Abstract
Background
Peritoneal dialysis (PD) technique failure is often accompanied by complications that increase risks of hospitalization and death. Planned transition to hemodialysis may improve outcomes. Transitioning patients from PD to home hemodialysis (HHD) may improve continuity of lifestyle and facilitate delivery of more frequent treatment.
Methods
We analyzed United States Renal Data System (USRDS) data to compare the incidence of death and kidney transplant in patients who transferred from PD to HHD and matched patients who transferred from PD to in-center HD (IHD). We used Fine-Gray regression to estimate hazard ratios (HRs) of death and transplant for HHD versus IHD.
Results
We identified 521 patients who transferred from PD to HHD. Survival in HHD patients was 89.1% at 1 year and 80.5% at 2 years. In intention-to-treat analysis, the HR of death for HHD versus matched IHD patients was 0.76 (95% confidence interval [CI] 0.65 – 0.90). In subsets of non-Medicare and Medicare patients, corresponding HRs were 0.57 (95% CI 0.43 – 0.75) and 0.92 (95% CI 0.75 – 1.13), respectively. Kidney transplant incidence in HHD patients was 10.6% at 1 year and 21.0% at 2 years. In modified intention-to-treat analysis, the HR of transplant for HHD versus matched IHD patients was 1.36 (1.14 – 1.61).
Conclusions
Transfer to HHD after PD technique failure was rare, but associated with lower risk of death and higher incidence of transplant than transfer to IHD. Heterogeneity in relative risks by Medicare coverage suggests uncertainty about the magnitude of benefit. Still, these data suggest that clinical outcomes after PD technique failure can be improved.
Patient outcomes following PD technique failure are typically poor, because the transfer interval is saddled with complications that often necessitate inpatient care and may imminently lead to death. It is plausible that accurate prediction of PD technique failure could enable planned and safer transition to HD. Notably, transitioning patients from PD to home hemodialysis (HHD) may ease the psychosocial burden of the transfer interval and facilitate delivery of more frequent treatment, which may be especially efficacious in patients with hypervolemia, hypertension, and heart failure (6,7). However, there are few data about the trajectory of patients who transfer from PD to HHD (8–10).
In this study, we used data from the United States Renal Data system (USRDS) to assess the incidence of death and kidney transplant in patients who transferred from PD to HHD and to compare outcomes in those patients and matched patients who transferred from PD to in-center HD (IHD). We matched patients according to both demographic factors and comorbidity. Although such matching is unlikely to fully account for healthcare processes that differentiated transfer to HHD versus IHD, the results of this study illustrate the potential for improved outcomes when PD technique failure does occur.
Methods
Study Cohort
Home HD patients were identified in a NxStage System One user registry that is managed by NxStage Medical, Inc. (Lawrence, MA) and has been linked to the USRDS database. Each registry record included beginning and ending dates of System One use in the home setting (i.e., private residence) and prescribed number of dialysis sessions per week. For each patient, we defined the index date as the date of first HD session in the home setting. We retained patients according to criteria in Figure 1. This cohort construction essentially imposed a 6-month entry period comprising a first half mostly covered by PD and a second half that was partially covered by HHD training.

Inclusion and exclusion criteria. USRDS = United States Renal Data System; IHD = in-center hemodialysis; PD = peritoneal dialysis.
In-center HD patients were identified in the USRDS database. We ascertained all patients who began PD for the first time and who later experienced technique failure, which was defined by IHD for ≥ 2 months following the last day of PD. For each patient, we defined the index date as the date that was exactly 2 months after the last day of PD. We retained patients according to criteria in Figure 1.
We categorized patients into 3 strata: those without Medicare coverage during the entire 6-month interval preceding the index date (stratum 1); those with Medicare coverage during the entire 6-month interval preceding the index date, but without Medicaid coverage (stratum 2); and those with both Medicare and Medicaid coverage during the entire 6-month interval preceding the index date (stratum 3). Medicare coverage permits detailed characterization of disease severity. Medicaid coverage is a proxy for low socioeconomic status.
