Abstract

Keywords
Guideline committees under the sponsorship of the International Society for Peritoneal Dialysis (ISPD) periodically update best practices for optimal peritoneal access (1–4). Recent advances in our understanding of the key aspects of providing successful placement and maintenance of peritoneal catheters compels the current update. Assessment of evidence for guidelines recommendations is made using a modification of the Grades of Recommendation Assessment, Development and Evaluation (GRADE) system for classification of the level of evidence and grade of recommendations (5). Where scientific evidence is not available, recommendations are based on a consensus opinion. The bibliography supporting the recommendations is not intended to be comprehensive. When there are multiple similar reports on the same subject, the committee prefers to cite the more recent publications.
Within each recommendation, strength is indicated as Level 1 (we recommend), Level 2 (we suggest), or not graded, and the quality of the supporting evidence is shown as A (high quality), B (moderate quality), C (low quality), or D (very low quality). The recommendations are not meant to be implemented indiscriminately in every instance but adapted as necessary according to local circumstances and the clinical situation. While many of the general principles presented here may be applied to pediatric patients, the focus of these guidelines is on adults. Clinicians who take care of pediatric PD patients should refer to the latest ISPD guidelines covering this patient group (6).
Catheters for Chronic Peritoneal Dialysis
We recommend catheters made of silicone rubber (1B)
We recommend that standard catheters be provided with double Dacron (polyester) cuffs (1C)
We recommend the use of catheters with either a straight or coiled tip with either a straight segment or preformed arc bend in the intercuff section (1C)
We recommend the use of an extended catheter for remote exit-site location when standard catheters are unable to provide both optimal pelvic position and satisfactory exit-site location (1C)
Currently, most chronic catheters are constructed of silicone rubber, whereas some are fabricated from polyurethane rubber. A polyurethane catheter that ceased production in 2010 was made of a particular polymer extremely susceptible to oxidative stress fractures, softening, and rupture due to chronic exposure to polyethylene glycol present in mupirocin ointment used for long-term catheter exit-site prophylaxis (7). A polyurethane catheter continues to be marketed that is constructed from a higher-grade polymer that may be more resistant to oxidative degradation or softening plasticizers; however, published clinical experiences with this device are required. Erosion of silicone catheters due to the use of gentamicin cream at the exit site has been reported but appears to be a rare complication (8).
The most commonly used PD catheter types are illustrated in Figure 1. The standard double Dacron (polyester) cuff, straight- and coiled-tip catheters with straight or preformed arc bend intercuff segments constitute the mainstay of PD access around the world (Figure 1 A, B). No difference in functionality has been convincingly demonstrated between straight- and coiled-tip catheters with or without a preformed arc bend. There have been 2 meta-analyses of randomized controlled trials (RCT) comparing straight- and coiled-tip catheters (9,10), one of which also included an assessment of a straight versus preformed arc bend design in the intercuff segment (10). While both meta-analyses favored straight-tip catheters, the results were nonuniform with regard to catheter migration with or without flow dysfunction, and the catheter removal and survival data included causes other than flow failure. The meta-analysis evaluating intercuff straight and preformed arc bend segments showed no significant difference between the 2 configurations (10).
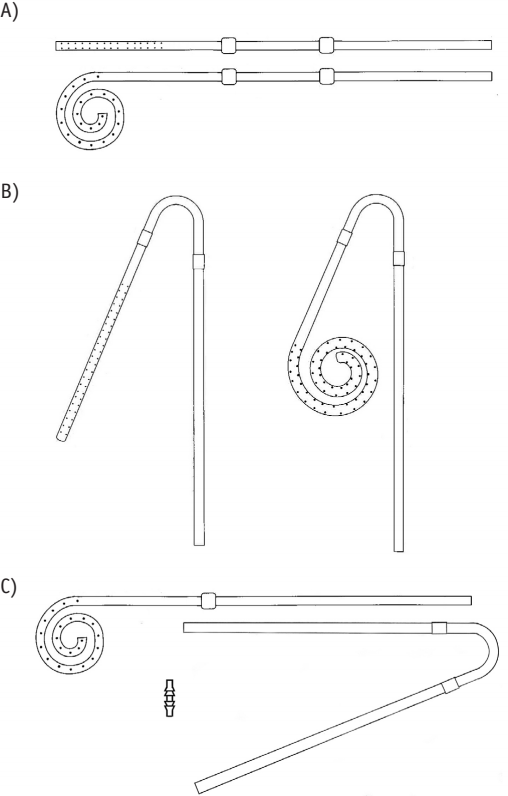
Commonly used peritoneal catheters. A) Catheter with straight intercuff segment, 2 cuffs, and straight or coiled tips. B) Catheter with preformed intercuff arc bend, 2 cuffs, and straight or coiled or tips. C) Extended catheter with 1-cuff, coiled-tip abdominal catheter, 2-cuff extension catheter with preformed intercuff arc bend, and titanium double-barbed connector.
Although standard catheters are available with single Dacron cuffs, it has been hypothesized that double-cuff catheters may be superior to single-cuff catheters in preventing peritonitis caused by periluminal entry of organisms. However, a small RCT showed no difference in peritonitis between single- and double-cuffed catheters (11), although this study seems to have been underpowered. A large retrospective cohort study suggested that the effect of the number of cuffs on peritonitis may be era related (12). Patients initiating PD from 1996 to 2000 had a significantly lower peritonitis rate with double-cuff than with single-cuff catheters, attributed mostly to lower rates of Staphylococcus aureus. In the later interval, 2001 - 2005, there was no difference in peritonitis rates based upon the number of cuffs. The widespread adoption of prophylactic exit-site and intranasal antibiotics during the later era may have reduced exit-site colonization and infection sufficiently to obviate the need for protection offered by the second cuff. The benefit of a double cuff may be particularly important where prophylactic antibiotics are not used. Given that compliance with prophylactic ointments is variable, having the added protection of a double-cuff catheter may be advantageous, especially among diabetic and immunosuppressed patients in whom the risk of Staphylococcus aureus catheter infection is higher (13).
Extended 2-piece catheters were originally designed to provide a presternal exit site (Figure 1 C) (14). The extended catheter consists of a 1-cuff abdominal catheter segment that attaches to a 1- or 2-cuff subcutaneous extension segment using a double barbed titanium connector to permit remote location of the exit site to the upper chest. Extended catheters are also used to provide remote exit-site locations to the upper abdominal and back regions (15,16). The abdominal catheter can be placed by any insertion method. The subcutaneous extension catheter is implanted using a vascular tunneling rod or similar device supplied by the catheter manufacturer.
Most currently manufactured chronic catheters possess a white radiopaque stripe along the longitudinal axis of the tubing that enables radiographic visualization. The stripe can also serve as a guide during implantation of the catheter to prevent accidental twisting or kinking of the catheter tubing. The majority of adult catheters have a 2.6-mm internal diameter. One catheter brand possesses a 3.5-mm internal diameter and can be identified by its blue radiopaque stripe. While the in vitro flow rate of the larger bore catheter is faster, any therapeutic advantage of this device has yet to be demonstrated in the in vivo state. The importance of recognizing the catheter bore size is to prevent accidental interchange of repair kits and replacement catheter adapters that can result in a loose fit and separation.
Various modifications of the standard catheter designs have been made in an attempt to address the common mechanical problems of tissue attachment, tip migration, and pericatheter leaks. However, none of these alternative configurations has persuasively shown to provide any benefit over the standard catheter designs shown in Figure 1, but they do increase device cost, add difficulty to insertion and removal, and they are not universally available. Concerns for common mechanical problems are more reliably addressed by proper implantation technique than by a catheter design.
Catheter Selection
Catheter choice should produce a satisfactory balance of pelvic position of the tubing tip, exit site in a location that minimizes the risk of infection and is easily visible and accessible to the patient, and resulting in minimal tubing stresses during the course of its passage through the abdominal wall (not graded).
We recommend that the PD access team be familiar with a basic inventory of catheter types that permit selection of the most appropriate device based upon body habitus and clinical conditions (1B).
We recommend that the PD team develop a protocol for preoperative mapping to select the most appropriate catheter type from their inventory of devices (1C).
Because patients present with a range of body sizes and shapes with a variety of medical conditions, 1 catheter type cannot be expected to fit all (17). Choice of catheter type should take into consideration the patient's belt line, obesity, skin creases and folds, presence of scars, chronic skin conditions, intestinal stomas, suprapubic catheters, gastrostomy tubes, incontinence, physical limitations, bathing habits, and occupation. If the patient prefers to sleep on a particular side, catheter placement may be better tolerated on the opposite side of the abdomen. It is imperative that the PD access team be familiar with a basic inventory of catheter types to enable customization of the peritoneal access to the specific needs of the individual patient that affords optimal pelvic position of the catheter tip and flexibility in exit-site location. Practical applications of a basic catheter inventory are illustrated in Figure 2. Poor catheter choice can result in flow dysfunction, flow pain, and exit-site locations prone to infection or inconvenience to the patient (4,17,18).
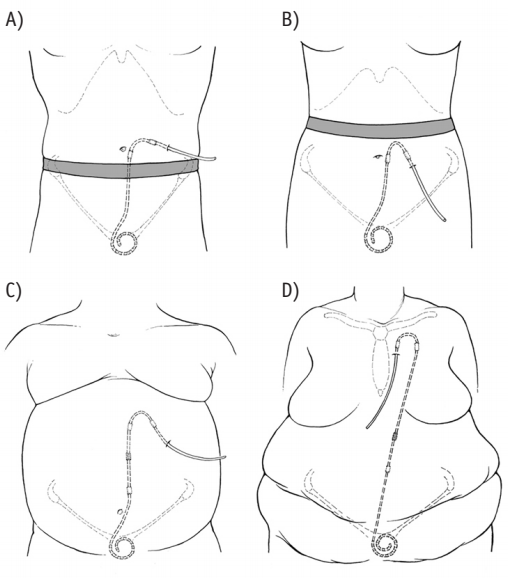
Practical applications of a basic catheter inventory. A) Straight intercuff segment catheter with laterally directed exit site emerging above a low-lying belt line. B) Preformed swan neck intercuff arc bend catheter with downwardly directed exit site emerging below a high-lying belt line. C) Extended catheter with upper abdominal exit site for an obese rotund abdomen, lower abdominal skin folds, or incontinence. D) Extended catheter with presternal exit site for severe obesity, multiple abdominal skin folds, intestinal stomas, or incontinence. Reprinted from Crabtree JH, Chow KM, Peritoneal dialysis catheter insertion. Seminars in Nephrology 2017; 37:17–29, with permission from Elsevier.
