Abstract
Safety of parenteral iron therapy is critical and has been demonstrated in several studies, but concerns persist on safety. We performed a retrospective single-center study investigating the safety and efficacy of parenteral iron administration using 2 iron preparations—Monofer and Cosmofer (Pharmacosmos A/S, Holbaek, Denmark)—in patients with chronic kidney disease (CKD), on peritoneal dialysis (PD) and non-dialysis. A database of CKD patients receiving intravenous (IV) iron was analyzed. Side effects were recorded during infusion, post-infusion, and after 48 hours. In a population of CKD patients (non-dialysis and PD), IV iron is safe with few major adverse effects for these 2 IV iron preparations studied with similar dosing schedules. These data provide reassurance on the relative short-term safety of IV iron preparations regarding acute infusion-related hypersensitivity reactions.
Correction of anemia in CKD patients using IV iron improves quality of life (3). However, it is associated with potential safety concerns, including short-term effects, such as acute hypersensitivity and anaphylactic and labile iron reactions, while longer term, there is the potential for inducing iron overload, oxidative stress, and increased infection risk (4). Severe acute hypersensitivity reactions are of most concern, but true anaphylaxis may be overestimated.
We aimed primarily to clarify the short-term safety and nature of adverse reactions related to Monofer (iron isomaltoside 1000) and Cosmofer (low-molecular-weight iron dextran) (Pharmacosmos A/S, Holbaek, Denmark) in CKD and peritoneal dialysis (PD) patients. Secondary outcomes included efficacy of iron therapy and impact on phosphate levels.
Methods
This retrospective, observational, single-center study investigated the safety of parenteral iron administration using Monofer and Cosmofer therapy in CKD and PD patients who received any amount of IV iron according to local protocol (ferritin < 200 μg/L and/or transferrin saturation < 20%). In our cohort of patients, anyone requiring intervention due to a medical problem soon after starting the IV iron and up to 48 hours later were documented as having an adverse reaction.
Data were obtained from a comprehensive database of patients receiving iron infusions since 2008, which recorded patients’ demographics, quantity of IV iron administered, hemoglobin (Hb), measures of iron status, and clinical observations, including blood pressure (BP), pulse, oxygen saturations (pre- and post-iron administration), any side effects during or after IV iron administration, and a follow-up discussion 48 hours later via telephone to record “late” adverse effects.
