Abstract

Keywords
One of the potential, although extremely rare, complications of long-term PD is encapsulating peritoneal sclerosis (EPS); it is associated with high morbidity related to bowel obstruction and malnutrition. The reported mortality of this condition is around 50%, usually within 12 months of the diagnosis (6,7). Mortality rates, however, depend upon severity of the disease, and not all deaths are due directly to EPS alone (7). It has been advocated by some that there should be a time limit for PD to prevent patients from developing this potentially devastating complication.
The principal aims of this paper are:
To review existing information about the epidemiology of EPS and its risk factors;
To determine whether there are any predictors for the development of EPS that would guide the decision to stop PD and transfer to HD; and
To reach a consensus that should be given to nephrologists and their patients about the length of time that is advisable to remain on PD.
Guidelines for EPS
Guidelines on the topic of EPS have been issued by the Japanese Society for Peritoneal Dialysis (8), the UK Renal Association (9), and the Dutch EPS Registry (10). When reading the guidelines, it is clear that issuing evidence-based guidelines on EPS is being hampered by:
A lack of well-defined diagnostic criteria, especially to determine early stages of EPS;
A lack of interventions that consistently improve outcome of EPS, even after PD has been stopped;
The fact that EPS may develop or symptomatically progress after discontinuation of PD (11) and transfer to HD or transplantation, making guidance about when to transfer patients electively from PD to HD particularly difficult;
A lack of epidemiological data in different dialysis populations relating length of time on PD to odds of developing EPS (EPS has, however, rarely been reported to occur before 3 years on PD); and
Difficulties determining individual risk and impact for transfer to HD given their comorbidities, personal situation, tolerance of HD, and need for vascular access.
Epidemiology of EPS
Encapsulating peritoneal sclerosis is a very uncommon complication of PD. Its risk of occurrence has been demonstrated to vary markedly among centers, among countries, and over time (Table 1). Based on the reports of various single-center, multi-center, and national registry observational cohort studies, the prevalence of EPS has been observed to vary between 0.4% and 8.9%, its incidence rate between 0.7 and 13.6 per 1,000 patient-years, and its risk of occurrence after 5 years on PD between 0.6% and 6.6% (Table 1). A variable (0 - 71%) proportion of EPS cases are not diagnosed until after completion of PD (12,13), including following kidney transplantation (14). The appreciable variability in observed EPS incidence, prevalence, and timing of occurrence may be related to genetic factors, differences in PD care (e.g. daily prescribed PD volumes, dialysate glucose exposure, use of biocompatible fluids, peritonitis prevention strategies, time spent on PD, etc.), or limitations of the studies published to date, including ascertainment bias, detection bias (e.g. under-diagnosing mild cases, over-diagnosing simple peritoneal fibrosis, lack of a reliable screening test, and failure to monitor patients following PD completion), inadequate length of follow-up (considering the usually long lag time to development of EPS following PD commencement), and inadequate sample size (thousands of patients need to be followed for many years to provide sufficiently precise statistical estimates of risk for informing clinical decision-making) (15). Most studies to date have also failed to consider the competing risks of death and kidney transplantation (15).
