Abstract
Background
There is an emerging practice pattern of automated peritoneal dialysis (APD) in China. We report on outcomes compared to continuous ambulatory peritoneal dialysis (CAPD) in a Chinese cohort.
Methods
Data were sourced from the Baxter Healthcare (China) Investment Co. Ltd Patient Support Program database, comprising an inception cohort commencing PD between 1 January 2005 and 13 August 2015. We used time-dependent cause-specific Cox proportional hazards and Fine-Gray competing risks (kidney transplantation, change to hemodialysis) models to estimate relative mortality risk between APD and CAPD. We adjusted or matched for age, gender, employment, insurance, primary renal disease, size of PD program, and year of dialysis inception. We used cluster robust regression to account for center effect.
Results
We modeled 100,351 subjects from 1,178 centers over 240,803 patient-years. Of these, 368 received APD at some time. Compared with patients on CAPD, those on APD were significantly younger, more likely to be male, employed, self-paying, and from larger programs. Overall, APD was associated with a hazard ratio (HR) for death of 0.79 (95% confidence interval [CI] 0.64 – 0.97) compared with CAPD in Cox proportional hazards models, and 0.76 (0.62 – 0.95) in Fine-Gray competing risks regression models. There was prominent effect modification by follow-up time: benefit was observed only up to 4 years follow-up, after which risk of death was similar.
Conclusion
Automated peritoneal dialysis is associated with an overall lower adjusted risk of death compared with CAPD in China. Analyses are limited by the likelihood of important selection bias arising from group imbalance, and residual confounding from unavailability of important clinical covariates such as comorbidity and Kt/V.
The increased global prevalence of APD has been most prominent in developed countries such as the United States and Canada (3). In China, as with other developing nations, uptake has been hampered by lack of reimbursement and the financial burden that the therapy imposes upon patients (5). The uptake of APD is expected to increase in China, however, given the increasing income of many citizens, and their changing expectations of advanced standards in healthcare.
In China, it is important to know whether the increased cost of APD is justified by improved outcomes. The weight of evidence suggests that APD has similar effects to continuous ambulatory PD (CAPD) in terms of residual renal function (6–17), and either lower (2,18–24) or similar (17,25–28) rates of peritonitis. Older studies have mostly shown similar all-cause mortality (2,7,25,29–33). There is, however, evidence of greater improvements in outcomes on APD over time relative to those on CAPD (34) and several more recent studies that show better survival with APD, especially in those with high peritoneal transport, and younger patients with less comorbidity (1,18,22,35–37). In this study, we explore this question further by comparing all-cause mortality between APD and CAPD in a modern cohort of Chinese patients, adjusting for a range of clinical and economic variables in cause-specific and competing risk models.
Methods
Study Design
We performed an observational cohort study, using an “as treated” framework (“did exposure that the patient actually received affect mortality?”) (38). Of note, all patients in this study were treated with twin-bag Dianeal PD systems or Homechoice APD systems (Baxter Healthcare International, Deerfield, IL, USA). Polyglucose PD fluid is not available in China, and only 2-L twin-bag Dianeal PD systems are sold in China.
Participants and Data Source
For this research, data were sourced from the Baxter Patient Support Program, in a broadly similar manner to previous studies from other countries (32,39–41). This database and its operation and governance are described in detail elsewhere (42).
From the larger dataset, we created a cohort of patients who initiated PD with Baxter products between 1 January 2005 and 13 August 2015 in China. Patients with records of less than 90 days were excluded, as were those less than 1 year of age. Those older than 95 were coded as missing age data on the basis of likely ascertainment error. The remainder were followed up until death, dropout from discontinuation of Baxter PD products (commencement on another brand of PD, transfer to hemodialysis), loss to follow-up for other reasons, or 13 November 2015, whichever occurred first.
For reasons of probity, the study was performed with a degree of external and independent oversight, using an established governance pathway that is applied to epidemiological studies using either primary Asia-Pacific Baxter Healthcare data, or secondary data from outside of Baxter Healthcare that is analyzed by Asia-Pacific Baxter Healthcare employees. As mandated by the pathway, the study was reviewed and approved by 3 independent groups. The first was the Arbor Research Collaborative for Health (www.arborresearch.org), which examined the technical aspect of research design and the statistical analysis plan and execution through direct inspection of source data and replication of the statistical programming code. In this way, they ensured the appropriateness of our statistical methods, confirmed the reproducibility of our results, and excluded any inappropriate manipulation of data or corrupted application of analytical techniques being applied to the research in question. The second was the Shanghai Clinical Research Center (http://www.scrcnet.org/IEC_en.asp), which considered ethical and social implications of the study. The third was an advisory board of representative Chinese subject matter experts, convened with the advice of the Chinese Society of Nephrology. This group reviewed the interpretation of study findings in the local healthcare context, and contributed to the development of insights. This process is described in detail in Appendix 1 (available as on-line supplementary material).
