Abstract
Background:
Herpes simplex virus (HSV) establishes latent infection in humans with periodic reactivation. Acyclovir, valacyclovir and foscarnet are in medical use today against HSV type-1 (HSV-1) and type-2 (HSV-2), inhibiting the DNA synthesis of the viruses. Additional drugs that will affect the growth of these viruses by other mechanisms and also decrease the frequency of appearance of drug-resistant mutants are required.
Methods:
Cationic polysaccharides were synthesized by conjugation of various oligoamines to oxidized polysaccharides by reductive amination. Polycations of dextran, pullulan and arabinogalactan were grafted with oligoamines of 2–4 amino groups forming Schiff-base imine-based conjugates followed by reduction with borohydride to obtain the stable amine-based conjugate. Evaluation of toxicity to BS-C-1 cells and antiviral activity against HSV-1 and HSV-2 of the different compounds was performed in vitro by a semiquantitative assay. A quantitative study with a selected compound followed.
Results:
Structure–activity relationship studies showed that the nature of the grafted oligoamine of the polycation plays an essential role in the antiviral activity against HSV-1 and HSV-2. Dextran-propan-1,3-diamine (DPD) was found to be the most potent of all the compounds examined. DPD did not decrease the infectivity of HSV upon direct exposure to the virions. The growth of HSV was significantly inhibited when DPD was added to the host cells 1 h prior to infection, thus preventing the adsorption and penetration of the virus into the cells.
Conclusions:
Our in vitro data warrant clinical investigation. DPD could have an advantage as a topical application in combination therapy of HSV lesions.
Introduction
Herpes simplex virus type-1 (HSV-1) and type-2 (HSV-2) establish latent infection in humans, with periodic reactivation. Herpes simplex labialis/facialis, also known as cold sore, is mainly a skin disease. Among illnesses caused by HSV-1 [1] and HSV-2 are: genital herpes [2], gingivostomatitis [3], herpetic keratoconjunctivitis [4], herpetic whitlow, herpetic encephalitis [5] and eczema herpeticatum [6]. Although herpes simplex labialis/facialis usually has a self-limited course, the disease affects quality of life, mainly because of painful disfigurement and to the psychological effect of recurrent herpes episodes.
During the past 40 years, various compounds affecting HSV infections have been identified [7]. Among the anti-herpesvirus drugs known today are acyclovir (ACV) [8], valacyclovir [9] and foscarnet [10]. These drugs appear to act primarily by inhibition of HSV replication. ACV, a nucleoside analogue of guanosine, inhibits HSV DNA polymerase activity, following three phosphorylations of the drug [11,12]. The first in the chain of phosphorylations, which is performed by the herpesvirus-encoded thymidine-kinase, enables ACV to become activated only in virus-infected cells. Although ACV and its analogues have dominated the therapy against herpesvirus infections for several decades because of their potency, their use suffers from several drawbacks, such as low aqueous solubility, low bioavailability following oral administration and appearance of drug-resistant mutants [11]. The sole FDA-approved treatment for infection with ACV-resistant HSV at present is foscarnet, an analogue of pyrophosphate. This drug binds DNA polymerase competitively and prevents chain elongation, but has poor oral absorption and might cause severe side effects [13].
The need for additional anti-herpesvirus drugs, acting by different mechanisms of action from that used by ACV, is of very high priority in antiviral drug research today. Several research teams have already developed a group of non-nucleoside polycationic compounds having anti-herpesvirus activity, such as neomycin, poly-L-lysine, poly-L-arginine and poly-L- histidine derivatives. Unlike the nucleoside-based antiviral agents, most of these compounds appear to act primarily by inhibiting early stages of virus replication. For example, a phosphoinositide-binding aminoglycoside, such as neomycin, inhibits binding of HSV-1 to receptors on baby hamster kidney cell line (BHK) cells, but not of HSV-2 [14]. Likewise, polyamino-acid-based derivatives, such as poly-L-lysine and poly-L-arginine, prevent HSV-1 binding to cellular receptors and consequently internalization of the virus into the cells [15]. In addition, an inhibitory effect of poly-L-lysine on other viruses, like tobacco mosaic virus, which causes viral envelope disruption, has been reported by Burger and Stahmann [16]. Docherty and Pollock [17] showed that poly-L-histidine conjugates inactivate HSV-1 and HSV-2 at pH 5 and 6 by 99%. A similar effect was obtained with poly-L-arginine, which inhibited HSV-1 and HSV-2 at all pH levels tested, whereas poly-L-lysine demonstrated a weaker effect on these viruses [17].
Prompted by these findings, we examined the antiviral activity of another class of polycations, that is, cationic polysaccharides, in which oligoamines are conjugated onto natural polysaccharide chains via reductive amination. These types of polyamine conjugates have already been extensively studied as gene vectors [18]. They are water-soluble, relatively non-toxic, biodegradable and biocompatible [19,20]. Dextran-oligoamine derivatives have also been tested as anti-prion agents and dextran-spermine was found to efficiently deplete the level of the protease-resistant isoform of the prion protein to an undetectable value [21].
Methods
Chemistry
Chemicals and equipment
Dextran, of an average molecular weight of 20 kDa, and pullulan, of 40 kDa, were purchased from Sigma–Aldrich (Rehovot, Israel). Arabinogalactan, with an average molecular weight of 19 kDa, was a gift from Larex International (St Paul, UK). Potassium periodate (KIO4), sodium borohydride (NaBH4), ethane-1,2-diamine, propane-1,3-diamine, butane-1,4-diamine, hexane-1,6-diamine, diethylenetriamine, N,N-bis(2-aminoethyl)-1,2-propanediamine, spermine and di-tert butyl dicarbonate were purchased from Sigma–Aldrich. All solvents and reagents were of analytical grade and used as received. A sage-metering pump model-365 (Orion, NJ, USA) was applied for slow and reproducible addition of reactants. Average molecular weights of polycations were estimated with a gel permeation chromatography (GPC)-Spectra Physics instrument (Darmstadt, Germany) containing a pump, column (Shodex KB-803) and refractive index detector [22]. Average molecular weights were estimated according to pullulan standards (PSS, Mainz, Germany) with values ranging between 5,800 and 212,000 Da. Eluents used were 0.05 M NaNO3 for the uncharged polymers and 5% (w/v) Na2HPO4 in 3% (v/v) acetonirile (pH 4) for the cationic conjugates [22]. The degree of conjugation was estimated with an elemental microanalysis of nitrogen (%N) using a PerkinElmer 2400/II CHN analyser (PerkinElmer, Waltham, MA, USA). 1H-NMR spectra were obtained on a Varian 300-MHz spectrometer in 5 mm optical density tubes. D2O/CDCl3/DMSO-d6 containing tetramethylsilane served as solvent and shift reference. FTIR spectra were recorded on a PerkinElmer 2000 FTIR (PerkinElmer).
Synthesis of cationic polysaccharides
Oxidized polysaccharides were prepared and characterized as previously described [23]. In brief, the desired polysaccharide (10 g, 62.5 mmol of glucose units) was dissolved in 200 ml of double-deionized water (DDW). Potassium periodate was added at either 1:1, 1:2, 3:1 or 5:1 M ratio (glucose/IO4−; eventually leading to the four polyaldehyde derivatives A, H, I and J, respectively), and the mixture was stirred in the dark at room temperature for 6–8 h. The resulting polyaldehyde derivatives were purified from iodate (IO3−) and unreacted periodate (IO4−) by Dowex-1 (acetate form) anion exchange chromatography, followed by extensive dialysis against DDW (3,500 cutoff cellulose tubing) at 4°C for 3 days. Purified polyaldehyde derivatives were freeze-dried to obtain white powder with 70% average yield. FTIR (KBr)=1,724 cm−1 (C=O). The aldehyde content was determined by the hydroxylamine hydrochloride method [24].