Studies Examining the Epidemiology of EPS *
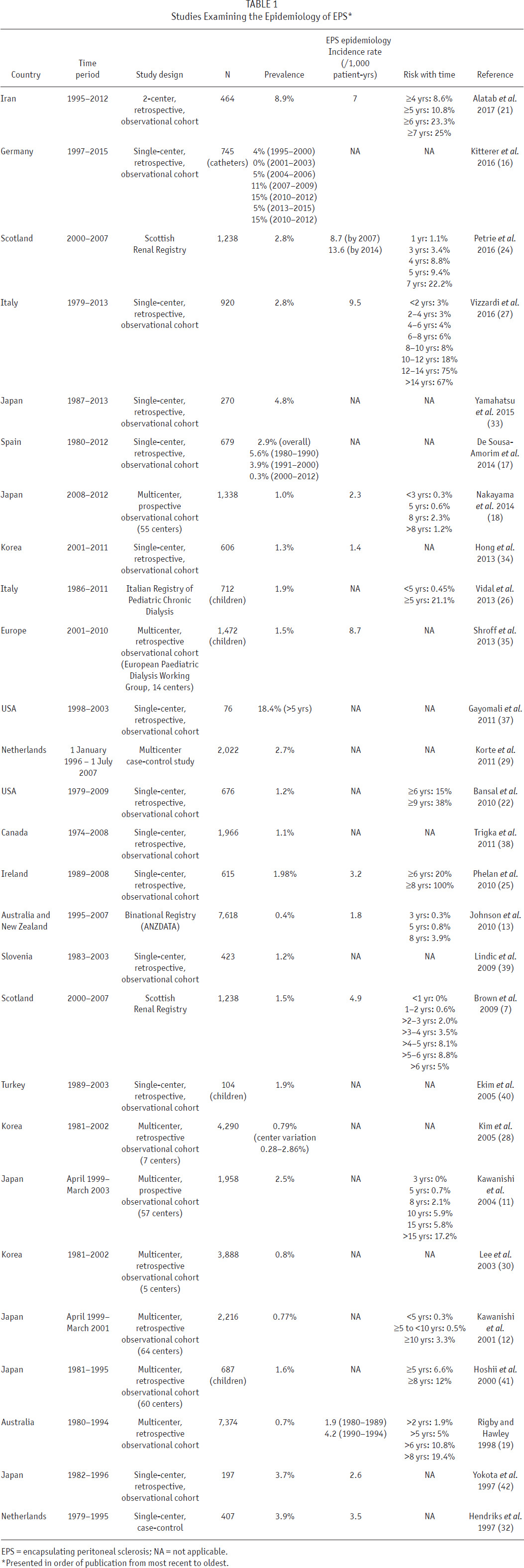
EPS = encapsulating peritoneal sclerosis; NA = not applicable.
Presented in order of publication from most recent to oldest.
Some studies from Germany (16), Spain (17), Japan (18), Australia (13,19), and the Netherlands (20) have further suggested that the incidence of EPS may be decreasing over time, although the reported incidence/prevalence estimates have been too imprecise to be certain of this. If EPS is indeed becoming less common, the reasons remain uncertain.
Most studies have consistently identified increasing PD duration as a key risk factor for development of EPS (Table 1) (11,12,18,21–27). Other parameters that have been identified in at least 1 study to be possible risk factors for EPS include higher dialysate glucose exposure, use of conventional PD solutions (as opposed to biocompatible PD solutions), peritonitis (frequent, severe, or prolonged), younger age (presumably because of lower competing risk of death), abdominal surgery, β–blocker use, icodextrin use, kidney transplantation, UF failure, and higher peritoneal solute transport rate (PSTR) (13,20–22,25,28–32). However, these data have been too imprecise and/or inconsistent to be considered reliable at this time.
It should be stressed that all current data indicate that the majority of patients receiving PD for a long duration do not develop EPS. Clinicians switching patients who have been on PD for several years to HD in an attempt to pre-emptively circumvent the risk of EPS (0.7 – 9.5 episodes per 1,000 patient-years) should consider the alternative risks of HD complications, including arteriovenous fistula failure (47% at 1 year) (42), bacteremia (137 episodes per 1,000 patient-years) (43), and endocarditis (1.7 – 4.8 episodes per 1,000 patient-years) (45,46). Other devastating complications of HD with consequences as potentially dire as EPS include embolic stroke from vegetations caused by endocarditis (47), other metastatic complications of bloodstream infections including osteomyelitis and spinal epidural abscess (48), and central venous stenosis (49). Although these conditions appear to be at least as prevalent as EPS in PD patients, the literature on these complications is scant.
Diagnosis of EPS
The diagnosis of EPS is based on a combination of structural (e.g. computed tomography [CT] scan appearance) and functional features (intermittent subacute bowel obstruction). It is important to be clinically aware of the possibility of EPS for many years after stopping PD; failure to listen to the patient and his/her symptoms may lead to a delay in diagnosis (50). Encapsulating peritoneal sclerosis presents after withdrawal from PD in the majority (70 – 90% in some series) of patients (11,51) and the time from cessation of PD until the development of EPS has been reported as up to 5 years (52). The diagnosis is clinical, relies on a constellation of symptoms, and can be confirmed radiologically. Changes in the peritoneum need to be differentiated from those of long-term PD. Only a fibrous cocoon wrapped around the bowel is diagnostic; a thickened peritoneal membrane and intra-abdominal adhesions are common in long-term PD and after peritonitis, particularly tuberculous peritonitis, and are therefore not diagnostic (52).
