Abstract
Background:
A combination of pegylated interferon and ribavirin is the current standard therapy for hepatitis C virus (HCV) infection, but this combination provides relatively low efficacy, especially in some patients with HCV genotype 1 infection; therefore, the development of novel therapeutic agents is required for further improvement in the treatment of chronic HCV infection.
Methods:
HCV pseudotype and subgenomic replicon assays were used in this study. The interaction of compounds with HCV receptors was examined using flow cytometry. Intracellular RNA levels were determined by semi-quantitative reverse transcriptase PCR.
Results:
Procyanidin B1 (PB1), a dimer of (−)-epicatechin and (+)-catechin, purified from Cinnamomi cortex, inhibits infection by vesicular stomatitis virus and HCV pseudotype virus in Huh-7 cells, with 50% effective concentrations of 29 and 15 μM, respectively. No inhibitory effects were observed in each component of PB1. We found that PB1 does not interfere with viral entry or receptor expression, but inhibits HCV RNA synthesis in a dose-dependent manner.
Conclusions:
These results indicate that PB1 suppresses HCV RNA synthesis, possibly as a HCV RNA polymerase inhibitor. Our results might contribute towards the development of more effective inhibitors for HCV infection from natural plants.
Introduction
Hepatitis C virus (HCV) is an enveloped, single-stranded, positive RNA virus and is the only member of the Hepacivirus genus of the Flaviviridae. HCV infection, affecting approximately 170 million people worldwide, leads to chronic hepatitis, liver cirrhosis and, ultimately, hepatocellular carcinoma [1]. Currently, no protective vaccine against HCV is available and treatment is mainly restricted to a combination of pegylated interferon (PEG-IFN) and ribavirin [2]. The sustained virological response (SVR) of treatment is dependent on the HCV genotype. For HCV genotype 1, the SVR rates are between 44% and 56% at 48 weeks of the therapy, whereas for genotypes 2 and 3, they are between 61% and 76% at only 24 weeks of therapy [3,4], indicating that the current standard therapy is insufficient for HCV-infected patients, especially for patients infected with HCV genotype 1. Moreover, the regimen has poor tolerability, especially for long-term treatment – up to 48 weeks – often leading to dose reductions or treatment interruptions [3]. In addition, recombinant IFN is expensive and requires injections for administration. Considering the limited efficacy, serious side effects and high cost, the development of novel, potent and tolerable antiviral agents is urgently needed.
To date, many specifically targeted antiviral compounds designed to inhibit HCV serine protease or RNA-dependent RNA polymerase have been examined in clinical trials, such as telaprevir (an HCV protease inhibitor [5]) and filibuvir (a non-nucleoside HCV polymerase inhibitor [6]) and many other promising compounds. Telaprevir monotherapy for 2 weeks resulted in a reduction of up to 5 log10 IU/ml in plasma HCV RNA levels in patients infected with HCV genotype 1 in Phase Ib trials [5]. Currently, telaprevir is in a Phase III clinical trial. Filibuvir allosterically inhibits HCV RNA polymerase through binding at the ‘thumb 2’ site of RNA polymerase. Filibuvir has been recently tested in a Phase II clinical evaluation in patients with HCV genotype 1. Up to 75% of patients treated with filibuvir plus PEG-IFN/ribavirin had undetectable viral load at week 4.
In our previous study, procyanidins, a class of flavanols, purified from Cinnamomi cortex inhibited severe acute respiratory syndrome coronavirus (SARS–CoV) replication in vitro [7]. We examined the effects of procyanidin B1 (PB1) on the entire HCV replication process using two HCV assays (HCV pseudotype and subgenomic replicon systems) and further studied the mechanisms of action of PB1 on HCV replication. Our results might contribute towards the development of novel effective compounds from natural plants for HCV therapy.
Methods
Cells and reagents
Human hepatocarcinoma Huh-7 cells were maintained in Dulbecco's modified Eagle's medium supplemented with 10% heat-inactivated fetal bovine serum (Gibco BRL, Grand Island, NY, USA), 100 U/ml penicillin G, 100 μg/ml streptomycin and L-glutamine (Gibco BRL). PB1 was purified from Cinnamomi cortex as described previously [8] and its structure is shown in Figure 1A. Chlorpromazine, methyl-β-cyclodextrin (MβCD), ribavirin, (−)-epicatechin and (+)-catechin were purchased from Sigma (St Louis, MO, USA), dissolved in water and filtrated.
Pseudotype virus infection assays
We previously reported the generation of vesicular stomatitis virus (VSV)-based pseudotype viruses bearing HCV envelope proteins E1 and E2 (HCVpv) or VSV glycoprotein (VSVpv), which exhibited high infectivity in Huh-7 cells, as shown in Figure 1B [9]. Briefly, 293T cells were transfected with an expression plasmid encoding the HCV E1 and E2 genes of Con1 strains (HCV genotype 1b) and then infected with a VSV envelope protein G (VSV-G)-complemented pseudotype virus (VSVΔG/GFP-G). The HCVpv released from 293T cells were able to infect Huh-7 cells but were unable to produce infectious progeny virus. VSVpv was produced in 293T cells transfected with pVSVΔG/GFP and pCAG-VSV-G and used as controls. The green fluorescent protein (GFP) in each pseudotype virus was replaced by a firefly luciferase gene. Prior to HCVpv or VSVpv infection, Huh-7 cells were incubated with PB1 at 37°C for 1 h. After 48 h of the pseudotype virus infection, cells were harvested, lysed and subjected to luciferase assays using a Mithras LB940 Reader (Berthold Technologies, Bad Wildbad, Germany). The 50% effective concentration (EC50) was defined as the concentration that reduced viral replication by 50%.