Exposure Variables
The primary exposure was time-varying sub-modality of PD (APD vs PD). We modeled the following patient-related factors: age, gender, primary kidney disease (diabetes, glomerulonephritis, hypertension, other/unknown), occupation (employed, student, unemployed, unknown), medical insurance (Medicare for migrant workers, New Rural Cooperative Medical Scheme/System [NCMS], self-pay (assuming unknown to be self-pay), Urban Employee Basic Medical Insurance [UEBMI], Urban Resident Basic Medical Insurance [URBMI]), size of the PD program as defined by their cumulative clinical experience over the period of observation (1 – 132, 133 – 310, 312 – 523, 565 – 2,042 patients), quartiles of era of dialysis inception (2005 – 2007, 2008 – 2009, 2010 – 2012, 2013 – 2015), and their de-identified center of treatment. Continuous covariates other than age were modeled as clinically relevant quantiles in order to avoid the assumption of linear relationships.
Outcome Variable
The primary outcome for the survival analysis was a recorded event of “patient death.” The recorded event of “withdrawal from dialysis” was included in models as death. Competing events recorded in the database are “transplantation,” “transfer to HD,” “transfer to another brand of PD products,” and “loss to follow-up.” The events recorded above are fixed categories in drop down menus: precise reason for loss to follow-up is recorded as free text in some but not all cases, with the most common reason being transfer out of the area.
Statistical Methods
We constructed time-varying exposure-outcome models to estimate the effect of modality on mortality, handled by episode-splitting within patients, using multiple approaches to analysis, described in Table 1. The main reasons for using multiple models was to ensure convergent validity of our results when modeling the data under different assumptions. In models 1 and 2, confounder imbalance was addressed by adjustment for covariates in multiple regression models. In models 3 and 4, this was dealt with through matching. In models 1 and 3, transfer to HD was modeled as a censoring event, ignoring the possibility (and high probability) that it is informative as an event, with a risk of death before the event that is different from that after the event. In models 2 and 4, transfer to HD was modeled as a competing event, making no assumptions around the distribution of risk of death before and after the event. Convergent validity can be established if the approaches correspond with one another.
Modeling Approaches Used for Analysis
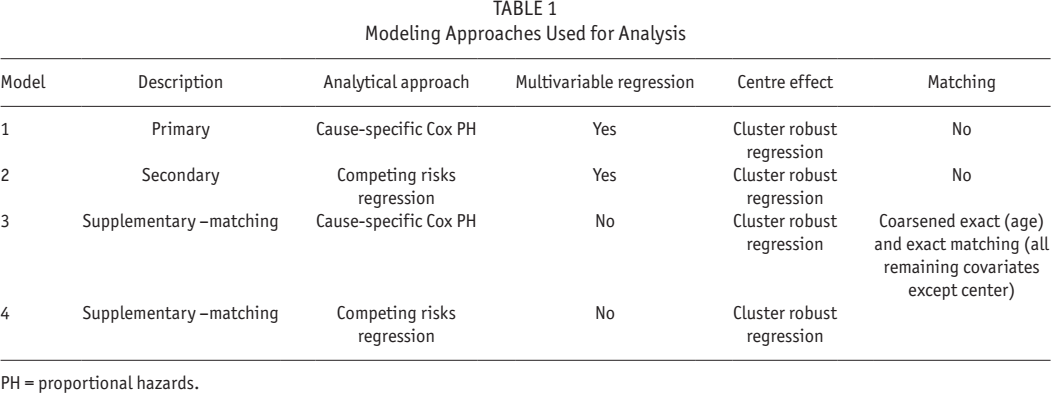
PH = proportional hazards.
Given the etiological, rather than prognostic, nature of the research (43), model 1 was designated as our primary analysis, using Cox proportional hazards analysis censoring for switch to HD, kidney transplantation, discontinuation of Baxter PD products, and loss to follow-up for unrecorded reasons. Hazard ratios (HRs) calculated using this approach can be interpreted as actuarial probabilities: mortality rate ratios “among those patients who are alive, but had not (yet) experienced a competing event.” We accounted for correlated data within centers of dialysis by robust cluster estimation, rather than a formal shared frailty model, which cannot be fit when there is delayed entry into a model, as is the case with episode-splitting.
In model 2, we modeled switch to HD and kidney transplantation as competing risks based on Fine and Gray's proportional subhazards model (44). Patients were censored in models at the time of discontinuation of Baxter PD products and loss to follow-up, assuming independent and non-informative censoring (time to event and time to censoring are independent conditional on the level of covariates). Subhazard ratios (SHRs) calculated using this approach can be interpreted as actual probabilities: mortality risk ratios “among those patients who are alive including those who had been switched to HD or been transplanted, but had not (yet) discontinued Baxter PD products or been lost to follow-up.” In this model, we again accounted for correlated data within centers of dialysis by robust cluster estimation.