Clinical Features of EPS
The diagnosis of EPS is based on a combination of bowel obstruction and features of encapsulation due to peritoneal fibrosis. Symptoms such as anorexia, nausea, vomiting, and weight loss are common. In addition, the step-wise process of symptom progression is important. If the capsule formed is too thin to impair intestinal peristalsis, EPS does not develop (encapsulating stage). However, if the capsule thickens with time, bowel obstruction symptoms appear (ileus stage). These symptoms improve by temporary fasting, but recur several months later. If the time to recurrence gradually shortens, EPS is diagnosed. Clinically, this progression manifests itself with early symptoms (bloody ascites, appetite loss, nausea, diarrhea, and abdominal pain), progressing to more severe symptoms, including constipation and abdominal mass accompanied by severe malnutrition and weight loss. Sometimes, early EPS presents with an inflammatory state including fever, general fatigue, and slight weight loss, with an elevated C-reactive protein, anemia, and hypoalbuminemia. The intermittent progression, and therefore symptoms, of EPS is a useful distinguishing feature from other gastrointestinal disorders (53).
Radiological Diagnosis of EPS
Of the techniques available, computed tomographic (CT) scanning has been reported as having the most discriminant value (52). It is also widely available and has the greatest reproducibility. Computed tomographic scanning is therefore recommended as the first investigation. The features that have been shown to have a high degree of agreement among radiologists are peritoneal calcification, bowel thickening, bowel tethering, and bowel dilatation (54,55). It should be stressed that finding these changes on CT scan does not suffice to make the diagnosis of EPS, especially not in the absence of the symptoms described above. Many long-term PD patients will have thickening of the peritoneal membrane without any features of EPS.
Pathological Features of EPS
A characteristic macroscopic appearance is observed at laparotomy or laparoscopy. Histological changes that are characteristic of EPS have been described but are not specific and overlap with membrane changes that occur with UF failure and infectious peritonitis in long-term PD (56). Thus, an “opportunistic” peritoneal biopsy at the time of incidental abdominal surgery in the absence of other features of EPS may be misleading and should not be used to make a diagnosis of EPS.
Membrane Transport Characteristics
Changes in PSTR are frequent in PD. It has been recognized for many years that UF capacity falls and there is an increase in PSTR prior to development of EPS (57). These membrane changes are, however, also commonly observed in patients on long-term PD who do not develop EPS. The Pan-Thames EPS study showed that, while the majority of patients had high PSTR, some developed EPS in the absence of these membrane changes and had good UF (6). These changes are discussed in more detail in the section on predicting EPS.
Management of EPS
It is generally accepted that, after a diagnosis of EPS, PD should be discontinued and the patient transferred to HD. However, it should be considered that some cases of EPS are clinically less severe and potentially could worsen upon stopping PD. The eventual risks of HD (access, access-related infection, hemodynamic intolerance, lifestyle issues, and patient preference) should also be considered carefully in a discussion with the patient about the best future renal replacement therapy option. Generally, the PD catheter is removed on discontinuing PD. Some patients in Japan have been managed by leaving the catheter in situ to apply regular peritoneal lavage (58,59). It is unclear whether this process has any beneficial effects by removing mediators of the peritoneal fibrotic process, or whether the catheter and irrigation fluid could act as a further stimulus to the EPS process.
Nutritional support (often by parenteral nutrition) is crucial in patients with EPS, many of whom will recover with conservative treatment (60). In addition, drug therapies that have been reported to have beneficial effects in EPS include corticosteroids (11,61), tamoxifen (62–64), and immunosuppression (65–67). The fact that EPS can actually develop following renal transplantation in patients already receiving corticosteroids and/or other immunosuppressive agents may, at least theoretically, argue against any therapeutic benefit of these drugs. Most of these reports are limited to isolated cases or relatively small series, have not been uniformly successful, and are potentially limited by other interventions, selection, and positive publication biases. The 2 larger studies gave conflicting results. The study from the Netherlands (63 patients) found that the mortality of the tamoxifen group was lower than those not given the drug (64). In contrast, in a large UK series (111 patients), there was no difference in outcomes for patients treated with steroids, immunosuppression, tamoxifen, or combinations of these compared with patients that were not (6). No definitive conclusions can therefore be drawn at this time about their value in the management of EPS.
