Abstract
Background:
Aurintricarboxylic acid (ATA) and ethacrynic acid (ECA) have been reported to exhibit antiviral activity against vaccinia virus infections in cell culture by inhibiting early and late gene transcription, respectively. The purpose of this work was to determine if these inhibitors would effectively treat vaccinia virus infections in mice, which has not previously been studied.
Methods:
ECA was investigated by cell culture plaque reduction assay for the inhibition of cowpox and vaccinia virus infections to clarify issues regarding its potency and selectivity. Mice infected intranasally with vaccinia virus were treated by intraperitoneal route twice daily for 5 days with ATA (10 and 30 mg/kg/day) and ECA (15 and 30 mg/kg/day) or once daily for 2 days with cidofovir (100 mg/kg/day).
Results:
ECA caused 50% inhibition of virus plaque formation at 20–79 μM in four cultured cell lines, with 50% cytotoxicity at 84–173 μM, giving low (1.3–4.2) selectivity index values. Preliminary toxicity tests in uninfected mice indicated that ATA and ECA were both overtly toxic at 100 mg/kg/day. No protection from mortality was afforded by treatment of vaccinia virus infections with ATA or ECA, but 100% survival was achieved in the cidofovir group. ATA- and ECA-treated mice died significantly sooner than placebo-treated animals, indicating that these compounds exacerbated the infection.
Conclusions:
Both ATA and ECA lack antiviral potency and selectivity in cell culture. The compounds were ineffective in treating mice at intraperitoneal doses of ≤30 mg/kg/day. These compounds do not appear to have potential for the treatment of poxvirus infections in vivo.
Introduction
Aurintricarboxylic acid (ATA) and ethacrynic acid (ECA) have been reported to inhibit vaccinia virus replication in cell culture [1,2]. ATA inhibits an early transcriptional event of vaccinia virus replication by targeting cellular and viral factors [1]. The compound blocks the phosphorylation of extracellular signal-regulated kinase 1/2 (a cellular enzyme) and the phosphatase activity of the viral AH1L enzyme. ATA caused a 90% reduction in virus titre at concentrations of 25–200 μg/ml (60–475 μM), depending upon the cell line used, and toxicity was not evident at 500 μg/ml (1,185 μM) [1]. ECA was shown to inhibit late viral gene expression, with no effect on other virus targets, such as virus entry into the cell, early gene expression or viral DNA synthesis [2]. ECA caused a 90% reduction in vaccinia virus yield at concentrations of 19–61 μM, depending upon cell type. Cytotoxicity was evident at 35–290 μM. Selectivity index (SI) values of 2.5–32 were based upon 50% reductions in virus titre (at 5.4–19.3 μM) rather than on 90% effective concentration (EC90) values [2]. Both compounds have modes of action that differ from nucleotide analogues such as cidofovir that inhibits viral DNA synthesis [3] or the non-nucleoside ST-246 that prevents virus assembly [4].
The authors of the reports of ATA and ECA inhibition indicate that these molecules might be drug candidates or provide new directions in pursuing antipoxvirus compounds that could be useful as drugs [1,2]. Thus, the compounds appear to merit further study, particularly as no data of their activities in animal models have been reported. Because of the need for antipoxvirus agents, owing to bioterrorism concerns surrounding the deliberate release of smallpox or monkeypox viruses into human populations [5,6], studies to identify new agents that have potential for treating humans are warranted. These studies include the determination of compound efficacy in animals [7,8].
From a review of the published literature [1,2], it was our impression that ATA would be too weak in potency in vitro to show efficacy in vivo. Although ECA is more potent than ATA, we were concerned that ECA might be poorly selective (that is, the 50% antiviral and cytotoxic activities might not be far enough apart). Thus, we performed some cell culture analyses with ECA to confirm our hypothesis. We felt that the antiviral activity of ATA was sufficiently documented not to pursue further in vitro studies with that material. Next, we performed animal experiments using intranasally infected mice treated with ATA and ECA and made comparisons with cidofovir treatment. The lack of utility of ATA and ECA in treating vaccinia virus infections in mice was clearly demonstrated.