In models 3 and 4, we performed cause-specific Cox proportional hazards analysis and competing risks regression between matched samples of patients on APD versus CAPD, respectively. The samples were created with exact matching on all the covariates listed in Table 2, apart from coarsened exact matching which was performed on age. This latter process was done by categorizing age into approximately 4.5-year strata, and matching within those strata. Exact and coarsening exact matching was implemented using the “cem” facility of Stata (45 46-47), and important advantages over other probability-based systems such as propensity-score matching (48,49). Exact matching creates samples that are balanced at both subject and population levels rather than just the latter, and avoids modeling assumptions in defining what constitutes a suitable match. Due to information loss, we were not able match for center, but instead matched for center size, accounting for correlated data within centers of dialysis by robust cluster estimation.
Clinical Characteristics of APD and CAPD Patients at Inception of Each Modality Episode
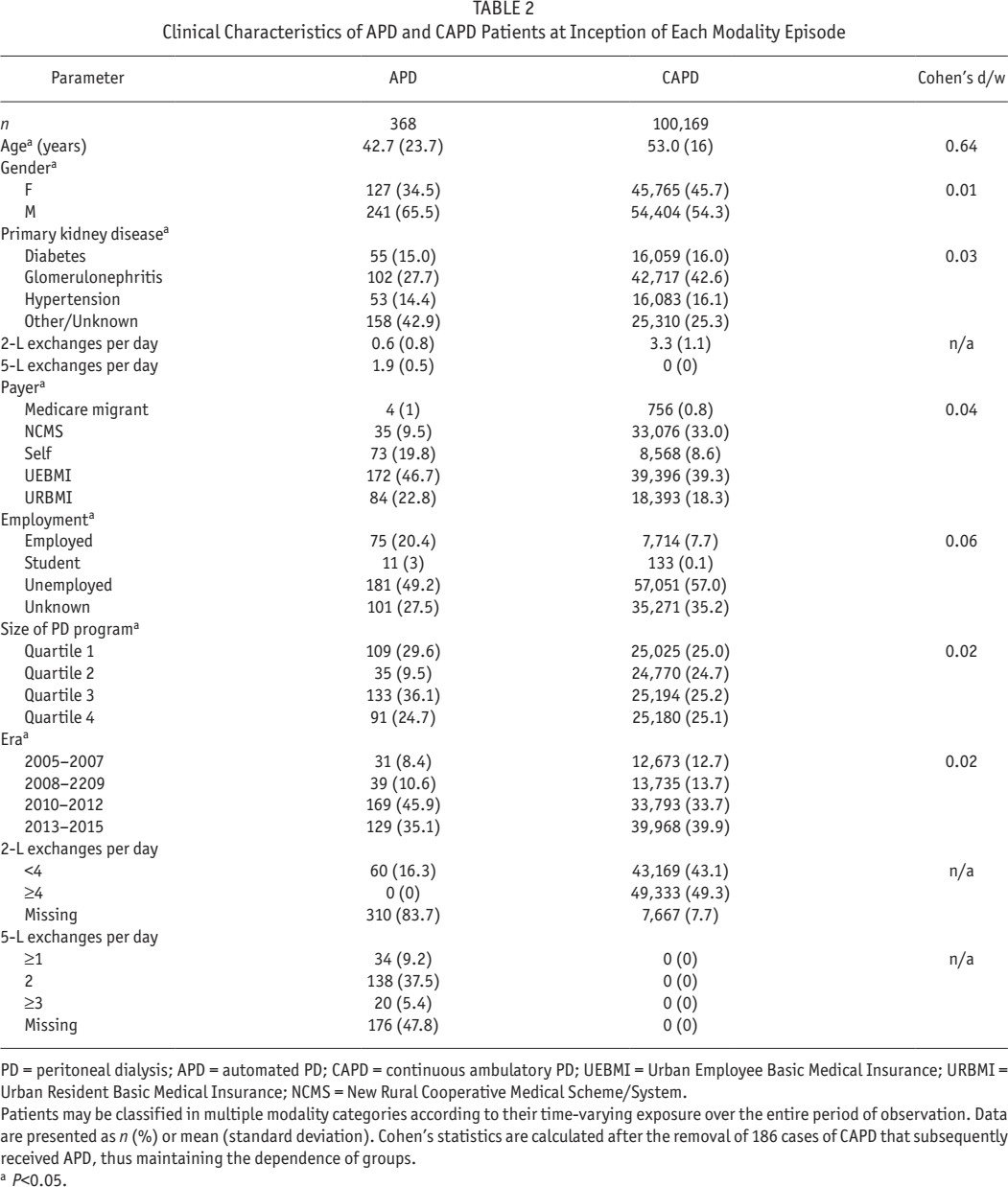
PD = peritoneal dialysis; APD = automated PD; CAPD = continuous ambulatory PD; UEBMI = Urban Employee Basic Medical Insurance; URBMI = Urban Resident Basic Medical Insurance; NCMS = New Rural Cooperative Medical Scheme/System.
