Abstract
In natural conditions the oocyte and embryo are subjected to ever-changing dynamic processes. However, the routine assisted reproductive technologies today involve the use of static in vitro culture systems. The objective was to determine whether there is any difference in the viability of embryos after in vitro culture under static and mechanical microvibration conditions. The viability of embryonic cells (9,624 embryos) generated from 4,436 couples after in vitro culture was evaluated. For groups ≤29, 30-34, 35-39, and ≥40 years, the following rates of high-quality embryos without fragmentation (two to four blastomeres on day 2; six to eight blastomeres and compacting morula on day 3; blastocyst, expanded and hatching blastocyst on day 5) were detected (static vs. vibration, respectively): 65% versus 71%, 44% versus 69%, 67% versus 76% (for statistically significant differences between respective rates in these three groups, p <0.05), and 67% versus 66% (p > 0.1). The following baby-take-home rates were determined for groups ≤29, 30-34, 35-39, and ≥40 years (static vs. vibration, respectively): 30% versus 31% (p > 0.1, increasing only on the level of tendency), 28% versus 37%, 23% versus 29%, and 9% versus 15% (differences between respective rates in these three groups with p < 0.05). It was concluded that in vitro culture of embryos under microvibration (with a mimic of conditions in nature whereby oviductal fluid is mechanically agitated by the epithelial cilia) significantly increases the baby-take-home rate for patients 30 years and older.
Introduction
Infertility is the inability of a person to reproduce by natural means. It is usually not the natural state of a healthy adult organism 1 . In that way infertility is a disease, in essence. A reasonable question to propose is whether cell transplantation may be a viable option for the treatment of this disease. In fact, the most effective method of treatment for sterility is harvesting and transplanting embryonic cells (embryo).
Embryo transplantation refers to a step in the process of assisted reproduction in which embryos are placed into the uterus of a female with the intent of establishing a pregnancy. This technique is often used in connection with in vitro fertilization (IVF). In vitro embryo culture is a stage of IVF in which embryos are allowed to grow in an artificial medium 1 .
IVF is a process by which an oocyte is fertilized by sperm in vitro. The process involves monitoring and stimulating a woman's ovulation, removing oocytes from the ovaries, and letting sperm fertilize them in vitro. The fertilized oocytes are cultured for 2-6 days in a growth medium and are then transplanted into the uterus with the intention of establishing a successful pregnancy. It is a technique of assisted reproductive technology for treatment of sterility 1 .
All fertilization steps in nature (receiving the ovulated oocytes, fertilization of oocytes, and embryonic development with subsequent transport to the uterus) take place inside the fallopian tube. The tubal mucosa is arranged as longitudinal folds and consists of a single layer of cuboidal or columnar epithelium. The major cell types of this epithelium are secretory and ciliated cells 2 , whose cilia have a vibration 3 (beat). Some investigations have shown an increase in ciliary beat frequency to 5.8 ± 0.3 Hz in the fimbrial section of the tube during the secretory phase 2 compared with the proliferative phase (4.9 ± 0.2 Hz).
What is known about microvibration in general? Vibration is a natural phenomenon that refers to mechanical oscillations about an equilibrium point. Since life began, the Earth has subjected all living things to a natural pulsation frequency. This natural phenomenon was predicted in 1952 4 and named the global electromagnetic resonance phenomenon or Schumann resonances. Schumann resonances are quasi-standing electromagnetic waves that exist in the Earth's “electromagnetic” cavity (the space between the surface of the Earth and the ionosphere). Schumann resonances are the principal background in the electromagnetic spectrum between 3 and 69 Hz and appear as distinct peaks at extremely low frequencies of around 7.83 (strongest), 14, 21, 27, 39, and 45 Hz (weakest). In daily life, this vibration could be “desirable” (e.g., musical instruments), but more often is undesirable (wasting energy and creating unwanted sound—noise).
The stimulating effect of vibration on living systems is well known and plays a relevant role in mechanical transduction, which is essential for the survival of both cells and higher organisms.
