Abstract
Background:
A number of compounds were examined for their inhibitory effects on bovine viral diarrhoea virus (BVDV), a surrogate model of hepatitis C virus, in cell cultures. Among them, some diphenylmethane derivatives were found to be selective inhibitors of BVDV.
Methods:
Determination of compounds for their anti-BVDV activity was based on the inhibition of virus-induced cytopathic effect in Madin–Darby bovine kidney cells and reduction of infectious virus particles in culture supernatants. To gain insight into the mechanism of action, the inhibition of viral entry and RNA synthesis in the host cells was also determined by real-time reverse transcription-PCR.
Results:
Among the test compounds, four diphenylmethane derivatives significantly inhibited BVDV replication with a 50% effective concentration ranging between 6.3 and 10.8 μM. They were not cytotoxic at concentrations up to 100 μM. The representative compound, SH-595A, reduced the virus titre of culture supernatants in a dose-dependent manner. In addition, the compound appeared to somewhat affect viral entry to the host cells. Although SH-595A was inhibitory to viral RNA synthesis, the inhibition was achieved only at high concentrations and was not comparable to its antiviral activity.
Conclusions:
The novel diphenylmethane derivatives are effective against BVDV replication and might have a unique mechanism of action.
Introduction
Human chronic hepatitis is often caused by persistent infection with hepatitis C virus (HCV). This persistent infection commonly leads to liver fibrosis, cirrhosis and hepatocellular carcinoma [1]. Pegylated interferon-α in combination with the nucleoside analogue ribavirin is currently used for the treatment of HCV infection [2]; however, the treatment outcome is dependent on the genotype of HCV. Indeed, the treatment is not effective in approximately 50% of patients who are infected with genotype 1b. Furthermore, this combination therapy is expensive and is often associated with unacceptable side effects in many patients; therefore, novel therapies aiming at complete and permanent eradication of HCV from the patient are still mandatory.
HCV belongs to the family Flaviviridae, which include three genera: hepacivirus, flavivirus and pestivirus. Bovine viral diarrhoea virus (BVDV), a member of the pestivirus genus is a causative agent of mucosal disease in cattle. The virus possesses a positive sense RNA genome of approximately 12.6 kb. All members of Flaviviridae share similarities in virion structure, genome organization and replication machinery. Because BVDV shares many important properties with HCV, BVDV provides a surrogate model of HCV [3,4], in particular, for evaluation of antiviral compounds [5]. Both HCV and BVDV utilize an internal ribosomal entry site within the 5′-non-translated region (NTR) for translating the viral polyprotein. Because of some similarities in their non-structural proteins, antiviral agents active against BVDV are likely to inhibit HCV replication.
The subgenomic HCV RNA replicon cell system has been recognized as a useful tool for discovery of novel anti-HCV agents [6,7]. However, this system does not produce infectious progeny; therefore, it cannot be used for the identification and characterization of antiviral agents acting on early and late steps in the viral replication cycle, such as viral attachment, entry, uncoating, maturation or release. In addition, antiviral agents that reduce the infectivity of progeny cannot be identified in this system [3]. By contrast, a cell culture system of productive HCV infection has recently been established with the replication-competent strain, JFH-1, which makes it possible to identify inhibitors of every step in the viral replication cycle [8–10]. However, this strain was isolated from a patient of fulminant hepatitis C and classified as genotype 2a with several mutations; thus, BVDV is still widely used as a surrogate model for the investigation of anti-HCV agents.