Patients may be classified in multiple modality categories according to their time-varying exposure over the entire period of observation. Data are presented as n (%) or mean (standard deviation). Cohen's statistics are calculated after the removal of 186 cases of CAPD that subsequently received APD, thus maintaining the dependence of groups.
P<0.05.
In all the multivariable models, we initially included all covariates in the models, which we then removed in a backward stepwise fashion beginning with the covariate with the highest p value from 2-tailed Wald tests of the individual coefficients. We based final confounder selection upon both biological plausibility and contribution to the comprehensibility of the model, and also the significance of the covariate within the model as assessed by the 2-tailed partial likelihood ratio test, p value at a level of 0.2 when jointly adjusted for other covariates. We modeled primary renal disease, occupation, and type of medical insurance using deviation from means coding (50), where there is no particular rationale for the selection of one group over the other as a reference group.
We used 2-way interaction terms in the primary main-effects model to test effect modification by age, primary renal disease, occupation, and medical insurance. We chose these interactions as being clinically plausible, on the basis of both published literature as well as cumulative clinical experience. Interactions were assessed using the 2-tailed Wald test, p values as a guide to selecting interaction terms for testing, with significance within the model using the likelihood ratio test (p < 0.05).
The assumption for proportional hazards for the final models was assessed formally by the use of scaled Schoenfeld residuals, and visually by-In [-ln(survival)] versus In (analysis time) plots for modality, adjusted for confounders. We assessed overall goodness of fit visually by comparing plots of Kaplan-Meier observed survival curves to the Cox predicted curves for modality.
Where necessary, we made comparisons between groups using the Fisher's exact test, and Mann-Whitney U (or Kruskal-Wallis) tests as appropriate. For the description of correlated data (e.g. a given patient may have contributions to each modality), we used linear or logistic regression with clustered robust standard errors. We computed effect size statistics using Cohen's d for continuous variables and Cohen's w for categorical ones (51,52).
A number of factors were not measured in this study (e.g. other comorbidity, residual renal function, medication, functional status, health literacy, etc.), and these might confound any identified relationship between modality and mortality. We therefore performed simple sensitivity analyses to estimate the extent to which unmeasured, hence uncontrolled, confounding may have over- or under-estimated the true effect (53 54 55 56-57). We defined various levels of a hypothetical unmeasured confounder, both in terms of prevalence among patients on CAPD (reference group) versus those on APD (comparator group), as well as the size of the association between the unmeasured confounder and mortality risk. We used previously published bias formulas to calculate the effect of these different levels of the unmeasured confounder on the modeled effect of APD from the primary analysis (54,57). If the point estimate of the effect of APD could be offset to 1.0 by only a modest confounder (i.e. one with a small association with mortality risk, and a similar distribution in each group), the estimates of the primary model could be deemed to be sensitive to unmeasured confounding, and therefore not robust. Alternatively, if the point estimate of the effect of APD could only be offset to 1.0 by an implausibly strong confounder (i.e. one with a very Large association with mortality risk, and a very unbalanced distribution between groups), estimates of the primary model could be deemed to be insensitive to unmeasured confounding, and therefore robust. Analyses were performed using Stata Intercooled MP/14.1 (StataCorp LP, College Station, TX, USA).
Results
Participants
The original inception cohort included 106,734 subjects with 25,744 deaths over 258,008 patient-years follow-up. There were 100,351 subjects from 1,178 centers meeting inclusion criteria with sufficient data for modeling meeting inclusion criteria w, with 23,881 deaths and 15,840 who were transferred to HD or transplanted over 240,801 patient-years. Table S1 summarizes the study cohort and excluded cohort due to missing data at dialysis inception (the point of commencement of dialysis). While the excluded cohort comprised only about 0.2% of the potential study cohort, subjects with missing data were more likely to be male, self-pay, with an unknown/other cause of primary kidney disease, and from smaller PD programs. Differences between the included and excluded dataset were small in terms of effect size and clinical significance, although some differences were still considerable and might need to be considered by the reader in the interpretation of results. Figure 1 shows patient disposition in the cohort, both in terms of assembly of the cohort, and also outcomes according to both sub-modality at 90 days, and also “as treated” and as analyzed in this study.