The most appropriate choice of catheter is the one that produces the best balance of pelvic location of the catheter tip, exit site in a low infection-risk zone easily visible and accessible to the patient, and permitting insertion through the abdominal wall with the least amount of tubing stress. This choice must not only take into consideration the patient's body habitus and clinical conditions but also the dimensions of the catheter device.
It has been demonstrated by computerized tomographic (CT) peritoneography that 30% - 55% of dialysate rests in the pelvis when the patient is supine (19), thereby supporting the concept of preferably positioning the catheter tip in the pelvis for optimal hydraulic function. On the other hand, excessively deep pelvic placement of the catheter, wedging the tip between the rectum and bladder or uterus can lead to extrinsic compression of the catheter side holes by these structures resulting in flow dysfunction and end-of-drain pain, especially in combination with the hydraulic suction of automated PD (APD) (18). It is the catheter insertion site and the length of intraperitoneal tubing that determines the pelvic position of the catheter tip. Overly deep placement of tubing in the pelvis can be frequently attributed to using the umbilicus as a landmark for catheter insertion and not taking into account the dimensions of the catheter tubing. To avoid this error, the pubic symphysis is recommended as a reliable reference for ideal location of the catheter tip in the upper part of the true pelvis (20,21). With the patient supine and the catheter tubing positioned in the paramedian plane, the upper extent of the catheter tip end that is to rest in the upper portion of the true pelvic bowl is aligned with the upper border of the pubic symphysis bone (Figure 3). For straight-tip catheters, ideally a design with 15 cm of tubing length beyond the deep cuff, a point 5 cm from the tip of the catheter is aligned with the pubic symphysis upper border. With coiled-tip catheters, the upper border of the coil is aligned with the upper border of the pubic symphysis. The insertion incision is indicated by marking the upper border of the deep cuff of the catheter in the paramedian plane. This skin incision site will intercept the musculofascial layer at the proper distance above the true pelvis (21).
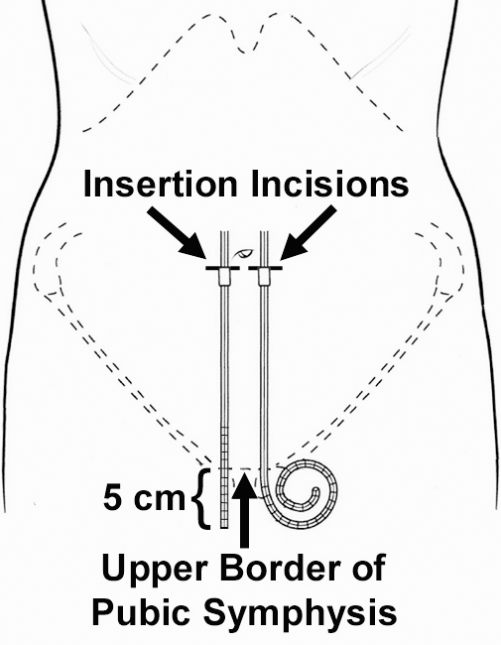
Schematic of a supine patient showing the method in which the catheter insertion site and deep cuff location are determined in order to achieve proper pelvic position of the catheter tip. For straight-tip catheters, ideally a design with 15 cm of tubing length beyond the deep cuff, a point 5 cm from the tip of the catheter, is aligned with the pubic symphysis upper border. With coiled-tip catheters, the upper border of the coil is aligned with the upper border of the pubic symphysis.
The insertion incision site will also determine the range of reachable exit sites. Catheters with a preformed arc bend in the intramural segment must precisely follow the arc configuration to avoid inducing tubing stress from shape memory resiliency forces, selecting an exit-site location 2 to 4 cm beyond the superficial cuff in line with the external limb of the catheter. To avoid excessive shape memory resiliency forces that can cause intraperitoneal catheter tip migration or superficial cuff extrusion, catheters with straight intramural segments are best limited to a gentle arc to produce a laterally directed exit site 2 to 4 cm beyond the superficial cuff (21,22). If the catheter needs to be bent more than to produce a laterally directed exit site, use a catheter with a preformed arc bend instead. A prospective cohort study demonstrated no difference between downward and laterally directed exit sites with regard to rates of exit-site and tunnel infections, peritonitis, and catheter loss (23).
After determining the insertion site to achieve optimal pelvic position of the catheter tip and the exit site that can be reached from this location, the patient is examined in a sitting position. Verify that the selected exit site of the catheter being tested produces a site easily visible to the patient, not located within the belt line, inside a skin crease, or on the blind side or apex of an obese skin fold. If the available inventory of single-piece catheters cannot produce both satisfactory pelvic position and exit-site location, device selection properly shifts to a 2-piece extended catheter system to remotely locate the exit site away from the problematic lower abdominal region to the upper abdomen or upper chest while maintaining optimum position of the catheter tip (24,25). Alternatively, single-piece catheters with long intercuff segments have been designed to reach the upper abdominal wall (26).
Upper abdominal and chest exit sites have the advantage of being located in regions where the subcutaneous fat layer is relatively thin, even in obese individuals, thereby minimizing tubing stresses from mobility of the subcutaneous fat layer with postural changes that can kink the catheter at the subcutaneous-fascial interface or tear the flat granulation tissue lining the sinus track external to the superficial cuff from amplified piston-like catheter motion. Prospective and retrospective cohort studies have demonstrated significantly longer survival times until first exit-site infection and/or lower exit-site infection rates with extended catheters compared with standard abdominal catheters (26–28). This is notable in that extended catheters enable peritoneal access for patients in whom conventional catheter placement would be difficult or impossible. Indications for extended catheters include obesity, incontinence, presence of intestinal stomas, gastrostomy tubes, suprapubic catheters, and those who desire to take a deep tub bath without risk of exit-site contamination (24,27).
The PD access team of each center should agree on a basic catheter inventory and assure that these specific items are made available for the peritoneal access procedure. A protocol for preoperative mapping of the patient should be developed to select the most appropriate catheter type from this inventory. Instead of the cumbersome use of sample catheters, a process of stencil-based preoperative mapping is emerging using marking stencils to provide a reliable and reproducible method of catheter selection (29).
Marking stencils are provided by some dialysis catheter manufacturers for the most commonly used coiled-tip catheter designs. Properly constructed stencils contain critical catheter design information, including the distance between the deep cuff and the catheter coil, suggested subcutaneous tunnel configurations, and recommended exit-site locations relative to the position of the superficial cuff. Additional features of a well-designed stencil plate permit its precise orientation on the trunk region according to fixed anatomical landmarks, such as the upper edge of the pubic symphysis and the anatomical midline of the torso. Stencils permit accurate and reproducible association of the catheter design elements to these anatomical landmarks to help determine the best catheter style and insertion site that will produce optimal pelvic position of the catheter tip and ideal exit-site location. In addition to the preoperative evaluation for catheter selection, the marking stencil is used again at the time of the catheter placement procedure to retrace the previously determined insertion incision, tunnel configuration, and exit-site location (30).
Catheter Placement Procedures
Best Practices in Patient Preparation and Peritoneal Catheter Implantation

Suggested Guidelines for Selecting a Peritoneal Dialysis Catheter Insertion Approach
We recommend that laparoscopic PD catheter implantation employ advanced adjunctive procedures that minimize the risk of mechanical complications (1B)
We recommend that percutaneous needle-guidewire insertion of PD catheters utilize image guidance (ultrasonography and/or fluoroscopy), when such means are available, to improve outcomes and minimize complications (2C)
Independent of the catheter implantation approach, adherence to a number of details is required to assure the best opportunity for creating a successful long-term peritoneal access. A best practice checklist for preoperative preparation and peritoneal catheter placement is presented in Table 1. Omission of any 1 of these components can lead to loss of the PD catheter. Some implantation techniques do not incorporate all of these best practices, such as percutaneous needle-guidewire approaches performed through the midline or positioning the deep cuff above the level of the fascia. It is essential that the practitioner be aware of deviations from recommended practices and be observant for the potential complications that may arise from such departures.
Percutaneous Needle-Guidewire Technique
Placement of catheters by blind percutaneous puncture is performed using a modification of the Seldinger technique. The convenience of this approach is that it can be performed at the bedside under local anesthesia using prepackaged self-contained kits that include the dialysis catheter. Often, the technique includes prefilling the abdomen with dialysis or saline solution instilled through an introducer needle inserted through an infraumbilical or paramedian incision (41,51). Alternatively, a Veress needle may be used to perform the prefill or the prefill step may be skipped altogether (52). A guidewire is passed through the needle into the peritoneal cavity and directed toward the pelvis. The needle is withdrawn. A dilator with overlying peel-away sheath is advanced through the fascia over the guidewire. The guidewire and dilator are withdrawn from the sheath. Optionally, to facilitate insertion, the catheter can be straightened and stiffened by insertion of an internal stylet. If a long guidewire is used, it can be left in the peel-away sheath and the catheter is threaded over the guidewire. The dialysis catheter is directed through the sheath toward the pelvis. As the deep catheter cuff advances, the sheath is peeled away. The deep cuff is advanced to the level of the fascia.
The addition of fluoroscopy to the procedure permits confirmation of needle entry into the peritoneal cavity by observing the flow of injected contrast solution around loops of bowel (36). Ultrasonography can be used in conjunction with fluoroscopy with the additional advantage of identifying and avoiding injury to the inferior epigastric vessels and bowel loops (53). Use of imaging techniques obviates the need to perform a prefill. The retrovesical space is identified by contrast pooling in the appropriate location. The guidewire and catheter are advanced to this site. The remainder of the procedure is conducted as described for blind placement. Although the radiopaque tubing stripe permits fluoroscopic imaging of the final catheter configuration, the proximity of adhesions or omentum cannot be assessed. Percutaneous guidewire placement techniques often leave the deep catheter cuff external to the fascia. After testing flow function, the catheter is then tunneled subcutaneously to the selected exit site.