A retrospective analysis was done using a cohort of patients who received intracellular sperm injections (ICSIs) at a German private fertility center about 3 years ago 5 . In vitro culture was performed either in a static environment with single oocyte/embryo culture (n = 291 patients) or under microvibration and group culture (n = 244 patients). In the static group, oocytes/ embryos were cultured individually, while in the microvibration all the oocytes were cultured together and up to four embryos were cultured in the drop with a three-dimensional vibration of 56 Hz for 5 s/60 min. The authors observed a significant increase in the fertilization rate in oocytes cultured in groups and under microvibration conditions compared to oocytes cultured individually in a static culture (82% vs. 78%); there was also an increase in the implantation rate under these conditions (42% vs. 35%). At the same time, authors noted that the clinical pregnancy rate showed a tendency to be higher in the microvibration group, but it did not reach significance (47% vs. 43%) 5 .
The aim of our investigations was to determine whether there was any difference in the viability of embryos after in vitro culture under static and mechanical microvibration conditions.
Materials and Methods
The authors confirm that all ongoing and related trials for this drug/intervention are registered (clinical trial registration: ISRCTN13773904, “In vitro culture and transfer of human embryos”).
The current work was performed from January 2011 to December 2015 at a private medical center (Endo-krinologikum Ulm, Praxisklinik Frauenstraβe, Ulm, Germany). Permission was granted by the ethical commissions of the medical faculties of the University Ulm, Germany (permission 321/10-UBB/bal. from 12. 11.2011) and the University Cologne, Germany (permission 13-147 from 20.11.2013) for the in vitro culture of embryos under mechanical microvibration. In vitro culture of embryos of each odd couple was performed in accordance with routine methodology in a static system (without microvibration). Each even couple were offered the choice of the in vitro culture of oocytes and embryos according to the standard routine or with mechanical agitation (microvibration) until transplantation. Written informed consent was obtained from all the participating couples. Eight participants did not agree to the in vitro culture of their embryos with microvibration, and their embryos were cultured in a static “traditional” system.
All patients with infertility were stimulated for IVF cycle (IVF) or intracytoplasmic spermatozoa injection cycle (ICSI) with triptorelin (Decapeptyl®; Ferring, Kiel, Germany) and recombinant follicle-stimulating hormone [FSH; Puregon® (MSD Sharp & Dohme GmbH, Haar, Germany), or Menogon® (Merck Serono GmbH, Darmstadt, Germany), or Gonal-f® (Merck Serono)] according to the standard protocol. Ovulation was induced by the administration of 5,000 IU of human chorionic gonadotropin (hCG; Brevactid®; Ferring), and oocytes were retrieved 34-36 h later and inseminated with the partner's sperm through conventional IVF and ICSI techniques.
Patients were alternately assigned to the two embryo culture groups. Only two or three embryos per patient were cultured, as according to German law no more than three pronuclear oocytes/embryos from one patient (usually two) can be cultured in vitro, and all cultured oocytes/ embryos must be later transferred to the patient independently of the developmental rate of these embryos.
Oocytes for the culture of pronuclear embryos were obtained from 4,436 patients who provided informed consent (aged 26-44 years, median age: 32.8 years). Pronuclear embryos (two or three per patient) were cultured in vitro under two different conditions: group 1 (n=4,821), without mechanical agitation of the culture medium (standard routine conditions); group 2 (n = 4,803), with mechanical agitation (44 Hz delivered over 5 s once every hour) and acceleration (660 mV/g at 3.3 V: X=± 1.0 g, Y= ±0.7 g, Z= ±0.15 g).
Mechanical agitation was achieved using the developed device Viboviduct 1500 (SimSoTec GmbH, Cologne, Germany). Before using the device, it was calibrated by measurement of vibration with a special device PCE-VT 2700 (PCE Instruments UK Ltd., Southampton, UK). Viboviduct 1500 generates microvibrations by use of a special electric motor with low electromagnetic noise. The generated vibrations are forwarded directly to the plate containing affixed Petri dishes with embryos. Harmful high frequencies are dampened and smoothed by the intelligent control software developed on the microprocessor. The control software monitors the motor movements. The device was designed and developed for use in a CO2 incubator.
Embryo development rates were determined on the day of transfer (day 2, day 3, or day 5). The embryos were cultured in 50 μl of culture medium (Sage, Los Angeles, CA, USA) under mineral oil (Sigma-Aldrich, St. Louis, MO, USA) for their transfer.
The embryos were graded on day 2 and day 3, as described by Steer et al. 6 , as follows: grade A, equal-sized symmetrical blastomeres; grade B, uneven blastomeres with <10% fragmentation; grade C, 10%-50% blastomeric fragmentation; and grade D, >50% blastomeric fragmentation. Day 5 embryos were graded according to Veeck and Zaninovic 7 .