Methods
Antiviral compounds
ATA and ECA were purchased from Sigma (St. Louis, MO, USA). Cidofovir was kindly provided by M Hitchcock of Gilead Sciences (Foster City, CA, USA). The acids were dissolved in 2% sodium bicarbonate solutions. ECA was further diluted into cell culture medium for studies conducted in vitro. Prior to discovering a suitable solvent for the compounds (ECA being the first one examined), animal experiments were performed with ECA suspended in saline for treatment of mice. ATA was dissolved in 2% sodium bicarbonate buffer for mouse experiments. Cidofovir, a positive control, was dissolved in saline. The placebo for animal studies was saline for the evaluation of ECA or 2% sodium bicarbonate for the study of ATA.
Viruses and cells
Vaccinia virus IHD and WR strains were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cowpox virus (Brighton strain) was obtained from the US Army Medical Research Institute of Infectious Diseases (Fort Detrick, Frederick, MD, USA). The origin of the cowpox virus was the Centers for Disease Control and Prevention (Atlanta, GA, USA). The viruses were initially propagated in African green monkey kidney (Vero) cells (ATCC). Higher virus titres for later use were obtained by propagating the viruses in a second line of African green monkey kidney (MA-104) cells, purchased from BioWhittaker (Walkersville, MD, USA). Other cells used for in vitro assays included human lung carcinoma (A549) and rhesus monkey kidney (LLC-MK2) cells. The three monkey cell lines were propagated in minimal essential medium (MEM) containing 0.22% sodium bicarbonate and 5% (Vero cells) or 10% (LLC-MK2 and MA-104 cells) foetal bovine serum (FBS). A549 cells required Ham's F12K medium with 0.22% sodium bicarbonate and 10% FBS. The medium for all antiviral assays was MEM with bicarbonate, 2% FBS, and 50 μg/ml of gentamicin.
Plaque reduction assays
Sensitivities of cowpox and vaccinia viruses to ECA were determined in 12-well microplates of cells [9,10]. The cells were infected with about 75–100 plaque forming units (PFU) of virus per well, the virus was adsorbed for 1 h, then twofold dilutions of antiviral compounds were applied for 3 days. Cells were fixed and stained in 5% buffered formalin containing 0.2% crystal violet for 15 min. The aspirated plates were rinsed with water and plaques were counted. Concentrations of compounds reducing plaque numbers by 50% (50% effective concentration [EC50] values) were determined by plotting the percentage of plaques relative to untreated cultures versus inhibitor concentration on semi-log10 paper.
Cytotoxicity assays
ECA was overtly toxic to uninfected cells at particular concentrations. Percent cytotoxicity was quantified by staining cell monolayers in 12-well plates with crystal violet after 3 days of ECA treatment. After 15 min, the cells were rinsed of excess dye and the plates were allowed to dry. The dye was eluted from the cells with ethanol and quantified spectrophotometrically at 590 nm [11]. Absorbance values were converted to percentages of the untreated control. The 50% cytotoxic concentration (CC50 value) was determined as described above.
Mouse experiments
Specific pathogen-free BALB/c mice weighing 14–15 g were obtained from Charles River Labs (Wilmington, MA, USA). Mice were infected intranasally with 50 μl of vaccinia (IHD strain) virus (approximately 1×105 PFU/mouse) following anaesthesia with ketamine (100 mg/kg, by intraperitoneal [ip] injection). Treatments with compounds were given by ip injection starting 24 h after virus exposure. The treatment schedules and doses of antiviral compounds that were used for the experiments were based upon our previous work [12]. The animals were kept for 21 days to record deaths and to monitor body weight.
Statistical analyses
Survival curves of data from mouse studies were assessed for all groups by the Mantel–Cox log rank test. Because statistical significance among groups was seen (P<0.001), pairwise comparisons were made using the Gehan–Breslow–Wilcoxon test. Evaluations were made using Prism software (GraphPad Software, San Diego, CA, USA).
Results
Cell culture experiments
The variability in antiviral potency and SI values reported previously for ECA [2] prompted us to carry out our own investigation into the antiviral activity of the compound, using one human and three monkey cell lines. Table 1 shows the results of plaque reduction experiments. Cowpox virus was inhibited at 40–79 μM and vaccinia virus was inhibited at 20–46 μM. Toxicity was observed in the cells at 84–173 μM, yielding SI values of 1.3–4.2. Thus, ECA was a moderately potent and poorly selective inhibitor of orthopoxviruses.