Data Elements
On the index date of each patient, we identified age, race, sex, primary cause of end-stage renal disease (ESRD), ESRD Network of residence, and ESRD. For patients in strata 2 and 3, we also identified Elixhauser comorbid conditions and cumulative cause-specific hospitalized days during the 6-month interval preceding the index date, as a proxy for the cause of technique failure. Comorbid conditions were defined from all claims during the 6-month interval preceding the index date; we required ≥ 1 Medicare Part A claim or ≥ 2 Part B claims with relevant diagnosis codes to define conditions. Hospitalized days were ascertained from inpatient claims; cause of hospitalization was defined by principal discharge diagnosis and categorized as cardiovascular disease, infection, and other morbidity. Regarding follow-up data, we identified dates of kidney transplant and death from USRDS data. In HHD patients, we defined technique failure as ≥ 2 months after the ending date of System One use without kidney transplant or death and set the date of technique failure as the date that was exactly 2 months after the last day of such use.
Matching Algorithm
We used propensity score matching (11). For each of stratum 1 and the pooled set of strata 2 and 3, we fit a logistic regression model of HHD initiation to the set of HHD and IHD patients. Predictors included main effects for age, race, sex, primary cause of ESRD, ESRD duration, ESRD Network, and the number of days between December 31, 2005, and the index date. In the pooled set of strata 2 and 3, predictors also included main effects for comorbid conditions and hospitalized days; these predictors theoretically reduced residual confounding between HHD and IHD in the pooled set of strata 2 and 3, relative to stratum 1. Age, ESRD duration, calendar time, and hospitalized days were parameterized with quadratic polynomials. From the fitted model, we calculated the logit of the probability of HHD initiation in each HHD or IHD patient.
We ordered HHD patients by index date and arbitrarily within index date and applied a greedy matching algorithm in the specified order. For each HHD patient with propensity score logit equal to r, we identified the index date d and Medicaid coverage status (for patients in the pooled set of strata 2 and 3). We retained IHD patients whose index date was within 6 months of d and whose Medicaid coverage status was the same as in the HHD patient (as was applicable). From this subset, we selected 3 IHD patients with propensity score logits equal to s1, s2, and s3, such that the absolute differences between r and s1, r and s2, and r and s3 were minimized; we applied no caliper. We did not permit resampling of IHD patients.
Statistical Analysis
We calculated mean summaries of measured factors in HHD patients, unmatched IHD patients, and matched IHD patients. We assessed the match quality with absolute standardized differences; differences less than 10% indicate sufficient similarity to obviate adjustment. For follow-up, we used intention-to-treat (ITT), modified ITT, and as-treated rules. By the ITT rule, we followed patients from the index date to the earlier of death or December 31, 2013. By the modified ITT rule, we followed patients to the earlier of kidney transplant, death, or December 31, 2013. Finally, by the as-treated rule, we followed patients to the earlier of kidney transplant, death, HHD technique failure, or December 31, 2013. We classified kidney transplant and death as competing events and HHD technique failure as a censoring event.
To compare risk of death between HHD and both unmatched and matched IHD patients, we estimated Kaplan-Meier survival functions in ITT analysis, overall and within strata defined by Medicare coverage. We also fit Fine-Gray regression models to estimate the relative hazard of death for HHD versus both unmatched and matched IHD patients, with stratification by each matched cluster of 1 HHD patient and 3 IHD patients (12). We fit models overall and within strata defined by Medicare coverage. We estimated both aggregate hazard ratios (i.e., risk during all of follow-up) and interval hazard ratios (i.e., risk during follow-up years 1, 2, and ≥ 3). In modified ITT and as-treated analyses, kidney transplant was classified as a competing event. To compare risk of kidney transplant between HHD and matched IHD patients, we estimated cumulative incidence functions in modified ITT analysis, with death classified as a competing event. We again fit Fine-Gray regression models to estimate the relative hazard of kidney transplant for HHD versus matched IHD patients, with aforementioned stratification. We fit models overall and within strata defined by Medicare coverage, with death classified as a competing event. Finally, we estimated the cumulative incidence of HHD technique failure, with both kidney transplant and death classified as competing events. We performed analyses in SAS, version 9.4 (Cary, NC) and R, version 3.4 (Vienna, Austria).
