Abstract
Peritonitis is the leading cause of transfer from peritoneal dialysis (PD) to hemodialysis (HD). It is also the leading cause of hospitalization of PD patients. The usual treatment of peritonitis for automated PD (APD) patients consists of antibiotics given once daily in the long dwell. However, the once-daily antibiotic dosing recommendations are based primarily on studies with continuous ambulatory PD (CAPD) regimens. Published studies on antibiotic dosing in APD are very limited. We will review the scant literature on this topic. It is possible that extrapolating once-daily dosing from CAPD to APD may lead to underdosing. There is a need for further pharmacokinetic studies of antibiotic dosing in APD.
Even as peritonitis rates have decreased over time, infection is still the leading cause of transfer from PD to hemodialysis (HD) in the United States (4,5). The 2017 United States Renal Data System (USRDS) reveals that all-cause hospital admissions were slightly higher for HD than PD (1.73 vs 1.69 admissions/pt year), but that admissions for infection are more common in PD vs HD (0.56 vs 0.44 episodes/pt year) and infection is the leading cause of hospital admission for PD patients (6). Infections in PD patients account for one third of the admissions and are higher than cardiovascular (CV) causes, in contrast to in-center HD patients. This makes the prevention and treatment of peritonitis a top priority among PD practitioners.
The empiric treatment recommendations for peritonitis are: gram-positive coverage with first-generation cephalosporin or vancomycin; gram-negative coverage with third-generation cephalosporin or an aminoglycoside (7). Intraperitoneal (IP) dosing is preferred over intravenous dosing of antibiotics, unless signs and symptoms of systemic sepsis are present (7,8). Unfortunately, data regarding antibiotic dosing for PD peritonitis are extremely limited, especially for APD (8). Li
First Generation Cephalosporins, Specifically Cefazolin
There are very limited papers studying dosing of first-generation cephalosporins in APD as shown in Table 1. Perhaps because of this, the most recent International Society for Peritoneal Dialysis (ISPD) guidelines (7) eliminated the table for APD antibiotics and only showed dosing for CAPD as opposed to the guidelines in 2010 (13–15). The most recent guidelines suggest that continuous dosing of cephalosporins in APD may be preferable (7). This recommendation may have been made due to the paucity of pharmacokinetic data available on intermittent dosing of first-generation cephalosporins in APD. Tosukhowong
First Generation Cephalosporins: Cefazolin
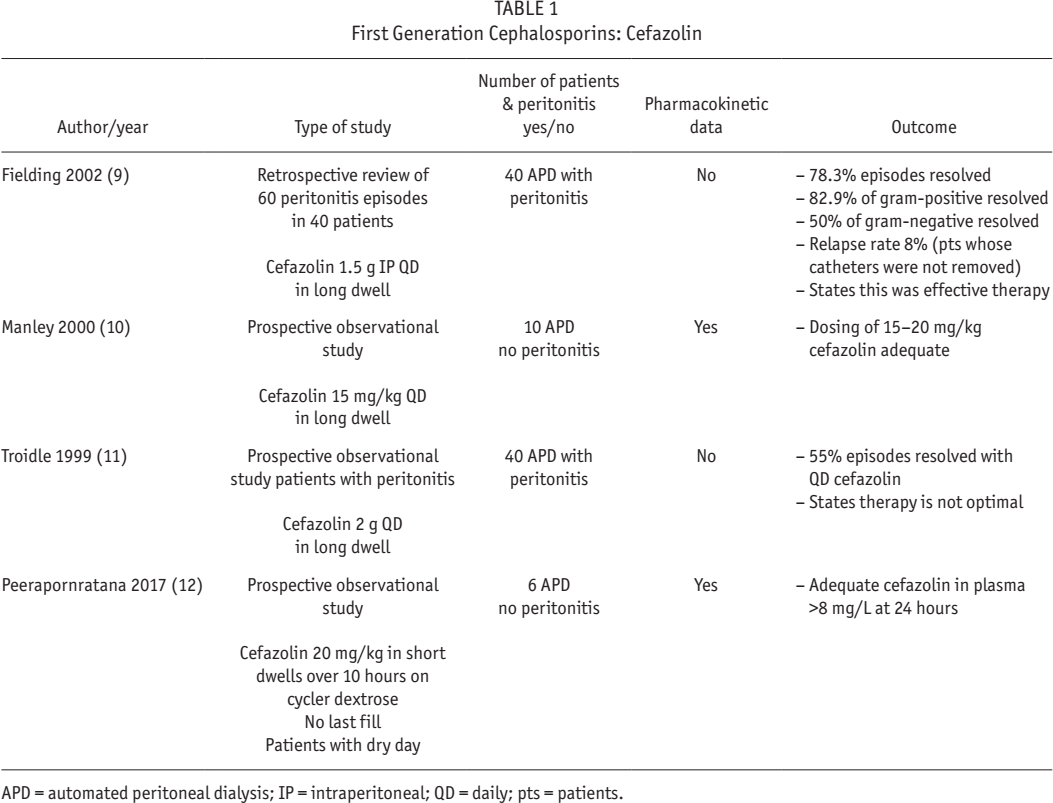
APD = automated peritoneal dialysis; IP = intraperitoneal; QD = daily; pts = patients.