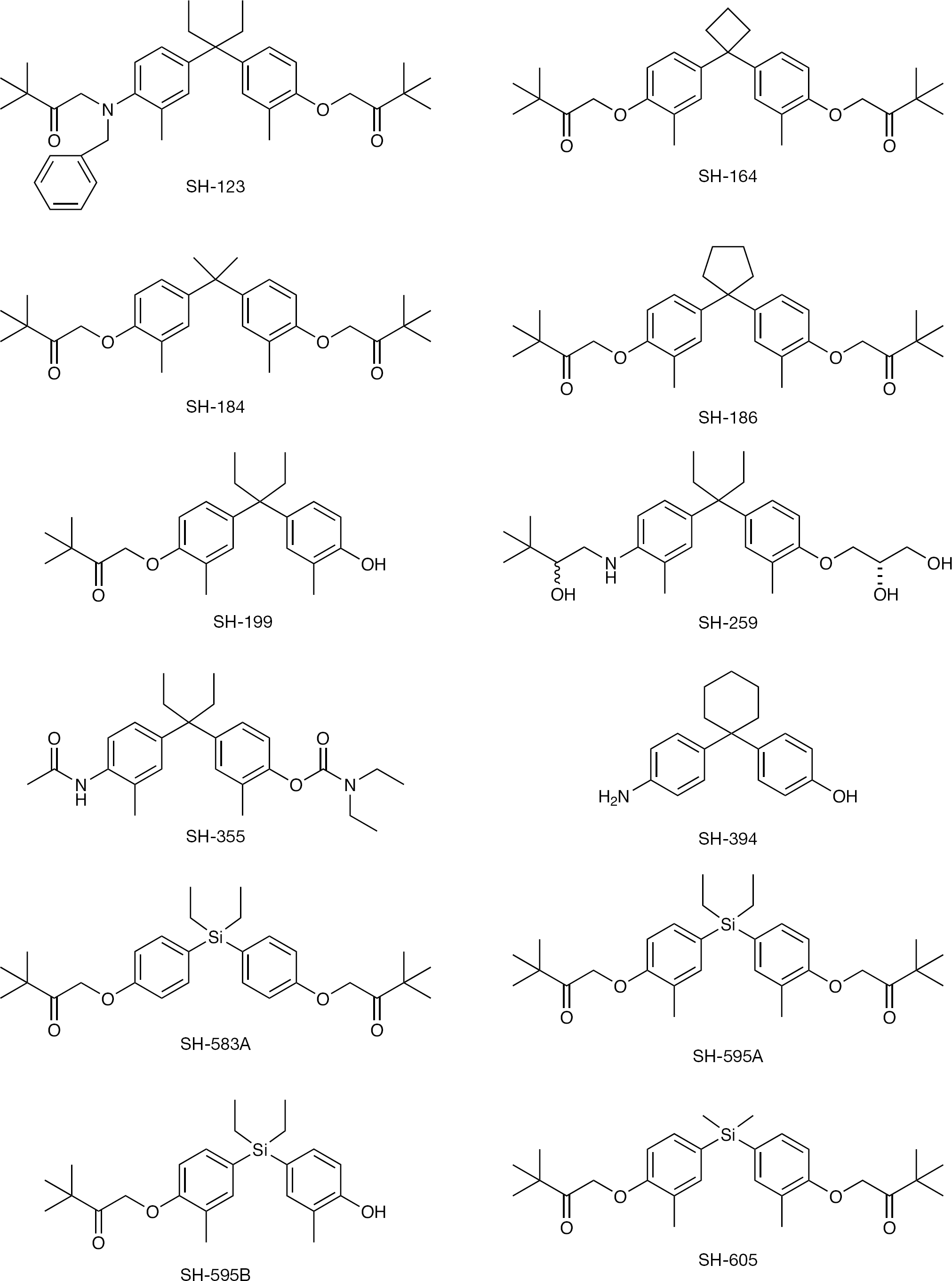
Structures of diphenylmethane derivatives
A simple and sensitive colorimetric assay of compounds, which evaluated their anti-BVDV activity, has previously been reported by our group [11]. In this study, we examined novel diphenylmethane derivatives (Figure 1) for their inhibitory effect on BVDV replication and found that some compounds were selective inhibitors of the virus. To gain insight into their mechanism of action, assays for viral entry and RNA inhibition were also conducted.
Methods
Compounds
Twelve diphenylmethane derivatives (Figure 1) and the reference compound, γ-carboline, were selected for antiviral assay. The synthesis of these compounds has been described elsewhere [12,13]. All compounds were dissolved in dimethyl sulfoxide at a concentration of 20 mM and stored at −20°C until use.
Cells and virus
Madin–Darby bovine kidney (MDBK) cells were purchased from Japan Health Sciences Foundation (Health Science Research Resources Bank, Osaka, Japan). The cells were grown and maintained in Dulbecco's modified Eagle's medium with high glucose (4.9 mg/ml; Gibco/BRL, Grand Island, NY, USA). The medium was supplemented with 10% heat-inactivated horse serum (Gibco/BRL), 100 units/ml penicillin G and 100 μg/ml streptomycin. The cells were certified as BVDV-contamination-negative. For antiviral assays, medium supplemented with 3% heat-inactivated horse serum and antibiotics was used. The cytopathic BVDV strain Nose was obtained from Kyoto Biken (Kyoto, Japan). BVDV was harvested from culture supernatants of virus-infected cells after incubation for 3 days. Virus stocks were stored at −80°C until use. The infectivity of the stocks was determined in MDBK cells by an end point serial dilution method and expressed as the 50% tissue culture infectious dose per ml (TCID50/ml).
Anti-BVDV assays
Determination of compounds for their anti-BVDV activity was based on the inhibition of virus-induced cytopathic effect (CPE) in MDBK cells, as described previously [11]. Briefly, the cells (1×105 cells/ml) were infected with BVDV at a multiplicity of infection (MOI) of 0.01 and 100 μl of the cell suspension was brought into each well in a microtitre plate. The cells were incubated in the presence of various concentrations of test compounds, including the reference compound, γ-carboline, for 3 days at 37°C in a humidified CO2 incubator. After incubation, culture supernatants were collected to determine their lactate dehydrogenase (LDH) levels by an LDH detection kit (Takara Biochemicals, Otsu, Japan) according the manufacturer's instructions. The cytotoxicity of the compounds was evaluated in parallel with their antiviral activity. The mock-infected MDBK cells (1×104 cells/well) were incubated in the presence of various concentrations of test compounds for 3 days. The viability was determined by a dye method using water-soluble tetrazolium Tetracolor One® (Seikagaku Corporation, Tokyo, Japan).