Entry assays based on intracellular p24 gag
Chlorpromazine, a cationic amphiphilic compound, inhibits clathrin-dependent endocytosis by preventing the assembly of clathrin-coated pits at the cellular membranes [10]. MβCD disrupts cholesterol-rich microdomains and inhibits both caveola and lipid-raft-dependent endocytosis [11]. To determine the effects of the compounds on viral entry, HIV type-1 (HIV-1)-based pseudotype virus bearing VSV-G (VSV-G/HIVpv) was produced in 293T cells transfected with pCMV-VSV-G, pCAG-HIVgp and pCS-II-luc. Huh-7 cells were pretreated with various concentrations of compounds (PB1, chlorpromazine or MβCD) for 1 h at 37°C in a 48-well plate, inoculated with VSV-G/HIVpv at 5 ng/well of p24. After 4 h of inoculation, the cells were washed twice with phosphate-buffered saline (PBS) and treated with 0.25% trypsin-EDTA (Gibco BRL) at 37°C to degrade the unincorporated virus particles. Intracellular p24 of the HIV-1 gag protein was measured using a Retro-Tek HIV-1 p24 ELISA kit (ZeptoMetrix Copororation, Buffalo, NY, USA).
Time-of-addition assays
A time-of-addition experiment was carried out in Huh-7 cells. PB1 (100 μM) was added at 0, 1, 4, 8, 12 and 24 h after VSVpv or HCVpv infection in Huh-7 cells. The luciferase activity was measured after 48 h of infection as described previously [12,13].
Cytotoxicity assays
The cytotoxity of the compounds was examined using the Cell Counting Kit-8 (Dojindo Molecular Technologies, Inc., Kumamoto, Japan). Huh-7 cells were incubated with various concentrations of the compounds for 72 h to determine the cell viability. The 50% cytotoxicity concentration (CC50) was defined as the concentration that reduced cell viability by 50%.
Flow cytometric analyses
Huh-7 cells were pretreated with or without PB1 (100 μM) at 37°C for 1 h. After washing with fluorescence-activated cell sorting (FACS) buffer (PBS with 2% fetal calf serum and 0.1% sodium azide), the cells were detached using 1 mM EDTA (Gibco BRL) for 30 min on ice. Cells were incubated with phycoerythrin (PE)-conjugated CD81 (Beckman Coulter, Fullerton, CA, USA) or CD71 (BD Pharmigen, San Diego, CA, USA) for 30 min at 4°C, subsequently washed and subjected to flow cytometry analysis (Cytomics FC500; Beckman Coulter). PE mouse immunoglobulin G2aκ monoclonal antibody (BD Pharmigen) was used as the isotype control. 7-Amino-actinomycin (BD Pharmigen) was used as a viability probe for dead cell exclusion.