Open Surgical Dissection
Placement of the PD catheter by open surgical dissection (mini-laparotomy) can be performed under local, regional, or general anesthesia (22,46). A transverse or vertical paramedian incision is made through the skin, subcutaneous tissues, and anterior rectus sheath. The underlying muscle fibers are split to expose the posterior rectus sheath. A small hole is made through the posterior sheath and peritoneum to enter the peritoneal cavity. A purse-string suture is placed around the opening. The catheter, usually straightened over an internal stylet, is advanced through the peritoneal incision toward the pelvis. Despite being an open procedure, the catheter is advanced mostly by feel, therefore blindly, into the peritoneal cavity. The stylet is partially withdrawn as the catheter is advanced until the deep cuff abuts the posterior fascia. After satisfactory placement has been achieved, the stylet is completely withdrawn and the purse-string suture is tied. Encouraging the catheter tip to remain oriented toward the pelvis is achieved by oblique passage of the catheter through the rectus sheath in a craniocaudal direction. The catheter tubing is exited through the anterior rectus sheath at least 2.5 cm cranial to the level of the purse-string suture and deep cuff location. Attention to detail in placement of the purse-string suture and repair of the anterior fascia is imperative to prevent pericatheter leak and hernia. The catheter is tunneled subcutaneously to the selected exit-site following a satisfactory test of flow function.
Peritoneoscopic Procedure
The peritoneoscopic approach, also known as the Y-TEC procedure, is a proprietary laparoscopic-assisted technique of peritoneal catheter placement (Y-TEC; Merit Medical, South Jordan, UT, USA). Peritoneoscopy and laparoscopy are synonymous terms; however, the word peritoneoscopic has been retained by interventional nephrologists to indicate the Y-TEC approach (54,55). The procedure is typically performed in a treatment room under local anesthesia. A 2.5-mm trocar with an overlying plastic sleeve is inserted percutaneously into the peritoneal cavity through a paramedian incision. The obturator of the trocar is removed, permitting insertion of a 2.2-mm laparoscope to confirm peritoneal entry. The scope is withdrawn and 0.6 to 1.5 L of room air is pumped into the abdomen with a syringe or hand bulb. The scope is reinserted and the overlying cannula and plastic sleeve are visually directed into an identified clear area within the peritoneal cavity. The scope and cannula are withdrawn, leaving the expandable plastic sleeve to serve as a conduit for blind insertion of the catheter over a stylet toward the previously identified clear area. The plastic sleeve is withdrawn, and the deep cuff is pushed into the rectus sheath. After testing flow function, the catheter is tunneled subcutaneously to the selected exit site.
Surgical Laparoscopy
Laparoscopy provides a minimally invasive approach with complete visualization of the peritoneal cavity during the catheter implantation procedure. Laparoscopic procedures are performed under general anesthesia in an operating room environment. Surgical laparoscopy uses either a basic or advanced approach to providing PD access. Basic laparoscopic catheter placement has come to mean using the laparoscope to simply monitor the positioning of the catheter tip within the peritoneal cavity (44,56), whereas advanced laparoscopic implantation utilizes additional preemptive procedures to minimize subsequent risk of mechanical catheter complications (57 58 59 60 61-62). With either approach, a pneumoperitoneum is created by insufflating gas through a lateral abdominal wall puncture site using a Veress needle or optical trocar device distant from the point of intended catheter insertion. Alternatively, and especially when patients have had previous midline abdominal surgery or peritonitis, initial port placement can be performed by cutdown to the peritoneal cavity through an incision just inside the lateral border of the rectus sheath in the mid- or upper-abdominal region. The laparoscope is inserted at this remote location to guide placement of the PD catheter into the pelvis through a second abdominal wall entry point. Completion of catheter positioning is the juncture between the basic and advanced laparoscopic PD access procedure.
Advanced laparoscopic catheter placement employs proactive adjunctive techniques that significantly improve catheter outcomes. Laparoscopically guided tunneling of a port device through the rectus sheath permits placement of the catheter in a long musculofascial tunnel directed toward the pelvis and effectively prevents catheter tip migration, eliminates pericatheter hernias, and reduces the risk of pericatheter leaks (57 58 59 60 61-62). Observed redundant omentum that lies in juxtaposition of the catheter tip can be displaced from the pelvis into the upper abdomen and fixed to the abdominal wall or falciform ligament, or folded upon itself (omentopexy) (43,63,64). Compartmentalizing adhesions that may affect completeness of dialysate drainage can be divided. Intraperitoneal structures that siphon up to the catheter tip during the intraoperative irrigation test can be laparoscopically resected, including epiploic appendices of the sigmoid colon and uterine tubes (43,65). Redundant and bulky rectosigmoid colon blocking the pelvic inlet can be suspended along the lateral abdominal wall (colopexy) (43,66). Previously unsuspected abdominal wall hernias can be identified and repaired at the time of the catheter implantation procedure (43,61).
Other variations of rectus sheath tunneling, including the use of a third laparoscopic port site, have been described but the effect is the same, with immobilization of the catheter in a craniocaudal direction through the rectus sheath toward the pelvis (58,60,62). Alternatively, immobilization of the catheter toward the pelvis has been accomplished with a suture sling placed around the tubing through the lower abdominal wall (67). Laparoscopically suturing the catheter tip to a pelvic structure has been associated with failure from erosion of the stitch from the tissue (68 69-70) or having to return to cut the suture in order to remove the catheter (71). Table 3 summarizes best practices for advanced laparoscopic placement of PD catheters.
Best Practices for Laparoscopic Peritoneal Catheter Implantation

The deep cuff of the catheter is positioned in the rectus muscle just below the anterior fascial sheath. A purse string fascial suture is placed around the catheter at the level of the anterior sheath to further minimize the risk of pericatheter leak (43). The pneumoperitoneum is released, but laparoscopic ports are left in place until a test irrigation of the catheter demonstrates successful flow function. After any indicated adjunctive procedures are completed, the catheter is tunneled subcutaneously to the selected exit site.
Catheter Implantation Outcomes
It is often argued that no single implantation approach has been shown to produce superior outcomes. Operator performance aside, when catheter placement by percutaneous needle-guidewire with or without image guidance, open surgical dissection, peritoneoscopy, and laparoscopy are compared side to side on identical study populations, the outcomes reported in the literature are not that different (44,52,56,72–74). Previous systematic reviews and meta-analyses comparing laparoscopic with open dissection produced nonuniform results and erred by including basic and advanced laparoscopic procedures under a single category (75–78). More recently, a meta-analysis of prospective and retrospective cohort studies comparing open dissection, basic, and advanced laparoscopic catheter implantation procedures demonstrated significantly superior outcomes for advanced laparoscopy over the other 2 approaches with regard to catheter tip migration, flow obstruction, and catheter survival (79). These data emphasize that simply using the laparoscope to witness catheter tip position is underutilization of this modality. This is further supported by studies showing that basic laparoscopy used only to observe catheter tip location produces results no better than fluoroscopically guided placement with radiologic verification of catheter position (73,80). The strength of advanced laparoscopic implantation is the adjunctive procedures that are enabled by this approach, producing outcomes superior to all other catheter placement methods.
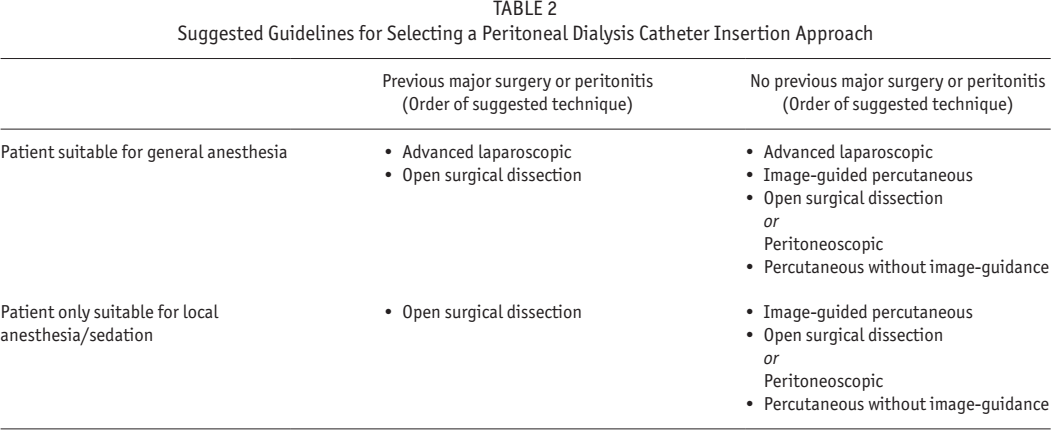
Percutaneous needle-guidewire placement with or without image guidance and peritoneoscopic catheter insertion methods may be inadvisable for patients with obesity, multiply operated abdomen, prior peritonitis, inability to lay flat, or poor tolerance to procedures under local anesthesia, even with conscious sedation. However, where technical expertise exists, a comprehensive preprocedural assessment utilizing ultrasound may permit objective case selection for safe percutaneous or peritoneoscopic insertion of PD catheters in patients who may have otherwise been excluded because of prior abdominal surgery, large bilateral polycystic kidneys, or central obesity (53). General anesthesia may be required for some cases of open surgical dissection and all laparoscopic procedures. Advances in anesthesia pharmacology, techniques, and monitoring have improved the safety of general anesthesia for high-risk patients. It is the magnitude of the surgical procedure itself that confers the most risk. Fortunately, PD catheter insertion is minimally invasive. Nevertheless, consideration must be given to the patient's comorbidities and the capability of the anesthetist when choosing the safest manner of conducting the procedure. Based upon patient factors, resource availability, and the expertise of the operating team, Table 2 offers guidelines for selecting a PD catheter insertion approach.