Virus yield reduction assays were conducted for the representative compound, SH-595A, and for γ-carboline. Briefly, MDBK cells (1×105 cells/ml) were infected with BVDV at a MOI of 0.01 and 500 μl of the infected cell suspension was brought into each well of a 24-well plate in the presence of various concentrations of SH-595A or γ-carboline. The cells were washed with culture medium at 24 h after virus infection and replaced with fresh culture medium containing appropriate concentrations of the test compounds. The plate was incubated for 2 days at 37°C. The culture supernatant of each well was collected and stored at −80°C until virus titration. The virus titre for each sample was determined and expressed as TCID50/ml.
ELISAs
To confirm the anti-BVDV activity of SH-595A, its inhibitory effect on viral antigen production was determined by a BVDV antigen ELISA kit (Bio-X Diagnostics, Jemelle, Belgium). MDBK cells (1×104 cells/well) were infected with the virus at a MOI of 0.01 and 100 μl of the cell suspension was brought into each well in a microtitre plate. The cells were incubated in the presence of various concentrations of the compound. After incubation for 3 days at 37°C, the supernatants were collected and their BVDV antigen levels were determined by the ELISA kit according to the manufacturer's instructions.
Real-time reverse transcription-PCR
The anti-BVDV activity of SH-595A and γ-carboline was also determined by the inhibition of viral RNA synthesis in MDBK cells by real-time reverse transcription (RT)-PCR. The cells (2×104 cells/well) were infected with the virus at a MOI of 1.0 and cultured in the presence of various concentrations of the test compounds. After incubation for 12 h, the cells were washed with phosphate-buffered saline (PBS), treated with lysis buffer of TaqMan® Gene Expression Cell-to-CT™ kit (Applied Biosystems, Branchburg, NJ, USA) and subjected to real-time RT-PCR. The BVDV RNA level was determined using the sense primer 5′-TGGTCCGACGCCTTAGTATAAAGG-3′, the antisense primer 5′-GGCTGTATTCGTAACAGTTGGTTAAA-3′ and the fluorescence probe 5′-ACGAGGGCACGCCCAAAGCA-3′ (Applied Biosystems). The primer pair amplifies the 5′-NTR of BVDV RNA. RT and PCR reagents of the kit were used for real-time RT-PCR according to the manufacturer's instructions. Non-specific inhibition of host cellular messenger RNA (mRNA) synthesis by SH-595A and γ-carboline was determined by amplification of a part of the bovine β-actin mRNA using the sense primer 5′-GCCCTGAGGCTCTCTTCCA-3′, the antisense primer 5′-GCGGATGTCGACGTCACA-3′ and the fluorescence probe 5′-CATGGAATCCTGCGGCATTCACG-3′ (Applied Biosystems).