It may be that with a single dose given in the long dwell of APD, dialysate cephalosporin levels are low during the short nighttime cycles. Cephalosporins are bactericidal when the concentration exceeds the MIC of bacteria, usually 1 – 8 mg/L. Concentration of the antibiotic at the site of infection is most relevant for killing bacteria (17). Whether this has a clinical impact on relapsing/repeat peritonitis and biofilm formation is unclear. Biofilm may be the cause of both relapsing and repeat peritonitis (7). Peritoneal dialysis catheters have been shown to be colonized by bacterial biofilms and may lead to recurrent infections (18–20). Biofilms have an innate lack of antibiotic susceptibility compared with the planktonic form of the organism (21). The MIC is the concentration of an antibiotic to eradicate the planktonic form of the bacteria (21). However, the MIC concentration is ineffective on organisms in their biofilm state (21). Eradication of the organism in the biofilm state may take 100 – 1,000 times the concentration of the antibiotic MIC level to be effective (21). The minimum biofilm eradication concentration (MBEC) is defined as the minimum concentration of an antimicrobial that eradicates biofilm (22). Girard has proposed testing for MBEC levels when patients have a longstanding or recurrent peritonitis (20). This requires further investigation.
Manley
Vancomycin
Vancomycin is routinely given intermittently intraperitoneally (IP) for peritonitis for APD and CAPD. This is a very convenient approach and is usually dosed every 3 – 7 days. However, Manley
Vancomycin
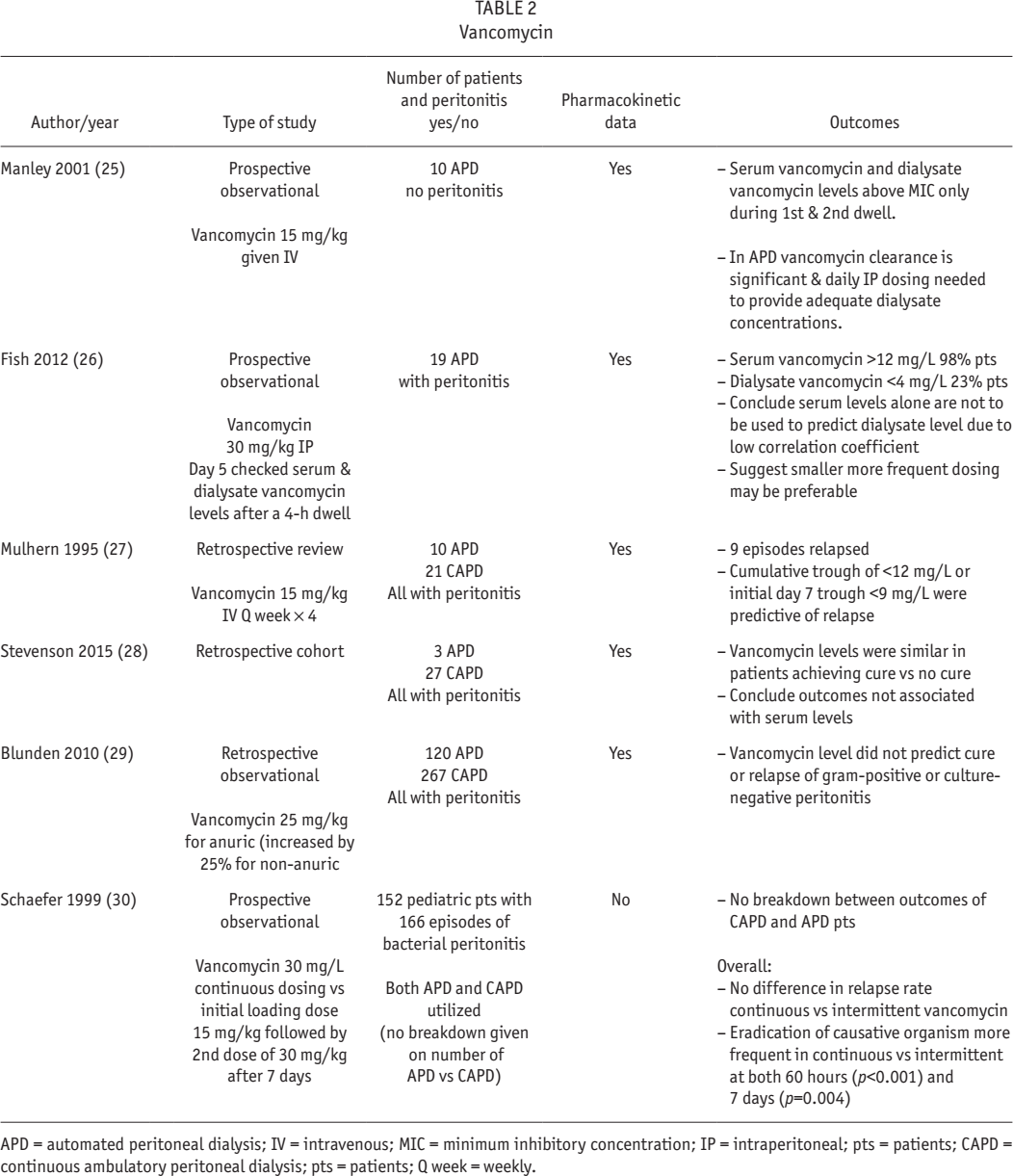