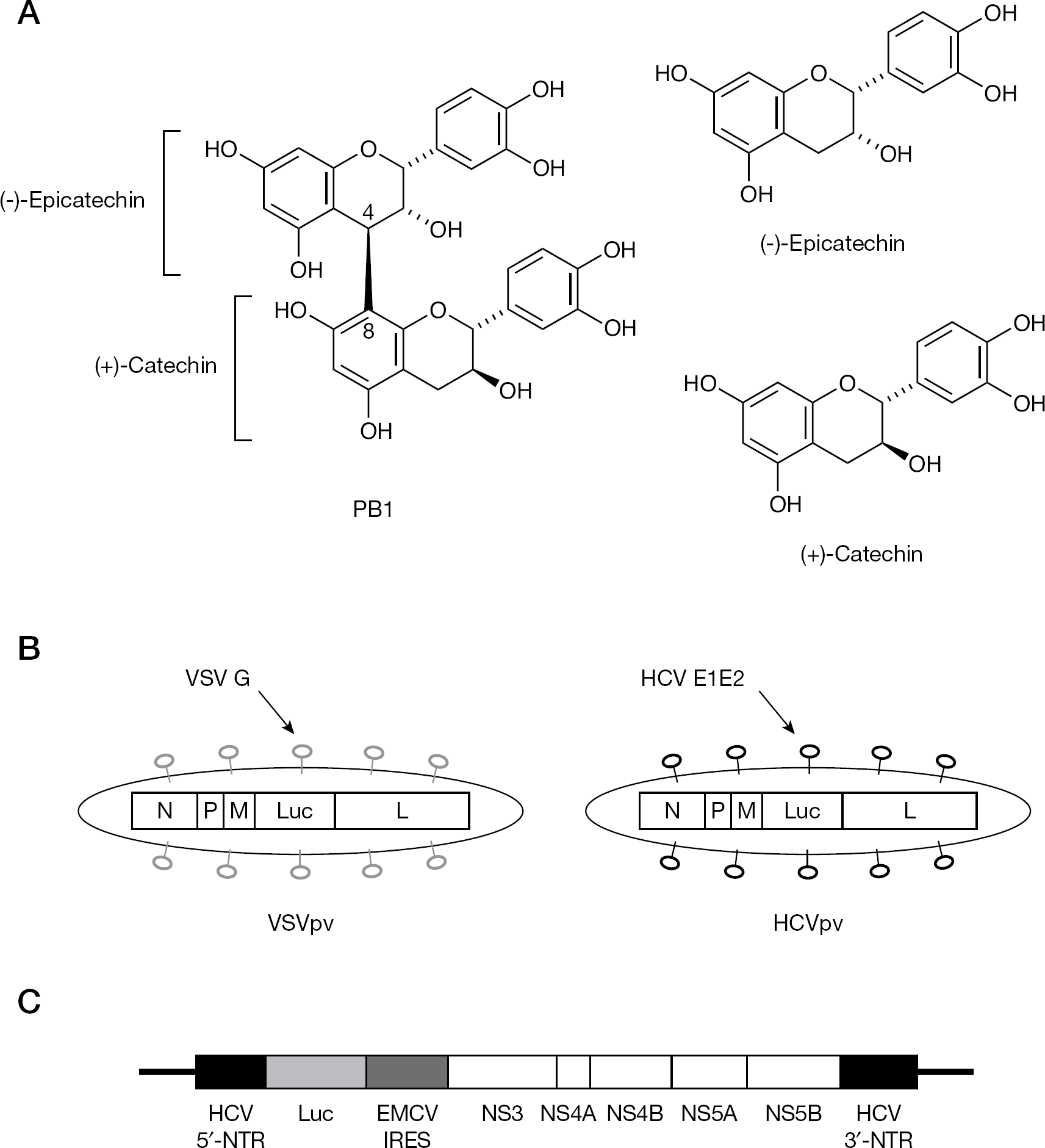
Structures of PB1 and its components, and schematic diagrams of pseudotype viruses and the HCV replicon
HCV replicon assays
The plasmid pLMH14 contains the 5′-non-translated region (5′-NTR), the first 36 nucleotides of the core region fused directly with the firefly luciferase reporter gene and the internal ribosome entry site (IRES) element from encephalomyocarditis virus (EMCV) that directs translation of the HCV non-structural (NS) proteins from NS3 to NS5B and the 3′-NTR as shown in Figure 1C. HCV replicon assays were performed as described previously [14]. Briefly, linearized pLMH14 at the restriction enzyme site XbaI was used for in vitro transcription of HCV replicon RNA using the SP6/T7 transcription kit (Roche, Mannheim, Germany).
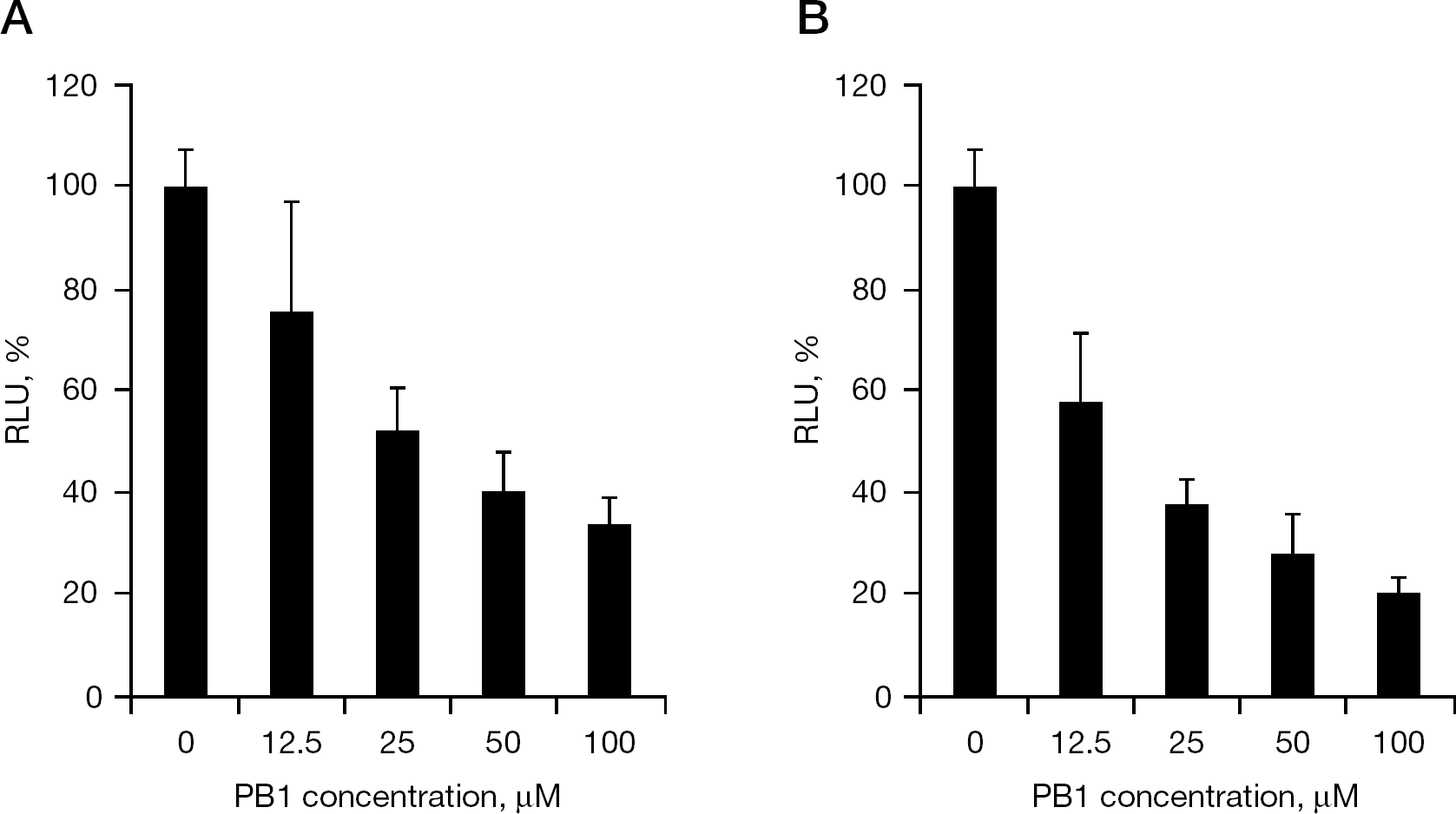
Effects of PB1 on VSV-based pseudotype virus infection
Replicon RNA (0.5 μg) was transfected into Huh-7 cells in 24-well plates with lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). After 5 h, the medium was replaced and serially diluted PB1, ribavirin, (−)-epicatechin or (+)-catechin was added to Huh-7 cells. The luciferase activity was determined at 3 h and 72 h post-transfection. The luciferase activities at 3 h were used for normalization of the transfection efficiency.
Semi-quantitative RT-PCR
At 24 h prior to transfection of HCV replicon RNA (0.5 μg), Huh-7 cells were seeded at a density of 2×105 cells/well in 12-well plates. After 72 h of PB1 treatment, total cellular RNA was extracted using TRIzol reagent (Invitrogen) and subjected to reverse transcription coupled PCR (RT-PCR) with a One Step RNA PCR kit (TaKaRa Bio, Inc., Shiga, Japan). Reactions were incubated at 50°C for 30 min for reverse transcription, for 2 min at 94°C to inactivate the avian myeloblastosis virus reverse transcriptase and subsequent PCR amplification of 30 cycles of denaturation at 94°C for 30 s, annealing at 55°C for 15–45 s and extension at 72°C for 1 min.