Special Peritoneal Access Methods
Extended 2-Piece Catheters
The abdominal segment of 2-piece extended catheters (Figure 1C) can be implanted by any of the above-mentioned insertion techniques (24,25,81,82). A secondary incision is made in the vicinity of the planned upper abdominal, presternal, or back exit site. A marking stencil is invaluable in devising the location of the secondary incision and exit site. The measured distance between the abdominal insertion incision and the secondary incision is used to calculate how much tubing length will be trimmed from one or both of the catheter segments in order to correctly span the distance. The trimmed catheters are joined with a supplied double barbed titanium connector and the linked catheter segments are tunneled on the surface of the fascia from the abdominal insertion site to the remote secondary incision with a tunneling rod. The extension catheter is then passed from the secondary incision through the exit site using a stylet to complete the procedure.
Catheter Embedding
Commonly referred to as the Moncrief-Popovich technique (83), catheter embedding consists of implanting a PD catheter far in advance of anticipated need. Instead of bringing the external limb of the catheter out to the surface, it is embedded under the skin in the subcutaneous space. When renal function declines to the point of needing to initiate dialysis, the external limb is brought to the outside through a small skin incision.
Because the catheter has been afforded extended healing time within the abdominal wall, the patient is able to proceed directly to full volume PD without the necessity of a break-in period that ordinarily accompanies a newly placed catheter. Firm tissue ingrowth of the cuffs and absence of biofilm formation have been speculated to reduce catheter infection-related peritonitis. Another important attribute of catheter embedding is greater patient acceptance for earlier commitment to PD by catheter placement ahead of time. The patient is not burdened with catheter maintenance until dialysis is needed. The need for insertion of vascular catheters and temporary hemodialysis can be avoided in patients previously implanted with an embedded catheter. The embedding technique permits more efficient surgical scheduling of catheter implantation as an elective non-urgent procedure and helps to reduce stress on operating room access. Disadvantages of the catheter embedding strategy include the need for 2 procedures (implantation and externalization) as opposed to 1 and the possibility of futile placement in the event of an adverse change in the patient's condition during the time period that the catheter is embedded or if the patient undergoes a preemptive kidney transplant and the catheter is never used (84,85).
Catheter embedding can be incorporated into any of the implantation approaches using any catheter device. The catheter is temporarily externalized through the future skin exit site prior to embedment. The exit-site scar serves as a landmark to know where to come back to for externalization. After acceptable flow function of the catheter is confirmed, the tubing is flushed with heparin, plugged, and buried in the subcutaneous tissue. To minimize the risk of hematoma or seroma and to facilitate subsequent externalization, the catheter should be embedded in a linear or curvilinear subcutaneous track using a tunneling stylet (86,87) as opposed to curling the tubing into a subcutaneous pocket (88). Embedding should not be performed if anticipated need for dialysis is <4 weeks or if the patient has had previous major abdominal surgery or peritonitis where adhesiolysis may likely leave blood in the peritoneal cavity. Externalization of embedded catheters is easily accommodated in the office provided that a suitable procedure room is available. Catheters have been embedded for months to years with an 85% to 93% immediate function rate upon externalization (84,87,89,90). Catheter dysfunction is usually due to adhesions or intraluminal fibrin clots. Overall, 94% to 99% are successfully used for dialysis after radiologic or laparoscopic revision of nonfunctioning catheters (84,87,90).
Simultaneous Abdominal Surgical Procedures
Hernia Repair
Abdominal wall hernias can be safely repaired at the time of the catheter placement procedure (43,61,87,91). If the hernia is complicated and a prolonged healing time is anticipated prior to initiating PD, consider combining the repair with catheter embedment.
Repair of hernias with prosthetic mesh is essential for PD patients to minimize risk of recurrence (92 93-94). Laparoscopic placement of intraperitoneal mesh is widely practiced for repair of abdominal wall defects; however, the safety of intraperitoneal mesh in PD patients has not been established. The question is whether or not the neoperitoneum that develops over the intraperitoneal mesh after 2 to 4 weeks is a sufficient barrier to prevent seeding of the material in the event of dialysis-related peritonitis. At the time of this guidelines publication, there has been only a single case report where intraperitoneal hernia mesh was not infected after a patient experienced 2 separate episodes of dialysis-related peritonitis that required catheter removal on each occasion (95). Until more evidence becomes available, extraperitoneal mesh repairs are suggested.
Cholecystectomy
Patients with symptomatic biliary tract disease without signs of active infection can safely undergo cholecystectomy at the time of catheter placement (43,87,96). Following sound surgical principles, the clean procedure (catheter placement) should precede the clean-contaminated portion (cholecystectomy) with closure and protection of PD catheter-related wounds and exit site.
Abdominal Vascular Prostheses
The two major concerns with performing PD in patients with an abdominal vascular prosthesis are, in the event of PD-related peritonitis, the graft may become infected by direct extension into the retroperitoneum, and an associated bacteremia may result in intravascular seeding of the prosthesis. While both of these routes of graft infection are possible, the occurrence appears to be quite rare.
Published reports describe placement of PD catheters and initiation of dialysis simultaneous with repair of ruptured abdominal aortic aneurysms (97) or at intervals between vascular graft placement and start of PD as early as 1 month (98), 3 months (99), and 4 months (100) without infection of the prosthesis. It would seem prudent to allow, at minimum, a 2-week period of retroperitoneal epithelialization following an intraabdominal graft placement before starting PD (100). The Kidney Disease Outcomes Quality Initiative (K/DOQI) guidelines recommend a waiting period of 4 months after intraabdominal vascular graft placement before initiating PD (101). Increasing the use of endovascular aortic and iliac artery stent grafting altogether avoids the problem of direct retroperitoneal contamination and allows patients already on PD to continue therapy uninterrupted (102).
The resistance to hematogenous infection of a prosthetic vascular graft increases with time due to the formation of a pseudointimal layer inside the graft. In addition, the significantly lower incidence of bacteremia associated with PD, as opposed to hemodialysis, makes it a more logical modality choice in patients with prosthetic grafts (100,103,104).
Gastrostomy Tubes
The use of percutaneous endoscopic gastrostomy (PEG) tubes in patients receiving PD is debated due to frequent infectious complications. Leakage of peritoneal fluid around the PEG leads to a high rate of fatal peritonitis, especially by fungal organisms (105,106). If a PD patient requires a PEG, it is recommended that the PD catheter be removed with staged reinsertion after the gastrostomy has had time to heal (106). There are reports of successfully retaining catheters without the occurrence of infection by suspending PD for 3 to 6 weeks’ healing time under the cover of prophylactic antibiotics, but failures using this approach should be expected (105,107,108). Inserting a PD catheter into a patient with an existing PEG is considered relatively safe. The catheter exit site should be located remote from the PEG, on either the opposite side of the abdomen or a presternal exit-site location to reduce the risk of catheter infection (106).
Autosomal Dominant Polycystic Kidney Disease
Peritoneal dialysis is often avoided in polycystic kidney disease (PKD) patients because of concerns about limited peritoneal space, peritonitis, and hernias. Recent studies support the feasibility of PD in most PKD patients (109-114). Despite crowding of the peritoneal space with enlarged cystic kidneys, there is no significant difference between PKD patients and non-PKD patients without diabetes for dialysis adequacy and patient and technique survival. Therefore, PD is successful as renal replacement therapy for many PKD patients, whatever their kidney size, even in patients who need a pre-transplant nephrectomy (115). In addition, there is no significant difference between PKD patients and non-PKD patients without diabetes for incidence of peritonitis overall and occurrence of enteric peritonitis (109,110,113,114).
Patients with PKD are at higher risk of abdominal wall hernias (115). The occurrence of hernias may not be directly related to increased intraperitoneal pressure but is possibly linked to collagen defects (116). Repair of hernias with prosthetic mesh will minimize risk of recurrence and permit continuation of PD.
To prevent injury to the massively enlarged kidneys during catheter implantation, caution must be exercised with insertion of laparoscopic ports, trocars, and needles. Open surgical cutdown to the peritoneal cavity for initial laparoscopic port placement or ultrasound-guided percutaneous insertion of trocars and needles is indicated (53).
Colonic Diverticulosis
Controversy exists concerning the association between colonic diverticulosis and the risk of experiencing enteric peritonitis while on PD. The presence of diverticulosis was once considered a relative contraindication to PD (117,118). There are studies clearly associating risk of enteric peritonitis with diverticulosis (118,119), while others find no such relationship (120,121). The difference in findings may be related to the wide variation in diverticulosis prevalence and patient characteristics among different countries. Diverticular disease is primarily found in the sigmoid colon in Western patients and predominantly right-sided in Asian populations, although the reason for this is unclear (119,122).
The prevalence of diverticulosis increases with age; however, age is not considered a risk factor for enteric peritonitis (118–120,123). The number of diverticula, their size, and the extent of colonic involvement appear to be the most important factors linked with the risk of enteric peritonitis (118,121). A study performing barium enemas as a predialysis investigation suggested that the presence of 10 or more diverticula or 1 or more diverticula greater than 10 mm in size was associated with increased risk of developing enteric peritonitis (118).
It is generally agreed that asymptomatic diverticulosis or a remote history of resolved diverticulitis is not a contraindication for PD (118,120,121,124). Preoperative imaging studies are warranted in patients with gastrointestinal symptoms. The presence of diverticular disease may be incidentally documented in patients undergoing routine colorectal cancer screening exams. Research is needed to better define the risk of infectious complications for potential PD candidates with diverticular disease.
Peritoneal Dialysis and Bariatric Surgery
Morbidly obese PD patients have a crucial need for effective weight management interventions to qualify for kidney transplantation and to improve obesity-related morbidity and overall mortality. Although limited published experiences are available, laparoscopic bariatric surgery has enabled PD patients desirous of kidney transplantation to reach their qualifying weight goal (125,126). Before submitting patients to surgery, it is advisable that they receive conditional approval for inclusion into regional kidney transplant programs, contingent upon achieving a center-specified target weight. Laparoscopic bariatric procedures producing the best weight reduction include gastric sleeve resection and Roux-en-Y gastric bypass. It is essential that operations are performed by an experienced bariatric surgeon with a low incidence of complications. Caution must be exercised in laparoscopic port placement to avoid damage to the catheter tubing in its abdominal wall track, especially for patients with an extended catheter to the upper abdomen or chest. With watertight closure of laparoscopic port sites, PD can be resumed immediately utilizing a recumbent low-volume intermittent PD protocol for the first 2 postoperative weeks (125,126).