Results
From the USRDS-linked user registry of 13,643 HHD patients, we identified 521 patients (3.8%) who transitioned from PD to HHD. Almost 92% were prescribed either 5 or 6 sessions per week. Mean age was 53.2 years, roughly 3 in 4 patients were white, roughly 2 in 3 patients were male, and mean ESRD duration was 4.3 years (Table 1). Over 57% had Medicare coverage. We identified 32,871 patients who transitioned from PD to IHD. Compared with HHD patients, mean age was 4.2 years older and mean ESRD duration was 1.0 year lower in IHD patients. Over 67% had Medicare coverage. In that subset, the prevalence of peripheral vascular disorders, hypertension, and fluid and electrolyte disorders were far higher (absolute standard differences > 20%) in IHD than HHD patients. Importantly, whereas HHD patients with Medicare coverage accumulated 9.1 hospitalized days during the 6-month interval preceding the index date, IHD patients with Medicare coverage accumulated 18.5 days; hospitalized days due to infection were especially imbalanced between IHD and HHD. Nevertheless, matching successfully balanced all factors (absolute standardized differences < 10%).
Patient Characteristics, by Dialytic Modality, in Unmatched and Matched Cohorts
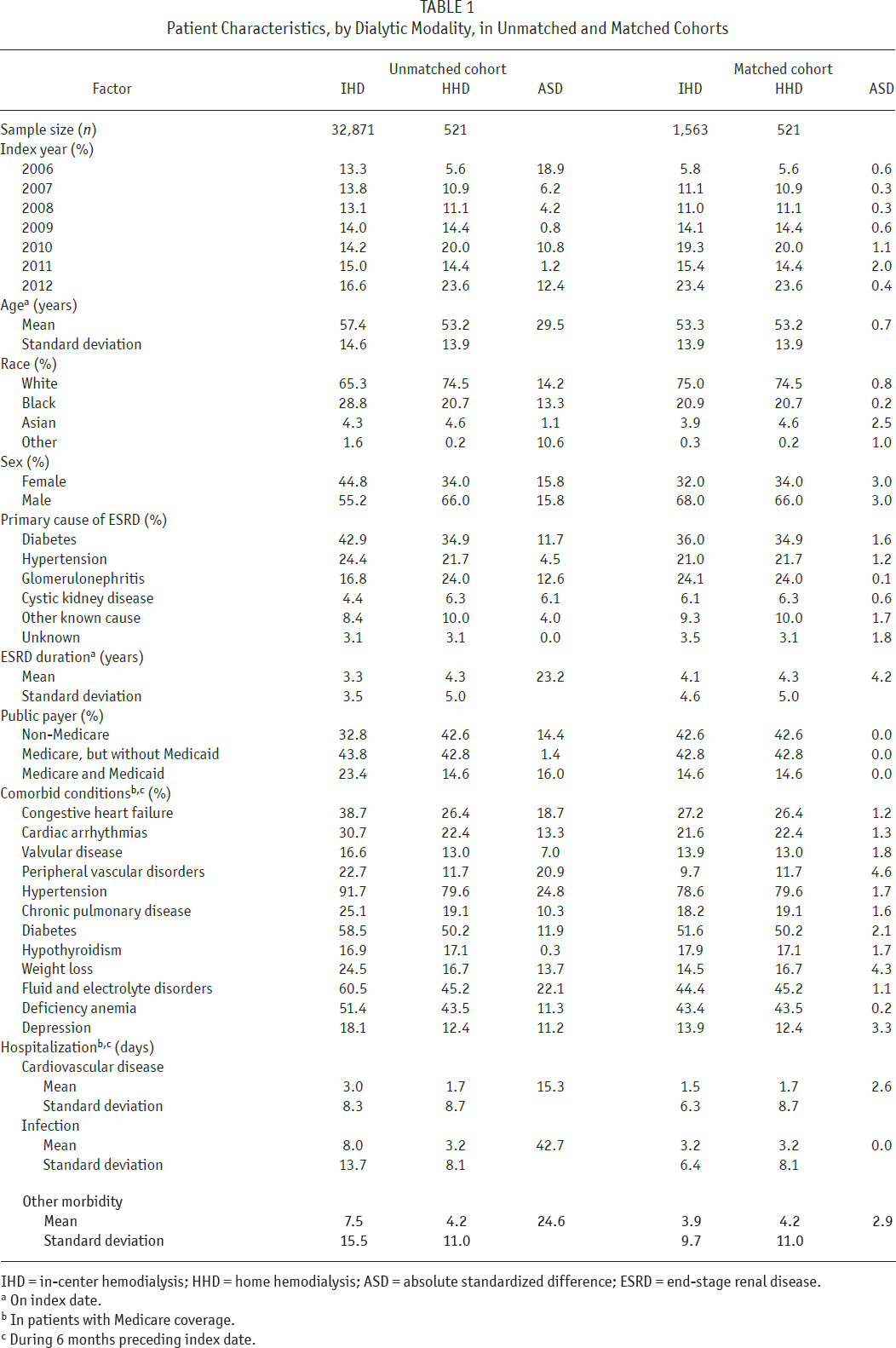
IHD = in-center hemodialysis; HHD = home hemodialysis; ASD = absolute standardized difference; ESRD = end-stage renal disease.