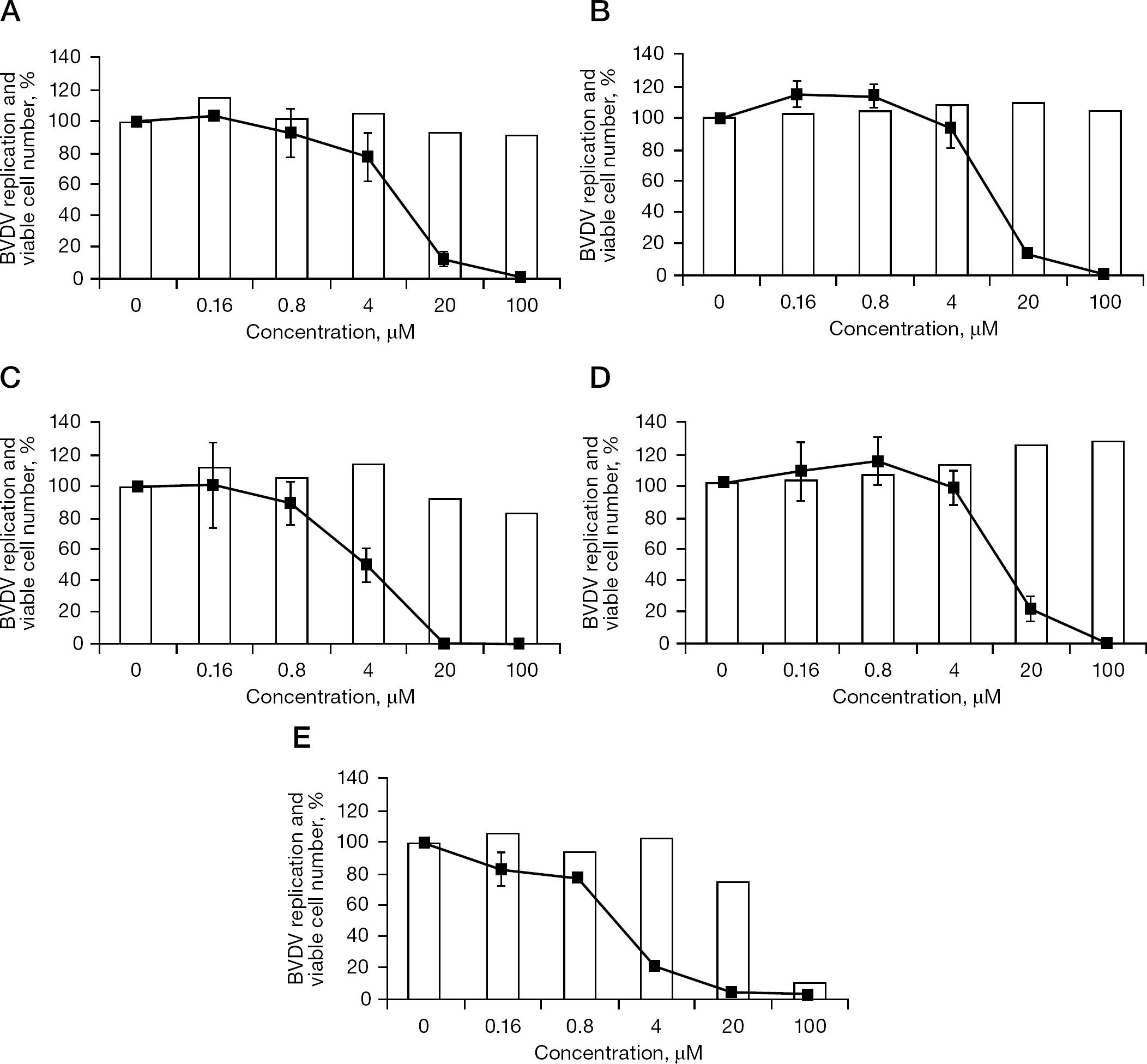
Inhibitory effect of diphenylmethane derivatives and γ-carboline on BVDV replication in MDBK cells
Viral entry inhibition assays
MDBK cells (2×104 cells/well) were seeded in a microtitre plate and incubated at 37°C for 24 h. The cells were infected with BVDV at a MOI of 2.0. SH-595A (100 μM) and γ-carboline (20 μM) were added at the time of infection and incubated without their removal, added at the time of infection but removed after incubation for 2 h, or added only after incubation for 2 h. The cells were incubated for 12 h after viral infection, washed with PBS and subjected to real-time RT-PCR, as described above.
Results
When 12 diphenylmethane derivatives (Figure 1) were examined for their inhibitory effect on BVDV replication in MDBK cells, 4 compounds displayed dose-dependent inhibition of virus-induced CPE (Figure 2). The 50% effective concentrations (EC50) of SH-164, SH-184, SH-583A and SH-595A were 9.0, 10.8, 6.3 and 10.5 μM, respectively (Table 1). None of the compounds reduced the viability of the mock-infected MDBK cells at a concentration of up to 100 μM. Compounds SH-123, SH-186, SH-199, SH-259, SH-355, SH-394, SH-595B and SH-605 also showed some inhibition of BVDV replication; however, their selectivity indices based on the ratio of 50% cytotoxic concentration (CC50) to EC50 were marginal. The EC50 and CC50 values of the reference compound γ-carboline were 1.9 and 42.6 μM, respectively (Table 1). In the next experiment, SH-595A and γ-carboline were examined for whether they could reduce the amount of infectious virus particles in culture supernatants of the cells. Dose-dependent reduction of the virus titre was observed for both compounds (Figure 3). These results indicate that diphenylmethane derivatives selectively inhibit BVDV replication in cell cultures. The antiviral activity of SH-595A against BVDV was also examined by a sandwich ELISA. It reduced the amount of viral antigen in culture supernatants of the infected cells in a dose-dependent manner (Figure 4). Its EC50 was 9.2 μM, which is comparable to the EC50 obtained by the CPE inhibition assays (Table 1).