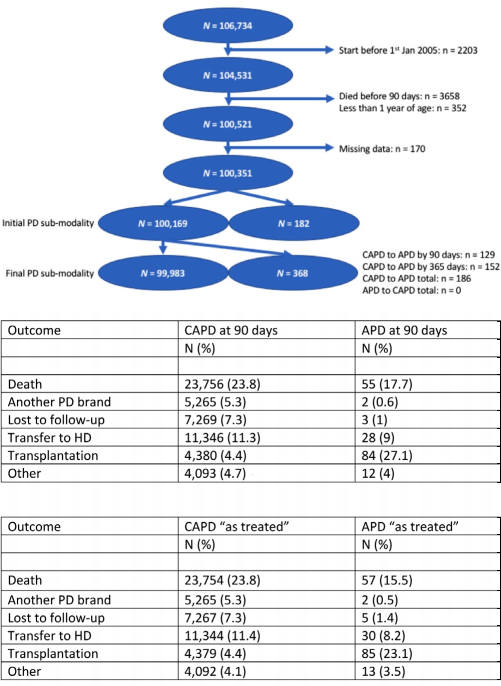
Cohort assembly and disposition. PD = peritoneal dialysis; CAPD = continuous ambulatory PD; APD = automated PD.
Descriptive Data
Table 2 summarizes the clinical characteristics of the final dataset for the primary and secondary analyses, and also compares these characteristics by sub-modality at PD inception. Of the 100,351 patients, 368 received APD from 99 centers and the remainder only CAPD. Those on APD were significantly younger, more likely to be male, employed, self-pay, and from larger PD programs. These differences were statistically significant, but other than age, associated with a very small effect size. In models 3 and 4, the matched cohort was restricted to 518 patients in total, 259 of whom were on APD, with 104 deaths. Table S2 summarizes the clinical characteristics of the final dataset for the supplementary analyses, using the matched samples. As might be expected from the algorithm we utilized, the samples are identical on measured matching criteria.
Main Results
Figure 2 illustrates actuarial and actual probabilities of death. Both suggest a beneficial effect associated with APD relative to CAPD. Figure 3 shows the primary multivariable model for the entire period of follow-up, with an overall significant 21% reduction in mortality risk associated with APD relative to CAPD. However, there is clear non-proportionality of hazards in the primary model (Figure S1), with poor agreement between actual and modeled mortality risk (Figure S2), and formal violation of the proportional hazards assumption (formal testing of scaled Schoenfeld residuals, p = 0.002). Given the prominent change in HR at 4 years, we estimated separate models for the periods of follow-up of < 4 and > 4 years. Within these periods of follow-up, there was no violation of the proportion hazards assumption (formal testing of scaled Schoenfeld residuals model, p = 0.8 for < 4 years, p = 0.6 for > 4 years). The full models from the primary analysis, separated by periods of follow-up, are shown in Figure S4.
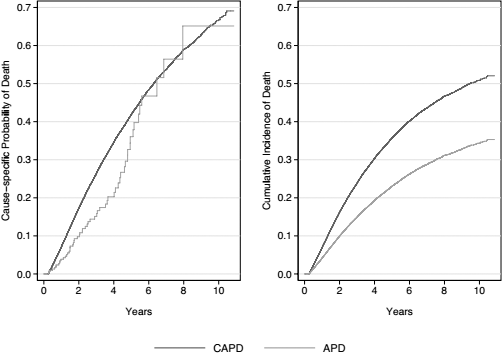
Mortality from the primary and secondary models, illustrating actuarial (right) and actual (left) probabilities, respectively. PD = peritoneal dialysis; CAPD = continuous ambulatory PD; APD = automated PD.
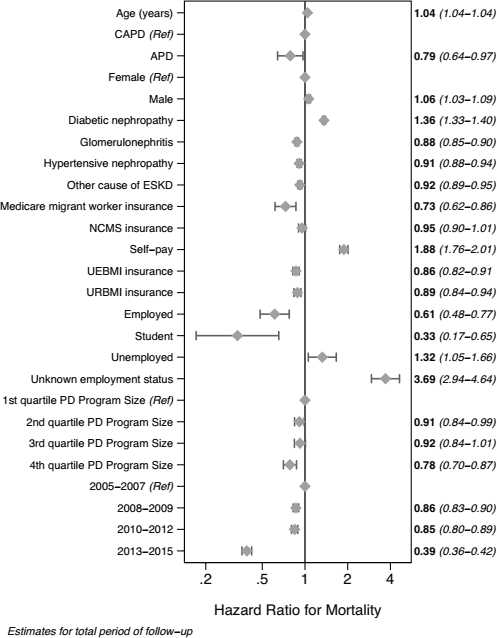
Main effects of the primary multivariable model over the entire period of follow-up, fully adjusted for the confounders listed in Table 2 (the marker represents point estimates, the whiskers, 95% confidence intervals). PD = peritoneal dialysis; CAPD = continuous ambulatory peritoneal dialysis; APD = automated peritoneal dialysis; ESKD = end-stage kidney disease; UEBMI = Urban Employee Basic Medical Insurance; URBMI = Urban Resident Basic Medical Insurance; NCMS = New Rural Cooperative Medical Scheme/System.