The oligoamine conjugation was conducted as follows: oxidized polysaccharide (1 g, 1–9.4 mmol of aldehyde groups) was dissolved in 100 ml of DDW. The dialdehyde solution was slowly added for several hours to a basic solution containing 1.5 equimolar amount of oligoamine dissolved in 50 ml of borate buffer (0.1 M, pH 11). The mixture was stirred at room temperature for 24 h. NaBH4 (1 g, 4 equimolar) was added to reduce the imine bonds to amines and stirring was continued for 24 h under the same conditions. The reduction was repeated with additional portion of NaBH4 (1 g, 4 equimolar) at the same conditions for 24 h. The resulting light-yellow solution was poured into a dialysis membrane (3,500 cutoff cellulose tubing) and dialyzed against DDW at 4°C for 3 days. The dialysate was lyophilized to dryness and stored under nitrogen atmosphere. Average yield was 50% (w/w). Carbon-hydrogens (CH) that are related to the peak (ppm) are underlined in the following paragraphs.
1H-NMR (D2O): 1.5 ppm (m, 2H, Dextran-NHCH2-
FTIR (KBr): 1,468 cm−1 (-CH2- aliphatic), 1,653 cm−1 (-NH2, primary amine), 2,935 cm−1 (C-C, aliphatic) and 3,297 cm−1 (-NH, -OH groups).
The primary amine content was determined by trinitrobenzene sulfonic acid (TNBS) method [23].
Synthesis of N-BOC-dimethylamino propyl amine
Solution of N,N-dimethylamino propyl amine (5.9 g, 0.058 mol) in methanol (30 ml) was reacted with di-tert-butyl dicarbonate (12.15 g, 0.96 mol) for 1 h at 0°C and overnight at room temperature to complete the reaction. The solvent was removed under reduced pressure. The residue was dissolved in 50 ml of DDW and protected N,N-dimethylamino propyl amine, was extracted with dichloromethane (3×50 ml), dried over anhydrous MgSO4, filtered and evaporated to dryness. The crude white powder was further dried under vacuum over solid NaOH pellets. Average yield was 90%.
1H-NMR (CDCl3, ppm): 1.37 (s, 9H, COOC(CH3)3), 1.5 (qvint, 2H, (CH3)2-NH-CH2-CH2-), 2.1 (s, 6H, -NH-(CH3)2), 2.2 (t, 2H, (CH3)2-NH-CH2-) and 3.05 (t, 2H, -CH2-NH-BOC).
Synthesis of monoquaternary ammonium propyl-1,3-diamine (MQ-propane-1,3-diamine)
A quantity of 6.62 g of N-BOC-dimethylamino propyl amine (33 mmol) dissolved in 40 ml of acetonitrile was reacted with 4.22 g of methyl iodide (2 equimolar, 66 mmol) at room temperature overnight. Solvent and traces of unreacted methyl iodide were removed by reduced pressure. Resulting product was redissolved in 100 ml of 2 N HCl. Obtained solution was stirred at room temperature for 3 h to deprotect the amino group. Aqueous HCl was removed by reduced pressure yielding yellow precipitate that was further dried under vacuum over solid NaOH pellets. Average yield was 74%.
1H-NMR (D2O, ppm, iodide form): 2.09 (m, 2H, -CH2-CH2-N(CH3)3), 2.9 (m, 2H, -CH2-N(CH3)3), 3.0 (s, 9H, -N(CH3)3) and 3.6 (m, 2H, -CH2-N(CH3)3). Elemental analysis (found): %N=9.23, %I=42.92, %C=24.96. Elemental analysis (calculated): %N=9.98, %I=45.23, %C=25.68.
Synthesis of dextran-MQ-propane-1,3-diamine conjugate
Oxidized polysaccharide (1.0 g, 4.3 mmol of aldehyde groups) was dissolved in 100 ml of DDW. The dialdehyde solution was slowly added for several hours to a basic solution containing 1.5 equimolar amount of monoquaternary ammonium propane-1,3-diamine dissolved in 50 ml of borate buffer (0.1 M, pH 11). The mixture was stirred at room temperature for 24 h. NaBH4 (1.0 g, 4.0 equimolar) was added to reduce the formed imine bonds and stirring was continued for 24 h at room temperature. The reduction was repeated with an additional portion of NaBH4 (1 g). The resulting solution was poured into a dialysis membrane (3,500 Da cutoff; Membrane Filtration Products, Inc., San Antonio, TX, USA) and dialyzed against DDW at 4°C for 3 days. The dialysate was lyophilized to dryness. Average yield was 50% (w/w).
1H-NMR (D2O, ppm):1.8 (m, 2H, -CH2-CH2-N(CH3)3), 2.1–2.9 (m, 4H, -CH2-CH2-CH2-N(CH3)3), 3.0 (m, 9H, -CH2-N(CH3)3), 3.2–4.3 (m, glucose hydrogens) and 4.8 (m, 1H, anomeric hydrogen).
Virology
Tissue culture media and reagents
Dulbecco modified Eagle's medium (DMEM), newborn calf serum (NBCS), penicillin G (10 units/ml), streptomycin (0.01 mg/ml), nystatin (1.25 units/ml) and a trypsin (0.25%)-EDTA (0.05%) solution, were purchased from Biological Industries (Beit Haemek, Israel). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was obtained from Sigma–Aldrich. Noble Agar was purchased from Difco Laboratories (Detroit, MI, USA), formaldehyde was from Bio Lab (Jerusalem, Israel) and crystal violet from BDH Chemicals Ltd (Poole, England). Plasticware (flasks and dishes) for tissue cultures were produced by Nunc (Roskilde, Denmark).
Viruses and cells
HSV-1 (HF strain) and HSV-2 (Curtis strain) were grown in BS-C-1 cells, a cell line originated from green monkey kidney. The cells were cultivated in DMEM supplemented with 7% NBCS and antibiotics and were incubated at 37°C in a humidified atmosphere supplied with 5% CO2. When cell monolayers were formed, the medium was removed and a solution of trypsin-EDTA was used in order to disperse the cells. The cells were diluted and transferred to new dishes or flasks with growth medium and further incubated at 37°C.
Infection procedure
Monolayers of BS-C-1 cells, were infected with virus at the indicated multiplicity of infection. After adsorption for 1 h at 37°C, the cells were washed and medium containing 2% NBCS was added. The infected culture was further incubated at 37°C until harvesting.
Plaque assay
BS-C-1 cell monolayers, in 35 or 60 mm diameter plastic dishes, were infected with a dilution of virus stock. After adsorption for 1 h at 37°C, the cells were overlaid with DMEM containing 0.7% Agar Noble, supplemented with 5% NBCS, and incubated for 4–5 days at 37°C. The cultures were then fixed with 20% formaldehyde in buffered saline and stained with 0.1% crystal violet in 0.1 M citric acid, washed with water and dried at room temperature. Virus plaques were then counted.