Antiviral activity and cytotoxicity of ECA in four cell lines
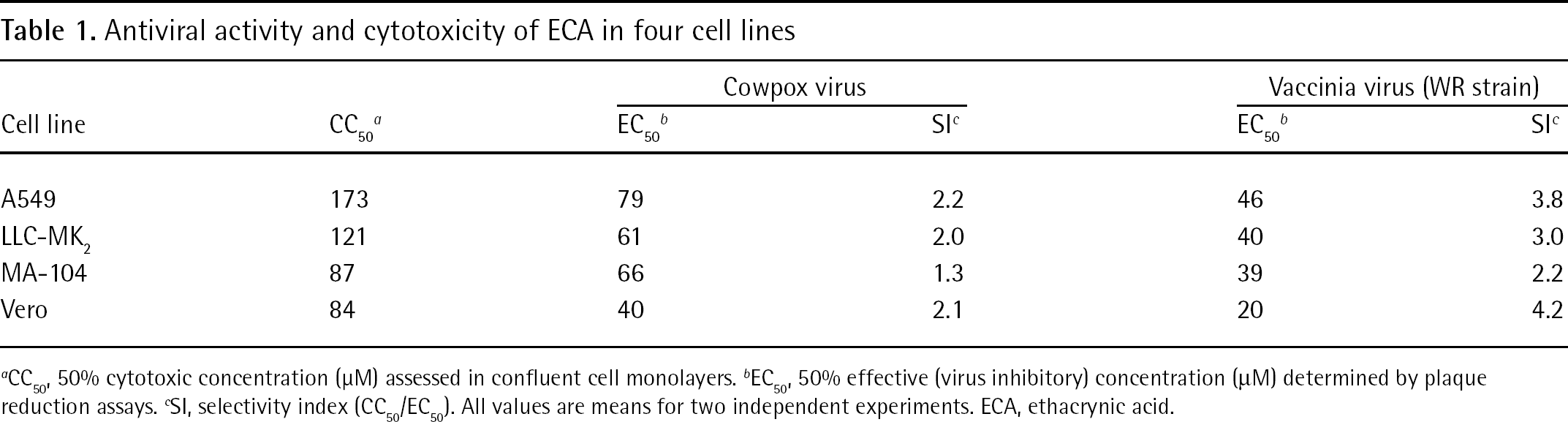
CC50, 50% cytotoxic concentration (μM) assessed in confluent cell monolayers.
EC50, 50% effective (virus inhibitory) concentration (μM) determined by plaque reduction assays.
SI, selectivity index (CC50/EC50). All values are means for two independent experiments. ECA, ethacrynic acid.
Mouse studies
Prior to conducting antiviral experiments in mice, it was important to determine the maximum tolerated doses of ATA and ECA in uninfected animals (Table 2). The compounds were evaluated by ip treatment route at doses of 3–100 mg/kg/day. Both ATA and ECA were overtly toxic at 100 mg/kg/day, with rapid death occurring in the ECA group. Mice treated with ATA for 5 days did not die until after the last treatment was given. Weight loss without death was observed in the 30 mg/kg/day ECA group. All other groups of ATA- and ECA-treated mice showed increases in body weight. Based on the results of these experiments, it was decided to test each compound for antiviral activity at a maximum dose of 30 mg/kg/day.
