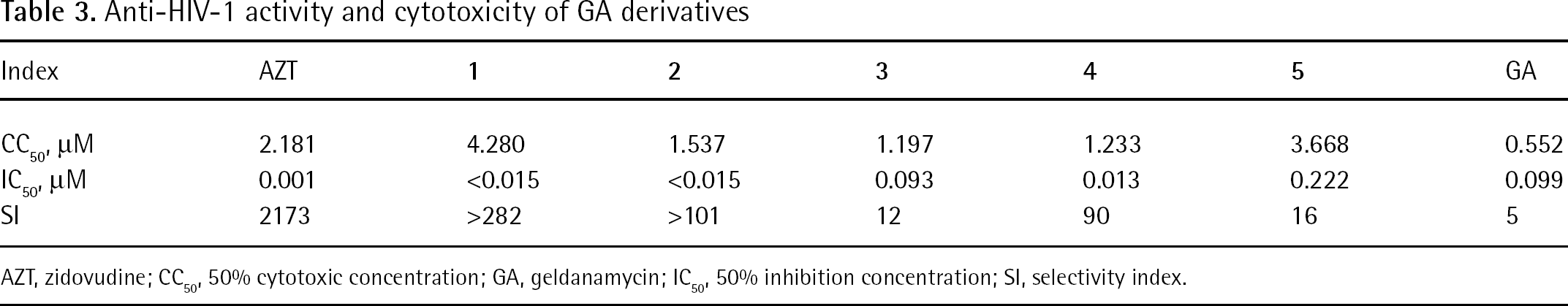
AZT, zidovudine; CC50, 50% cytotoxic concentration; GA, geldanamycin; IC50, 50% inhibition concentration; SI, selectivity index.
In vitro Coxsackie virus inhibitory activities
The lack of effective antiviral drugs against picornavirus led us to investigate the anti-Coxsackie virus activity of these GA derivatives (Table 4). Except for compounds
Anti-Coxsackie virus activity and cytotoxicity of GA derivatives
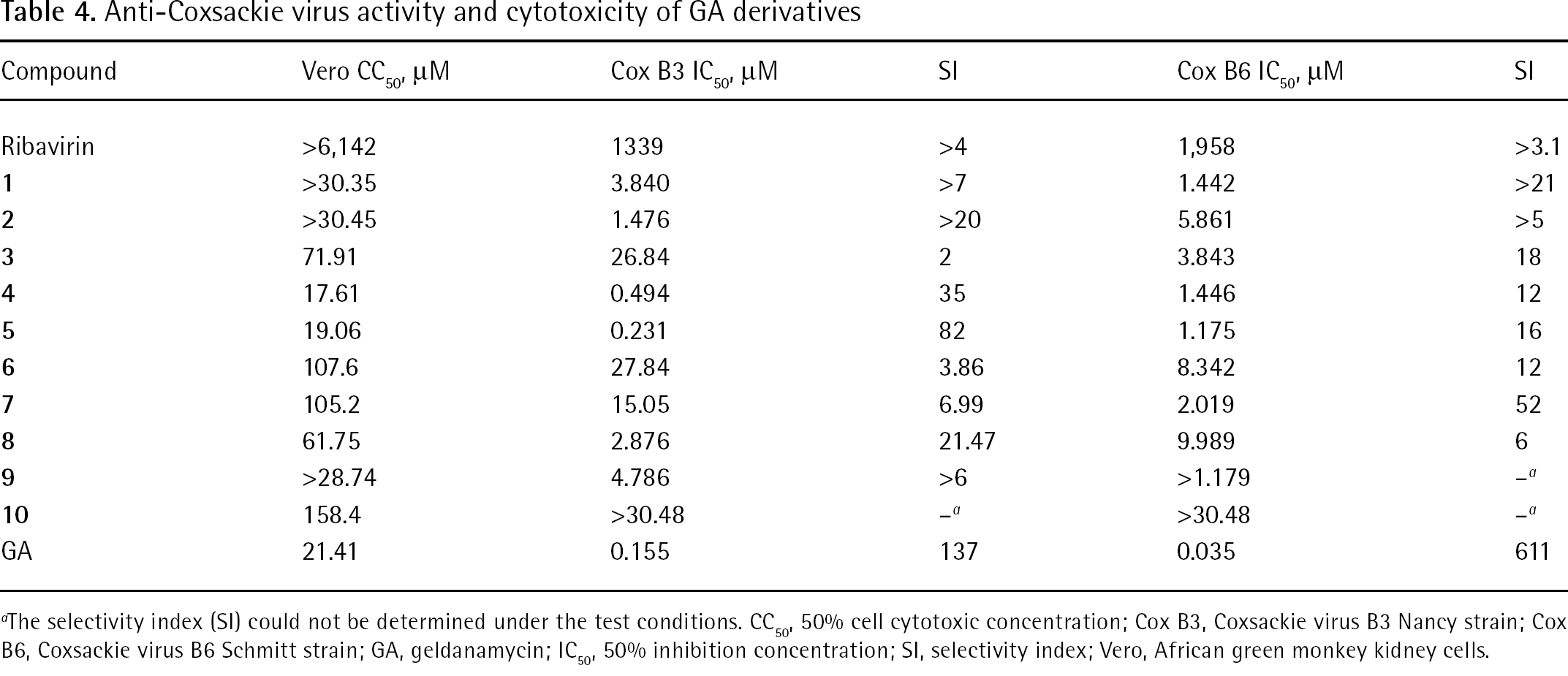
The selectivity index (SI) could not be determined under the test conditions. CC50, 50% cell cytotoxic concentration; Cox B3, Coxsackie virus B3 Nancy strain; Cox B6, Coxsackie virus B6 Schmitt strain; GA, geldanamycin; IC50, 50% inhibition concentration; SI, selectivity index; Vero, African green monkey kidney cells.
Acute toxicity in mice
To further evaluate the toxicity of GA derivatives, we investigated the acute toxicity of compounds
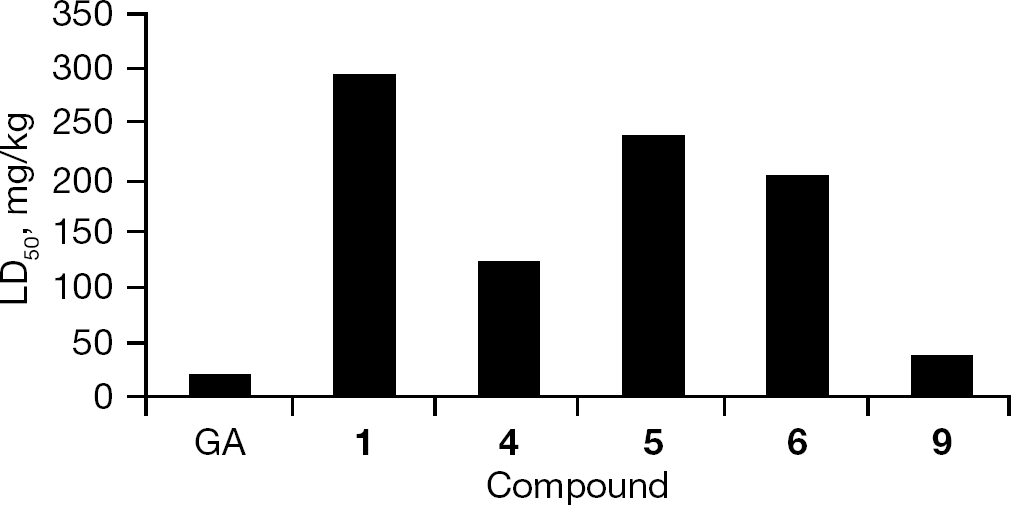
The acute toxicity of GA derivatives in mice
In vivo inhibitory activity of DHBV DNA replication in a DHBV infectious model
Based on the in vitro anti-HBV activity results and the relationships between chemical structure and antiviral activity as well as toxicity, compounds