Anti-BVDV activity of diphenylmethane derivatives in MDBK cells
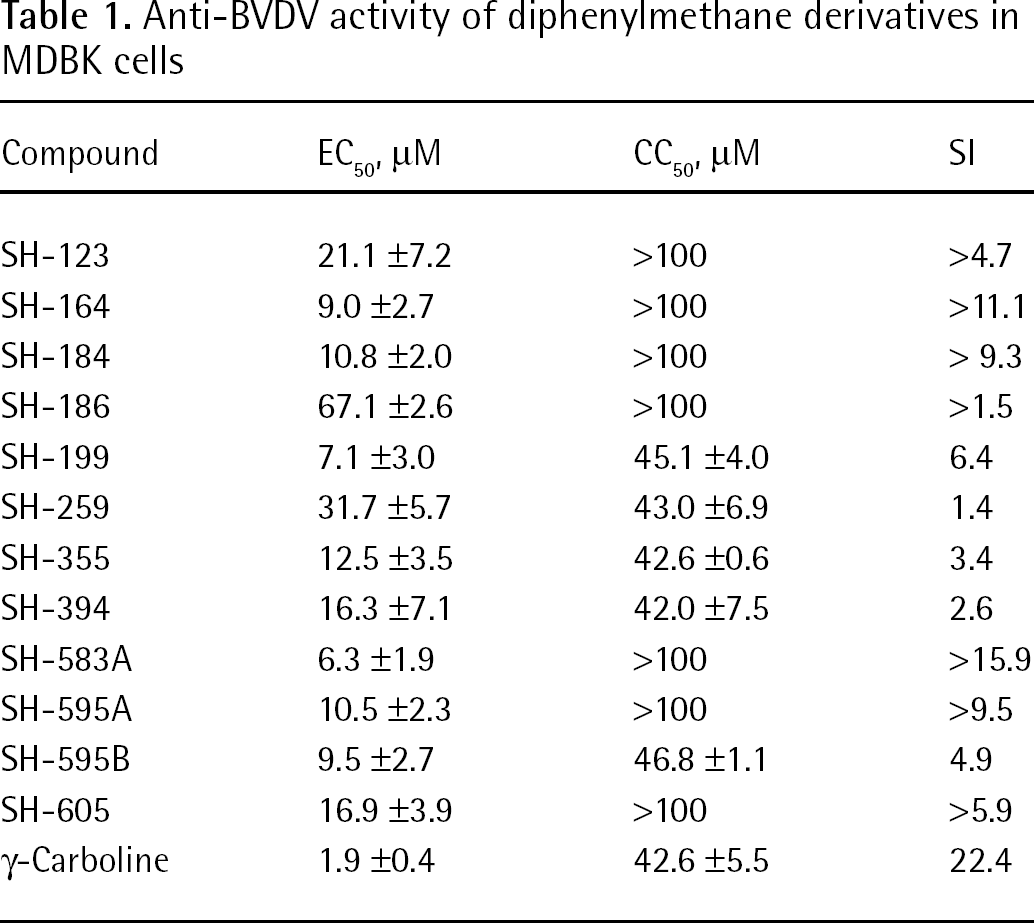
All data represent means ±SD for at least three separate experiments. CC50, 50% cytotoxic concentration based on the reduction of viable cell number; EC50, 50% effective concentration based on the reduction of cell destruction induced by bovine viral diarrhoea virus (BVDV) replication; MDBK, Madin–Darby bovine kidney; SI, selectivity index (a ratio of CC50 to EC50).
When the inhibitory effect of SH-595A and γ-carboline on BVDV RNA synthesis was examined, SH-595A showed modest inhibition of viral RNA synthesis. Although it achieved approximately 70% inhibition of viral RNA synthesis at a concentration of 100 μM, little inhibition was observed at a concentration of 20 μM (Figure 5A). By contrast, γ-carboline almost completely inhibited viral RNA synthesis at 4 μM without affecting β-actin mRNA synthesis (Figure 5B). Considering the finding that both SH-595A and γ-carboline achieved approximately 80% inhibition of BVDV replication at a concentration of 20 and 4 μM, respectively (Figure 2), the mechanism of BVDV inhibition by SH-595A seems to differ from that by γ-carboline. To gain further insight into the mechanism of BVDV inhibition by SH-595A, the effect of delayed compound addition on the viral RNA synthesis was investigated. The concentrations 100 and 20 μM were used for SH-595A and γ-carboline, respectively. When the compounds were added to the cell cultures simultaneously with the virus and were not removed during the whole culture period (condition 1), 87% and almost complete inhibition of viral RNA synthesis was observed for SH-595A and γ-carboline, respectively (Figure 6). When the compounds existed in the cell cultures during only the first 2 h after virus infection (condition 2), SH-595A and γ-carboline generated 47% and 19% inhibition, respectively. When the compounds were added after the virus adsorption period for 2 h (condition 3), SH-595A and γ-carboline achieved 64% and almost complete inhibition of viral RNA synthesis, respectively. Again, these results indicate that the mechanism of BVDV inhibition by SH-595A seems to differ from that by γ-carboline and that it might interfere with the viral adsorption step to some extent in addition to a step after virus adsorption.
