Abstract
Graft-versus-host disease (GVHD) is the limiting complication after bone marrow transplantation (BMT), and its pathophysiology seems to be highly influenced by vascular factors. Our study aimed at elucidating possible mechanisms involved in vascular GVHD. For this purpose, we used a fully MHC-mismatched model of BALB/c mice conditioned according to two different intensity protocols with total body irradiation and transplantation of allogeneic (C57BL/6) or syngeneic bone marrow cells and splenocytes. Mesenteric resistance arteries were studied in a pressurized myograph. We also quantified the expression of indoleamine 2,3-dioxygenase (IDO), endothelial (eNOS), and inducible NO synthase (iNOS), as well as several pro- and anti-inflammatory cytokines. We measured the serum levels of tryptophan (trp) and kynurenine (kyn), the kyn/trp ratio (KTR) as a marker of IDO activity, and adiponectin (APN). The myographic study showed a correlation of GVHD severity after allogeneic BMT with functional vessel alterations that started with increased vessel stress and ended in eccentric vessel remodeling, increased vessel strain, and endothelial dysfunction. These alterations were accompanied by increasing IDO activity and decreasing APN levels in the serum of allogeneic animals. The mRNA expression showed significantly elevated IDO, decreased eNOS, and elevation of most studied pro- and anti-inflammatory cytokines. Our study provides further data supporting the importance of vessel alterations in GVHD and is the first to show an association of vascular GVHD with hypoadiponectinemia and an increased activity and vascular expression of IDO. Whether there is also a causative involvement of these two factors in the development of GVHD needs to be further investigated.
Keywords
Introduction
Graft-versus-host disease (GVHD) is an often lethal complication and the main limitation on a wider use of allogeneic bone marrow transplantation (BMT) in the treatment of hematological disorders 1 . Over the past few years, the hypothesis of a vascular form of GVHD has emerged, which was based on different clinical and experimental findings in acute and chronic GVHD2,3. Recently, we could, for the first time, characterize functional and structural vascular alterations in the early phase after allogeneic transplantation in an ex vivo setting 4 . In our animal model of acute GVHD, small vessel arteries of allogeneically transplanted BALB/c mice showed endothelial dysfunction and a geometry-independent increase in vessel strain. Both findings were supported by electron microscopy, which showed severely injured endothelium and altered vessel walls. As a possible mechanism of endothelial dysfunction, we found dysregulated expression of endothelial (eNOS) and inducible nitric oxide (NO) synthase (iNOS) in the vasculature. Our data further substantiated the hypothesis of vascular GVHD and the assumption that vasoprotective therapy may ameliorate systemic GVHD. Therefore, the underlying mechanisms of vascular injury need to be further elucidated. Endothelial function and integrity are influenced by many different factors 5 . Great importance in this context is attached to all kinds of inflammatory processes. On the one hand, inflammation acutely and chronically alters endothelial function; on the other hand, the endothelium is a main regulator of inflammation in turn. GVHD is characterized by an immense inflammatory response, but factors affecting endothelial function and causing vascular injury are not yet understood.
Therefore, we developed two different intensity transplantation protocols, both of them causing GVHD, but only the more intense generating vascular injury and endothelial dysfunction. In this model, we investigated potential pathophysiological mechanisms of vascular injury by analyzing factors that possibly link endothelial function to inflammation in acute GVHD, namely, indoleamine 2,3-dioxygenase (IDO), adiponectin (APN), and NO. Furthermore, vascular GVHD should further be characterized by quantifying the expression of several pro- and anti-inflammatory cytokines.
Materials and Methods
Experimental Bone Marrow Transplantation
All animal experiments had been approved by the local institutional animal committee of the University of Regensburg and were conducted in accordance with German animal protection laws. Transplantations were carried out according to a well-established protocol of our laboratory as recently described elsewhere4,6. In brief, female C57BL/6N (H-2b) and BALB/c (H-2d) mice purchased from Charles River Laboratories (Sulzbach, Germany) were transplanted at an age of 11 to 12 weeks. On the day of BMT, BALB/c recipient mice were conditioned using a linear accelerator with 150 cGy/min. After that, the mice received a mixture of bone marrow cells and splenocytes from either syngeneic (BALB/c, synTx) or allogeneic (C57BL/6, alloTx) donors. The irradiation dose and the count of transplanted bone marrow cells and splenocytes were diversified according to two different intensity protocols as shown in Table 1 [low- intensity protocol (LIP) and high-intensity protocol (HIP)]. Animals were housed for 4 weeks after transplantation and then euthanized for further investigation of their mesenteric arteries. Of the eight allogeneically transplanted animals in each group, one from the LIP and three from the HIP group died during the 4 weeks. Of all syngeneically transplanted animals, only one in the HIP group died (Table 1). Untreated animals served as controls to detect possible effects of irradiation and transplantation procedures (n = 5, ctrl).