Semiquantitative determination of toxicity and antiviral activity
This method, which aimed to determine toxicity and antiviral activity semiquantitatively of a wide range of chemically related compounds, was developed and extensively used at the Wellcome Laboratories of Tropical Medicine, Beckenham, England. When a certain compound was found to be promising using this method, then a quantitative assay was performed. Monolayers of BS-C-1 cells in 60 mm diameter dishes were infected with a virus suspension, in order to result in partial confluency of plaques (approximately 200 plaques per dish). After adsorption for 1 h at 37°C, 5 ml of agar overlay medium was added. When the medium was solidified, a disc (5 mm in diameter) of Whatman 3 MM paper, which was immersed in a solution containing the examined compound at a concentration of 5 mg/ml, was layered on top of the overlay, in the centre of the culture. A gradient of concentrations of the compound was then created with the highest in the centre of the culture and concentrations gradually decreasing towards its periphery. After incubation of the cultures for 4–5 days at 37°C they were fixed with formaldehyde and stained with crystal violet, as described above. Data were obtained by measurement of two diameters: the first was of the central area, in which the cell monolayer was destroyed in case the tested compound had toxicity, and the second was of the more external area, in which virus plaques failed to appear, reflecting antiviral activity. The solvent dimethylsulfoxide (DMSO) or DDW, in which the tested compound was dissolved in, did not exhibit toxicity to the cells, nor antiviral activity, using this assay. Only when a compound had a specific antiviral activity, while exhibiting minimal toxicity to the host-cells, was a quantitative assay then performed.
Results
Anti-herpesvirus activity of various dextranoligoamine conjugates
Oxidized polysaccharides were derived from cationic polysaccharides by conjugation of oligoamine, as previously described [21]. The polycations were characterized for their structure (1H-NMR), nitrogen content (%N) and primary amine content (TNBS test), as shown in Figure 1.
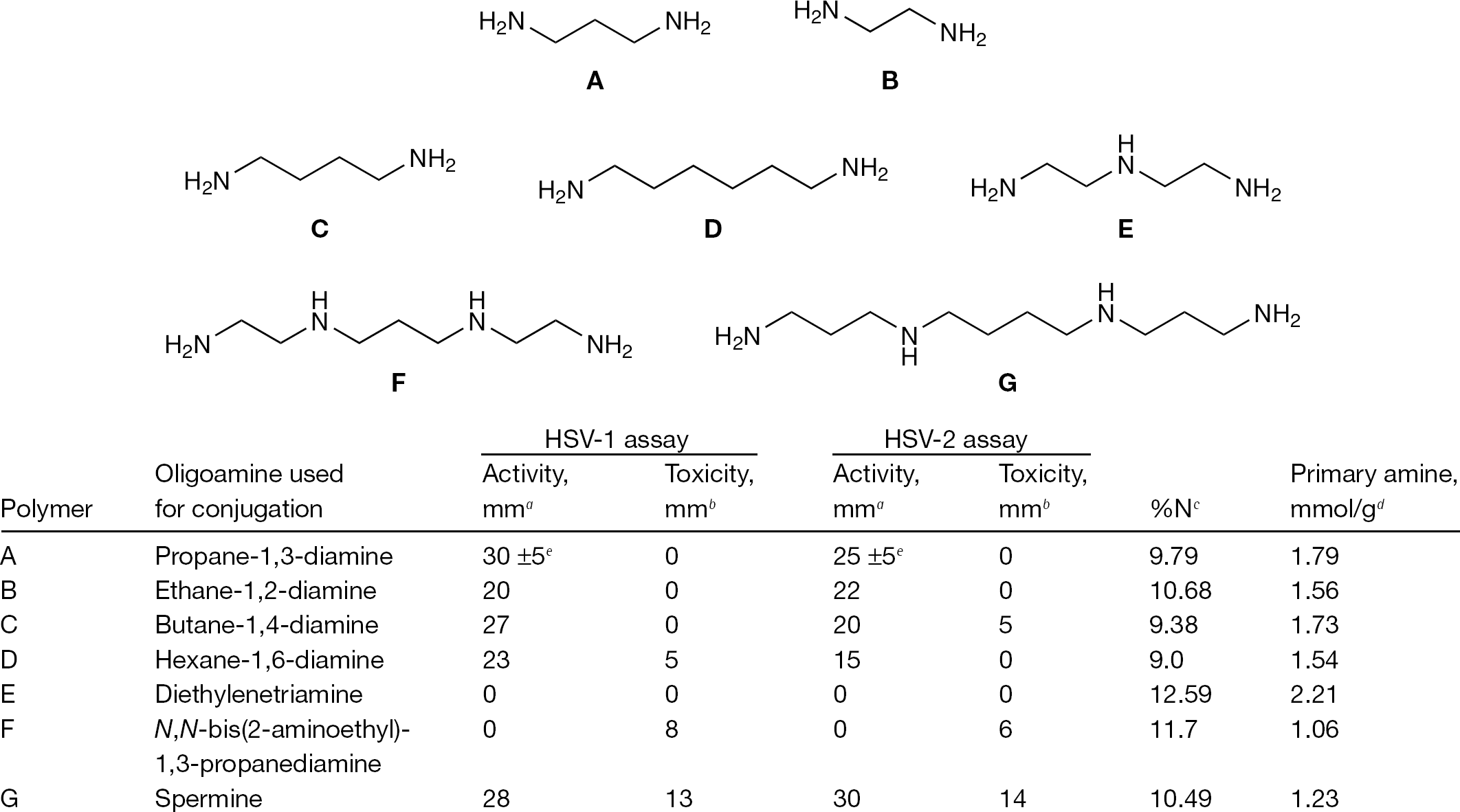
Chemical characterization and biological activity of dextran-oligoamine conjugates
The overall yield obtained was at the range of 50% w/w because of the formation of low molecular weight chains resulting from extensive aminolysis of the glycoside linkages during the conjugation, which are lost during dialysis purification [25]. The polycations had similar nitrogen content, ranging from 9.0% to 12.59%. HSV-1- and HSV-2-infected BS-C-1 cells were exposed to various polycations in the semiquantitative assay. The antiviral effect against these viruses was evaluated by measurement of the diameter of the plaque inhibition area (Figure 1). Most effective were diamine-based conjugates, in particular dextran-propane-1,3-diamine (DPD) conjugate, which resulted in a significant inhibition (25–30 mm) of HSV, whereas triamines, such as dextran-diethylenetriamine and dextran-N,N-bis(2-aminoethyl)-1,3-propanediamine had no effect on these two viruses (Figure 1). By contrast, tetramines, such as dextran-spermine, were effective against HSV-1 (28 mm) and HSV-2 (30 mm) but were also toxic to BS-C-1 cells (13–14 mm; Figure 1). Based on these results, DPD was selected for further study.
Activity of dextran-propane-1,3-diamine with increasing propane-1,3-diamine content
To determine whether the nitrogen content contributes to the potency of the compounds, DPD conjugates with various degrees of diamine substitution (prepared by conjugation of increasing amounts of propane-1,3-diamine onto oxidized dextran), were tested. The polycations were characterized for their average molecular weight (GPC), structure (1H-NMR), nitrogen content (%N) and primary amine content (TNBS test), as shown in Table 1. The activity against HSV-1 and HSV-2 was determined by the semiquantitative assay (Table 1). Interestingly, the most efficient compound was the moderately substituted conjugate (A) whereas both lower (that is, I and J) and higher degrees (that is, H) of substitution reduced the anti-herpesvirus efficiency of the conjugates. Based on these results, a moderately substituted conjugate was chosen for further study.