Perioperative Management of the Pd Catheter
We suggest a trial irrigation of the catheter before final placement is accepted with a sufficient volume of solution to demonstrate unimpeded inflow and outflow (not graded)
We suggest that postoperative flushing of the catheter be tailored to specific patient conditions, i.e., timing and frequency, based upon presence or absence of blood in the trial irrigant at the time of catheter placement or persistence of blood-tinged effluent during postoperative flushes (not graded)
We suggest that the PD center's preferred catheter adapter and transfer or extension set be attached at the time of the catheter placement procedure (not graded)
We suggest a nonocclusive gauze surgical dressing sufficient in size to immobilize the catheter, absorb drainage, and prevent trauma and contamination of the exit site (not graded)
We suggest that when possible, postoperative dressing changes be restricted to experienced PD nursing staff (not graded)
Testing Hydraulic Function
It is important to test catheter patency and flow function before accepting intraperitoneal placement of the catheter and ending the procedure. If the catheter has poor flow function at the outset, it is unreasonable to presume that somehow it will improve during the postoperative period. Catheter position should be revised until satisfactory flow function is achieved.
There are no established protocols for hydraulic testing, and a wide variety of clinical practices exist. A minimalist approach is to inject 60 mL of saline into the catheter. Easy return of some of this fluid and changes in the level of an air-fluid interface in the catheter during respiration confirm that the catheter is located in the peritoneum and has no kinks. A more thorough test of flow function consists of infusing 500 to 1,000 mL of saline or dialysate and observing for unimpeded inflow and outflow, allowing a 100- to 200-mL residual volume to remain to avoid leaving peritoneal structures siphoned up to the side holes of the catheter. The larger irrigation volumes may permit an opportunity for redundant omentum, epiploic appendices, vermiform appendix, or uterine tubes to drift up to the catheter tip and manifest as a cause for slow or low volume drainage. Repositioning the catheter may potentially resolve the flow dysfunction, while laparoscopic techniques can definitively deal with these identified sources of obstruction and reduce the risk for future mechanical complications. The larger irrigation volume also provides an assessment of hemostasis and washes out any accumulation of blood from the procedure.
Postoperative Catheter Flushing
As is the case with hydraulic testing, there is a wide range of postoperative catheter flushing policies among PD centers, if performed at all (127,128). The most common practices include flushing with dialysate or saline solution weekly, using 500- to 1,000-mL volumes, until dialysis is initiated (128). The primary reason for flushing is to prevent fibrin or blood clot obstruction of the catheter. The argument offered against flushing is that no high-level evidence exists that it does in fact prevent blockage. The proponents of a no-flushing policy assert that embedded catheters are not flushed and still function upon exteriorization months to years later. However, it is often overlooked that 10% to 15% of embedded catheters are obstructed by fibrin clots and adhesions when first exteriorized (84,87,90). In a recent RCT concerning PD start times, in which 1 of the catheter groups was not flushed for 4 weeks following placement, technique failure from flow dysfunction was 17% in the intention to treat analysis and 20% when analyzed per protocol (129,130).
The catheter implantation procedure may be accompanied by the accumulation of blood in the peritoneal cavity, especially when performance of adhesiolysis, omentopexy, hernia repair, cholecystectomy, and other adjunctive procedures represent additional sites of bloody seepage. Intraperitoneal blood can lead to catheter blockage from intraluminal clots and formation of adhesions. An early flushing protocol to clear the blood and leaving residual solution in the peritoneal cavity have been shown in a retrospective cohort study to significantly reduce the incidence of catheter failure (131).
While the need for RCTs evaluating the role of PD catheter flushing in preventing catheter malfunction is clearly justified, a flexible approach based upon patient conditions can be suggested. If bloody effluent is recognized during hydraulic testing and/or the patient undergoes multiple interventions during catheter placement that increase the risk of bleeding, it is advisable to flush the catheter within 24 hours, repeating the lavage until clearing of blood is noted. Heparin, 1,000 units/L, may be added to the irrigant to help prevent blood clots and fibrin plugs. Unless there is persistence of blood in the effluent, flushes can be extended to weekly intervals until PD is started. If catheter placement is uneventful with negligible blood in the test irrigant, initial flush is performed at 1 week and then weekly until dialysis is initiated. In the event that the catheter is unused for a period of time, flushing can be increased to 2- to 4-week intervals after the first month.
The additional benefit of postoperative flushing is that it represents an opportunity to detect catheter malfunction early in order to facilitate timely intervention prior to the scheduled start of patient training or to review clinical status, including care of the exit site (128).
Catheter Adapters and Transfer/Extension Sets
The access provider should ascertain the PD center's preferences for type of catheter adapter and transfer/extension set and attach these devices at the time of the catheter implantation procedure. Although manufacturers include a plastic adapter with the catheter, some PD centers prefer a separately supplied titanium catheter adapter. There is no better place than the sterile environment of the operating room to make these necessary connections, sparing the PD nursing staff from having to go through meticulous sterile preparation procedures to make these attachments and risk iatrogenic peritonitis.
Surgical Dressings
Properly applied surgical dressings achieve immobilization of the catheter and prevent trauma and contamination of the exit site. Nonocclusive gauze dressings are preferred because drainage is wicked away from the insertion incision and exit site (132,133). Transparent occlusive dressings should not be used alone because drainage tends to pool underneath them. The dressing must be large enough to cover the insertion incision and exit site and contribute to immobilizing the catheter tubing to prevent traction injury. The transfer/extension set should be taped securely to the abdomen, separate from the dressing so that the PD nursing staff have access for catheter flushing without disturbing the dressing. The surgical dressing should not be changed for 5 to 10 days unless there is obvious bleeding or signs of infection (4,50). It is generally agreed that postoperative dressing changes should be restricted to experienced PD staff, or trained patients if they live far from the center (50). To prevent contamination and infection of the healing exit site, patients are not to resume showering until instructed by the PD nursing staff that it is safe to do so. Postoperative and long-term exit-site care, including frequency of dressing changes, types of dressings (if any), cleansing agents, and use of topical prophylactic antibiotics at the exit site, have been described in recent ISPD guidelines (134,135).
Catheter Break-In Procedures
We recommend a break-in period of at least 2 weeks before elective start on PD (1B).
We recommend a modified PD prescription using low volume exchanges with the patient in the supine position if urgent start on PD with a break-in period of < 2 weeks is needed (1C).
The break-in period is defined as the time interval between PD catheter insertion and initiation of PD. Procedures to prevent and treat catheter-related infections, peritonitis, and mechanical complications during the break-in period are covered elsewhere in the present or latest ISPD guidelines (134,135).
One randomized trial (129), a number of observational studies (136-139), and many smaller mainly retrospective single-center studies have constantly shown that urgent start on PD with a break-in period of less than 2 weeks may be associated with a minor increased risk of mechanical complications but apparently no detrimental effect on patient survival, peritonitis-free survival, or PD technique survival compared with elective start on PD. In most studies, the apparent increased risk of mechanical complications was managed conservatively without the need to remove the PD catheter. Although our present knowledge is mainly based on non-randomized studies (136-139) with marked variability in study design, definition of urgent start, sample size, duration of follow-up, basic demographics of patients included, and geographical locations, the overall results are remarkably uniform.
As intraperitoneal pressure is linearly related to dwell volume (140) and is increased in the upright position, we recommend a modified PD prescription using low dwell volumes with the patient in the supine position to minimize the risk of leakage if urgent start on PD is needed. Presently, there are no convincing data to support any particular prescription for urgent start on APD or continuous ambulatory PD (CAPD) except for low-volume exchanges in the supine position. Furthermore, there are no convincing data to support the use of any particular type of PD catheter or insertion approach when urgent PD start is needed. Accordingly, each center should establish peritoneal access using their standard procedures based upon facility resources and operator expertise.
There are no RCTs comparing urgent start on PD with urgent start on hemodialysis. In the urgent setting, the choice of modality has to be balanced between the potential for increased risk of mechanical complications related to urgent start on PD and the increased risk of bloodstream infections and central venous stenosis and thrombosis known to be associated with urgent start on hemodialysis using a central venous catheter.
Complications of Peritoneal Catheters
Infectious and mechanical complications of the peritoneal catheter are the 2 most common reasons for PD failure. With early and appropriate intervention, many catheters can be saved, often without interruption of therapy. On the other hand, in the event of certain infectious complications, it is important to know when urgent removal of the catheter is essential to preserving the peritoneal membrane so patients may return to PD (134,135).
Infectious Complications and Management
We suggest that superficial cuff extrusion be managed by cuff shaving (2C)
We recommend ultrasonographic evaluation of the transmural catheter segment in cases of chronic exit-site infection or when the exit-site infection is responding slowly to treatment, especially for infections involving Staphylococcus aureus and Pseudomonas aeruginosa, and that these findings be used to direct definitive treatment (1B)
We suggest splicing a new catheter segment to the intercuff section of the existing catheter and tunneling it to a more satisfactory exit-site location where an ultrasound exam shows absence of fluid around the superficial cuff and the location of the exit site was a contributing factor to the chronic infection (2C)
We recommend unroofing/cuff shaving or simultaneous catheter replacement for clinical or ultrasonographic findings of tunnel infection with fluid around the superficial cuff and the intercuff tubing segment (1C)
We recommend catheter removal, interim hemodialysis, and staged reinsertion of the PD catheter for clinical or ultrasonographic evidence of tunnel infection with fluid around the deep cuff or concurrent peritonitis (1B)
We recommend simultaneous catheter replacement for relapsing peritonitis caused by Staphylococcal species if antibiotic therapy resolves abdominal symptoms and the peritoneal cell count is < 100/μL (1A)
Recommendations for prevention and antimicrobial treatment of catheter-related infections and peritonitis are detailed in separate ISPD guidelines (134,135). The present guidelines will focus on interventional therapy to preserve the PD access or, in the event of catheter loss, to minimize the interval before returning to PD.