On the basis of successful surgical results from Japan (51,68,69), aggressive surgical treatment for when there was no improvement of bowel obstruction was started in the UK (70) and Germany (71,72). From these reports, it appears that with surgery, EPS mortality rate declines to 32 – 35%. It must be stated, though, that for surgical results to be successful, the surgical team must have a thorough understanding of the pathology of EPS. Such surgery should therefore only be done in specialist regional centers that can provide appropriate surgical training and patient support.
Renal Transplantation and EPS
Although there were reports from the UK and the Netherlands (73 74-75), the numbers from these reports are small and there is still no evidence of a genuine sustained increase in the occurrence of post-transplant EPS. Balanced against this, there have been isolated reports of dramatic resolution of established EPS following renal transplantation, possibly as a result of immunosuppression (76). Indeed, a prior diagnosis and treatment of EPS is not a contraindication to transplantation. Encapsulating peritoneal sclerosis occurs after transplantation only in patients who have been exposed to PD for several years; there appears to be no risk if patients have been on PD for a short time. Ideally, therefore, patients should be transplanted within 3 – 4 years of starting PD. This requires appropriate patient education, efficient workup, access to the national deceased organ waiting list, and encouragement of living donation. The same is true for patients on HD so that patients can benefit from the improved survival and quality of life associated with successful transplantation.
Screening for EPS
There is no reliable screening tool established for EPS. Change in PSTR across the peritoneal membrane is not particularly helpful. A rise in PSTR is common in patients on long-term PD and therefore is commonly found in patients subsequently developing EPS. As discussed, though, EPS can also occur in patients with slow PSTR. Similar observations and conclusions apply to loss of UF.
Computed tomographic scanning has been proposed as a screening tool, but EPS can occur within a year or less of a normal CT scan in asymptomatic patients (77). In contrast, long-term patients on PD with minor abdominal symptoms in this study were sometimes found to have minor CT scan abnormalities; all such patients then progressed to the full EPS clinical syndrome on stopping PD.
Predicting Eps
Epidemiology
When predicting the absolute risk of EPS, for both clinical and statistical reasons, it is not possible to ignore the competing risk of death. Methods such as the Kaplan-Meier estimator will not account for competing risks and therefore will overestimate the level of risk (78). Work for the Peritoneal Dialysis Competitive Risk Analysis for Long-Term Outcomes (PD-CRAFT) study using data from the ANZDATA registry and the Scottish Renal Registry has verified this phenomenon, showing increasing disparity with longer follow-up. As is clearly shown in the studies of the epidemiology of EPS, duration of PD is strongly associated with EPS risk. In competing risks prediction models for patients in ANZDATA 3 and 5 years after the start of PD using age and primary renal disease as predictors of mortality and duration of PD as a predictor of EPS, most of the variability in EPS incidence was explained (unpublished data from PD-CRAFT), i.e. the ‘real-world’ risk of EPS is almost entirely due to the risk of death and the duration of PD. No prediction models are available for clinical usage currently as the baseline risk is not yet clear due to the variability in EPS incidence described previously.
Despite the very strong effect of the duration of PD and risk of mortality, clinicians may wish to further stratify patients at risk of EPS. Data on this are not robust enough for strong guidance as there are no datasets with sufficiently detailed information on the very large populations required to study EPS. There are several small, mostly single-center, studies that have examined membrane function testing and dialysate biomarkers for this purpose, and they have all been predicated on the same pathophysiological model for membrane damage.