Chemical characterization and biological activity of dextran-propane-1,3-diamine conjugates at different degrees of substitution
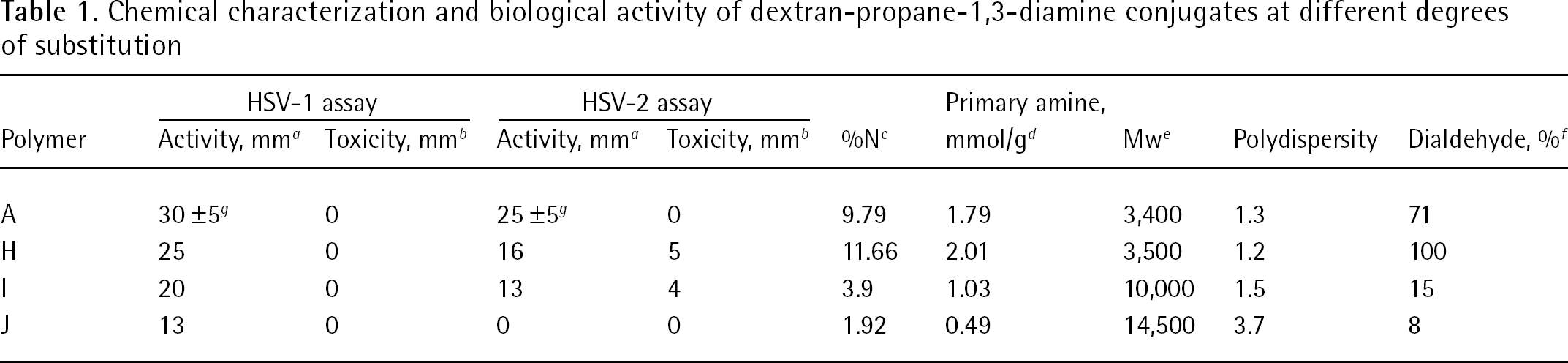
Oxidized dextran was reacted with propane-1,3-diamine at 1:1.5 aldehyde/oligoamine mol ratio under the same conditions as described in the Methods section.
Antiviral activity of the compounds is represented by the diameter (mm) of the external area in the semiquantitative assay in which plaques of herpes simplex virus (HSV) were not formed.
Toxicity is represented by the diameter (mm) reflecting toxicity of the tested compound to the BS-C-1 cells. An acyclovir solution (5 mg/ml) was used as a positive inhibitory control for HSV. A paper, which was immersed in it and layered in the centre of the infected culture, caused 50 mm diameter of plaque inhibition (activity) without any (0 mm) toxicity.
The percentage nitrogen content found by elemental analysis.
Amount of primary amine (mmol/g) in conjugates, determined by the trinitrobenzene sulfonic acid method.
Average molecular weight (Mw) and polydispersity (P=Mw/number average molecular weight) were determined by gel permeation chromatography.
Percentage dialdehyde content of the oxidized dextran prior to diamine conjugation as determined by hydroxylamine hydrochloride titration method.
More than a single assay was done with dextran-propan-1,3-dianine; SD is presented. HSV-1, herpes simplex virus type-1; HSV-2, herpes simplex virus type-2.
Effect of the polysaccharide backbone
To follow the effect of the polysaccharide structure on the activity of the agents, propane-1,3-diamine grafted on pullulan, arabinogalactan and dextran, were examined. Dextran and pullulan are linear chains with glucose units connected by 1,4 and 1,6-glycoside bonds [24], respectively. Arabinogalactan is a branched polymer with its saccharide units connected by 1,3-glycoside bonds [24]. The polycations were characterized for their average molecular weight (GPC), structure (1H-NMR), nitrogen content (%N) and primary amine content (TNBS test), as shown in Table 2. According to the semiquantitative assay, dextran-based derivatives efficiently inhibited both HSV-1 and HSV-2, whereas pullulan- and arabinogalactan-based derivatives were less effective (Table 2).
Chemical characterization and biological activity of polysaccharide-propane-1,3-diamine conjugates

Appropriate oxidized polysaccharide (approximately 70% dialdehyde) was reacted with propane-1,3-diamine at 1:1.5 aldehyde/oligoamine mole ratio under the same conditions as described in the Methods section.
Antiviral activity of the compounds is represented by the diameter (mm) of the external area in the semiquantitative assay in which plaques of herpes simplex virus (HSV) were not formed.
Toxicity is represented by the diameter (mm) reflecting toxicity of the tested compound to the BS-C-1 cells. An acyclovir solution (5 mg/ml) was used as a positive inhibitory control for HSV. A paper, which was immersed in it and layered in the centre of the infected culture, caused 50 mm diameter of plaque inhibition (activity) without any (0 mm) toxicity.
The percentage nitrogen content found by elemental analysis.
Amount of primary amine (mmol/g) in conjugates, determined by the trinitrobenzene sulfonic acid method.
Average molecular weight (Mw) and polydispersity (P=Mw/numbers average molecular weight) were determined by gel permeation chromatography.
More than a single assay was done with dextran-propan-1,3-dianine; SD is presented. HSV-1, herpes simplex virus type-1; HSV-2, herpes simplex virus type-2.
Quaternary ammonium effect on the activity of the compound
We then evaluated the charge effect of the quaternary ammonium propane-1,3-diamine on inhibition of both HSV-1 and HSV-2. Highly charged polycations can interact with negatively charged residues of viral glycoproteins or of cellular membrane. In both cases, this charge interaction could be responsible for HSV inhibition. Therefore, oxidized dextran was modified with monoquaternary ammonium propane-1,3-diamine, to obtain a highly positively charged compound. The semiquantitative assay demonstrated that monoquaternary ammonium conjugate failed to inhibit the two viruses at the tested concentration (Figure 2).
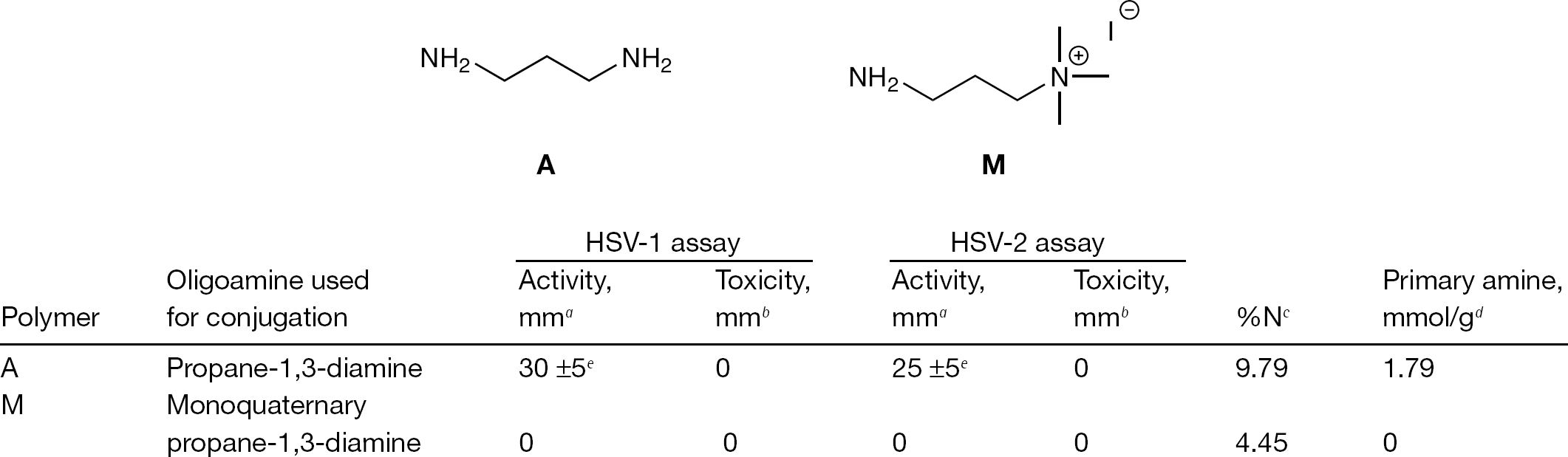
Chemical characterization and biological activity of dextran-oligoamine conjugates
Quantitative determination of the toxicity of dextran-propane-1,3-diamine for BS-C-1 cells
Toxicity of DPD for BS-C-1 cells was determined using MTT assay, which is a reliable test for determination of cell viability [26–30]. This assay is based on conversion of the yellow tetrazolium salt MTT into its blue formazan product performed by live cells; the enzymes involved are rapidly inactivated upon cell death. The intensity of the blue colour of the formazan is proportional to the number of live cells in the culture and can be determined colorimetrically by an ELISA spectrophotometer. The results demonstrated that DPD was not toxic to BS-C-1 cells at a concentration of 156.3 μg/ml, whereas higher concentrations significantly affected the viability of the cells (Figure 3). Therefore, a concentration of 150 μg/ml of DPD was chosen as an optimal concentration in all subsequent experiments.