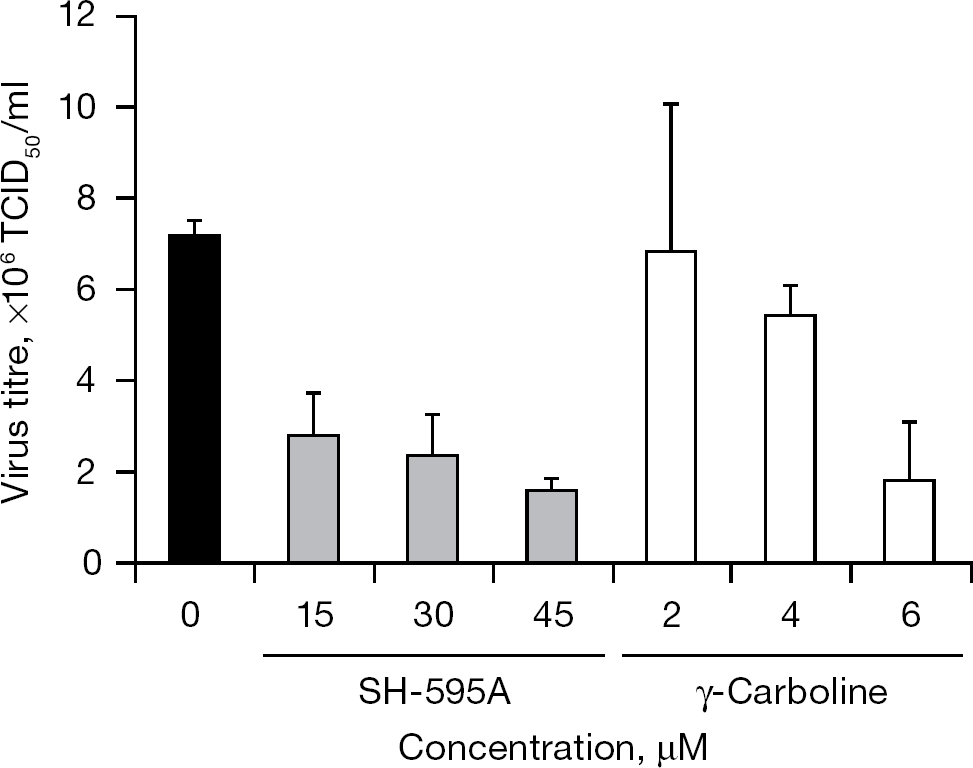
Inhibitory effect of SH-595A and γ-carboline on viral infectivity in culture supernatants
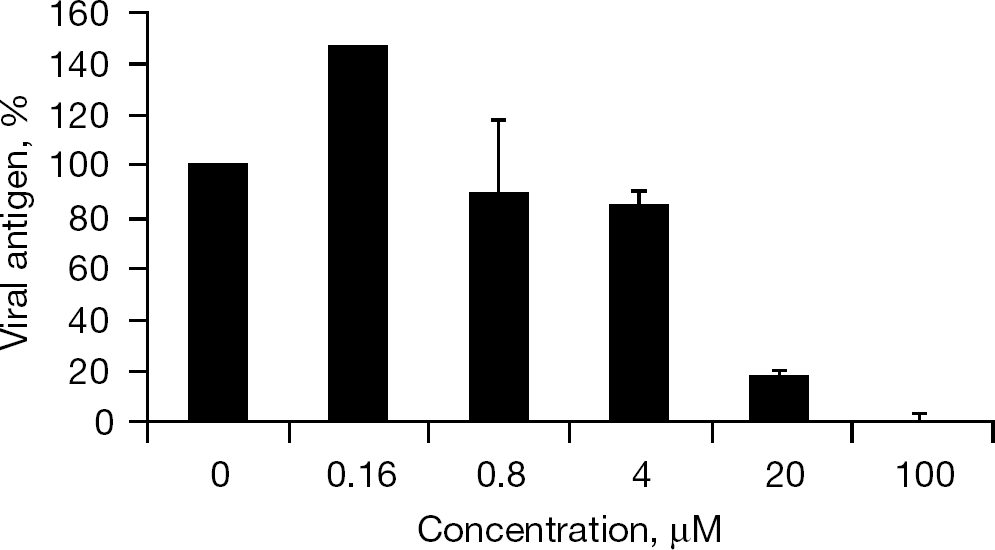
Inhibitory effect of SH-595A on viral antigen production in culture supernatants
Discussion
BVDV is considered to be a valuable surrogate for identifying and characterizing anti-HCV agents [14]. Our strategy is to discover novel anti-BVDV compounds that can be developed as anti-HCV agents. Recently, we have reported the synthesis of novel diphenylmethane derivatives having anti-BVDV activity in cell cultures [12]. In this study, we attempted to further investigate their antiviral activity and mechanism of action.