On index date.
In patients with Medicare coverage.
During 6 months preceding index date.
In ITT analysis, there were 142, 538, and 15,125 deaths in HHD, matched IHD, and unmatched IHD patients, respectively. Survival functions are displayed in Figure 2. For HHD versus matched IHD patients, survival was 89.1% versus 86.8% at 1 year, 80.5% versus 76.7% at 2 years, and 70.1% versus 62.4% at 4 years, respectively; for unmatched IHD patients, survival was 81.3%, 69.6%, and 53.2% at 1, 2 and 4 years, respectively. Survival differences varied by Medicare coverage status. In non-Medicare patients, survival in HHD patients was immediately higher than in matched IHD patients, and after 4 years, survival was 77.6% and 65.1% in HHD and IHD patients, respectively (Figure 3A). In contrast, in Medicare patients, survival in HHD patients was approximately equal to survival in matched IHD patients for 2 years (Figure 3B). Later, HHD patient survival was modestly higher; after 4 years, survival was 64.2% and 60.2% in HHD and matched IHD patients, respectively.
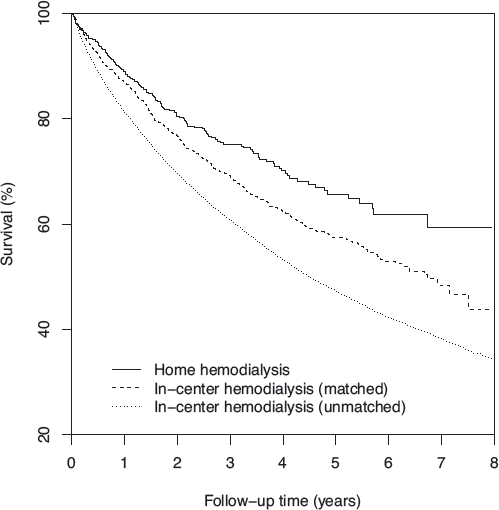
Survival among home hemodialysis patients, matched in-center hemodialysis patients, and unmatched in-center hemodialysis patients, in intention-to-treat analysis following peritoneal dialysis technique failure.
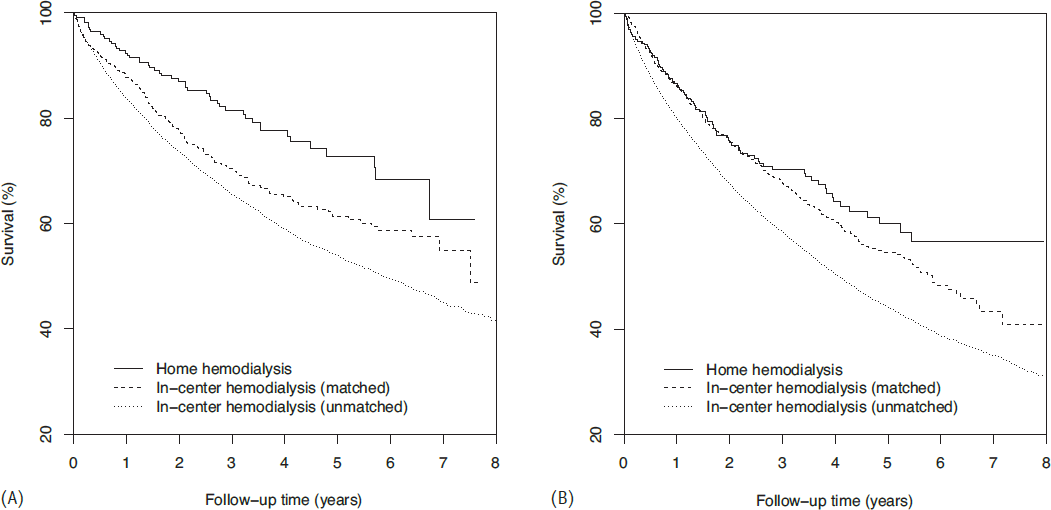
Survival among home hemodialysis patients, matched in-center hemodialysis patients, and unmatched in-center hemodialysis patients without Medicare (A) or with Medicare (B) coverage, in intention-to-treat analysis following peritoneal dialysis technique failure.