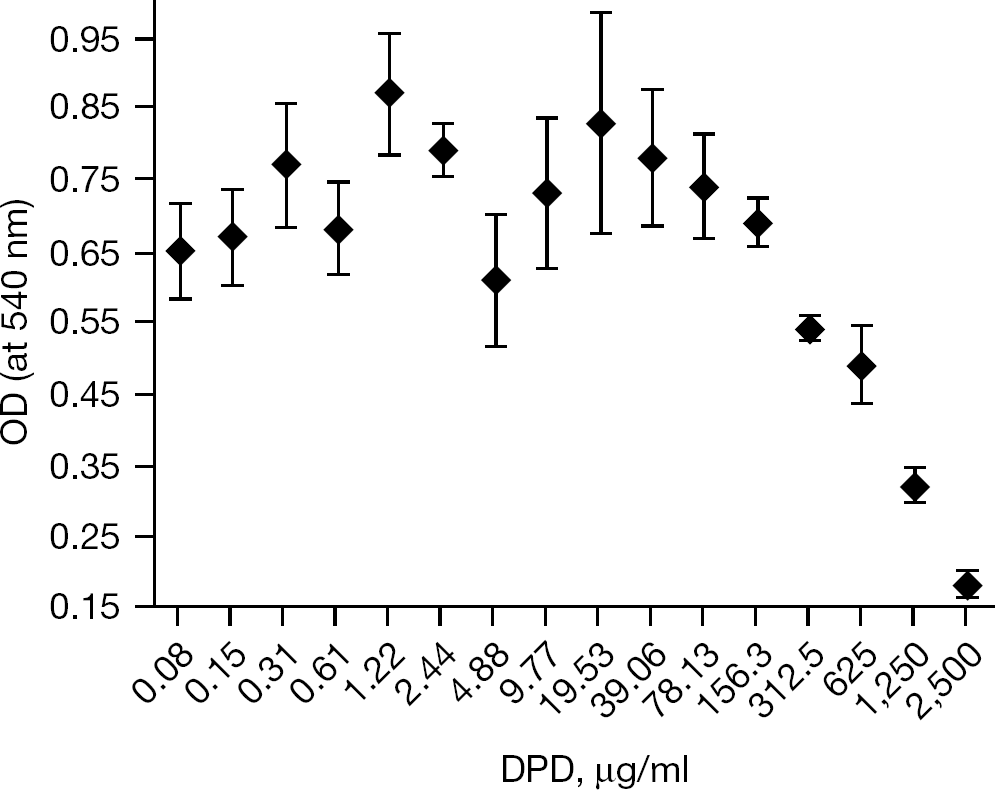
Toxicity of DPD for BS-C-1 cells
Inhibition of the growth of herpes simplex virus
Because a significant inhibition of plaque formation of HSV-2 resulted by DPD was observed using semiquantitative assay (Figure 1), we wished to determine this inhibition quantitatively. Preliminary experiments indicated that when DPD (150 μg/ml) was added to BS-C-1 cultures at the end of the adsorption period (1 h after infection), only a slight inhibition of the growth of HSV-1 and HSV-2 was observed (data not shown). This led us to determine the preferred time of addition of DPD to the cells that will result in the most efficient inhibition of HSV. The growth of HSV-2 was followed in the presence of DPD, added to the cells at time intervals before or after infection. The greatest inhibition was achieved when DPD was added to the cells 1 h before infection and was maintained in the cell culture thereafter (Figure 4). By contrast, significantly lower levels of inhibition were observed when DPD was added to the cells at other time intervals: 24 or 2 h before infection, at the end of the adsorption period (1 h), or 6 h afterwards (Figure 4). The one-step growth curves of HSV-1 and HSV-2 were then followed when treatment of the cells with DPD started 1 h before infection (at a multiplicity of infection of 3 plaque-forming units [PFU] per cell) and was maintained thereafter (Figure 5). Under these conditions, significant delays in the initial growth of both HSV-1 and HSV-2 were observed, and a somewhat lower yield of infectious virus was also obtained, especially in respect to HSV-1 (Figure 5).
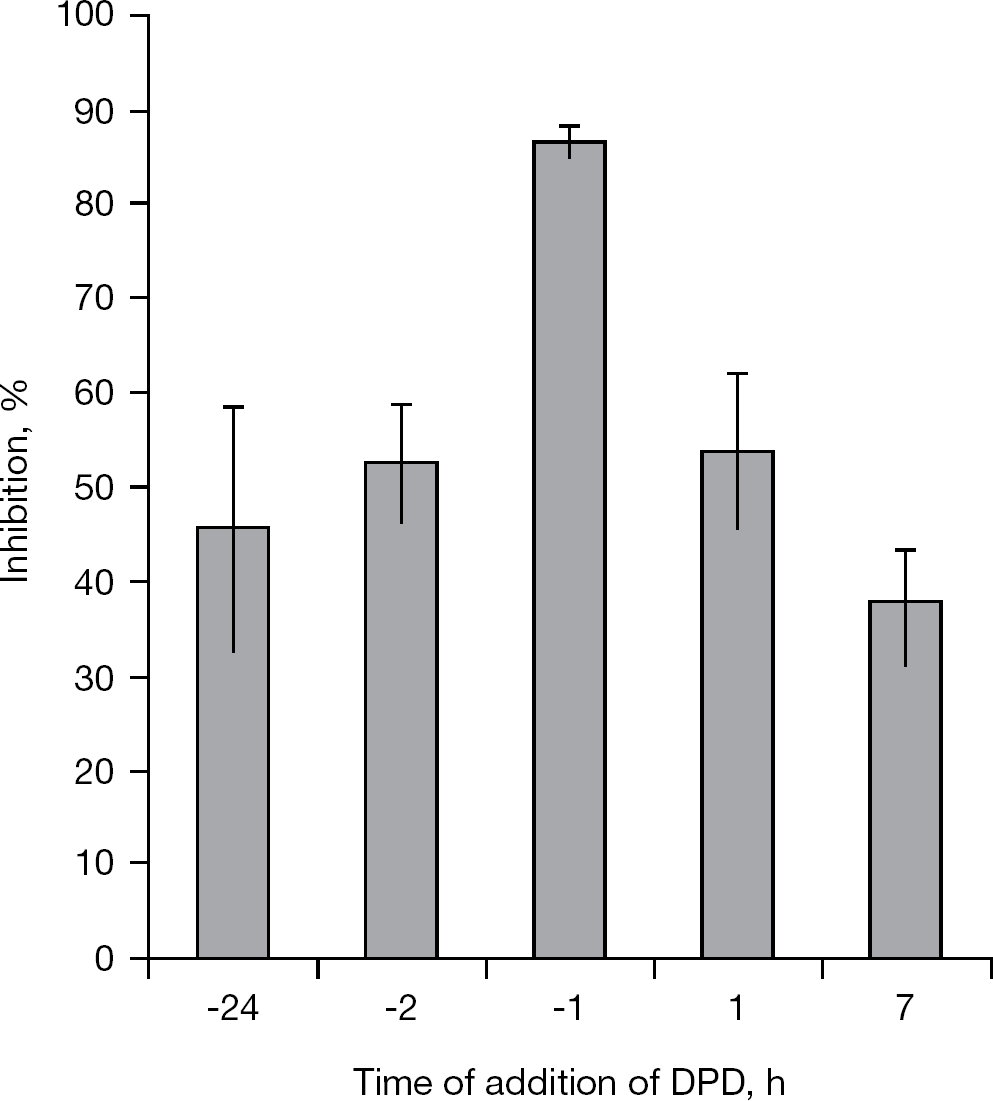
Inhibition of virus growth when DPD is added at time intervals before or after infection
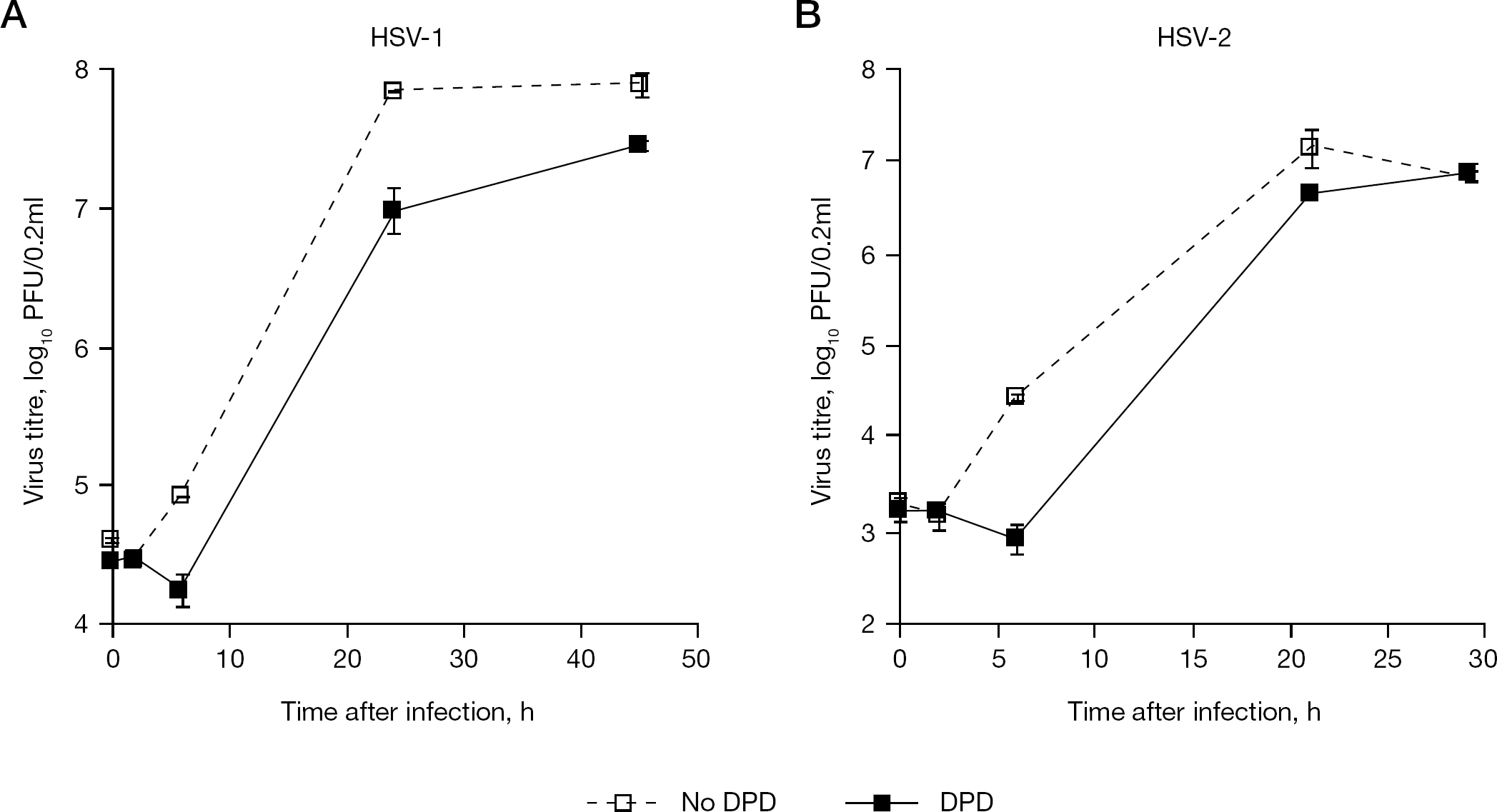
HSV growth in cells treated with DPD starting at 1 h before infection