The final estimates from models 1 to 4 are shown in Figure 4, separated by periods of follow-up. The results of all these models support convergent validity—an overall benefit associated with APD relative to CAPD, which is more marked in the earlier subset of follow-up time, and attenuated after 4 years of follow-up. Of note, the power of analyses in the later period is low for all the models due to the relatively small number of patient risk at that time; for instance, only 79 APD and 17,162 CAPD patients remain at risk in the primary model at 4 years. As such, estimates in this later period from all the models must be interpreted cautiously.
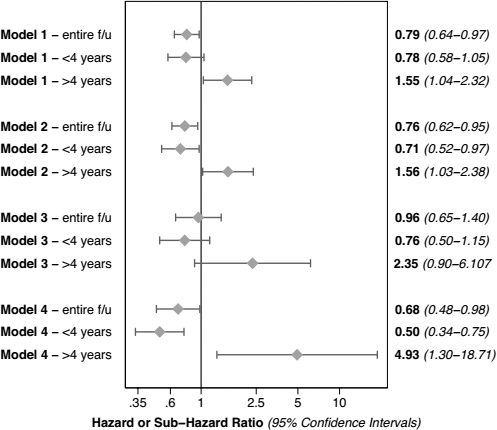
Effects of automated peritoneal dialysis (APD) relative to continuous ambulatory peritoneal dialysis (CAPD) from all models listed in Table 1, by period of follow-up (the marker represents point estimates, the whiskers, 95% confidence intervals). Model 1 is the primary model (cause-specific Cox proportional hazards on the whole cohort); model 2 is the secondary model (Fine and Gray competing risks regression on the whole cohort); model 3 is the first supplementary model (cause-specific Cox proportional hazards on the matched cohort); model 4 is the second supplementary model (Fine and Gray competing risks regression on the matched cohort). f/u = follow-up.
There were significant interactions (p < 0.05) involving age and gender in the primary analysis, with clear effect modification in those who were younger and males. There were no interactions involving occupation and medical insurance. Figure 5 shows the mortality risk of APD relative to CAPD within subgroups of age and gender, by period of follow-up for each. As can be seen in this figure, the beneficial effect associated with APD is greater in younger patients and in males.
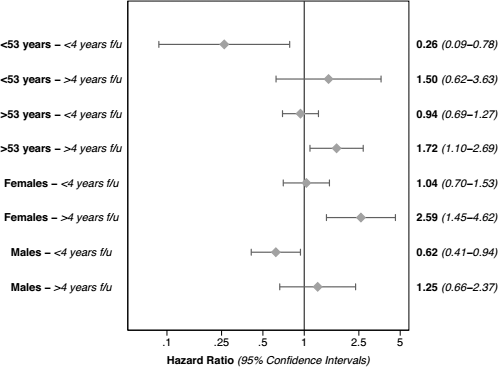
Effects of automated peritoneal dialysis (APD) relative to continuous ambulatory peritoneal dialysis (CAPD) for subsets of patient age and gender, by period of follow-up (the marker represents point estimates, the whiskers, 95% confidence intervals). f/u = follow-up.
Table 3 summarizes the results of our analyses for unmeasured confounding. These suggest that the associations that we observed for APD are unlikely to have been due to confounding only. It is only after adjustment for a strong confounder (i.e., one that produces a > 2 – 5-fold decrease in risk of death), with very different distribution between APD and CAPD groups (i.e., one that differs in prevalence between each group by > 20 - 50% more), that we see attenuation of the association between modality and mortality risk. Adjustment for more moderate confounding produced little change in the results.
Sensitivity of Main Effects Model to Inclusion of Hypothetical Unmeasured Confounder, CD
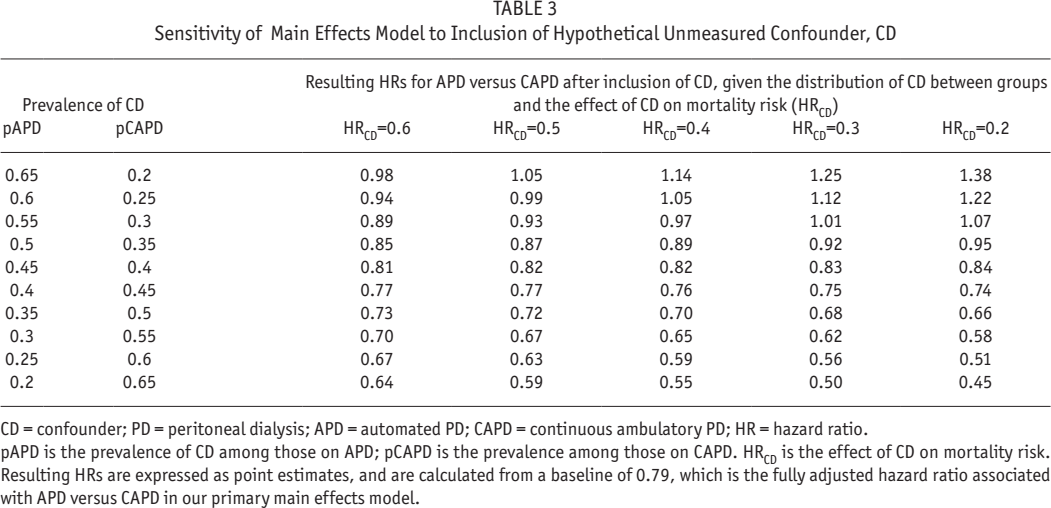
CD = confounder; PD = peritoneal dialysis; APD = automated PD; CAPD = continuous ambulatory PD; HR = hazard ratio.