Superficial Cuff Extrusion
Extrusion of the superficial Dacron cuff through the exit site usually begins as a mechanical complication caused by shape memory resiliency forces induced by bending a catheter in the subcutaneous track that has a straight intercuff tubing segment. Depending on the magnitude of these shape memory forces and the proximity of the cuff to the exit site, straightening of the tubing may cause the cuff to extrude through the exit site. If the extruding cuff is not managed, it soon becomes seeded with bacteria and predisposes the patient to exit-site infection (141). A cuff that has completely extruded still remains a reservoir of bacteria in the vicinity of the exit site. During routine exit-site care, unavoidable wetting of an extruded bacterial-laden cuff leads to constant exit-site contamination. If not completely extruded, the cuff should be gently delivered through the sinus and shaved off of the catheter with a scalpel or avulsed from the tubing with forceps. In the presence of purulent drainage, specimens for culture and Gram stain should be collected, empiric antibiotics instituted, and exit-site care adjusted to handle the degree of inflammation and drainage. Rapid stabilization of the exit site can be expected with elimination of the extruded cuff.
Chronic Exit-Site Infection
An exit-site infection becomes chronic if it persists or relapses after 2 to 3 weeks of appropriate antibiotic therapy and intensified exit-site care as outlined in the ISPD guide-lines recommendations for catheter-related infections (135). There maybe pain and tenderness at the exit site, presence of exuberant granulation tissue with associated scab and crust, and purulent or bloody discharge from the exit sinus. The epithelium within the exit sinus has usually receded but the skin around the exit site may be normal color or pale pink (142). The majority of these patients, especially when Staphylococcus aureus or Pseudomonas aeruginosa are the infecting organisms, have superficial cuff and tunnel involvement as demonstrated by ultrasound (143,144). Presumably, microbial seeding of the cuff material leads to the chronic expression of the infection. If not appropriately treated in a timely fashion, the infection will track along the catheter to the peritoneal cavity, with the development of peritonitis.
Ultrasonography is a useful tool in planning operative intervention for chronic exit-site infections through its capability of detecting superficial cuff and tunnel involvement before clinical signs of pain, tenderness, induration, erythema, and swelling appear. If the ultrasound exam shows an absence of fluid around the superficial cuff and the chronic infection is due to poor exit-site location, splicing a new catheter segment to the intercuff section of the existing catheter and routing it to a more satisfactory exit-site position is an option that does not interrupt PD therapy. Variations of the splicing procedure have been described with equally successful results (145-147); however, it would appear prudent not to cross the midline with the splice segment, reserving the opposite side of the abdomen for catheter replacement should it be required.
If ultrasonography reveals fluid around the superficial cuff, with or without fluid in the intercuff section but without deep cuff involvement or concurrent peritonitis, and the exit-site location is not a contributing cause, the chronic infection can be managed by excising the exit-site skin and extending the skin incision over the subcutaneous track until the superficial cuff is exposed. The superficial cuff is shaved, the catheter immobilized with the shaved segment external to the wound, and the incision left open to heal by secondary intention (148). Another variation of the procedure when infection is limited to the superficial cuff consists of excising the exit site and the skin overlying the subcutaneous track en bloc with the underlying tissue around the catheter segment containing the cuff to avoid spillage of infected material. The wound is closed around the clean exposed intercuff catheter segment and protected from contamination. The en bloc resected infected tissue is removed from the catheter and the cuff shaved. The catheter is immobilized to facilitate wound healing (149). These options work best when the superficial cuff is within several centimeters of the exit site. Advantages are low cost, minimal invasiveness, and no interruption of PD.
An alternative to splicing or unroofing/cuff shaving is simultaneous catheter insertion and removal. This option is indicated when the exit-site location is unsatisfactory and flow function of the existing catheter is suboptimal. The clean step, insertion of the new catheter on the opposite side of the abdomen, is performed first, followed by the dirty step, removal of the old catheter, with care to avoid cross contamination of wounds. Removal of the catheter with staged insertion of a new catheter at a later date is indicated if there is deep cuff involvement or concurrent peritonitis.
Pd-Related Peritonitis
Diagnosis and antibiotic therapy for PD-related peritonitis are covered in separate ISPD guidelines (134). Importantly, there must be a low threshold for removal of the PD catheter for peritonitis that is not responding appropriately to treatment. The goal is to preserve peritoneal membrane function. Peritonitis can cause peritoneal adhesions that may result in catheter obstruction, limit the dialyzable space, or produce loculations that cause incomplete dialysate drainage. Fibrosis of the peritoneal membrane may affect its capacity for ultrafiltration and transfer of solutes.
Most patients with PD-related peritonitis will show considerable clinical improvement within 48 to 72 hours of initiating appropriate antibiotic therapy. If patients have not shown definitive clinical improvement by 5 days, catheter removal should be performed. Immediate catheter removal is indicated for fungal peritonitis. Antimicrobial therapy should be continued for at least 2 weeks after catheter removal for refractory peritonitis (150,151). Reinsertion of the dialysis catheter can be performed as early as 2 to 3 weeks after catheter removal if resolution of peritoneal symptoms is complete (150-152), although some would recommend waiting longer for fungal peritonitis (153,154).
Relapsing Peritonitis
Simultaneous catheter insertion and removal without interruption of PD can be performed for selected cases of relapsing peritonitis. Relapsing peritonitis is defined as an episode that occurs within 4 weeks of completion of therapy of a prior episode with the same organism or 1 sterile episode (134). Relapsing peritonitis related to the peritoneal catheter is due to bacteria harbored in a biofilm covering the intraperitoneal portion of the tubing or to seeding of the peritoneum by direct extension of a deep cuff and tunnel infection. Although antibiotics may temporarily control the infection, the residual bacterial nidus within the biofilm will eventually proliferate and lead to a recrudescence of overt infection. Relapsing episodes of peritonitis related to catheter infection must be differentiated from other intraperitoneal causes, such as diverticulitis or abscess. Ultrasound evidence of deep cuff infection should be managed by catheter removal and staged reinsertion. Simultaneous catheter insertion and removal can be considered if antibiotic treatment resolves clinical signs of infection, the dialysate leukocyte count is < 100/μL, and the infecting organisms are not mycobacteria, fungi, enteric, or Pseudomonas species in origin.
Following surgical principles, the clean step, insertion of the new catheter, is performed first. The risk of seeding of the new catheter by planktonic bacteria is exceptionally low if the procedure is timed when clinical symptoms are absent and the dialysate leukocyte count is < 100/μL. Best results are seen when simultaneous catheter insertion and removal is performed for Staphylococcal species, with success rates ≥ 95% (155,156). The simultaneous replacement procedure should be carried out under perioperative antibiotic coverage (156).
Peritoneal Leakage and Management
We recommend that initiation of dialysis following catheter placement be delayed for 2 weeks when possible to minimize the risk of leaks (1B)
We recommend that acute and urgent start of PD< 2 weeks following catheter placement utilize a recumbent, low-volume, intermittent dialysis regimen, leaving the peritoneal cavity dry during ambulatory periods to minimize the risk of leak (1C)
We recommend the use of CT peritoneography or peritoneal scintigraphy to investigate suspected peritoneal boundary dialysate leaks (1A)
Peritoneal leaks, defined as any dialysate loss from the peritoneal cavity other than through the lumen of the catheter, are arbitrarily classified as early (< 30 days) or late (> 30 days), following catheter implantation and the start of PD (157). The time period in which the leak occurs may suggest its etiology. However, some peritoneal boundary leaks may occur at any time during the course of PD therapy.
Early Pericatheter Leaks
Early leaks are usually related to catheter implantation technique, the timing of PD initiation, dialysate volumes used, and the strength of abdominal wall tissues. Delay in performing the catheter insertion procedure may be advisable in patients with the recent onset of a persistent cough to avoid the risk of pericatheter leak. When PD is initiated, subcutaneous leakage may occur at the catheter insertion site and manifest as fluid appearing through the incision or at the exit site. Questionable leaks can be verified by a positive glucose test strip indicating high glucose concentration of the seeping fluid.
The incidence of pericatheter leaks is higher with a midline approach to catheter placement than with a paramedian site (37,39). Pericatheter leaks may occur as a consequence of early institution of PD. Delaying start of dialysis for 2 weeks following catheter placement minimizes developing a leak (157-159). Temporarily discontinuing dialysis for 1 to 3 weeks usually results in spontaneous cessation of an early leak. Dramatic early leaks may indicate purse string suture failure or technical error in wound repair and demands immediate exploration. Leakage through the exit site or insertion incision is prone to tunnel infection and peritonitis. Prophylactic antibiotic therapy should be considered (159, 160). Persistent leaks warrant catheter replacement.
Late Pericatheter Leaks
Pericatheter hernias, pseudohernias, or occult tunnel infections with separation of the cuffs from the surrounding tissues are pathways for late leakage around the catheter (157,159–161). A pseudohernia is a dialysate-filled peritoneal sac that extends alongside the catheter into the subcutaneous tissues, suggesting a hernia bulge at the catheter insertion site. Pericatheter hernias and pseudohernias are best managed by simultaneous catheter replacement and repair of the fascial defect. Separation of infected catheter cuffs from adjacent tissues allows free egress of dialysate. Tunnel infections can be occult without signs of exit-site infection or active peritonitis. Imaging studies (ultrasound or CT scan) help differentiate between pericatheter hernia or pseudohernia and occult tunnel infection. Dialysate leakage resulting from a tunnel infection requires catheter removal and interim hemodialysis.