Multivariate analyses of variance indicated that 3TC and compound
Discussion
The toxicity of GA has been a marked obstacle for its development for clinical use. This has spurred pharmaceutical chemists to pay great attention to developing less toxic GA derivatives. 17-allylamino-17-demethoxygeldanamycin has been reported to be a less toxic HSP90 inhibitor for treatment of tumours and has been enrolled into multiple clinic trials [25]. In our study, the majority of tested compounds also appeared to be less cytotoxic than GA in the cell cultures; the LD50 values of the selected six compounds were higher than that of GA in mice, giving further evidence of the low toxicity of these derivatives. We also found that the 17,19-disubstituted derivative (
17-Substituted GA derivatives contained both stronger and weaker antiviral activity than GA, but all had a largely decreased toxicity. According to the crystal structure of the GA–HSP90 complex [26], the substituent in the 17-position of GA is exposed to the outside cavity of the HSP90 protein, which suggests that the difference of substituents at the 17-position of GA are not crucial for the interaction of the test compounds with the HSP90 protein. This observation also helps us to understand why there was no obvious regularity observed between antiviral activity and structural difference in the 17-position in this study. Noticeably, compounds
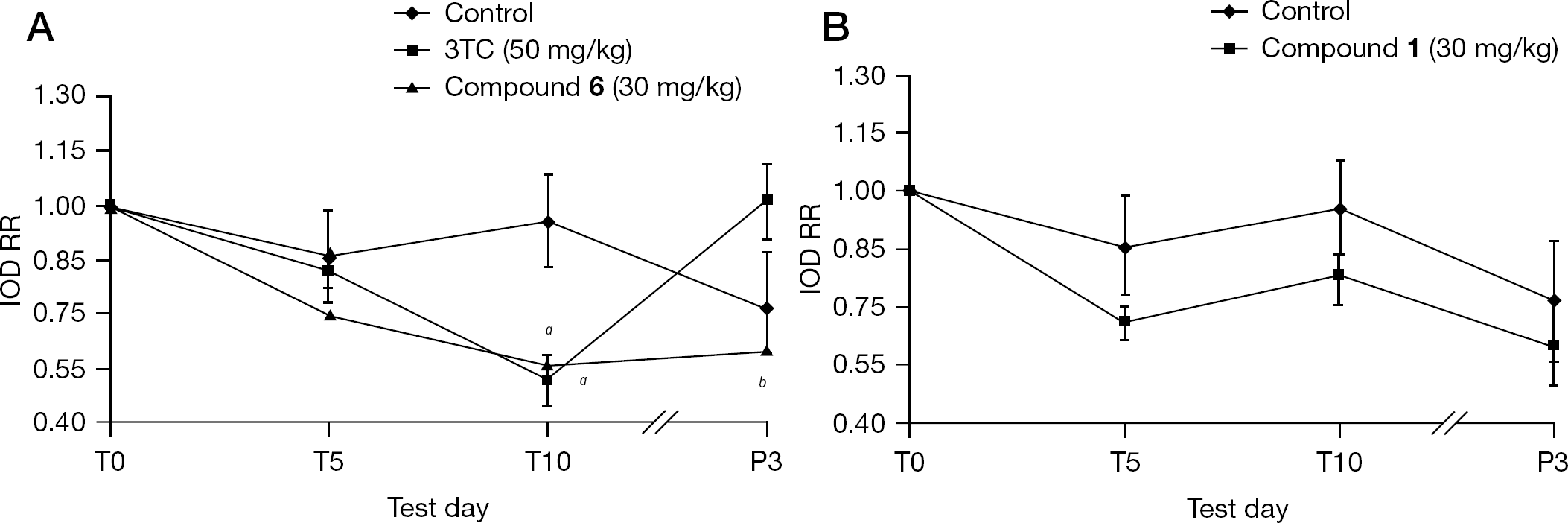
Mean changes in DHBV DNA levels of DHBV-infected duckling serum during different treatments
In China, the high incidence of HBV infection and the large number of HBV carriers is a severe problem. With the expectation of exploiting the potency of HSP90 inhibitor(s) against HBV, we investigated in vivo inhibitory activity of compounds
Unexpectedly, GA derivatives showed weaker or even no activity against HCV in Huh7.5 cells infected with the full-length viral genome. According to our previous study, GA and compounds
In summary, the broad-spectrum antiviral activity, low toxicity and good bioavailability of 17-amino-17-demethoxygeldanamycin derivatives were presented in this work, in comparison with GA. In particular, compound
Footnotes
Acknowledgements
This work was supported by the National Nature Science Foundation of China (30472076 and 30772600), the ‘863’ programme of the Ministry of Science and Technology of China (2006AA02Z414), and was partly supported by the National Major Science and Technology Project of China (‘Innovation and Development of New Drugs’; 2009ZX09301-003).
The authors declare no competing interests.