5′-NTR and firefly luciferase genes in the HCV replicon RNA (Figure 1C) and cellular β-actin were amplified with the following primers: 5′-NTR (forward 5′-ACACTCCACCATAGATCACTCCCCT-3′ and reverse 5′-CGGGGCACTCGCAAGCACCCTATCA-3′), firefly luciferase (forward 5′-CACATCTCATCTACCTCCCG-3′ and reverse 5′-TCCACAACCTTCGCTTCA-3′) and β-actin (forward 5′-ATCTGGCACCACACCTTCTACAATGAGCTGCG-3′ and reverse 5′-CGTCATACTCCTGCTTGCTGATCCACATCTGC-3′).
Results
PB1 suppressed VSVpv and HCVpv infection
A pseudotype virus can make only a single round of infection without producing infectious progeny virus in infected cells, making it an excellent system for studying the viral replication steps between entry and RNA genome synthesis. Two pseudotype viruses were used: VSVpv comprising VSV envelope protein G and env-deleted VSV genome (ΔG-VSV), and HCVpv comprising HCV E1E2 and ΔG-VSV. The only difference between VSVpv and HCVpv is the envelope (Figure 1B). In this assay, PB1 inhibited both VSVpv and HCVpv infection in a dose-dependent manner (Figure 2). The mean ±SD EC50 for VSVpv and HCVpv was 29 ±7.3 μM and 15 ±3.9 μM, respectively. These results indicate that PB1 blocks VSV and HCV pseudotype virus infection by interference with the viral entry step through HCV or VSV envelope proteins and/or intracellular VSV genome replication process.
PB1 inhibited viral replication at post-entry step(s)
To determine the mechanism of action of PB1 on HCV replication, a time of addition experiment was performed. The assay provided time-dependent activity that enabled estimation of the inhibitory step(s) of compounds. As shown in Figure 3, the luciferase activity was suppressed to <50% even when PB1 was added after 12 h of VSVpv or HCVpv pseudotype virus infection. A previous study showed that nearly 90% of VSV and HCV was internalized into Huh-7 cells within 1.5 and 3 h, respectively [15]. These results suggest that PB1 blocks viral replication after the internalization.