APD = automated peritoneal dialysis; IV = intravenous; MIC = minimum inhibitory concentration; IP = intraperitoneal; pts = patients; CAPD = continuous ambulatory peritoneal dialysis; pts = patients; Q week = weekly.
Mulhern demonstrated that patients who relapsed had lower serum vancomycin levels (7.8 ± 0.6 mg/L during relapse vs 13.7 ± 0.9mg/L during relapse-free episodes
A recent paper looked retrospectively at a group of 35 Canadian patients with 58 episodes of coagulase-negative
Vancomycin-resistant
Third Generation Cephalosporins, Specifically Ceftazidime
Table 3 shows the limited data on the use of third- generation cephlosporins in APD. Of the 4 papers listed, 1 was simulated data (Monte Carlo) (33). The Monte Carlo analysis demonstrated the inability of intermittent dosing with ceftazidime to achieve an IP antibiotic level of > 5 x MIC. The levels were < 5 x MIC for 75% – 100% of the time. The same was true with intermittent dosing of cefazolin (33).
Third-Generation Cephalosporins: Ceftazidime
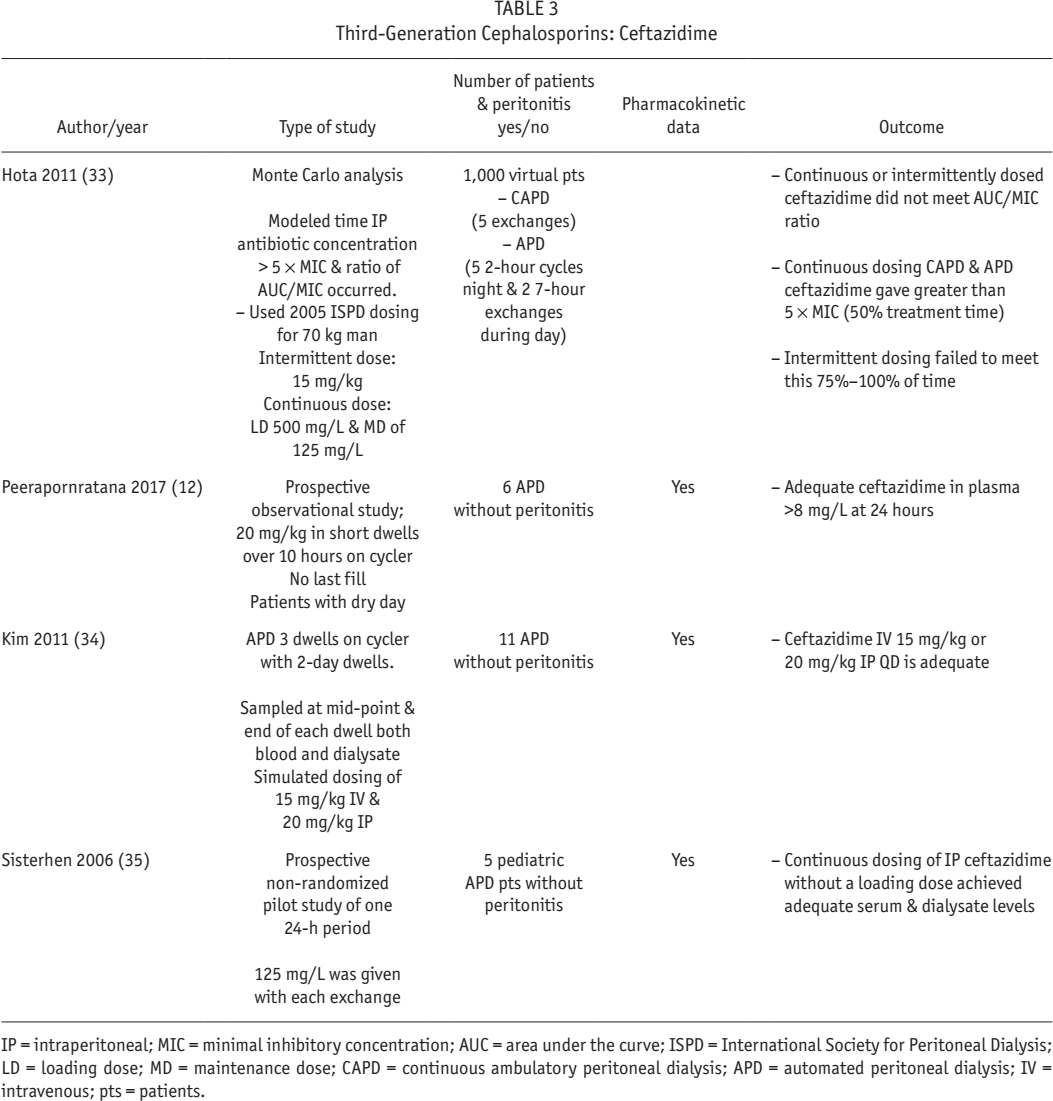
IP = intraperitoneal; MIC = minimal inhibitory concentration; AUC = area under the curve; ISPD = International Society for Peritoneal Dialysis; LD = loading dose; MD = maintenance dose; CAPD = continuous ambulatory peritoneal dialysis; APD = automated peritoneal dialysis; IV = intravenous; pts = patients.
Third generation antibiotics are almost always given in the long dwell daily. The guidelines recommend an intermittent once-daily dose (7). However, this daily dose was studied in CAPD but is widely extrapolated to APD. Very little data support this approach. More studies are needed.