Hazard ratios (HRs) of death for HHD versus both unmatched and matched IHD patients are displayed in Table 2. Without matching, the aggregate HR was 0.56 in ITT analysis. With matching, but without comorbidity factors in propensity scoring, the aggregate HR was 0.67 in ITT analysis. Alternatively, with comorbidity factors in propensity scoring, aggregate HRs were 0.76, 0.74, and 0.68 in ITT, modified ITT, and as-treated analyses, respectively (p < 0.05 for all, Figure 4). Interval HRs became more protective with elapsing follow-up. In non-Medicare patients, HHD was associated with between 43% and 55% lower risk of death, and interval HRs exhibited no clear trend. In Medicare patients, HHD was associated with between 8% and 13% lower risk of death, and interval HRs indicated significant protective associations only after 2 years of follow-up. However, differences between HHD patients with and without Medicare coverage were modest (Supplementary Table 1); only mean ESRD duration (4.9 years in Medicare patients and 3.5 years in non-Medicare patients) was significantly different (p < 0.01 from Wilcoxon test). Inclusion of comorbidity factors in propensity scoring attenuated the aggregate HR in ITT analysis, entirely because of a large shift in interval HRs during follow-up years 1 and 2 in Medicare patients.
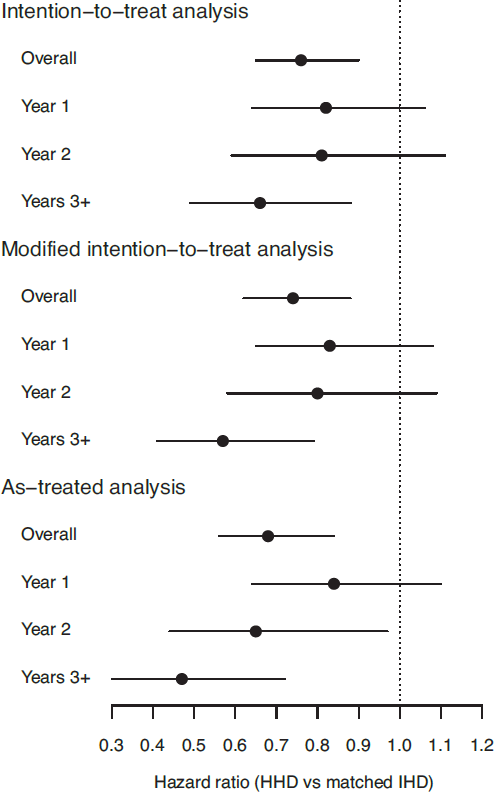
Hazard ratios of death for home hemodialysis versus matched in-center hemodialysis patients. HHD = home hemodialysis; IHD = in-center hemodialysis.
Overall and Interval Hazard Ratios of Death for Home Hemodialysis Versus Matched In-Center Hemodialysis Patients, in Aggregate and by Medicare Coverage Status

PS = propensity scoring.
Follow-up to the earliest of death, kidney transplant, or December 31, 2013, with kidney transplant classified as a competing event.
Follow-up to the earliest of death, kidney transplant, home hemodialysis technique failure, or December 31, 2013, with kidney transplant classified as a competing event and home hemodialysis technique failure classified as a censoring event.