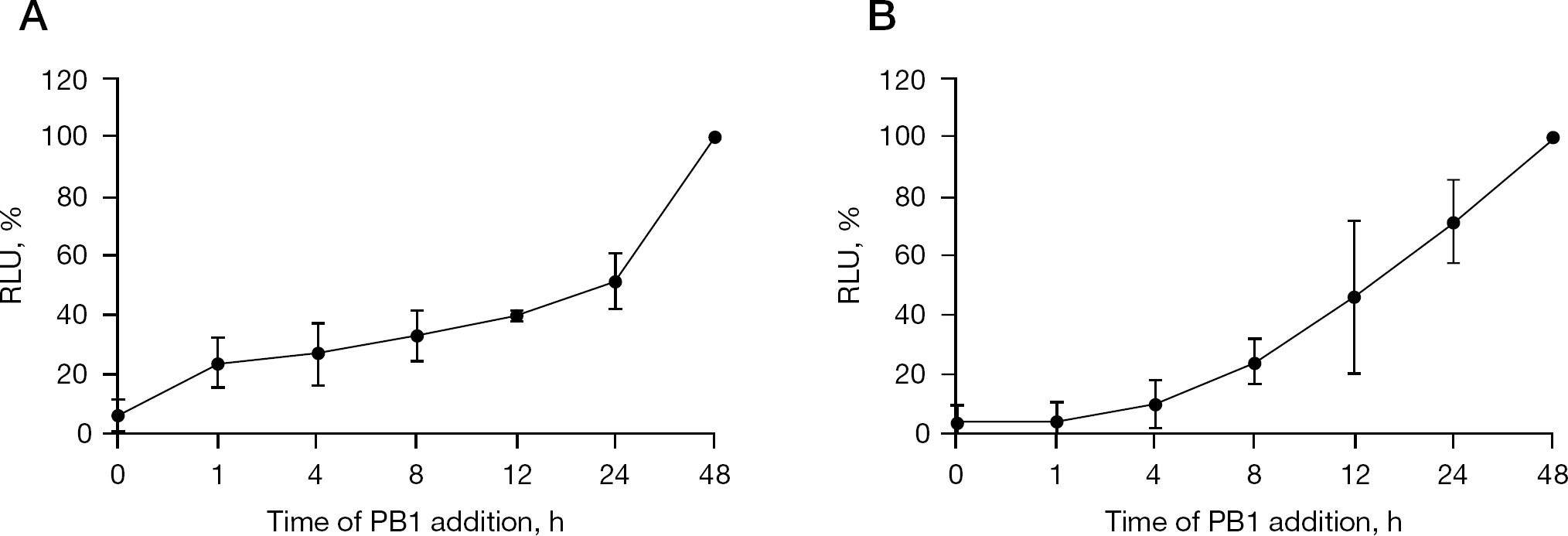
Time of addition analysis of PB1 in Huh-7 cells
PB1 had no inhibitory effects on viral entry
To further determine whether PB1 affects the expression of CD81, one of the main receptors of HCV [16], flow cytometric assays were performed. CD71 (transferrin receptor) was used as a negative control as PB1 does not affect CD71 expression [7]. The proportion of positive-staining cells and mean fluorescence intensity (MFI) were little changed by PB1 treatment (Table 1).
Flow cytometric analysis of CD81 expression

Huh-7 cells were pretreated with or without procyanidin B1 (PB1; 100 μM) at 37°C for 1 h. Cells were washed, detached, incubated with phycoerythrin-conjugated CD81 or CD71 (transferrin receptor) for 30 min at 4°C and then subjected to flow cytometric analysis. Positive-staining cells and mean fluorescence intensity (MFI) are shown.
Entry of both VSV and HCV depends on clathrin-dependent endocytosis [10,17]. To examine the effects on viral entry, we therefore used VSV-G/HIVpv. After 4 h of infection during the reverse transcription step of the HIV-1 genome, we determined the amount of intracellular HIV-1 gag protein p24, which is highly correlated with that of entered virus particles [18]. Consistent with previous reports [11], chlorpromazine decreased the intracellular p24 level in a dose-dependent manner (Figure 4A), whereas MβCD did not (Figure 4B). PB1 had little effect on the intracellular p24 levels (Figure 4C). Moreover, the binding of VSV-G/HIVpv at 4°C was maintained even in the presence of PB1 (Figure 4D). These results indicate that PB1 inhibits the virus infection rather than the entry; thus, at least for HCV and VSV infection, it is unlikely that PB1 blocks viral replication through inhibition of clathrin-dependent endocytosis.
PB1 inhibited HCV RNA synthesis
We next examined the effects of PB1 on HCV RNA replication. Although ribavirin has been shown to inhibit RNA synthesis in vivo, it has little effect on HCV RNA replication in the replicon system in vitro [19]. Consistent with the previous report, the inhibitory effect of ribavirin seems to be induced by its cytotoxity (Figure 5A); however, PB1 suppressed the replication of HCV replicon in a dose-dependent manner (Figure 5B). The mean ±SD EC50 and CC50 values were 72 ±7.4 μM and 465 ±79 μM, respectively, with the result that the selective index (CC50/EC50) was calculated to be 6.5. Because PB1 is composed of (−)-epicatechin and (+)-catechin by C4–C8 linkage (Figure 1A), we next examined both components solely for anti-HCV activity. The single components, (−)-epicatechin or (+)-catechin, had little inhibitory effects on HCV replicon (Figure 5C and 5D) indicating that the dimer structure of PB1 is required for inhibitory activity.
The effects of PB1 on the synthesis of HCV RNA in the cells were examined by semi-quantitative RT-PCR. As shown in Figure 6, the signal intensity for 5′-NTR and luciferase correlated with the amount of intracellular HCV RNA copies; the signal intensity was weak at higher concentrations (100 μM and 50 μM of PB1) compared with the signal intensity at lower concentrations. In parallel experiments, the signal intensity for β-actin was little changed up to 100 μM PB1 indicating that PB1 directly inhibited the intracellular HCV RNA replication without affecting cellular messenger RNA synthesis. These results demonstrated that PB1 blocks HCV replication through the inhibition of RNA synthesis.