pAPD is the prevalence of CD among those on APD; pCAPD is the prevalence among those on CAPD. HRCD is the effect of CD on mortality risk. Resulting HRs are expressed as point estimates, and are calculated from a baseline of 0.79, which is the fully adjusted hazard ratio associated with APD versus CAPD in our primary main effects model.
We performed a sensitivity analysis restricting the above analyses between those patients on CAPD using ≥ 4 x 2-L exchanges per day and those on APD using ≥ 2 x 5-L exchanges per day. In this restricted cohort, there was an even greater beneficial effect associated with APD relative to CAPD, compared to the main analysis in the unrestricted cohort (Figures S4 and S5).
Discussion
In this cohort study, survival on APD was found to be generally superior to that on CAPD, adjusted for a number of other important covariates. However, the results of this study are by no means conclusive, and the effect of APD per se on patient survival must still be a matter of speculation. This is because of 2 very important limitations which are discussed below.
Firstly, our data do not contain important recognized risk factors for mortality. We were, however, able to include and adjust for the usual core demographics and primary kidney disease. In addition, we were able to adjust for some important predictors that other studies do not usually include. In our model, reimbursement status and occupation were powerful predictors of outcome, as one would expect (58–61). Size of PD program was also a significant predictor of outcomes; this variable is accepted as a general indicator of cumulative clinical experience, program infrastructure, and general expertise (31,6263–64). The effects of covariates that we were able to include are therefore as expected, directionally speaking. However, important covariates such as residual renal function, details of clinical comorbidity, membrane transport characteristics, and Kt/V are missing.
Secondly, it is important to note the very large difference between the number of patients on APD and the number of patients on CAPD. In China, APD is not reimbursed by payers, and must be funded out of pocket by the patient or family, or directly by a hospital or charity. As such, the decision to use APD is very much a shared decision between patients and caring healthcare professionals, and is driven by compelling clinical and/or patient-centered reasons (e.g. pediatric indications, non-negotiable study or employment lifestyle needs), and access to care (patient or provider willingness to pay). This ecosystem is reflected in the characteristics of the APD cohort—patients receiving APD were likely to be younger, employed, self-paying, with better access to care as exemplified by their differentially greater transplantation rate and lower rate of loss to follow-up. We have tried to adjust for these factors as much as possible using the covariates available to us, such as patient insurance and employment status, and hospital cumulative clinical experience. Despite our efforts to account for differences, there is likely to be residual confounding that no amount of modeling can abrogate—a large but unquantifiable part of the demonstrated association between APD and better outcomes is likely to be from unmeasured differences between the cohorts, rather than APD itself. There is therefore a high likelihood of important selection bias, which remains as residual confounding.
The broader literature is inconsistent with respect to survival with APD versus CAPD. The weight of overall clinical opinion, however, is that the benefit of APD is mainly limited to the patient experience (4). However, many of the studies specifically comparing mortality risk are not particularly robust, and largely invalidated by informative censoring (29), or immortal time bias (65). In addition, these studies are often from older eras, and do not account for the improvements in the evolution of connection systems for CAPD in the 1990s (twin bag, flush before fill), and then later on for APD (connection assist devices, and the use of Luer lock connections). Numerous studies have shown that mortality rates are improving faster for some dialysis modalities than others, and recent evidence suggests that this is also the case for APD relative to CAPD (34). As such, it is timely to re-ask the question of differential outcomes by PD sub-modality, and attempt to answer it using up-to-date and robust analytical methods as we have done.