The mechanism of herpes simplex virus inhibition by dextran-propane-1,3-diamine
In order to find out whether DPD interacts directly with the virus particles and thus inactivates their infectivity, HSV-2 was incubated with DPD (375 μg/ml) in a test-tube for 5 h at 4°C or 21°C, and the infectivity of the virus was then determined. The results clearly demonstrated that DPD failed to inactivate the virions upon direct exposure (data not shown).
Based on previously reported findings, as well as on our data, and in view of the chemical nature of DPD, we hypothesized that DPD might prevent the adsorption of HSV to the cells by interacting with host cell receptors for the virus and consequently blocking the penetration of the virus into the cells [30]. In order to check this assumption, monolayers of BS-C-1 cells in 60 mm diameter dishes were treated with DPD (150 μg/ml) 1 h before infection with 100–200 PFU of HSV-2. The infected cells were washed at time intervals afterwards and agar-containing overlay was added. A parallel set of infected cultures was not treated with DPD. After incubation of the cultures for 5 days at 37°C, the cells were fixed, stained and virus plaques were counted. The results indicated that under these conditions of low multiplicity of infection, DPD inhibited by 95% the adsorption and penetration of the virus to the cells (Figure 6A). Photographs of an infected culture, treated with DPD (150 μg/ml), washed 1 h after infection and then overlaid with agar-containing medium, and in parallel, of untreated infected culture, are presented in Figure 6B. The adsorption/penetration of HSV-2 experiments was then followed in the presence of decreasing concentrations of DPD. It was found that although 150 μg/ml of DPD resulted in inhibition of >90% in the appearance of HSV-2 plaques, 75 μg/ml inhibited only 50% of the plaques, and 37.5 μg/ml of DPD did not affect the number of virus plaques at all (Figure 7).