Toxicities of ATA and ECA in uninfected mice
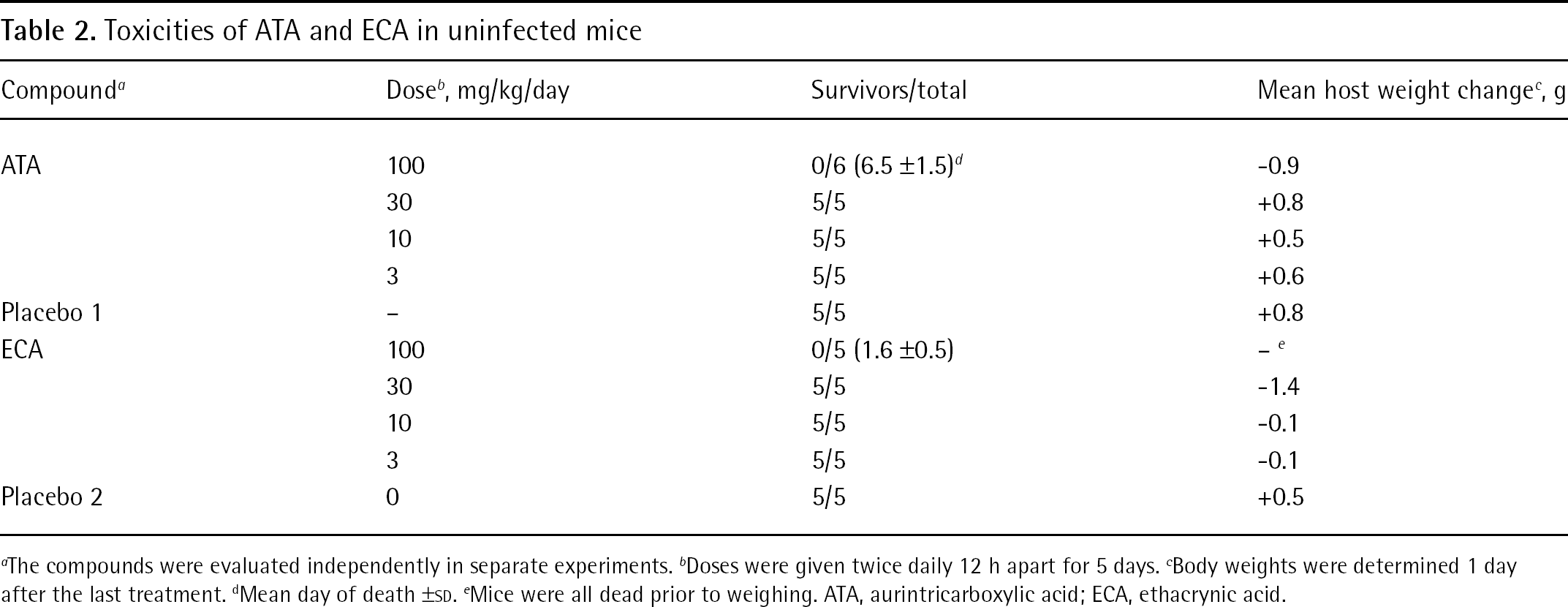
The compounds were evaluated independently in separate experiments.
Doses were given twice daily 12 h apart for 5 days.
Body weights were determined 1 day after the last treatment.
Mean day of death ±SD.
Mice were all dead prior to weighing. ATA, aurintricarboxylic acid; ECA, ethacrynic acid.
Antiviral studies were conducted in mice infected with vaccinia virus (IHD strain). This infection evolves slower than infections with the WR strain of vaccinia, and antiviral treatment is more effective [12]. Thus, any positive benefit of treatment with ATA or ECA would be evident. Mice infected intranasally were treated with the acids or with cidofovir starting 1 day after infection (Figure 1). Treatment with ATA did not prevent death. The 30 mg/kg/day dose accelerated the time to death compared with placebo. Cidofovir treatment for 2 days was 100% protective. Treatment with ECA also failed to prevent death and treatment with both doses shortened the time to death. Cidofovir was completely protective.
Discussion
In these experiments, two compounds purported to have potential as drug candidates or lead compounds for new directions in the development of drugs effective against poxvirus infections were evaluated for antiviral activity. ECA was found to be poorly selective in cell culture infected with cowpox and vaccinia viruses. The results confirmed those reported previously [2], if SI values are calculated based on 90% (10-fold) reduction in virus titre rather than the published method (50% or twofold reduction in virus titre). We have found that EC90 values for virus titre reduction correlate well with the EC50 value for plaque reduction (DFS and BLH, unpublished), which was the assay that we performed. A recalculation of the previous data [2] based upon EC90/IC50 (or CC50) gave SI values of <1–9.9, compared with 1.3 and 4.2 reported here. Thus, our results are consistent with the former results for the moderate potency and low selectivity of ECA against vaccinia virus in cell culture.