Membrane Function
As already discussed, longitudinal follow-up of peritoneal membrane function has shown that EPS is associated with progressive transport defects, including excessive increase in PSTR and loss of UF capacity as compared with control long-term PD patients (6,29,32,79–88) (summarized in Table 2). As PSTR predominantly reflects peritoneal inflammation (89,90) and EPS is an inflammatory condition, changes in PSTR appeared a promising risk marker for epidemiological and pathophysiological reasons. More recent longitudinal studies carefully matched for duration of PD to account for the association between PSTR and duration of PD (91) suggest that differences in PSTR are apparent only in the later stages prior to EPS, therefore limiting its application as a potential risk indicator (83,88).
Case Series and Registry Studies Reporting Data on Membrane Function in Patients with EPS *
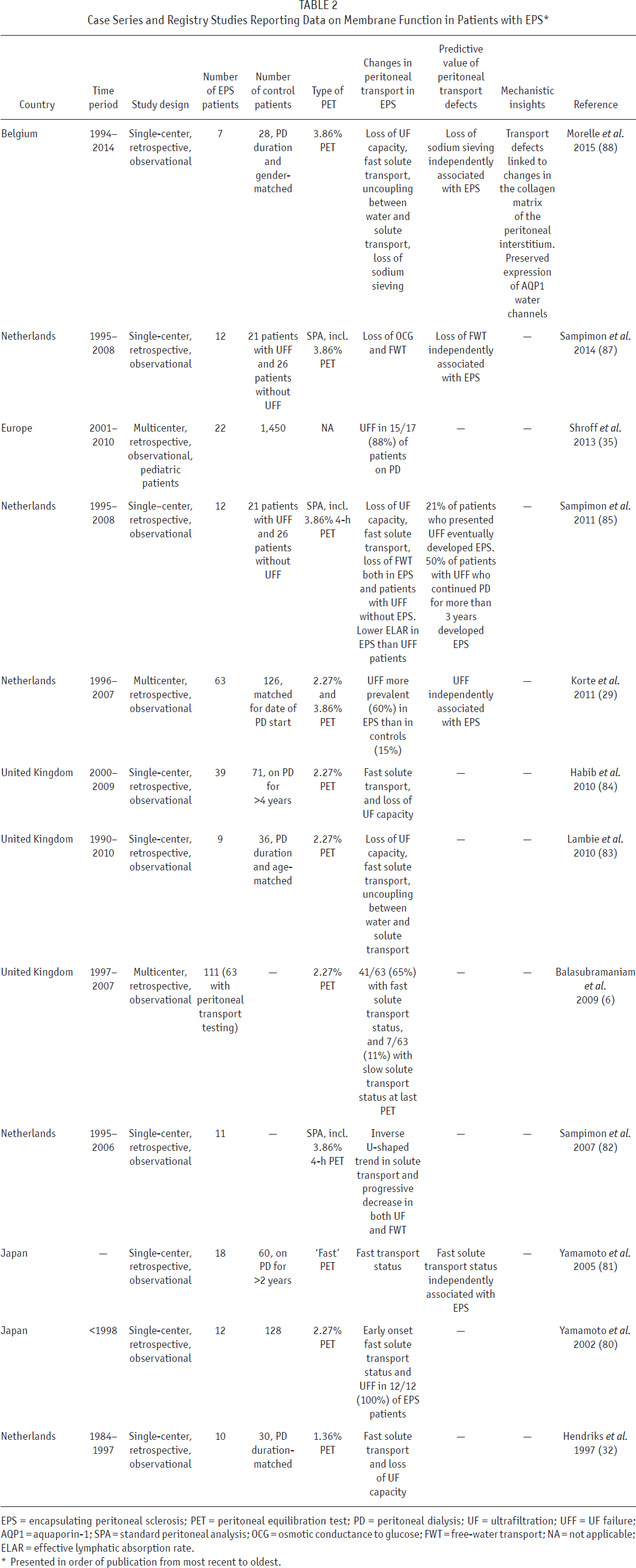
EPS = encapsulating peritoneal sclerosis; PET = peritoneal equilibration test; PD = peritoneal dialysis; UF = ultrafiltration; UFF = UF failure; AQP1 = aquaporin-1; SPA = standard peritoneal analysis; OCG = osmotic conductance to glucose; FWT = free-water transport; NA = not applicable; ELAR = effective lymphatic absorption rate.
Presented in order of publication from most recent to oldest.