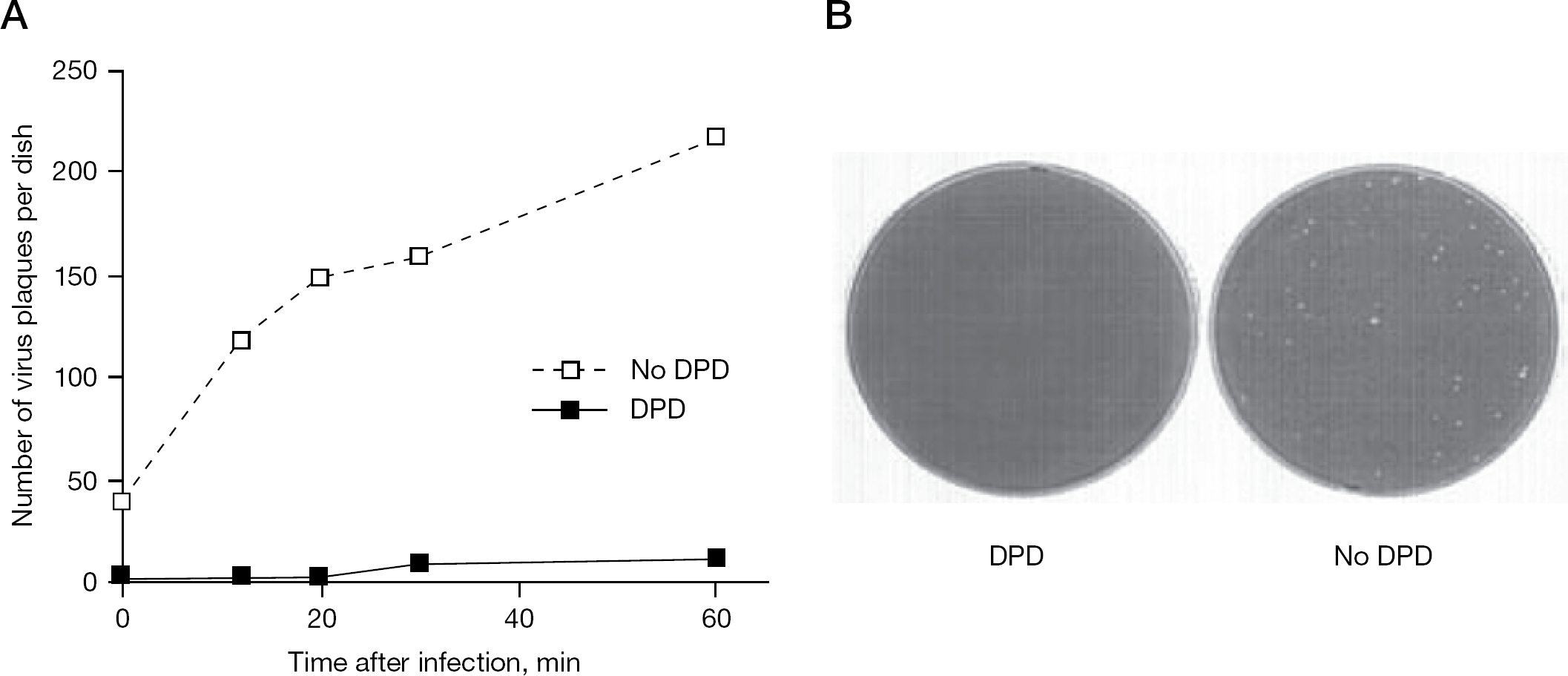
Effect of DPD on the adsorption/penetration of HSV-2 to the cells
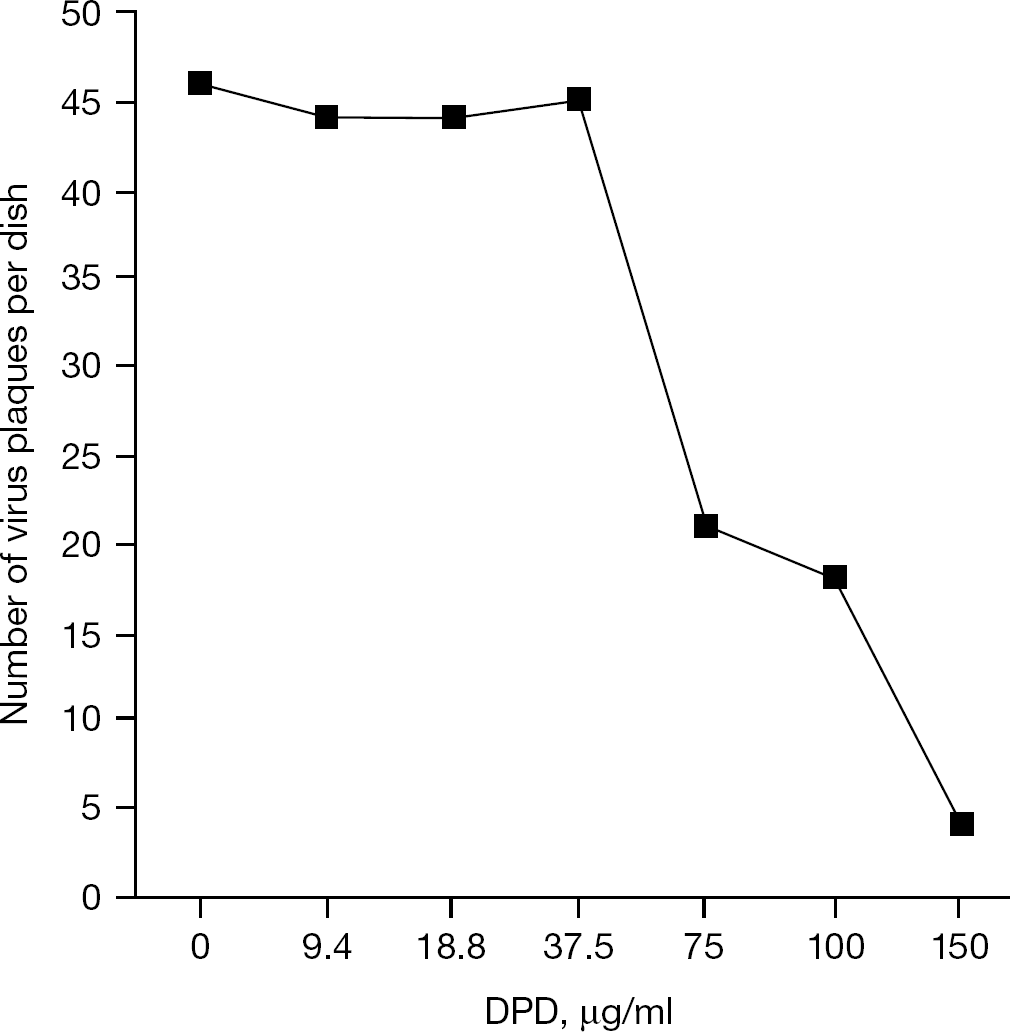
Effect of different concentrations of DPD on HSV-2 adsorption/penetration
Discussion
Biodegradable polycations, prepared by grafting linear (non-branched) oligoamine residues on natural polysaccharides (Figure 8) inhibited the growth of HSV-1 and HSV-2. The most effective and non-toxic dextran derivative was DPD, having one propane-1,3-diamine unit per 1.33 saccharide units. There was a clear relationship between the antiviral activity of the compounds and their chemical structure (Figure 1). Chemical structure integrates several quantitative features of the conjugate, including the molecular weight of the oligoamine molecule serving as a graft and the actual degree of conjugation in each particular conjugate. Although several structural parameters determine the anti-HSV potency of the various conjugates, the most important feature appears to be the identity of the oligoamine grafted onto the sugar backbone, whereas other parameters play a minor role.