The study was approved by the audit department, as part of service evaluation and improvement in the implementation and assessment of iron therapy. We adhered to the principles of the declaration of Helsinki, International Council of Harmonisation, and good governance.
Descriptive statistics detail the population and their demographics (Table 1). Continuous variables were summarized as mean values, standard error of mean (SEM), or ranges. Categorical variables (adverse reactions) were recorded as frequencies with percentages. Changes between Hb and ferritin pre- and post-Cosmofer and Monofer were compared using the 1-way ANOVA test. A
Demographic Data
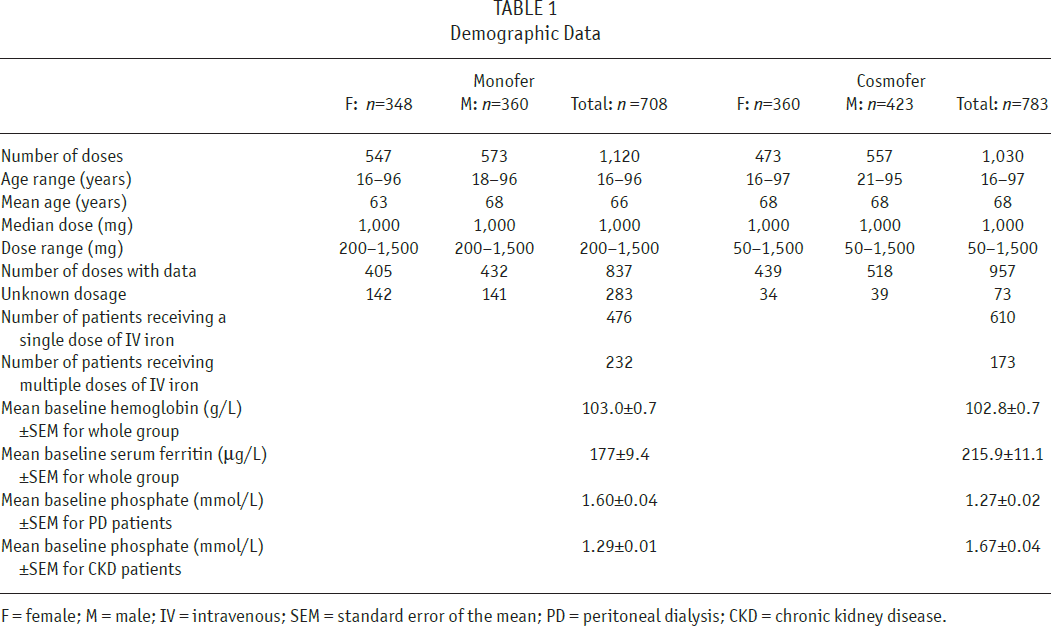
F = female; M = male; IV = intravenous; SEM = standard error of the mean; PD = peritoneal dialysis; CKD = chronic kidney disease.
Results
Overall, 1,120 doses of Monofer total dose infusion (1,000 – 1,500 mg) were administered to 708 patients (1,023 doses in 669 CKD and 97 doses in 64 PD patients) and 1,030 doses of Cosmofer total dose infusion (750 – 1,500 mg) were administered to 783 patients (893 doses in 708 CKD and 137 doses in 96 PD patients). In both cohorts, some patients received iron while in non-dialysis and when they subsequently progressed to PD. No patient had a previous known allergy to the 2 iron preparations.
For Monofer, there were 6 reactions: vomiting (1), hypertension (1), lethargy (1), constipation (1), and flare-up of eczema (1). The same patient with a respiratory infection who reacted to Cosmofer requiring oxygen support and salbutamol nebulizer also had a similar reaction to Monofer. None where classified as a
For Cosmofer, 7 reactions occurred post-administration or within 48 hours of dosing. On 2 occasions, there was a fall in BP requiring discontinuation of medication, with subsequent complete patient recovery. One patient suffered a severe adverse reaction requiring oxygen therapy, hydrocortisone, and salbutamol nebulizers. This patient had chronic obstructive pulmonary disease and a concurrent respiratory infection, and therefore it was not regarded as an anaphylactic response. Another patient had a suspected anaphylactic reaction leading to a collapse requiring medication discontinuation, oxygen support, intravenous hydrocortisone, and chlorphenamine. The other reactions consisted of hypertension (2) and rash (1). A single dose was given to 610 patients and multiple doses given to 173 patients. Six adverse reactions were after a single dose of Cosmofer and 1 reaction after multiple doses.