Discussion
Procyanidins, which are oligomers and/or polymers of monomeric flavonoids, exert strong antioxidant and radical scavenging activity compared with vitamins C and E [20]. A procyanidin derivative, PB1, purified from Cinnamomi cortex has inhibitory effects on the replication of herpes simplex virus [21], HIV-1 [22] and SARS–CoV [7]. However, to date, the anti-HCV activity of PB1 has not been documented; therefore, in this study, we examined the effects of PB1 on HCV replication and revealed that PB1 inhibited HCV replication. Procyanidins have many isomeric forms depending on the extent of polymerization (dimers to pentadecamers) and the nature of their constituent units [23]. Some biological activities are related to the degree of polymerization. In our study, (−)-epicatechin and (+)-catechin, which are components of PB1, alone showed little anti-HCV activity indicating that the polymerization of catechins plays an important role in antiviral activity.
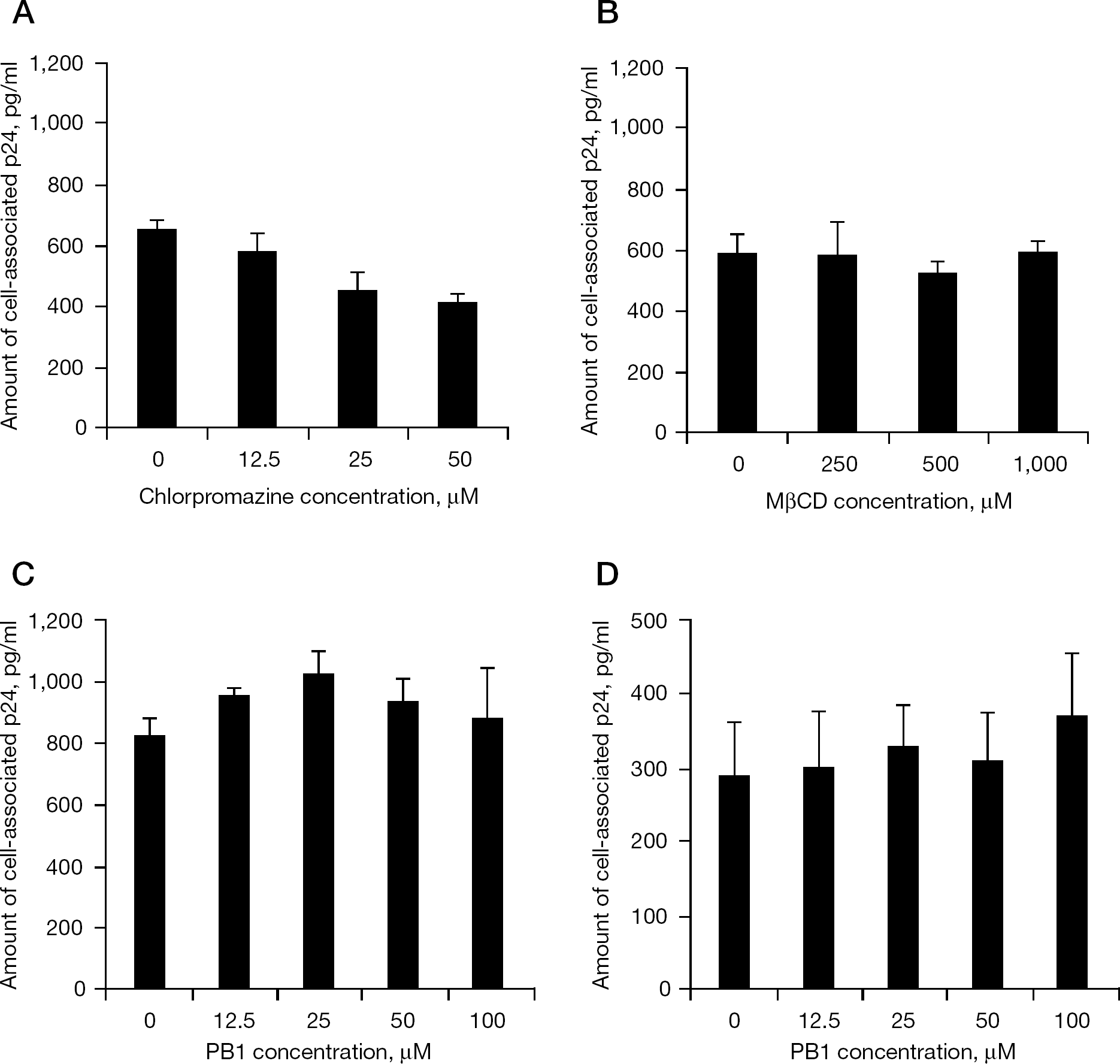
PB1 inhibits VSV-G/HIVpv binding and endocytosis in Huh-7 cells
The currently recommended therapy for chronic hepatitis C is a combination of PEG-IFN and ribavirin for 24 or 48 weeks. IFN has potent antiviral activity but indirectly inhibits viral replication. Rather, it induces IFN-stimulated genes, which establish a non-virus-specific antiviral state in the cell [19]. Several possible mechanisms of action of ribavirin in HCV have been proposed including: inhibition of inosine monophosphate dehydrogenase, leading to depletion of guanosine triphosphate (GTP) necessary for viral RNA synthesis [24,25]; induction of lethal mutagenesis in the viral RNA genome [26,27]; and alteration of the TH1/ TH2 balance favouring a TH1 CD4+ T-cell response and thus leading to viral clearance [19,28]. Some studies also demonstrated that ribavirin directly inhibits HCV RNA polymerase in vitro [19,25,29]. The guanosine analogue, ribavirin, is intracellularly phosphorylated into triphosphate forms that act as a chain terminator through misincorporation of ribavirin triphosphate by HCV RNA polymerase, resulting in the inhibition of HCV replication. However, it is unlikely to be its major mechanism of action against HCV in vivo. By contrast, the