Progressive uncoupling between solute and water transport across the EPS peritoneal membrane (a loss of UF capacity disproportionate to the rise in PSTR) (92), thought to be due to fibrosis, led to the hypothesis that functional measures of this fibrosis may be a more reliable and earlier indicator of the risk of EPS (83). This hypothesis was verified in case-control series demonstrating a reduction in osmotically-driven water flow across the peritoneal membrane (estimated by either sodium sieving, free-water transport, or direct assessment of osmotic conductance) in EPS patients (89,90). In daily clinical practice, the modified 3.86% glucose-based peritoneal equilibration test (PET) accurately evaluates UF capacity, allows the diagnosis of UF failure (defined as a net UF < 400 mL after 4 h), and provides the opportunity to determine sodium sieving, estimated either by the change in dialysate-over-plasma ratio of sodium or by the dip in dialysate sodium concentration during the first hour of the dwell (93). As a biochemical measure, it is theoretically more reliable than volumetric assessment of UF, which may be highly variable and influenced by other factors such as catheter patency.
The combination of functional and structural analysis of the membrane of patients with EPS demonstrated that reduced osmotic water transport is directly related to the degree of fibrosis and to changes in collagen density and structure in the peritoneal interstitium (88), in line with predictions based on the serial pore-membrane/fiber matrix and distributed models (94 95-96). Of note, the low osmotic conductance and abolition of sodium sieving are not associated with any change in the expression of aquaporin-1 (AQP1) water channels in the peritoneal capillaries of patients with EPS (97,98).
Biomarkers
As the current pathophysiological model for EPS risk is based on chronic inflammation and fibrosis within the peritoneum, the use of biomarkers from the peritoneal effluent has been suggested to improve the prediction of EPS. They have therefore been tested to assess the risk of EPS in 4 different case control studies either fully or partially matched for duration of PD using prospectively collected dialysate samples (99-102). The consistent finding is that several inflammatory cytokines (interleukin-6, tumor necrosis factor-α, monocyte chemoattractant protein-1, chemokine ligand 15, and plasminogen activator inhibitor-1) are slightly elevated up to several years before EPS, supporting a role for chronic peritoneal inflammation in the physiopathology of EPS. However, the levels of biomarkers in the effluent show significant variability (a lognormal distribution) and the differences between cases and controls, whilst statistically significant, have been small and therefore of debatable clinical significance. Whether functional measurements or biomarkers, when combined with PD duration and risk factors for mortality, improve the prediction of EPS has not been tested prospectively.
Prevention of EPS
As discussed, the incidence of EPS increases significantly with time on PD, particularly after 5 or more years of treatment, but the majority of long-term PD patients will not develop EPS. Importantly, EPS may develop or worsen after stopping PD. There are no prospective data demonstrating any benefit of pre-emptively switching long-term PD patients to HD. A modality switch could also have significant adverse psychosocial and medical implications for patients, which need to be considered on an individual basis. Long-term access for HD also needs to be discussed and planned with the patient; the risk of infection from temporary HD access is considerably higher than the low risk of EPS at some point in the future.
If considering switching patients from long-term PD to HD pre-emptively because of concern about risk of EPS, it may be appropriate to select those patients with potentially adverse features for a high risk of PD technique failure such as high and rising peritoneal permeability, low UF capacity, difficulty in fluid balance control, and requirement for high glucose concentration dialysate, as well as those with frequent episodes of peritonitis. These features would then possibly select those at greater risk of PD technique failure. The effect of this management on EPS risk is, however, unknown. It would be important to ensure that these patients are monitored specifically for clinical features of EPS, which can develop some time after switching to HD. Informing patients of the early signs of EPS was recommended in a study of patient experiences in particular at the time of transition to HD as symptoms were often subtle but not understood. Providing written information is recommended to give the patients control and enable them to discuss their concerns with the healthcare team (50).
Other strategies that have been suggested for EPS prevention include minimization of dialysate glucose administration and prescription of “biocompatible” PD fluids. In comparison studies of peritoneal biopsy specimens of low GDP and conventional acidic solutions, the neutral solutions with low GDP were associated with less peritoneal membrane fibrosis and vascular sclerosis through suppression of advanced glycation end-product accumulation (103). Moreover, the multicenter prospective observation study (the NEXT-PD study) confirmed fewer cases of EPS (18). The authors postulated that the use of neutral solutions with low GDP may have contributed to this observation along with other factors such as discontinuing PD in high-risk patients and minimizing use of high-glucose fluids with use of icodextrin.