Conditions of the Two Transplantation Protocols and Numbers of Transplanted Animals

Abbreviations: Gy, gray; BMC, transplanted bone marrow cells; SC, transplanted splenocytes; alloTx, number of allogeneically transplanted and surviving animals after 4 weeks; synTx, number of syngeneically transplanted and surviving animals after 4 weeks.
Clinical GVHD Score and Serum Samples
Clinical GVHD scores were assessed weekly after transplantation by a standard scoring system that incorporated five clinical parameters: weight loss, posture (hunching), mobility, fur texture, and skin integrity 7 . Each parameter was graded between 0 and 2 followed by the calculation of the cumulative score for each mouse. Blood samples were obtained immediately after euthanasia in week +4. We measured tryptophan (trp), kynurenine (kyn), and the kyn/trp ratio (KTR) (Immundiagnostik AG, Bensheim, Germany) as a marker of IDO activity as well as levels of APN in serum (R&D Systems, Minneapolis, MN, USA) via enzyme-linked immunosorbent assay (ELISA) according to the respective manufacturer's instruction.
Myographic Study
After euthanasia, the mesenteric vasculature was dissected for preparation of the small resistance arteries. The myographic study was then conducted according to protocols well established in our laboratory4,8. In brief, one second-order branch of the superior mesenteric artery was prepared in each animal, mounted onto a pressurized myograph (111 P; Danish Myo Technology, Aarhus, Denmark), and perfused with oxygenated 37°C KREBS solution (95% O2, 5% CO2) (pH 7.4) containing NaCl (118 mmol/L), KH2PO4 (1.18 mmol/L), KCl (4.7 mmol/L), MgSO4 (1.18 mmol/L), CaCl2 (2.5 mmol/L), D-glucose (5.5 mmol/L), NaHCO3 (25 mmol/L), and EDTA (0.026 mmol/L). First, the active vasodilatory properties of the resistance arteries were investigated. After precontraction with norepinephrine (NE; 10−5 mol/L) endothelium- dependent vasodilation was assessed by addition of acetylcholine (ACh) in increasing concentrations (10−10–10−4 mol/L) to the vessels in the chamber of the myograph. For evaluation of endothelium-independent vasodilation, the direct NO donor sodium nitroprusside (SNP) (10−9–10−2 mol/L) was used (all reagents for the myographic study were from AXXORA GmbH, Lörrach, Germany). Therefore, at each concentration of the given agent, the vessels lumen diameter and wall thickness were determined after 5 min of incubation. Thereafter, the intraluminal pressure of the vessels was equilibrated at 45 mmHg, their media thickness and lumen diameter were measured, and parameters of vessel geometry were calculated [media/lumen ratio and cross-sectional area (CSA)]. Finally, myogenic tone was inactivated by calcium depletion through incubation with Ca2+-free KREBS solution for 30 min. Intravascular pressure was raised from 3 to 10, 20, 30, and 40 mmHg and then in 20 mmHg steps up to 140 mmHg to assess passive mechanical properties (circumferential strain and stress). All vessel measurements for vasodilatory and mechanical properties were performed at three points along the vessel with the use of a calibrated video system, and the mean was calculated.
mRNA Expression in Mesenteric Resistance Arteries
The expression of IDO, eNOS, iNOS, tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), interleukin-1α (IL-1α), IL-1β, IL-2, IL-4, IL-6, IL-10, and IL-11 in small arteries was measured using real-time reverse transcription polymerase chain reaction (RT-PCR) as described previously4,8. Total RNA from mesenteric arteries of the second branch was extracted using an RNeasy kit (Qiagen, Hilden, Germany). Total RNA (1 μg) was used in the reverse transcription reaction (reagents were from Promega, Mannheim, Germany). Real-time RT-PCR was detected with the ABIPrism 7900 TaqMan (Applied Biosystems, Foster City, CA, USA) using β-actin as housekeeping gene for normalization. TaqMan probes (Table 2) were purchased from Applied Biosystems and Eurofins (Regensburg, Germany). After measuring each animal in triplicate, the mean was calculated.