In modified ITT analysis, there were 134 kidney transplants in HHD patients and 312 transplants in matched IHD patients. Transplant incidence functions are displayed in Figure 5. For HHD versus IHD patients, cumulative incidence estimates were 10.6% versus 8.8% at 1 year, 21.0% versus 15.2% at 2 years, and 28.8% versus 22.4% at 4 years, respectively. Hazard ratios of kidney transplant for HHD versus matched IHD patients are displayed in Table 3. The HRs of transplant in the matched cohort were 1.36, when HHD technique failure was ignored, and 1.34, when HHD technique failure was classified as a censoring event. Hazard ratios favored HHD in non-Medicare patients and were neutral in Medicare patients.
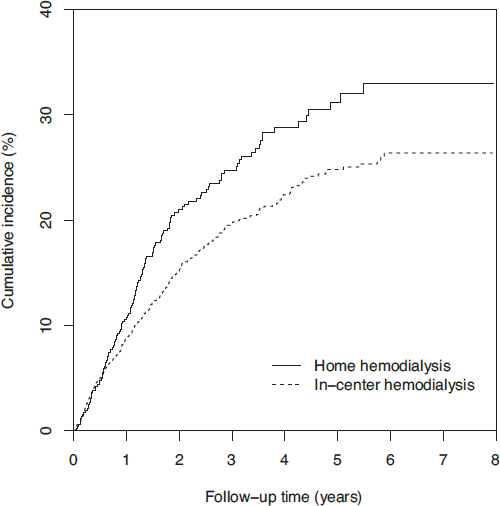
Cumulative incidence of kidney transplant among home hemodialysis and matched in-center hemodialysis patients, in modified intention-to-treat analysis following peritoneal dialysis technique failure. Death was classified as a competing event.
Overall Hazard Ratios of Kidney Transplant for Home Hemodialysis Versus Matched In-Center Hemodialysis Patients, in Aggregate and by Medicare Coverage Status
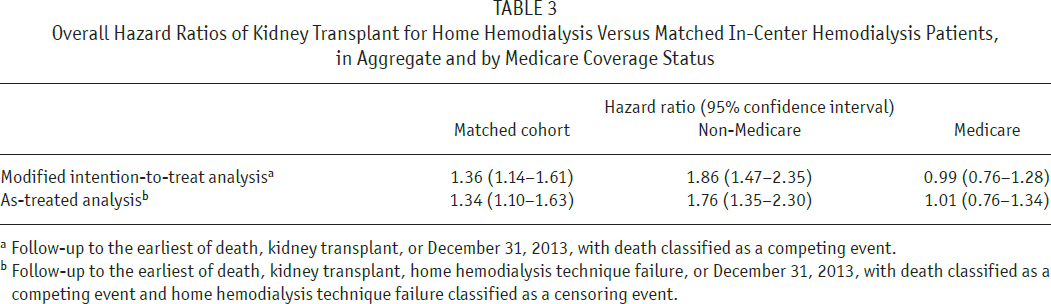
Follow-up to the earliest of death, kidney transplant, or December 31, 2013, with death classified as a competing event.
Follow-up to the earliest of death, kidney transplant, home hemodialysis technique failure, or December 31, 2013, with death classified as a competing event and home hemodialysis technique failure classified as a censoring event.
The cumulative incidence of HHD technique failure is displayed in Figure 6. Cumulative incidence estimates were 18.8% after 1 year, 26.7% after 2 years, and 29.8% after 3 years. There was little additional technique failure thereafter. In as-treated analysis, incidence estimates were 22.1%, 19.5%, and 31.0% for kidney transplant, death, and HHD technique failure, respectively, after 4 years; that is, slightly more than 27% of HHD patients remained on HHD after 4 years.
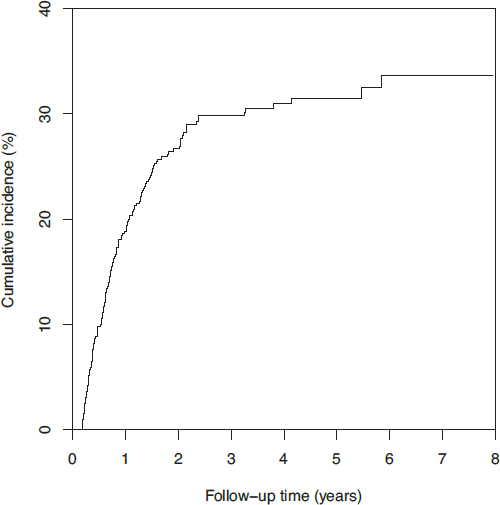
Cumulative incidence of home hemodialysis technique failure, in as-treated analysis following peritoneal dialysis technique failure. Both kidney transplant and death were classified as competing events.