Aminoglycosides, Specifically Gentamicin and Tobramycin
Aminoglycosides initially bind to the outside of the bacteria in a concentration-dependent manner, causing damage to the membrane and possible cell death. The antibiotic is then taken into the cell, where it attaches to ribosomes; this impairs the reading of messenger ribonucleic acid and kills the bacterium. This second bactericidal phase is not concentration-dependent. It is known as the post-antibiotic effect (PAE). Because of the PAE, aminoglycosides are suited for intermittent dosing (36).
Intermittent dosing of aminoglycosides may be more appropriate than continual dosing due to the phenomenon of adaptive resistance (AR). Adaptive resistance occurs in
If used for more than a brief period, vestibular and ototoxicity may become an issue. Aminoglycosides have different levels of toxicity in the order of gentamicin > tobramycin > netilmicin (39). Gentamicin and tobramycin can be toxic to both the cochlea (hearing loss) and the vestibular apparatus (ataxia, vertigo, and oscillopsia), and the damage may be acute or chronic (40). Both ototoxicity and vestibular toxicity have been reported in PD with the use of gentamicin, with some data indicating cumulative effect with repeated courses (41 42 43-44). Empiric dosing as initial treatment of peritonitis is likely safe but long-term use is more problematic. The 2016 ISPD guidelines provide intermittent and continuous dosing recommendations for both gentamicin and tobramycin (7). The studies cited in the 2016 guidelines were all done on CAPD patients (7). In the current literature, studies on APD dosing are scant as shown in Table 4.