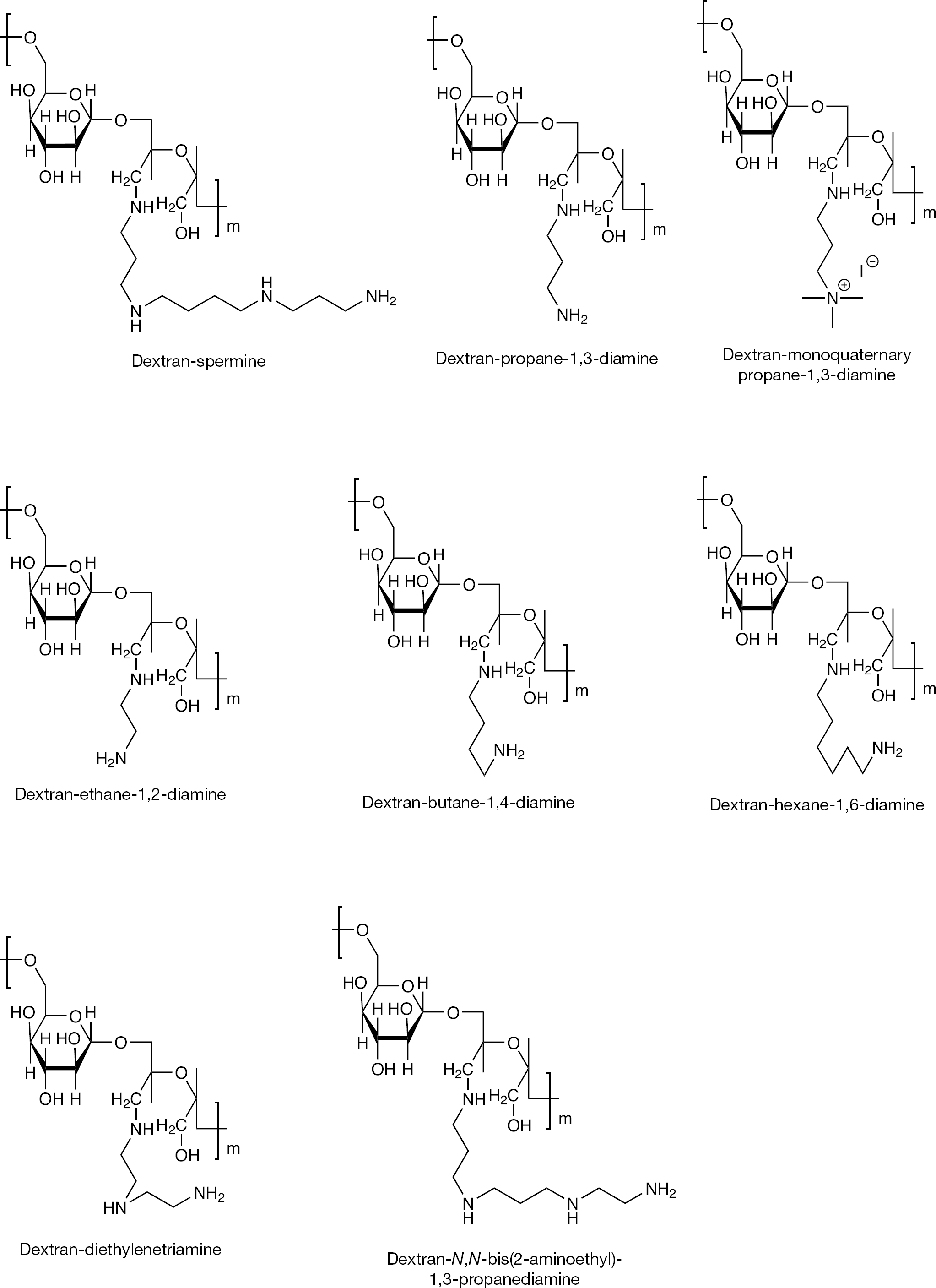
Structures of representative cationic polysaccharides
The antiviral activity against HSV seems to be related to the fact that the compounds are cationic. Dextran, as well as oxidized dextran, did not show any activity against HSV. By contrast, the lack of activity of propane-1,3-diamine supports the idea that the cationic charge itself is not sufficient for inhibiting HSV growth. Furthermore, the absence of activity of the unreduced imine derivative of DPD, which might be attributed to the reduced number of cationic groups, as the imine bond is not cationic, supports the role of the cationic groups in the conjugate. These findings confirm previously reported findings showing that specific spacing of the cationic groups in the polymer is required for the activity of the compound [31].
The oligoamine used for conjugation had an important role on the antiviral activity against HSV. DPD yielded by far the most active compounds (for example, A), whereas triamine (for example, E and F) had no detectable anti-herpesvirus activity in our assay, and tetramine (for example, G) was highly toxic to BS-C-1 cells. Most of the diamine-based compounds were not cytotoxic, as shown in the semiquantitative assay. Furthermore, the efficiency of the conjugates can be attributed to the nature of the polysaccharide backbone. DPD was the most efficient compound, whereas propane-1,3-diamine grafted in the same manner on pullulan and arabinogalactan resulted in slightly less active conjugates. The reason for the superior activity of the dextran compound remains to be determined.
Because anti-herpesvirus activity might be related to the fact that substances are cationic [31] and propane-1,3-diamine is clearly the active moiety, it was expected that increasing the level of diamine conjugation onto oxidized dextran would result in increased antiviral potency. However, differences in the anti-herpesvirus activity of nitrogen content >9.8% were negligible. Thus, the number of ionized amino groups is not the only characteristic determining the potency of the drug.
Furthermore, quaternary ammonium salt was also considered to be affecting the anti-herpesvirus activity of the compound. It has been suggested that cellular membrane phosphoinositides might be the poly-L-lysine target [31]. Therefore, in order to improve DPD binding properties, oxidized dextran was substituted with monoquaternary propane-1,3-diamine, which was expected to result in enhanced anti-herpesvirus activity. In contrast to the leading derivative DPD, quaternary ammonium graft completely abolished the activity of the conjugate. This contradicts the results obtained with poly-L-histidine, where anti-herpesvirus activity was pH-dependent. In brief, the ability of histidine peptides to inhibit the infectivity of HSV was influenced by the total cationic charge of the imidazole side chain of the histidine residue at an appropriate pH [17]. Additional studies are required in order to explain the unpredicted inactivity of the monoquaternary ammonium-based conjugate.
Cell surface heparan sulfate serves as a receptor for many viruses, including HSV-1 and HSV-2 [31]. However, HSV-1 and HSV-2 have also been reported to exhibit a number of type-specific differences in their interactions with host cells [32]. HSV-1 infection of cells was inhibited to a greater extent than HSV-2 by polycationic substances, such as neomycin and poly-L-lysine [33–35], but no significant difference in the inhibition of these two viruses was observed with regard to DPD. In contrast to the inhibitory effect of DPD on HSV observed in the present study, such an effect was not seen with regard to vaccinia virus (WR strain) and poliomyelitis virus (type-1; Sabin strain), grown in the same BS-C-1 cells (data not shown). This clearly indicates that the inhibitory effect of DPD on HSV is specific. This might also suggest that the latter two viruses have different receptors, which enable them to adsorb and penetrate into the cells in the presence of DPD.
Our data indicated that incubation of HSV-2 in a test tube for 5 h at 4°C or at 21°C did not decrease its infectivity, suggesting that DPD does not interact directly with the virions of herpes simplex and does not inactivate their infectivity.
According to the quantitative assay, DPD was not toxic to BS-C-1 cells at a concentration of 150 μg/ml (Figure 3). Furthermore, the growth of HSV-2 was inhibited efficiently when DPD (150 μg/ml) was added to the cells 1 h prior to infection and was maintained in the cultures thereafter (Figure 4). Under these conditions, DPD (150 μg) had a significant inhibitory effect on the initiation of the growth of both HSV-1 and HSV-2 (Figure 5). By contrast to the somewhat limited effect of DPD seen on the one-step growth curves of HSV-1 and HSV-2, infecting BS-C-1 cells at a high multiplicity of infection (3 PFU per cell; Figure 5), a relatively more pronounced effect was observed when DPD was added to BS-C-1 cultures infected with HSV-2 at a much lower multiplicity of infection (100–200 PFU per 5×106 cells), when the effect of DPD on the adsorption/penetration of the virus into the cells was followed (Figure 6). This can be explained by the finding that at the low multiplicity of infection, almost all the viruses can be inhibited by DPD, whereas during the one-step growth curve, despite the presence of DPD, some viruses can overcome the drug, adsorb and penetrate into the cells and complete their growth cycle, thus producing thousands of infectious progeny viruses (Figure 5).
Although DPD was shown in the present study to be less effective in inhibition of HSV than ACV (Figure 1), the combined topical use of ACV together with DPD, two drugs exhibiting different mechanisms of action against HSV, might have an advantage by restricting the enlargement of HSV lesions, in addition to lowering the risk of the appearance of ACV-resistant mutants. Extensive and continued efforts we made to isolate DPD-resistant mutants of HSV-1 and HSV-2 viruses in vitro failed (L Livne and EK, unpublished observations). This might be explained by the fact that DPD interacts with components of the membrane of the host cell that bind the virus, and not with specific viral components.
Footnotes
The authors declare no competing interests.