Physical strain can be either an early or late cause of pericatheter leakage. Strenuous physical activities can force dialysate through the abdominal wall around the catheter. Abdominal wall weakness, obesity, steroids, intraperitoneal pressure, and large dialysate volumes increase the risk of leakage from physical strain (157,159). The leak is managed by temporary suspension of dialysis or by supine low-volume dialysate exchanges with a dry peritoneal cavity during ambulatory periods. Lifting limitations of 7 to 10 kg are recommended for prevention, but weight and activity level are flexible based upon previous physical condition. The risk of leak can be minimized by performing sports and exercise activities with a dry abdomen (162).
Other Peritoneal Boundary Leaks
Leakage from previously undiagnosed hernias may present as obvious bulges, genital swelling, abdominal wall edema, weight gain, or apparent ultrafiltration failure (163,164). If not revealed on physical exam, occult hernias with leaks maybe identified by contrast CT peritoneography or technetium-99m peritoneal scintigraphy (164,165). Repair techniques must incorporate watertight closures to allow patients to continue PD postoperatively without interim hemodialysis. Risk of leak is minimized by using a supine, low-volume, intermittent PD regimen for 2 weeks following repair, leaving the peritoneal cavity dry during ambulatory periods (94).
Pleuroperitoneal connection with leakage of dialysate into the pleural space occurs in 1% - 2% of PD patients. Dyspnea is frequently the first clinical sign of leak; however, patients may present only with pleuritic pain or a decrease in ultrafiltration. The pleuroperitoneal leak is usually unilateral, most commonly on the right side, and occurs during the first year of PD. Diagnosis is confirmed by thoracentesis, with recovery of fluid low in protein and high in glucose concentration. Alternatively, the diagnosis can be established by contrast CT peritoneography or technetium-99m peritoneal scintigraphy. Conservative management (peritoneal rest, low-volume dialysis) is rarely successful. Thoracoscopic pleurodesis with talc poudrage or mechanical rub produces 85% - 100% success rate. Interim hemodialysis is required for approximately 3 weeks following the procedure (166-168).
Flow Dysfunction and Management
Diagnostic studies and treatment for catheter flow dysfunction should progress in a logical order from conservative or noninvasive approaches to more aggressive interventions (not graded)
Choice of intervention for catheter flow dysfunction (radio-logical manipulation, laparoscopic rescue, or simultaneous catheter replacement) should be based upon patient factors, facility resources, and operator expertise (not graded)
Catheter flow dysfunction is usually manifested as outflow failure; therefore, the volume of drained dialysate is substantially less than the inflow volume. The most common cause of outflow dysfunction is constipation (31). Distended rectosigmoid colon may block the catheter side holes or displace the catheter tip into a position of poor drainage function. Extrinsic bladder compression on the catheter due to urinary retention occurs less frequently (169). Mechanical kinking of the catheter tubing or an intraluminal fibrin clot is usually accompanied by 2-way obstruction. A flat-plate radiograph of the abdomen is often helpful in identifying a fecal-filled rectosigmoid colon, catheter displacement, or a kink in the catheter tubing.
Constipation
Constipation is treated with oral osmotic agents, e.g., lactulose, sorbitol, or polyethylene glycol solution. Stimulant laxatives such as bisacodyl and saline enemas are reserved for refractory cases since chemical and mechanical irritation of the colonic mucosa has been associated with transmural migration of bacteria and development of peritonitis (32).
Bladder Distention
Causes of urinary retention with bladder distention include bladder outlet obstruction, detrusor underactivity, and neurogenic bladder. The degree of dysfunction is not only influenced by bladder size, but the depth of the catheter tip in the pelvis and any coexisting rectal distention. Bladder scan or post-void catheterization should be performed for symptoms of urinary retention. Most urologists consider post-void urine volumes > 50 to 100 mL to be abnormal. Chronic urinary retention is often defined as a post-void residual > 300 mL (170,171).
Catheter Kinks
Catheter tubing kinks occur almost exclusively in the transmural segment and represent technical errors made in catheter insertion. Occasionally, the kink can be difficult to demonstrate and it may not always be apparent on a flat-plate radiograph. Lateral films of the abdomen with the patient supine and sitting (lateral chest X ray for presternal catheters) with arms down at the side may be necessary to identify a tubing kink. ACT scan can also be used to recognize a kink in the catheter tubing. The location of the kink will dictate whether revision or catheter replacement is required.
Intraluminal Debris
If the X ray eliminates tubing kinks or displacement, bladder distention is excluded, and flow function is not restored with correction of constipation, then fibrinolytic therapy with tissue plasminogen activator (tPA) may be attempted to clear presumed intraluminal fibrin or blood clots. Failure to dislodge intraluminal debris by brisk irrigation of the catheter with saline is followed by instillation of tPA. If catheter obstruction is due to a fibrin or blood clot, recovery of flow function with tPA has been reported at nearly 100% (172-174). Because of cost considerations, the dose of tPA (used in a dilution of 1 mg/mL) has been based upon the calculated volume of the catheter assembly; however, no adverse consequences have been documented for catheter overfill or repeat administration (173,174).
Catheter Migration and Tissue Attachment
When extrinsic compression of the catheter tip by distended pelvic structures and intraluminal blockage by fibrin have been excluded, the flow failure can be attributed to either catheter tip migration to a location of poor drainage function or obstruction by adherent intraperitoneal tissues. Both conditions may have the radiologic appearance of a catheter tip displaced from the pelvis, while the latter can also occur with normal pelvic position. Options for restoring catheter flow function include radiologically-guided manipulation, laparoscopically-directed interventions, and simultaneous catheter replacement.
Radiological Manipulation
Fluoroscopic guidewire, stiff rod, and aluminum bar manipulations have been used to resolve catheter tip migration and extraluminal and intraluminal obstructions (175-180). The procedures are minimally invasive, do not require anesthesia beyond the possible use of conscious sedation, are low in cost, and allow PD to be resumed immediately if technically successful. However, multiple sessions are often required to obtain long-term clinical success. The inability to definitively address the underlying cause of the flow dysfunction accounts for initial technical failures and recurrences. Clinical success has been described as 46% - 75% of cases in published reports (175-180). Radiological manipulation is difficult or impossible to perform through catheters with a preformed arc bend or through long presternal catheters. When laparoscopic backup is not available, technical failures are often managed by catheter replacement. When considering approaches for catheter salvage, it is important to recognize that patients often become frustrated with multiple interventions and interruption of therapy and elect to transfer permanently to hemodialysis.
Laparoscopic Rescue
Laparoscopy has the advantage of allowing identification of the underlying condition producing catheter flow dysfunction, permitting diagnosis-specific management. Laparoscopically enabled interventions have produced long-term clinical success in 63% - 100% of cases (43,61,87,181–184). For this reason, laparoscopic rescue is often considered the next step in the management sequence for catheter flow dysfunction after the diagnosis of constipation, bladder distention, and fibrin plug have been excluded. Although laparoscopy is a minimally invasive procedure that permits patients to immediately resume PD, it does require general anesthesia and procedural costs are higher compared with radiological interventions. However, the high success rate for laparoscopic rescue minimizes the need for multiple procedures and may reduce patient dropout.
Recurrence of catheter tip migration from shape memory resiliency forces is prevented with a suture sling through the lower abdominal wall (67). As was discussed in the section on laparoscopic catheter insertion, the use of pelvic anchoring sutures is discouraged because of suture erosion with remigration of the catheter or difficulty in removing the catheter from a firmly holding stitch. Depending on the intraperitoneal structure involved, extraluminal obstruction is treated by omentolysis with omentopexy, adhesiolysis, epiploectomy, salpingectomy, or appendectomy (43,61,65,87,181–184). Fibrin casts of the catheter are cleared by externalizing the catheter tip through a laparoscopic port site and stripping the clot from the tubing. At the conclusion of the procedure, all port sites, regardless of size, are sutured watertight so PD can be restarted immediately using the supine, low-volume, intermittent dialysis protocol.
Simultaneous Catheter Replacement for Flow Dysfunction
Simultaneous replacement of the catheter is the least favor-able option for the management of catheter flow complications. The replacement catheter is subject to all of the potential complications of a new catheter, e.g., leaks, bleeding, infection, and obstruction. Especially in the case of unsuccessful radiological manipulation, the new catheter may be exposed to the same underlying condition that caused the first catheter to fail. However, there may be no other option but simultaneous catheter replacement if backup laparoscopic intervention is not available or the patient is not a candidate for general anesthesia. Regardless of the salvage approach on hand, if a significant technical error is recognized in the placement of the original catheter, the best choice may be to replace it (156).
External Catheter Damage
Damage to the external catheter tubing may result from sharp instrument cuts or punctures, fracture from crushing clamps, catheter adapter tears, or chemical destruction of the catheter material from antibiotic ointments or organic solvents. External splicing repair by the PD nursing staff using commercially available repair kits is possible if at least 2 cm of tubing is present beyond the exit site (185). Catheter damage with leak is considered a contaminating event, and investigation for peritonitis is required and prophylactic antibiotics indicated. Internal splicing repair to the intercuff segment can be considered if the catheter tubing is too short for external repair, flow function has been satisfactory, and there is no concurrent peritonitis (165). Catheter replacement is the alternative if preexisting catheter flow dysfunction is present.
Catheter Removal
Catheters may be removed by either open surgical dissection or “pull technique” (not graded)
We suggest that open surgical dissection removal of the Dacron cuffs intact with the catheter be performed when removal is for a tunnel infection or catheter infection-related peritonitis, 2-piece extended catheters joined with a titanium connector, or devices equipped with a Dacron flange and silicone bead fixation components (2C)
We suggest that the “pull technique” is best suited when catheter removal is performed for noninfectious indications where retaining the Dacron cuffs in the tissues is of minimal risk (2C)
Indications for removal of the PD catheter include catheter-related infection, peritonitis, flow dysfunction, pericatheter leak and hernia, renal transplantation, improved renal function, dialysis inadequacy, and elective transfer to hemodialysis.
Catheter removal is ordinarily performed by surgical dissection in the operating room or a suitable procedure room under local or general anesthesia (186). An incision is made through the previous insertion site scar and the Dacron cuffs are dissected free of the tissues, permitting complete removal of the catheter device. The operator must have the skill and resources to manage hemorrhage from the inferior epigastric vessels in the event of injury during catheter removal. Fascial defects will require suture repair to prevent an abdominal wall hernia. When catheter removal includes drainage of a tunnel abscess or debridement of exuberant granulation tissue at the exit site, the wound is left open to heal by secondary intention. Wounds of catheters removed for peritonitis can usually be irrigated with saline and closed primarily.