Embryo transfer was performed on day 2, day 3, or day 5 after retrieval of oocytes. Pregnancy was defined as an increase in hCG serum concentration (20 IU/L), which was determined 11 and 13-15 days after embryo transfer. Clinical pregnancy was recorded when the fetal sac was visualized on an ultrasound at gestational weeks 7 to 8.
Statistical Analysis
Quality of embryos, amount of transferred embryos, amount of morphologically ideal developed embryos (grades A and B), amount of sacs, pregnancy outcome (ongoing pregnancy, abortion, abrasion, and biochemical pregnancy), and baby-take-home rate [the number of live births per number of IVF/ICSI treatments (cycles) (expressed as percentages)] were evaluated by analysis of variance (ANOVA). Various characteristics were summarized by mean and standard deviation (SD) within groups. The level of statistical significance was set at p <0.05. Clinical pregnancy rates were analyzed by ANOVA for categorical variables using the CATMOD Procedure of SAS Institute Inc. (Cary, NC, USA). Comparisons between age groups were performed by pairwise contrasts and Bonferroni-Holm adjustment for multiple comparisons using the MULTTEST procedure of SAS.
Results
The mean numbers of transferred embryos per patient for the static group and the microvibration group were 2.17 ™ 0.32 and 2.17 ™ 0.36, respectively.
For groups ≤29, 30-34, 35-39, and ≥40 years, the following rates of high-quality embryos without fragmentation (two to four blastomeres on day 2; six to eight blastomeres and compacting morula on day 3; blastocyst, expanded and hatching blastocyst on day 5) were detected (static vs. vibration, respectively): 65.2% versus 70.8%, 44.3% versus 69.3%, 67.% versus 76.4% (for statistic significant differences between respective rates in these three groups, p < 0.05) and 67% versus 66% (p > 0.1) (Tables 1 and 2).
High-Quality Embryos in Different Age Groups Just Before Transplantation: In Vitro Culture in Static System

2A, two blastomeres, no fragmentation; 4A, four blastomeres, no fragmentation; 6A, six blastomeres, no fragmentation; 8A, eight blastomeres, no fragmentation; 8A compacting, eight blastomeres, beginning of compacting; EB, early blastocyst; B3, blastocyst with small blastocoel; B2, blastocyst; B1, expanded blastocyst, B1h, fully expanded or hatching blastocyst. Only underlined rates (percentages) were compared (Table 1 vs. Table 2). Only rates (percentages) marked with an asterisk (*) have no significant difference (p > 0.1) (Table 1 vs. Table 2). The rest of the respective rates (percentages) are significantly different (p < 0.05) (Table 1 vs. Table 2).
High-Quality Embryos in Different Age Groups Just Before Transplantation: In Vitro Culture With Microvibration

2A, two blastomeres, no fragmentation; 4A, four blastomeres, no fragmentation; 6A, six blastomeres, no fragmentation; 8A, eight blastomeres, no fragmentation; 8A compacting, eight blastomeres, beginning of compacting; EB, early blastocyst; B3, blastocyst with small blastocoel; B2, blastocyst; B1, expanded blastocyst, B1h, fully expanded or hatching blastocyst. Only underlined rates (percentages) were compared (Table 2 vs. Table 1). Only rates (percentages) marked with an asterisk (*) have no significant difference (p > 0.1) (Table 2 vs. Table 1). The rest of the respective rates (percentages) are significantly different (p < 0.05) (Table 2 vs. Table 1).
The following baby-take-home rate was detected for groups ≤29, 30-34, 35-39, and ≥40 years, respectively (static vs. vibration): 30% versus 31% (p > 0.1, increasing only on the level of tendency), 28% versus 37%, 23% versus 29%, and 9% versus 15% (differences between respective rates in these three groups with p < 0.05) (Table 3).