Primer Sequences Used for Gene Expression Analysis

Data Analysis
All data are shown as mean ± standard error of the mean (SEM). A value of p < 0.05 was considered significant. Groups were compared using one-way analysis of variance (ANOVA) with the Holm–Šidák as a post hoc test. CSA of the media (Table 3), vessel vasodilation to ACh, and SNP circumferential strain, stress, and the strain/stress relation were calculated as described previously 4 . Curves for vasodilatory and mechanical vessel properties were compared with two-way ANOVA for repeated measurements with the Holm–Šidák method as a post hoc test.
Geometry of Mesenteric Arteries at 45 mmHg Intraluminal Pressure

Abbreviations: CSA, cross-sectional area of media; alloTx, allogeneic transplantation; synTx, syngeneic transplantation. The p values for one-way ANOVA comparing all animal groups.
Media thickness: Holm–Šidák without any significant differences between the individual groups.
Lumen diameter: Holm–Šidák *p < 0.01 versus synTx(HIP) and control; †p < 0.05 versus synTx(HIP) and control.
Media/lumen ratio: Holm–Šidák *p < 0.01 versus control, p < 0.05 versus synTx(LIP) and synTx(HIP).
CSA: Holm–Šidák *p < 0.01 versus synTx(HIP), p < 0.05 versus control.
Results
Body Weight and Clinical GVHD Score
Figure 1 shows the biometric animal data over the course of the animal experiment. The baseline body weight (week 1) in all groups was between 19 and 21 g. The weight of control animals had slightly increased over the 4-week experiment. In contrast, in the first week after BMT, body weight had significantly declined in all other groups. HIP allogeneic animals showed the highest weight loss (-26.4%). In the weeks after transplantation, syngeneically transplanted animals recovered and were even heavier in week +4 than at baseline [synTx(LIP): +5.3%, synTx(HIP): +6.9%]. Allogeneically transplanted animals had also shown a slight weight gain over the first 2 weeks after BMT but clearly lost weight in week +4, resulting in significantly reduced body weight at the end of the experiment [alloTx(LIP): −9.9%, alloTx(HIP): −24.6%] compared to all other animal groups. This finding was accompanied by the development of GVHD after allogeneic BMT as objectified by the clinical GVHD score. In week +4, HIP allogeneic animals reached a significantly higher score (6 ± 0.3) than LIP allogeneic animals (3.9 ± 0.7), indicating more severe GVHD after HIP transplantation. The scores of allogeneic animals were significantly higher than those for syngeneically transplanted animals [synTx(LIP): 1.4 ± 0.1, synTx(HIP): 2.2 ± 1.1].

Biometric animal data. (A) Percentage changes in body weight between controls and allogeneically and syngeneically transplanted animals. Significant loss of weight in allogeneically transplanted animals after BMT. Week +1: One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus all other groups; #p < 0.001 versus alloTx(LIP), p < 0.01 versus synTx(HIP), p < 0.05 versus synTx(LIP). Week +2: One-way ANOVA (p = 0.005) with Holm–Šidák: *p < 0.01 versus ctrl. Week +3: One-way ANOVA (p = 0.002) with Holm–Šidák: *p < 0.01 versus ctrl. Week +4: One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP), p < 0.05 versus alloTx(LIP); #p < 0.001 versus ctrl, p < 0.05 versus synTx(LIP) and synTx(HIP). (B) Clinical GVHD score. Development of severe GVHD after BMT in HIP allogeneic animals. Week +1: One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus synTx(LIP) and synTx(HIP), p < 0.05 versus alloTx(LIP); #p < 0.05 versus synTx(LIP). Week +2: One-way ANOVA (p = 0.002) with Holm–Šidák: *p < 0.01 versus synTx(LIP), p < 0.05 versus synTx(HIP). Week +3: One-way ANOVA (p = 0.01) with Holm–Šidák: *p < 0.05 versus synTx(LIP). Week +4: One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus synTx(LIP) and synTx(HIP), p < 0.05 versus alloTx(LIP); #p < 0.05 versus synTx(LIP).