Although employed much less frequently, catheter removal can be performed by the “pull technique” (187,188). The external part of the catheter tubing is grasped and, with steady gentle traction, the catheter is forcefully pulled from the abdominal wall. The Dacron cuffs shear off of the tubing during extraction and are retained in the tissues. The “pull technique” is commonly performed in the office or procedure room with or without local anesthesia or sedation. Infection of the retained cuffs necessitating later excision, almost always the superficial cuff, has been reported in 2.5% to 3.2% of cases (187,188). Therefore, when the procedure is being performed for chronic exit-site infection with superficial cuff involvement, it is advisable to open up the exit site sufficiently to allow removal of the superficial cuff during the pullout (188). In cases of tunnel abscess necessitating drainage and debridement or catheter infection-related peritonitis where the deep cuff is involved with the transmural propagation of the infection, surgical removal of the catheter and cuffs is preferred (189).
No tubing breaks have been reported with the “pull technique” during steady smooth traction on the catheter. The technique is not recommended for 2-piece extended catheters joined by a titanium connector due to separation of the tubing at the connector junction during attempted withdrawal. In addition, the “pull technique” is not appropriate for catheters equipped with a bead and flange below the deep cuff, e.g., the Missouri or Toronto-Western catheters (187,188), or when the catheter has been sutured in the pelvis. While no RCTs or cohort studies have compared the “pull technique” with surgical dissection, complications of bleeding and wound infection appear to be slightly higher with surgical dissection (187,188,190).
Secondary Embedding
We suggest secondary embedding of the PD catheter when renal function has improved enough to stop dialysis but recovery is not expected to be long-term, conditional to previously normal catheter flow function (2D)
On occasion, catheter removal is performed because patients regain sufficient renal function to discontinue dialysis, but recovery is not expected to be permanent. An alternative to removing the catheter is secondary embedding (191,192). The inconvenience and cost of catheter maintenance can be eliminated for the interim by secondary embedding while still preserving a readily available peritoneal access that can be immediately employed to its fullest extent without the complications of new catheter placement, e.g., flow dysfunction and pericatheter leak. The prerequisite requirement for secondary embedding is that the catheter has normal flow function.
The procedure is performed similar to catheter splicing, discussed under the section on complications, except that the spliced external segment is embedded. An incision is made through the previous insertion-site scar to expose the intercuff segment of the catheter. The catheter is divided in the intercuff section and joined with a double-barbed titanium connector to a new catheter that has been trimmed to form the splicing segment. The new external segment is tunneled into a subcutaneous bed as described in the section on embedded catheters. After the wounds are closed and protected, the remaining external segment of the former catheter is removed and the old exit-site wound is excised and closed. When renal function has declined to the point of needing to reinstitute treatment, the embedded catheter segment is externalized for immediate full-volume dialysis therapy.
Audit of Pd Access Procedures
We recommend an audit of catheter insertion outcomes on at least an annual basis as part of a multidisciplinary meeting of the PD team, including attendance of access operators when feasible (1B)
We suggest clinical goals specific for the PD access procedure include (2C):
Catheter patency at 12 months of > 95% for advanced laparoscopic placement and > 80% for all other catheter insertion methods
Exit-site/tunnel infection within 30 days of catheter insertion: < 5%
Peritonitis within 30 days of catheter insertion: < 5%
Visceral injury (bowel, bladder, solid organ): < 1%
Significant hemorrhage requiring transfusion or surgical intervention: < 1%
We suggest that incidences of pericatheter leaks within 30 days of catheter insertion be recorded separately for early PD starts (< 14 days) and late starts (≥ 14 days) (not graded)
There is substantial evidence that audit improves patient care (193,194). Audit is just one step in the continuous quality improvement approach to improving outcomes. Easily audit-able performance measures that are specific for the outcome of the PD access procedure include long-term catheter patency and occurrence of procedure-related complications, pericatheter leakage, infection, visceral injury, and hemorrhage. These measures are compared with an external standard or benchmark established from the literature to motivate improvement toward these specific goals.
Catheter patency is defined as the percentage or probability of catheter survival at 12 months following placement; therefore, the catheter has not been removed, replaced, or required some type of intervention (surgical or radiological) because of flow dysfunction or irremediable drain pain. Monitoring of catheter patency for embedded catheters begins at the time of externalization. Other causes of catheter loss are censored, including death, transplant, infection, pericatheter leakage, or transfers to hemodialysis because of inadequate dialysis, psychosocial reasons, or medical problems. Most studies describing catheter survival combine the losses from infections with mechanical complications and often suggest no differences in the 12-month postoperative outcomes among conventional insertion methods (open dissection, percutaneous with/without image guidance, peritoneoscopic, and basic laparoscopic) (44,56,73,80,195–197). However, the effect of infectious complications on catheter survival beyond 30 days is more likely due to causes unrelated to the insertion procedure per se. The few studies that provide data according to the above definition for 12-month catheter patency support a benchmark value of > 80% patency for conventional methods and > 95% for advanced laparoscopic procedures (43,57,61,62).
The definition of surgical-site infection provided by the United States Center for Disease Control and Prevention (CDC) is an infection that occurs within 30 days of the procedure and appears to be related to the procedure (198). To be consistent with the CDC's classification scheme, the performance measure for exit-site/tunnel infection is extended from the 2-week interval in previous PD access guidelines to 30 days. The goal for exit-site/tunnel infection within 30 days of catheter insertion is an occurrence rate < 5% (61,197).
Procedure-related peritonitis following catheter placement includes intraoperative break in sterile technique, extension of surgical site infection to the peritoneal cavity, pericatheter leak, and contamination from connections during postoperative catheter flushes. A reasonable goal for peritonitis within 30 days following catheter insertion is < 5% (52,55,61,197). Perforation of bowel or bladder or solid organ injury during catheter insertion should be a rare event, occurring < 1% (55,57,61,197,199–201). Hemorrhage necessitating blood transfusion and/or subsequent surgical intervention to control bleeding may occur during catheter placement. Care to prevent injury to the inferior epigastric vessels, avoiding forceful blind instrumentation in the peritoneal cavity, and meticulous detail to hemostasis should limit this complication to < 1% (57,61,197).
Pericatheter leakage during the first 30 days following catheter placement is often related to operator technique and constitutes an important performance measure to audit; however, there are a number of other factors not related to the insertion procedure that contribute to the wide range of reported incidence of this complication (55,56,129,136,197,202). Abdominal wall weakness due to previous abdominal surgeries, multiple pregnancies, long-term therapy with steroids, and obesity can cause leakage (159). The time interval after catheter placement before initiation of dialysis and the exchange volumes employed contribute significantly to the risk of pericatheter leakage (129,139). Peritoneal dialysis centers that have urgent-start programs may experience higher leak rates than those units that allow an extended period of wound healing before commencing dialysis. Setting audit standards for leak rates is further complicated by published reports that group pericatheter leakage with leaks from other anatomical sites, e.g., abdominal wall hernias and pleuroperitoneal fistulae, that have nothing to do with the catheter implantation method (139). We suggest that incidences of pericatheter leaks within 30 days of catheter insertion are tracked separately for early PD starts (< 14 days) and late starts (≥ 14 days), thereby establishing a baseline that reflects the clinical practice in that center.
The PD team should set up a structured observation process to capture performance measures at regular intervals. How often the data are analyzed and reviewed to determine practice status and plans for improvement should be guided by the magnitude of the disparity between current performance and established goals, but a meeting of the PD team ought to be conducted at least annually. While attendance of PD access providers at a multidisciplinary PD team meeting is ideal, communities exist where numerous operators supply services to a center; hence, expecting a satisfactory turnout for such a meeting may be unattainable. An effective alternative is to circulate a community report card among the PD access providers describing the audit outcomes for each individual according to a personal identification number. Providers are able to assess their performance relative to others in the community, hopefully motivating improved service from the underperformers. The audit process also permits the director of dialysis services to constructively interact with those producing suboptimal outcomes to suggest training opportunities, e.g., the ISPD's PD University programs for surgeons (www. pdusurgeons.com) and interventionalists (www.pduinir.com) or other comparable instruction.
Future Research
Clinical trials are sought to compare image-guided with non-image guided percutaneous catheter insertion and advanced laparoscopic with basic laparoscopic and/or other conventional catheter implantation methods. The value of purse string sutures in preventing pericatheter leakage should be formally studied. The safety of intraperitoneal prosthetic mesh hernia repair in PD patients requires investigation. Studies are needed to determine the optimal duration of antibiotic coverage after simultaneous catheter replacement for infectious complications. Criteria defining PD candidacy in patients with diverticular disease deserve further examination. The usefulness of the catheter embedding procedure as a strategy to increase PD penetration and lower central venous catheter usage should be explored by comparing a patient group undergoing early catheter placement with embedment with a group waiting for conventional timing of catheter insertion close to anticipated need. Investigations testing the efficacy of postoperative catheter flushes should be undertaken. Further studies are also needed to assess the effectiveness and safety of various salvage procedures for catheter-related infections.
Footnotes
JHC receives consultancy fees from Baxter Healthcare and Merit Medical and speakers’ honoraria from Baxter Healthcare, Merit Medical, Fresenius Medical Care, DaVita, and Medtronic. AEF has received speakers' fees from Baxter Healthcare. MW receives speakers’ honoraria from Baxter Healthcare and Fresenius Medical Care. AAA serves as a consultant for Baxter Healthcare, Boston Scientific, Bard Peripheral Vascular, Sirtex Medical, W. L. Gore, and Abbott Medical. BLG has received speakers' fees from Baxter Healthcare. VRB receives a Baxter CEC Grant. FJMFD serves as a consultant for Sandoz and has received speakers' honoraria from Astellas, TEVA, Chiesi, and Sandoz. The other authors have no financial conflicts of interest to declare.