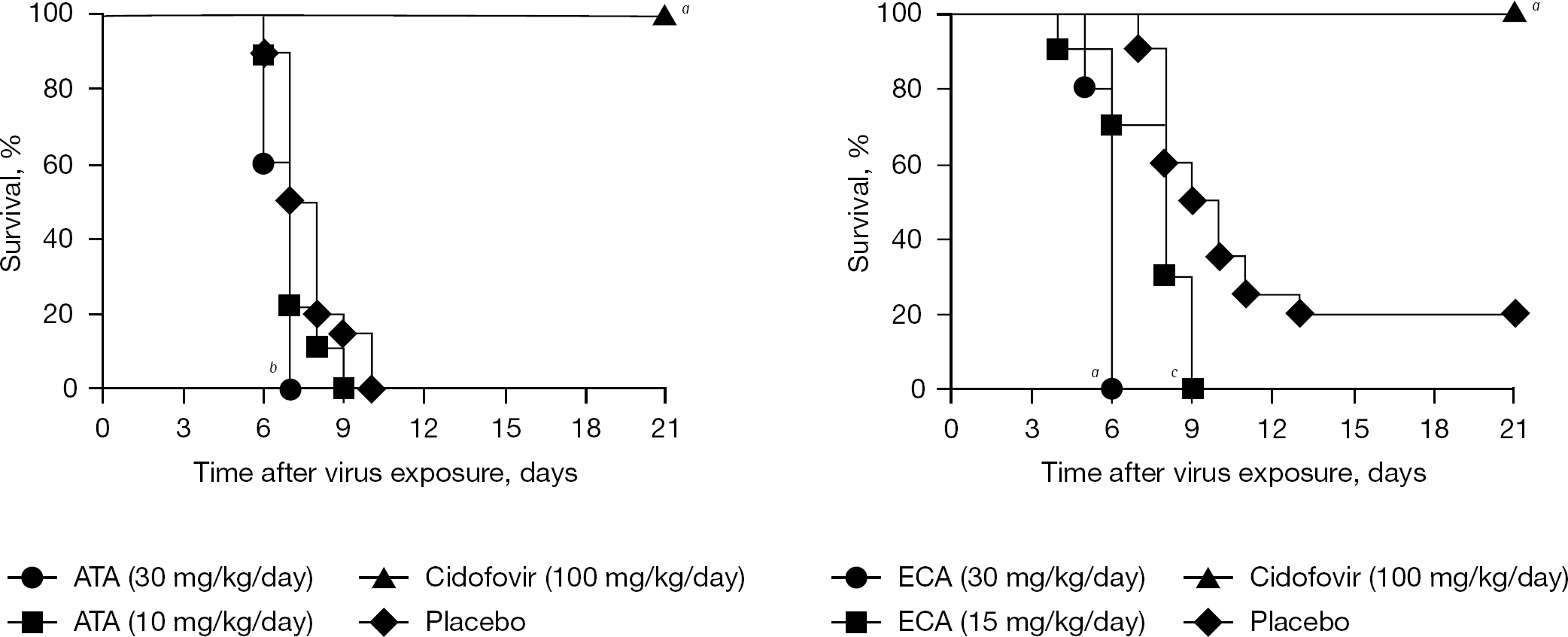
Effects of ATA, ECA and cidofovir on survival of a vaccinia virus (IHD strain) respiratory infection in mice
The low potency of ATA (60–475 μM) and the low selectivity of ECA (<1–9.9) in cell culture were initial indicators that, despite their uniquely identified modes of action, they might not exhibit antiviral activity in vivo. Indeed, the compounds exhibited no protective activity in mice infected intranasally with vaccinia (IHD strain) virus. In fact, mice treated with the compound fared worse (that is, died sooner) than animals treated with placebo. ECA showed some toxicity in uninfected mice at the 30 mg/kg/day dose by inducing weight loss, and this may have contributed to early death. Cidofovir was highly effective in this model, as was previously reported [10].
To date, ECA has only been reported to be inhibitory to poxvirus replication, specifically against vaccinia virus [2], but not other related or unrelated viruses. Here, we report that the related cowpox virus is also inhibited, which is not surprising. ATA has been reported to inhibit a number of different viruses, including dengue and Wesselbron flaviviruses [13], HCV [14], HIV [15], influenza virus [16], severe acute respiratory syndrome coronavirus [17], vaccinia virus [1] and vesicular stomatitis virus [18]. The possibility exists that ATA could inhibit some of these viruses in vivo wherein the antiviral potency is considerably better (that is, lower 50% virus inhibitory concentrations). The studies presented here establish the maximum tolerated dose of the compounds in mice and show that they are not effective against vaccinia virus infections.
Footnotes
Acknowledgements
This work was supported by contracts NO1-AI-15435 and NO1-AI-30063 (awarded to Southern Research Institute) from the Virology Branch, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, USA. The contents of this article do not necessarily reflect the position or policy of the government and no official endorsement should be inferred. The investigators adhered to the ‘Guide for the Care and Use of Laboratory Animals’, prepared by the Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, National Research Council and used facilities fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
The authors declare no competing interests.