Summary Statements
Encapsulating peritoneal sclerosis is recognized as a potential and rare complication of long-term PD, occurring in patients on PD for more than 5 years. Although the incidence of EPS then increases with further time on PD, the condition remains infrequent and the majority of long-term PD patients are not affected.
The decision about when to discuss EPS as a potential complication of long-term PD therapy should be undertaken at some point with the patient—not necessarily at the start of PD but more reasonably at the 3 – 4 year point of therapy.
Encapsulating peritoneal sclerosis is associated with considerable morbidity and mortality. It is therefore important to develop strategies to reduce the risk to an individual patient.
No single strategy to reduce the risk of EPS has been proven in clinical trials, but there is some evidence to support the following:
Minimizing dialysate glucose exposure, although it is important to ensure that fluid volume status is not compromised as a result
Preventing acute PD-related peritonitis using interventions recommended by the ISPD peritonitis guidelines (104)
Use of neutral-pH, low-glucose degradation product dialysis solutions (low-grade evidence only)
The more severe clinical features of EPS with bowel obstruction, poor nutrition, and ascites may develop even if PD is discontinued (patient transferred to HD or transplanted).
There are no specific predictors for the development of EPS:
Although many patients with EPS have high PSTR, this is not true for all patients and this is a common finding in patients on long-term PD.
There is no evidence that CT scanning has any value in predicting EPS.
Progressive loss of osmotic conductance to glucose (uncoupling between water and solute transport, altered sodium sieving, decreased free-water transport) may reflect the development of peritoneal interstitial fibrosis and may help identifying patients at risk of EPS. However, this needs to be confirmed in prospective studies.
Although changes in peritoneal membrane function, loss of UF and frequent peritonitis are poor predictors of EPS, they are factors suggesting that transfer to HD should be considered and discussed with the patient, if appropriate, to optimize dialysis delivery. Such patients should be monitored closely for possible development of EPS if changing dialysis modality to HD.
Older patients and those with comorbidities have a limited life expectancy when starting dialysis. Few will therefore survive long enough on PD to be at risk of developing EPS. Such patients are unlikely to be candidates for transplantation so their quality of life on dialysis is very important. In considering discussing the risk of EPS with such a patient, it is therefore important to consider realistically their life expectancy, the feasibility of HD for that patient, and how this would affect their quality of life. Discussions with the patient should be part of a shared decision-making process about overall prognosis and goals of care.
Conclusion
Encapsulating peritoneal sclerosis is a rare condition. There is no evidence to withhold PD as a treatment option because of fear of development of EPS. There is insufficient evidence to support a single rule about optimal length of time on PD to avoid the risk of EPS
Each long-term patient needs to be considered individually, taking into account the following factors:
Age and prognosis of patient;
Length of time on PD;
Quality of PD (dialysis adequacy, ultrafiltration, peritonitis frequency);
Access to and suitability for transplantation;
Potential risk of HD in the particular patient (hemodynamic stability, vascular access); and
Quality of life of the patient.
All these items should be discussed and any decision arrived at by shared decision-making.
Footnotes
We have read and understood Peritoneal Dialysis International's policy on disclosing conflicts of interest and declare the following interests: EB has received speaker fees from Baxter Healthcare and Fresenius Medical Care; TM has speaker, travel, and consultant fees from Baxter Healthcare; JB has consultant fees from Baxter Healthcare, Amgen, Otsuka, Keryx, Rockwell Scientific, speaker fees from Amgen, DaVita Healthcare Partners, Baxter Healthcare; JM has travel grants and speaker fees from Baxter Healthcare and Fresenius Medical Care, and research grant from Baxter Healthcare; WvB has speaker fees and research grants from Fresenius Medical Care, Baxter Healthcare and Gambro; DJ has consultancy fees, research grants, speaker's honoraria and travel sponsorships from Baxter Healthcare and Fresenius Medical Care.