Vasodilatory Vessel Properties
The vasodilatory response of mesenteric resistance arteries to increasing concentrations of ACh and SNP was tested ex vivo (Fig 2). Since ACh causes NO release from the endothelium, which in turn provokes relaxation of smooth muscle cells in the vessel wall, ACh-induced vasodilation depends on an intact endothelium. Usually, we expect that high concentrations of ACh (10–4 mol/L) are able to nearly completely reverse the precontraction of resistance arteries with NE (10–5 mol/L). However, in our experiments, endothelium-dependent vasodilation (Fig. 2A) was significantly decreased in HIP allogeneic animals, which indicated endothelial dysfunction. In contrast, LIP allogeneic animals did not show any endothelial dysfunction. In all other transplantation groups, the response to small doses of ACh (10–8 and 10–7 mol/L) was even more pronounced than in the control group, but vasodilation at higher concentrations remained unchanged. Maximum vasodilation reached at 10–4 mol/L of ACh was 83.3 ± 4.0% for ctrl, 93.0 ± 3.1% for alloTx(LIP), 96.7 ± 1.5% for synTx(LIP), 55.1 ± 5.2% for alloTx(HIP), and 96.7 ± 3.8% for synTx(HIP). Furthermore, we assessed the vasodilatory response to SNP (Fig. 2B). SNP acts as a direct NO donor, and therefore its vasodilatory effect does not depend on endothelium as a NO producer, but only on working smooth muscle cells. Endothelium-independent vasodilation in the investigated groups did not show any differences, indicating normal function of the smooth muscle cells.

Active vasodilatory properties of mesenteric resistance arteries. (A) Endothelium-dependent vasodilatory response to acetylcholine. Vasodilatory response to acetylcholine is significantly reduced in vessels of HIP transplanted animals. Two-way ANOVA for repeated measurements (p < 0.001) with Holm–Šidák: *p < 0.05 versus alloTx(LIP) and synTx(LIP); #p < 0.01 versus synTx(LIP) and synTx(HIP), p < 0.05 versus alloTx(LIP); §p < 0.001 versus synTx(HIP), p < 0.01 versus alloTx(LIP) and synTx(LIP); $p < 0.01 versus alloTx(LIP), synTx(LIP), and synTx(HIP); & p < 0.001 versus alloTx(LIP), p < 0.01 versus synTx(LIP) and synTx(HIP). (B) Endothelium-independent vasodilatory response to sodium nitroprusside. Vasodilatory response to sodium nitroprusside is unaffected after BMT. Two-way ANOVA for repeated measurements: p = 0.553.
Morphological and Mechanical Vessel Properties
The morphological data of mesenteric vessels assessed at an intraluminal pressure of 45 mmHg are shown in Table 3. The media thickness of the vessels significantly differed between the animal groups, and HIP syngeneic animals tended to have the thinnest media. Lumen diameters were highest in allogeneically transplanted animals, which resulted in a significant lower media and lumen ratio and thus eccentric vascular remodeling in HIP allogeneic animals. LIP allogeneic animals only showed a trend toward eccentric remodeling, but this trend was not significant. Figure 3 depicts circumferential strain and stress as well as the strain/stress relationship as further passive mechanical vessel properties. Strain (Fig. 3A) represents vessel compliance. Only HIP allogeneic animals showed a significant increase between 40 and 140 mmHg. In contrast, vessels of LIP allogeneic animals showed no increase in compliance, at best a slight trend in comparison to LIP syngeneic and control animals. Circumferential stress (Fig. 3B) matched the wall tension of vessels and was significantly elevated after allogeneic transplantation in both groups. Thereby, the strain/stress relationship (Fig. 3C) clearly indicated higher vessel strain at the same stress levels with a right shift of the curve for HIP transplanted animals.