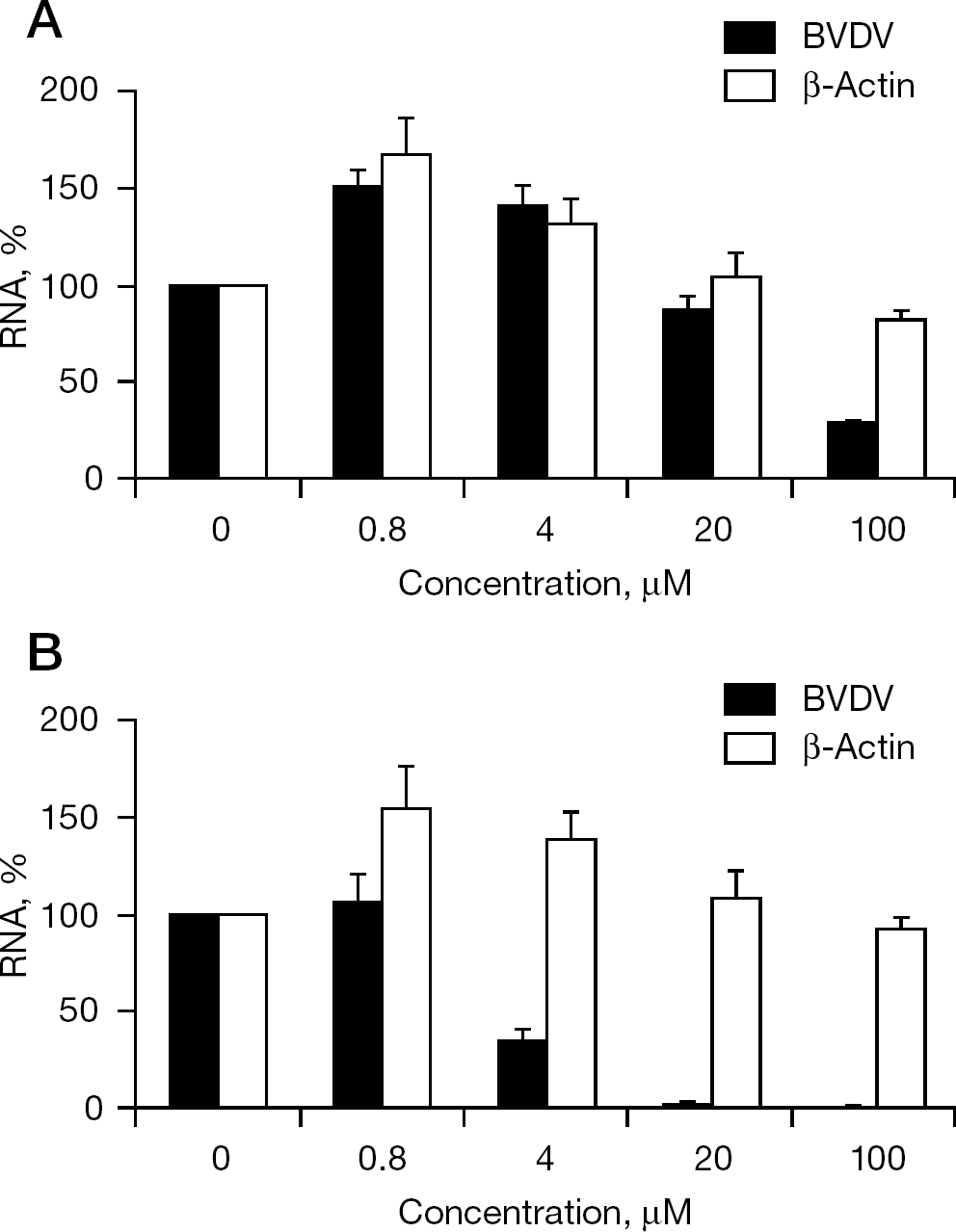
Inhibitory effect of SH-595A and γ-carboline on viral RNA synthesis in MDBK cells
Among the active compounds, SH-595A has been considered as a representative compound and was compared with γ-carboline [13]. SH-595A contains a silicon atom in its chemical structure; therefore, the compound is indeed novel and unique (Figure 1). According to the results obtained by real-time RT-PCR, SH-595A was found to be a modest inhibitor of BVDV RNA synthesis. It only marginally inhibited viral RNA synthesis at a concentration of 20 μM (Figure 5A), at which the compound achieved approximately 80% inhibition of viral replication as determined by the CPE inhibition assay (Figure 2D) and the viral antigen ELISA for culture supernatants (Figure 4). By contrast, γ-carboline was a potent inhibitor of viral RNA synthesis and its inhibitory concentration for viral RNA synthesis corresponded well with that for viral replication (Figures 2E and 5B). It is assumed that γ-carboline exerts its anti-BVDV activity through the inhibition of viral RNA polymerase activity (MTAS et al., unpublished data). These results suggest that SH-595A interacts, in part, with a step after viral RNA synthesis.