mechanism of action of PB1 appears to be distinct from those of IFN and ribavirin. In our study, PB1 inhibited both VSV and HCV pseudotype virus infection in Huh-7 cells. PB1 interfered little with viral entry but inhibited HCV RNA synthesis in a dose-dependent manner. It is possible that PB1 inhibits other targets including cellular factors required for RNA synthesis. However, PB1 can selectively block HCV RNA synthesis without influence on cellular RNA (β-actin) synthesis; therefore, it is likely that PB1 suppresses HCV RNA synthesis as a HCV RNA polymerase inhibitor. Unfortunately, HCV RNA dependent RNA polymerase is not commercially available at present. To strengthen our hypothesis, we examined whether PB1 inhibits the polymerase activity of the T7 RNA polymerase in an enzymetic assay and demonstrated that PB1 directly inhibits T7 RNA polymerase activity (SL et al., data not shown). The effects of PB1 on NS3 helicase or NS3/4A serine protease will also need to be determined because the inhibition of helicase or protease can lead to the suppression of HCV RNA synthesis. Further experiments are needed to reveal the detailed mechanisms of HCV inhibition in future; however, at present, HCV enzymes mentioned above are not commercially available.
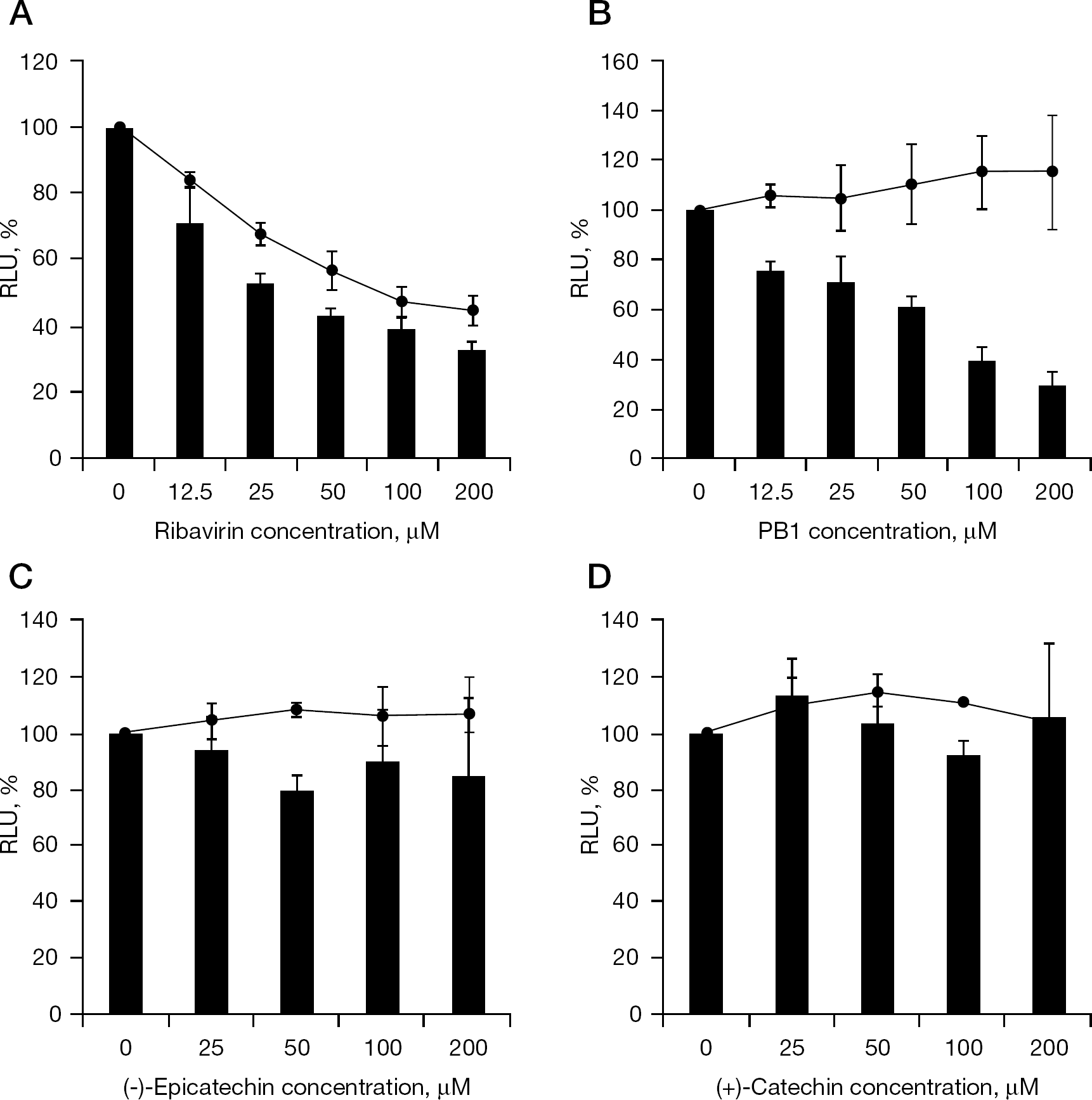
PB1 inhibits HCV replication
In contrast to tumour viruses that cause tumourigenesis, such as human T-cell lymphotropic virus type-1 and hepatitis B virus (HBV), the HCV genome is not integrated into its host genome and has a predominantly cytoplasmic life cycle [30]. Although the mechanisms of tumourigenesis still remain unclear, chronic immune-mediated inflammation and associated oxidative chromosomal DNA damage probably play an important role in HCV-induced hepatocellular carcinoma [31,32]. Procyanidins inhibit tumour growth and induce apoptosis in various tumour cells [23,33,34]. One of the possible mechanisms of the suppressive effects on carcinogenesis appears to be antioxidant and anti-inflammatory activities suggesting that procyanidins might prevent the development of HCV-related hepatocellular carcinoma. Procyanidins, abundant in natural plant products including berries, grapes, cinnamon and pycnogenol, have few adverse effects on normal cells compared with current therapeutic agents [35] suggesting that they could be good candidates as additional or supportive anti-HCV agents used in prolonged therapy.