Number of Formed Sacs and “Baby-Take-Home” Rate After Transplantation of Developed and Transferred Embryos in Different Age Groups: In Vitro Culture in Static System and With Microvibration
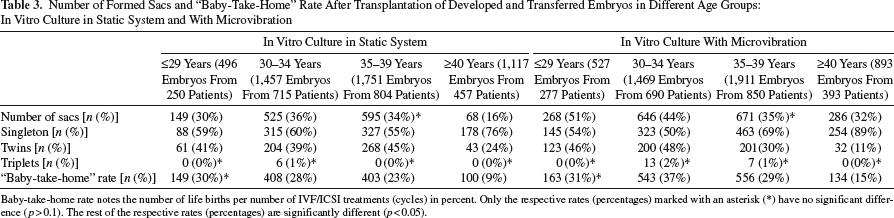
Baby-take-home rate notes the number of life births per number of IVF/ICSI treatments (cycles) in percent. Only the respective rates (percentages) marked with an asterisk (*) have no significant difference (p>0.1). The rest of the respective rates (percentages) are significantly different (p<0.05).
Discussion
There was no significant difference between the quality of embryos from some groups (e.g., in group ≥40 years), but the baby-take-home rate was significantly different. We believe the subjectivity of the process by which embryos are evaluated may affect the results, as interpretations vary depending on the embryologist performing the evaluation. However, the number of babies born is an absolutely objective rate that is free from subjectivity, and we have drawn our conclusions based on this parameter.
The conditions of embryonic development in vivo include ciliary beating from the moment ovulation begins, in fertilization, and during embryo transport via the oviduct to the uterus 8 . “Ciliary” refers to the cilia, which is Latin for vibrating hairs. Baseline cilia beating frequencies have been reported to vary widely between individuals in the range of 5-20 Hz9,10. Ciliary beat has the following characteristics: (i) its rate is remarkably uniform, and (ii) the beat of a particular cilium and its adjacent cilium appears to be well coordinated, and a definite metachronal wave is established 11 . This metachronism is defined as coordinated oscillation including a definite phasing of microvibration between the cilia of a single cell and a definite phasing of this vibration between the cilia of adjacent cells. The fluid that surrounds the cilia and forms a blanket above the tips of the cilia is a suspension of mucus 12 .
The positive effect of pulsatile mechanical microvibration for the cytoplasmic maturation of in vitro mature pig oocytes was described in a previous study 13 . These authors subjected cumulus-oocyte complexes cultured in microdrops to pulsatile mechanical vibration at a frequency of 20 Hz with acceleration (660 mV/g at 3.3 V: X= ™1.0 g, Y= ™ 0.7 g, Z= ™0.15 g; instruction of the manufacturer). During in vitro maturation, vibration did not affect the proportion of oocytes reaching the metaphase II stage. However, blastocyst formation rates after the activation of oocytes exposed to vibration were significantly higher than those obtained from oocytes matured without mechanical vibration (27% vs. 12% and 26% vs. 15%, respectively, for the 5- and 10-s pulses).
In medicine, the embryonic development rates and clinical results were compared between a static culture group (n = 159 cycles) and a microvibration culture group (n = 166 cycles) in poor responders. A microvibrator was set at a frequency of 42 Hz, 5 s/60-min duration of embryo development 14 . In poor responders, the embryonic development rate was improved to a limited extent under the microvibration culture conditions, but the clinical results were significantly improved15.
It should be noted that the vibration at a frequency of 44 Hz and the acceleration described above are the parameters of movement of the plate on which Petri dishes with culture medium and embryos are located. Our laboratory's observations on bovine oocytes have shown that the amplitude of vibration of embryos as well as acceleration of cells are lower than these rates with which the plate is vibrating (data not shown). This fact is due to the inertness of oocytes suspended in a liquid environment; vibration is drastically suppressed by the culture medium and is dependent on the composition and volume of this medium. Parameters of the “real” vibration of embryos are calculated mathematically and make up 33 Hz. It is necessary to emphasize that the observations in vivo and published rate of vibration of cilia are different because the embryo is “swimming.” Therefore, knowing the rate of embryonic development is a criterion for adjusting the frequency of the apparatus vibration.
The acceleration accompanying any movement (a = ra 2 and w=2πf, wherein a is the acceleration, r is the radius of movement, and f is the frequency) is crucially important for biological objects. The maximal rate of acceleration is achieved at a maximal frequency of 50 Hz. This is why it would not be correct to use the “optimal” rate of vibration frequency established for an apparatus. It is also important to note that apparatus with the same frequency may have a different acceleration rate that should be taken into consideration.
In conclusion, in vitro culture of embryos under microvibration (mimicking conditions in nature whereby oviductal fluid is mechanically agitated by the epithelial cilia) significantly increases the baby-take-home rate for patients 30 years of age and older.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