Aminoglycosides: Gentamicin and Tobramycin
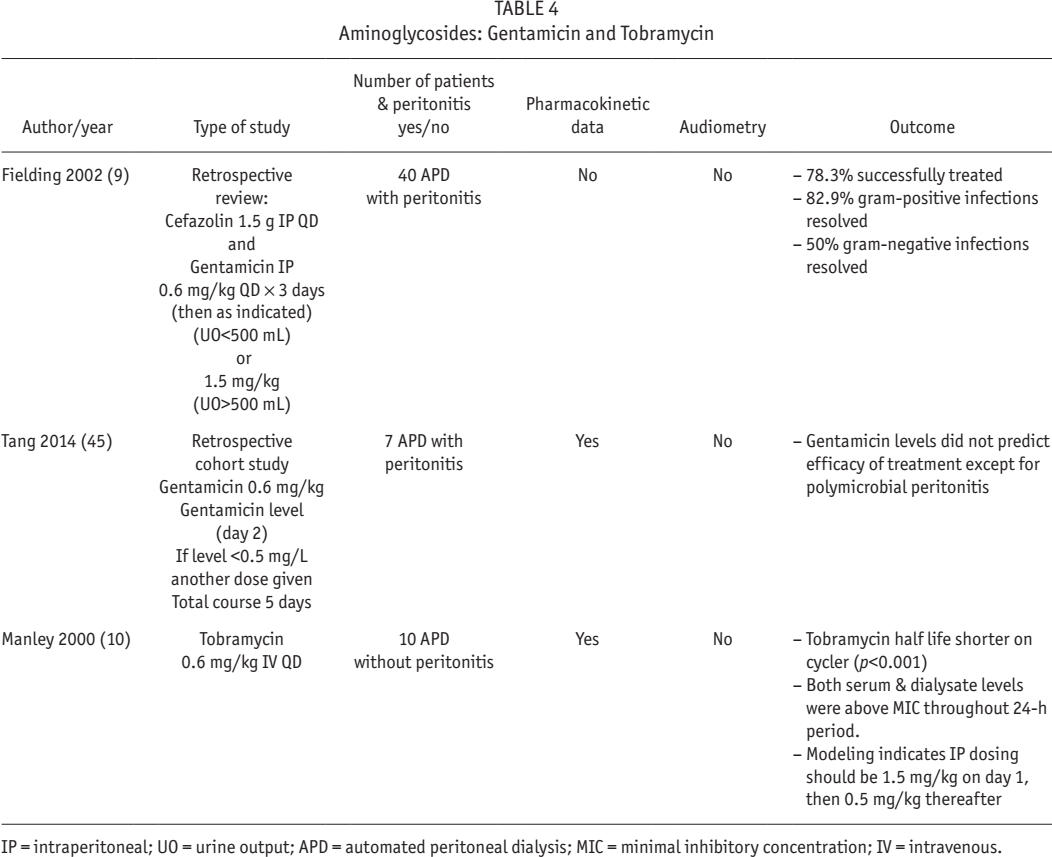
IP = intraperitoneal; UO = urine output; APD = automated peritoneal dialysis; MIC = minimal inhibitory concentration; IV = intravenous.
Conclusion and Recommendations
Few data are available on the pharmacokinetics of many commonly used antibiotics when given intraperitoneally in APD, yet this is the form of dialysis that most patients utilize in the western world. There are many areas where more research is needed. Pharmacokinetic and efficacy data are needed on continuous vs intermittent dosing, increased dosing for residual renal failure, effect of membrane transport status on dosing, and particularly dosing in APD vs CAPD.
Research is also needed to evaluate drug pharmacokinetics during peritonitis. A paper by Imada in 1988 demonstrated that CAPD patients with peritonitis who received IP doses of cefotaxime or cephalothin had lower levels of antibiotic in the dialysate than those who did not have peritonitis (46). The serum levels of cefotaxime were higher in patients with peritonitis than those without peritonitis. The serum level of cephalothin was not different between the 2 groups (46). The authors went on to say that high concentrations of antibiotic in the peritoneum can only be maintained by adding antibiotic to each CAPD exchange (46). Since peritoneal inflammation affects IP drug pharmacokinetics and absorption is increased into the blood, studies must also be done at different stages of peritonitis as the inflammation is resolving.
Also needed is more research into MIC vs MBEC to potentially reduce relapsing and repeat peritonitis. We urge further research on these areas to enhance treatment of peritonitis and improve outcomes. Nephrologists, nurses, and pharmacists should collaborate in designing and carrying out these studies. Both the ISPD and industry should help with funding of these studies.
In the absence of more data on dosing for APD, authors should carefully monitor levels of vancomycin, ensuring that the trough levels stay at 15 μg/mL as a minimum. Careful assessment of response to the antibiotics by repeated measurement of effluent cell count is useful in following the course of therapy of patients on APD in particular. In some circumstances, conversion from APD to CAPD may be ideal to ensure adequate delivery of antibiotics, but this is often impractical. Another option for peritonitis that is not resolving quickly may be to convert from intermittent dosing of antibiotics to continuous dosing while remaining on APD.
Footnotes
AM is an employee of Baxter Healthcare and BP has no conflicts of interest to declare.