In a sensitivity analysis, we relaxed inclusion criteria to permit patients with ≥ 1 day of PD treatment during the 6-month interval preceding the index date. We identified 1,017 HHD patients and 3,051 matched IHD patients. Hazard ratios of death (Supplementary Table 2) and kidney transplant (Supplementary Table 3) were qualitatively similar, although 95% confidence intervals were narrower.
Discussion
Increasing utilization of PD in incident ESRD patients will generate increasing counts of PD technique failures per year, unless the rate of technique failure falls more rapidly than the count of incident patients on PD rises. Although PD is associated with similar or better survival and quality of life relative to HD (13–15), effective strategies are needed to manage the transition from PD to HD in order to avoid induction of collateral risk when PD becomes either clinically ineffective or unviable. Home HD is a potentially effective strategy, because patients are already accustomed to dialysis not only in the home, but also on a daily basis. In the present study, we compared survival and kidney transplant incidence in patients who transferred from PD to HHD with matched patients who transferred from PD to IHD. We found that patients who transferred to HHD versus IHD had a lower risk of death and higher incidence of transplant. However, apparent benefits of HHD were attenuated in Medicare patients. Weaker associations in Medicare patients may have been attributable to superior control of confounding by comorbidity in this subset and differences in patient characteristics between non-Medicare and Medicare patients.
Technique failure remains a challenge in PD, although recent estimates of technique failure incidence in the US are surprisingly sparse. Guo and Mujais reported first-year technique failure rates between 18.7% and 20.5% in US cohorts from 1999 to 2001 (16), while Afolalu et al. reported first-year rates of 15.6% and 11.9% among small and large centers, respectively, in a regional cohort from 2001 to 2005 (17). A recent analysis of USRDS data showed that first-year technique failure rates in incident ESRD patients on PD varied between 19.5 and 20.8 events per 100 patient-years from 2005 to 2009 and between 18.1 and 18.9 events per 100 patient-years from 2010 to 2012 (18). In a matched cohort study, Weinhandl et al. reported that the risk of technique failure was higher with PD than HHD, even in incident ESRD patients (19). Nadeau-Fredette et al. corroborated this pattern among patients in Australia and New Zealand (20).
The switch from PD to HD is often forced by morbidity. The causes of PD technique failure in the Netherlands Cooperative Study on the Adequacy of Dialysis (NECOSAD) were infectious complications (41%), psychosocial or unknown reasons (30%), abdominal or catheter-related complications (19%), and under-dialysis or ultrafiltration failure (10%) (21). A similar distribution of causes was observed in a recent study of predominantly black and Hispanic patients in an urban US center (22). In our cohort, despite an inclusion criterion of survival for ≥ 2 months following the last day of PD, Medicare patients who transitioned from PD to IHD accumulated nearly 19 hospitalized days during a 6-month transfer interval, over 3 times the background rate of 11 hospitalized days per year among Medicare patients on dialysis in 2014 (1). Unsurprisingly, survival after PD technique failure is generally poor; Piraino et al. reported 15% mortality at 6 months after technique failure (23), whereas we reported 19% mortality at 14 months after technique failure in patients who completed ≥ 2 months of IHD. High rates of hospitalization and death around the time of PD technique failure suggest room for improvement in clinical care.
Planning a switch from PD to HD may improve outcomes. Boissinot et al. investigated differences between 37 patients who switched from PD to HD without planning and 23 patients who switched with planning (24). Those with planned transitions were less likely to be hospitalized and more likely to have a functioning arteriovenous fistula at 2 months after the last day of PD. Logically, planning may involve consideration of a switch from PD to HHD, as PD patients have already demonstrated a willingness to undergo dialysis in the home. However, transitions from PD to HHD have been relatively rare. In a cohort of patients from Australia and New Zealand, 5.4% of PD technique failure cases transitioned to HHD (10). In light of the numbers of IHD and HHD patients in our unmatched cohort, it appears that approximately 2% of PD technique failure cases in the US transitioned to HHD. Systematic investigation of barriers to transitioning patients from PD to HHD is needed.