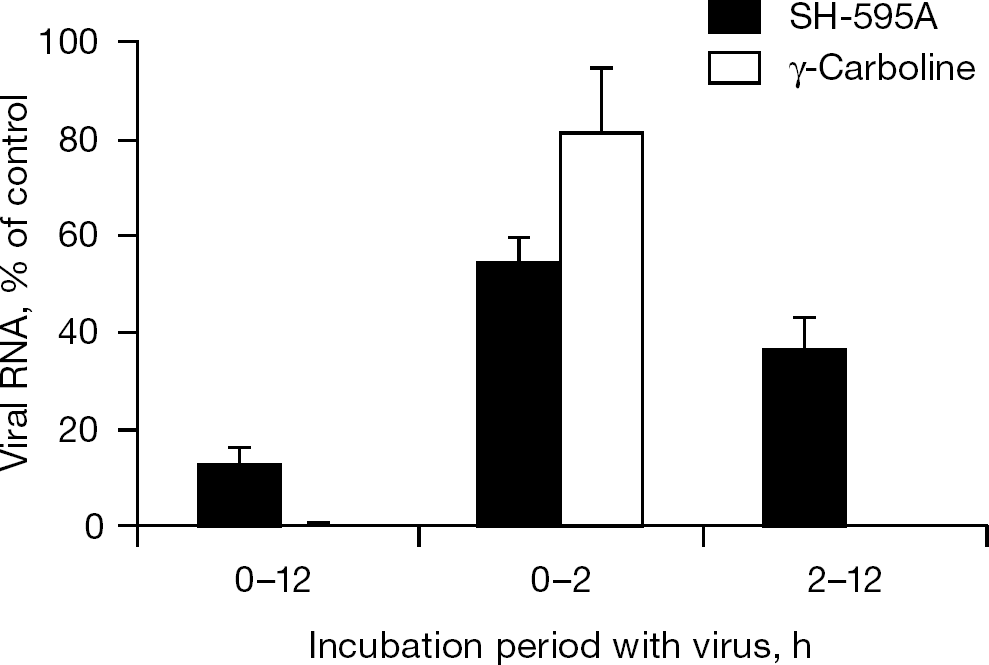
Inhibitory effect of SH-595A and γ-carboline on viral entry to MDBK cells
Another possible explanation for the discrepancy between viral RNA inhibition by SH-595A and its antiviral activity is the difference of the assay conditions used for the experiments. The CPE inhibition assay reflects the inhibitory effect of test compounds on multi-round viral replication because of the low MOI (0.01) used for the experiment, whereas the viral RNA inhibition assay reflects the inhibition of single-round viral replication by test compounds because of the high MOI (1.0). Indeed, no viral antigen could be detected in the culture supernatants of the infected cells with BVDV at a low MOI (0.01) and exposed to SH-595A (100 μM) after a virus adsorption period for 2 h (MTAS et al., data not shown). If SH-595A could interfere with an early step of the BVDV replication cycle, such as virus adsorption, the activity of the compound should be enhanced in a multi-round viral replication assay. To prove this hypothesis, a viral entry inhibition assay was examined for SH-595A and γ-carboline under a high MOI condition. It has been reported that a single cycle of BVDV replication takes 13 h on average and that gradual increase of intracellular viral RNA is noted at 6–8 h after virus infection [15]; therefore, like γ-carboline, viral RNA polymerase inhibitors are expected to suppress viral RNA synthesis, even when added after a virus adsorption period. However, SH-595A achieved only 65% inhibition, even at a concentration of 100 μM (Figure 6). In addition, if SH-595A could only inhibit the viral adsorption process, the viral RNA level in condition 2 (0–2 h) would be identical to that in condition 1 (0–12 h). By contrast, similar to γ-carboline, if SH-595A could not inhibit the viral adsorption process but could inhibit a process after viral adsorption, the viral RNA level in condition 1 (0–12 h) would be similar to that in condition 3 (2–12 h). These results suggest that SH-595A might have more than one target for inhibition of BVDV. The isolation and characterization of resistant mutants are also important for learning more about the mechanism of action; however, such studies are difficult to conduct because of the low potency of the current compounds. Further experiments are required to identify diphenylmethane derivatives that have higher antiviral activity and to determine their target molecule(s).
Finally, some diphenylmethane derivatives, including SH-595A, were examined for their inhibitory effect on HCV replication in HCV RNA replicon cells and were found to be inactive (MTAS et al., data not shown). Because the replicon cells do not involve early and late steps of the HCV replication cycle [6,7], the diphenylmethane derivatives might be worth testing for their anti-HCV activity in the cell-free viral replication system [8–10].
Footnotes
Acknowledgements
This work was supported in part by the Science and Technology Incubation Program in Advanced Regions, Japan Science and Technology Agency, Japan.
All authors are inventors of the patent currently submitted to Japan Patent Office.