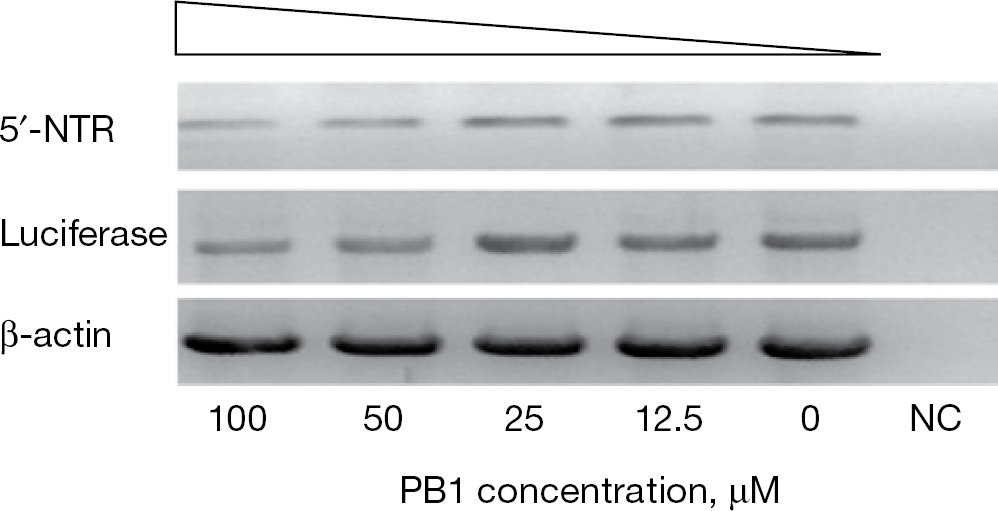
Expression of messenger RNA by RT-PCR
Several studies showed that procyanidins can be absorbed in rats and humans [36–39]. Procyanidins were detected in the human plasma as early as 30 min and reached the maximal concentrations by 2 h after oral administration [38]. Holt et al. [38] reported that 2 h after the ingestion of a procyanidin-rich cocoa (0.375 g/kg) containing 256 mg procyanidin, the concentration of procyanidin B2 in human plasma reached 41 nM on average, indicating that oral administration of 62 g of cocoa procyanidins is needed to reach a plasma concentration of 10 μM. In another study, the concentrations of procyanidin B2 in human plasma reached 100 nM after the consumption of cocoa (0.5 g/kg), of which 9.7 mg/g was procyanidins [39], indicating that 38 g of cocoa procyanidins is required to reach a plasma concentration of 10 μM at 2 h after oral administration. PB1 showed moderate antiviral activity not only in our study but also in other studies [7,21,22], which reported antiviral activity at concentrations (1–100 μM). To improve the physiological concentration, certain chemical modifications that enhance antiviral activity, plasma stability and/or absorption of PB1 are required. The development of drug delivery systems might also enable to improve bioavailability and antiviral efficiency.
In this study, we demonstrated that a catechin dimer from natural plants, PB1, suppresses HCV RNA synthesis possibly as a HCV RNA polymerase inhibitor. Our study could contribute to the development of novel therapeutic strategies for the prevention and treatment of HCV as well as other viral infections.
Footnotes
Acknowledgements
This work was supported, in part, by Grants-in-Aid for Special Educational Grant from the Ministry of Education, Culture, Sports, Science, and Technology, and for the Scientific Research Expenses from the Ministry of Health, and Welfare (Japan). LS has been supported by the Japanese AIDS Foundation (Tokyo, Japan).
The authors declare no competing interests.