We found that transitioning to HHD after PD technique failure was associated with a lower risk of death and higher incidence of kidney transplant than transitioning to IHD. The lower risk of death with HHD may be attributable to several factors that are inherent in the modality, including a likely planned transition from PD, cardiovascular benefits of intensive HD, and selection bias. Patients who “crash” into HD have poor outcomes, as these patients have not undergone ESRD planning and often initiate HD with vascular access via catheter (25). Many PD patients who suffer technique failure similarly “crash” into HD. It is plausible that the transition from PD to HHD was planned for most patients, so that positive associations reflect the positive impact of careful planning, rather than any clinical effect of HHD. Planning would likely involve placement of an arteriovenous fistula, the use of which is associated with a lower risk of infection than vascular access via catheter (26). Unfortunately, we lacked data about vascular access modality during the transition from PD to HD. It is also plausible that HHD was associated with improved clinical outcomes because HHD facilitated treatment for 5 to 6 sessions per week in most patients. Intensive HD lowers pre-dialysis blood pressure (7,27,28), ameliorates left ventricular hypertrophy (6,27,29), and reduces ultrafiltration rates (30). In addition, treatment for 5 or 6 sessions per week almost always eliminates 2-day gaps between consecutive sessions; such gaps are associated with higher risks of death and cardiovascular hospitalization (31–33). On the other hand, associations in ITT analysis were probably attenuated by a relatively high incidence of HHD technique failure.
We observed attenuated associations of HHD with clinical outcomes in Medicare versus non-Medicare patients. In the former group, we used claims to measure comorbidity factors, but in the latter group, we lacked claims and could not measure such factors. In Medicare patients, inclusion of comorbidity factors in propensity scoring shifted the aggregate hazard ratio of death for HHD versus matched IHD patients from 0.75 to 0.92 in ITT analysis. Thus, it is possible that reported hazard ratios of both death and kidney transplant in non-Medicare patients were biased in favor of HHD. Still, the shift of relative hazard in Medicare patients was due entirely to shifts in the first 2 years of follow-up. Detailed adjustment for comorbidity in non-Medicare patients might have little effect on relative hazards after the first 2 years of follow-up. It is also notable that ESRD duration at PD technique failure was 1.4 years greater in HHD patients with versus without Medicare. Theoretically, longer time on PD before technique failure may limit the potential benefit of HHD thereafter.
Our study has several limitations. First, selection bias is inherent in observational studies that compare dialytic modalities. We used propensity scoring to control measured confounding, but we could not address multiple factors, including cause of PD technique failure, biochemistry and residual kidney function, and vascular access modality. Only a randomized controlled trial can eliminate the threat of confounding, but the feasibility of such a trial is unclear. Second, statistical power was limited. We identified only 521 US patients who transferred from PD to HHD during a 7-year interval. Nonsignificant associations are not necessarily evidence of null associations. Third, HHD patients used low-volume dialysate, thus limiting the applicability of results. Finally, a transition from PD to HHD likely represents highly coordinated care; results should be interpreted in view of multiple mechanisms, including HHD itself.
In conclusion, we found that while transfer from PD to HHD has been relatively rare in the US, such a transfer was associated with a lower risk of death and higher incidence of kidney transplant than transfer from PD to IHD. Because PD patients are already accustomed to dialysis in the home, expanded prescription of HHD after PD technique failure may primarily demand anticipation of failure and planning of a clinical response. To refine the process of transitioning patients from PD to HHD, dialysis providers and researchers should continue to assess and report outcomes in patients who transition accordingly.
Footnotes
Dr. Kansal has received compensation as a physician consultant to NxStage Medical and was previously a member of the Scientific Advisory Board of NxStage Medical. Dr. Morfin has received compensation as a physician consultant to NxStage Medical and Satellite Healthcare and is currently a member of the Scientific Advisory Board of NxStage Medical. Dr. Weinhandl is an employee of NxStage Medical.
The data reported here have been supplied by the United States Renal Data System. The interpretation and reporting of these data are the responsibility of the authors and in no way should be seen as an official policy or interpretation of the US government.