So, are there any plausible clinical reasons for better patient survival with APD in our study? In some studies, investigators have suggested that APD may provide better control of extracellular volume. Automated PD does indeed achieve better fluid balance than CAPD in most high transporters, although the achievement of dry weight and blood pressure control is overall fairly similar (1,2). In the absence of any fluid control or peritoneal transport status data in our study, we cannot investigate this hypothesis further. Another potential reason for better patient survival with APD in our particular study may be the relatively low dose of dialysis in the CAPD patients—more than 50% of patients are on 3 exchanges a day. This practice pattern, however, is common in both Hong Kong and China, with historically good outcomes when carefully applied to suitable patients of low body weight or with adequate residual renal function (66 67-68). Moreover, our sensitivity analysis restricted analysis to those receiving what might be regarded as an optimal dialysis dose (≥ 4 exchanges a day for CAPD, ≥ 2x 5-L exchanges a day for APD); this did not abrogate the effect of observed benefit of APD.
Cumulative clinical experience suggests another reason for a potential clinical benefit with APD that might explain the better patient survival with APD in our study. For CAPD, prescription options are usually limited to variation in dialysate glucose concentration. Although there is an ability to alter fill volume and frequency, this is seldom exercised due to the logistic and cost burden of changing bag size and number—a default prescription of 4 x 2-L exchanges per day is the most common, globally (69). In contrast, APD provides a wider range of easily accessible and cost-neutral prescription options, which include not only fill volumes and frequency but also overall time on cycler, the use of a daytime dwell, prescription changes that deferentially increase, and sodium and volume removal. Such options allow suitably trained nephrologists to better tailor their prescription to patient clinical needs, and plausibly obtain better clinical outcomes. We cannot test this hypothesis in our study due to lack of granularity in our data. In our opinion and experience, however, this is a plausible scenario and potential explanation for the results of our study.
Another key finding in our study was the relatively greater associated benefit of APD in younger patients and males. This is consistent with other studies from Hong Kong and Taiwan (22,37). For the same reasons as stated above, this observation could be either causal or due to residual confounding. Notwithstanding, this appears to be a consistent finding in Asian cohorts and merits further examination.
A final noteworthy finding in our study is that of non-proportional hazards, with an early survival benefit associated with APD before 4 years of follow-up and no effect thereafter. Such non-proportionality has not been observed previously, although there is only a small body of literature in which the assumption of proportional hazards may have been tested. To our knowledge, only 1 paper has reported non-proportionality of hazards over time, and the pattern of the finding was different in that report compared with ours (31). Our speculation is that this non-proportionality arises from selection bias from loss to follow-up in the APD arm. Over time, eligible and probably healthier patients are differentially transplanted in the APD arm, leaving a progressively more comorbid cohort with comparatively poorer outcomes (70). A structural model with time-varying comorbidity would be required to tease this out, and we do not have the requisite data to perform this analysis.
Conclusion
There are limitations to our study in terms of internal validity, beyond those already mentioned above. First, associations per se do not prove causality. Second, our study is likely to be limited by the ascertainment error that is inherent in all registries around the world. For instance, patients may not always know the cause of their end-stage kidney failure. Because of reasons pertaining to patient privacy, there is no means of validating the clinical and outcomes data modeled in this study. Third, we used an “as -treated” approach, rather than “intention-to-treat” (38). This is not a weakness per se, since each approach is used to address different issues. In our study, we are most interested in “causal” associations between modality and outcome and less interested in clinical prognostication. Such aims are well served by the approach used in this study, so long as there is appropriate modeling of the attribution of mortality to modality. We did not lag the attribution of outcomes to modality in this study, which is usual in such “as treated” models—no patients in the dataset transferred from APD to CAPD, and the modality of record was able to be modeled directly in relation to outcome without a lag. As part of the review process, we did perform an intention-to-treat analysis modeling sub-modality as defined as APD versus CAPD at 90 days—estimates were weakened by information loss and a decrease in sample size, but were directionally similar to those which are presented for the main analysis (HR for APD versus CAPD 0.82 [95% CI 0.67 – 1.02], full analysis not shown).
There are also limitations to our study in terms of external validity that arise from the practice patterns in China, which are different to those in most published cohort studies that in themselves mostly come from International Monetary Fund advanced economies. For instance, icodextrin is unavailable in China. Icodextrin has an uncertain effect on patient mortality in randomized controlled trials reporting this outcome (71–86), but is associated with improved mortality in several cohort studies (40,87–89). As such, the external validity of our study is limited for developed or advanced economies, but still high for the majority of the world's PD population, which is to be found in Asia, and for whom icodextrin is similarly unavailable.
As a final note, the benefit of APD is certainly well established for patient experience (4), with generally better reported patient-centered outcomes (90,91). Such outcomes, in the wider picture, are likely to be just as important as clinical ones. Notwithstanding, our study suggests the possibility of an additional relationship between PD sub-modality and mortality risk in China.
Footnotes
Acknowledgments
We would like to thank Dr Jarcy Zee for her expertise and patience in providing an analytical review of our models and data, and the Arbor Research Collaborative for Health for their support of this initiative.
MM, GZ, QY, and JS are fully employed by Baxter Healthcare. The other authors have no financial conflicts of interest to declare.