Both Monofer and Cosmofer led to significant increases in Hb and ferritin (Figure 1). The Hb increases for Monofer and Cosmofer were similar (4.5g/L vs 3.5 g/L,
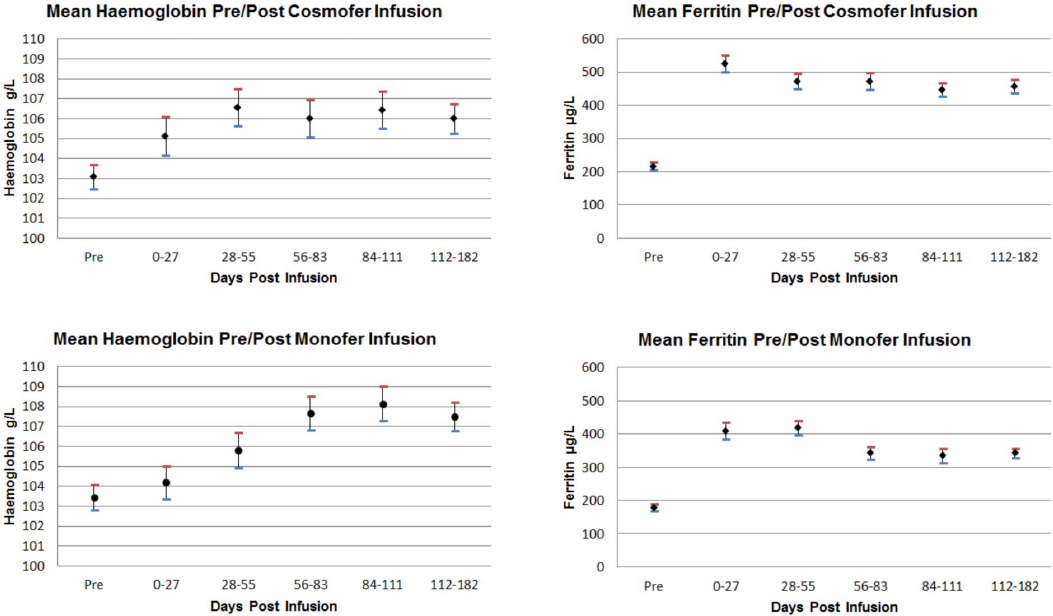
Summary of mean hemoglobin and ferritin levels pre- and post-therapy with Cosmofer and Monofer over the 182-day follow-up period of study. Results are mean values and SEM. SEM = standard error of the mean.
Discussion
Cosmofer and Monofer were associated with an incidence of adverse reactions of 0.68% and 0.54%, respectively, with 1 true anaphylactic reaction (Cosmofer), an average 4-g/L rise in Hb, and no impact on phosphate levels.
Wang
Anaphylaxis currently has no single accepted definition. The relative safety of IV iron preparations regarding acute infusion-related hypersensitivity reactions is not well characterized, and recording of adverse drug or anaphylactic reactions may not be accurate due to clinicians’ or nurses’ definitions, of the event (6), but recent algorithms to regulate, grade, and help manage anaphylactic and adverse reactions due to IV iron have been created (8–10). Retrospective identification of adverse reactions is difficult, and rather than “anaphylaxis,” it may be due to other more frequent reactions, such as the “Fishbane” reaction (6).
Monofer had an adverse reaction rate of 0.54%, with no confirmed episodes of anaphylaxis but a severe reaction requiring therapy. This is reassuring, given the relatively small numbers studied in this analysis and in line with a recent review from Kalra
Intravenous iron hypersensitivity rates are lower compared to other widely used medications such as non-steroidal anti-inflammatory drugs, paracetamol, and penicillin, with few concerns (9). Furthermore a recent meta-analysis that reviewed randomized trials of IV iron found no increased risk in adverse reactions, infections or mortality rate, consistent with our findings (13).
Our data add to the literature demonstrating an incidence of 1 in 1,000 doses for moderate to severe adverse drug reactions and 5 – 8 in 1,000 doses for any adverse event in line with the FIRM data (12), with 2 commonly used parenteral iron preparations, in non-dialysis and PD patients with CKD (6). Whilst such analysis is useful information, the data may be prone to possible bias and therefore confirmation, in randomized controlled studies with a larger data set, is needed. Despite reaction concerns, IV iron is safe, significantly improves quality of life, reduces morbidity and mortality (3,14), remains important in managing iron-deficiency anemia, and outweighs the risks (14,15).
At present, the balance between the benefits and the risks remains in favor of iron administration and patients should be reassured.
Footnotes
Acknowledgments
The authors gratefully acknowledge statistical support from research nurse Richard Cooper.
SB has received honorarium, participated in expert panels, and received funding to attend renal conferences from Pharmacosmos and Vifor Pharma. Pharmacosmos UK Ltd provided a fund to support open access of this article, but had no involvement in the analysis/ interpretation of data or composition of the manuscript. The authors have no financial conflicts of interest to declare.