Passive mechanical properties of mesenteric arteries. (A) Vessels strain between 3 and 140 mmHg intraluminal pressure. Strain is significantly elevated in mesenteric vessels of HIP allogeneic animals. Two-way ANOVA for repeated measurements (p < 0.001) with Holm–Šidák: *p < 0.05 versus ctrl and synTx(LIP); #p < 0.05 versus ctrl; §p < 0.05 versus ctrl, synTx(LIP), and synTx(HIP). (B) Vessel stress between 3 and 140 mmHg intraluminal pressure. Vessel stress is significantly increased after allogeneic transplantation. Two-way ANOVA for repeated measurements (p < 0.001) with Holm–Šidák: *p < 0.01 versus synTx(HIP), p < 0.05 versus ctrl; #p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP); §p < 0.01 versus synTx(HIP), p < 0.05 versus ctrl; $p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP); & p < 0.001 versus ctrl and synTx(HIP), p < 0.05 versus synTx(LIP); £p < 0.001 versus ctrl, synTx(LIP), and syn(HIP), p < 0.05 versus alloTx(LIP); £p < 0.001 versus ctrl, synTx(LIP), and syn(HIP). (C) Strain/stress relationship. Vessels after HIP allogeneic transplantation show increased compliance at the same level of wall tension. Slope of strain/stress relation: ctrl, 5.5 ± 0.4; alloTx(LIP), 4.7 ± 0.3; synTx(LIP), 4.9 ± 0.3; alloTx(HIP), 3.9 ± 0.5; synTx(HIP), 5.3 ± 0.2. One-way ANOVA (p = 0.03) with Holm–Šidák: *p < 0.05 versus ctrl and synTx(HIP).
Serum levels of IDO Metabolites, KTR, and APN
IDO is the rate-limiting enzyme for the catabolism of trp to kyn. Therefore, the KTR reflects IDO activity. Levels of trp (Fig. 4A) were significantly reduced in HIP allogeneic animals and only tended to be lower in LIP allogeneic animals [ctrl: 2,467 ± 112 μmol/L, alloTx(LIP): 2,034± 114 μmol/L, synTx(LIP): 2,392 ± 185 μmol/L, alloTx(HIP): 1,639 ± 98 μmol/L, synTx(HIP): 2,263 ± 181 μmol/L]. Concomitantly, levels of kyn (Fig. 4B) were significantly elevated in both allogeneic animal groups [ctrl: 0.37 ± 0.02 μmol/L, alloTx(LIP): 2.83 ± 0.42 μmol/L, synTx(LIP): 0.88 ± 0.1 μmol/L, alloTx(HIP): 2.67 ± 0.2 μmol/L, synTx(HIP): 0.8 ± 0.11 μmol/L]. This elevation significantly increased KTR and thus IDO activity in allogeneically transplanted animals in comparison to syngeneic and control animals (Fig. 4C). Among allogeneic animals, HIP animals also had a significantly higher KTR than LIP animals [ctrl: 164 ± 12 μmol/mol, alloTx(LIP): 1,095 ± 161 μmol/mol, synTx(LIP): 368 ± 25 μmol/mol, alloTx(HIP): 1,557 ± 103 μmol/mol, synTx(HIP): 349 ± 30 μmol/mol]. Levels of APN (Fig. 4D) were significantly reduced after allogeneic transplantation. Again, levels were even significantly lower in HIP allogeneic animals than in the LIP allogeneic group [ctrl: 010.8 ± 0.2 mg ml, alloTx(LIP): 9.5 ± 0.4 mg/ml, synTx(LIP): 12.9 ± 0.3 mg/ml, alloTx(HIP): 6.2 ± 0.6 mg/ml, synTx(HIP): 12.5 ± 0.6 mg/ml].

Serum levels of IDO metabolites, KTR, and APN. (A) Levels of tryptophan. Tryptophan levels are significantly decreased after allogeneic transplantation. One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.01 versus ctrl and synTx(LIP), p < 0.05 versus synTx(HIP). (B) Levels of kynurenine. Kynurenine levels are significantly increased after allogeneic transplantation. One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP); #p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP). (C) Kynurenine/tryptophan ratio (KTR). KTR as a marker of IDO activity is significantly increased after allogeneic transplantation. One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP); #p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP), p < 0.01 versus alloTx(LIP). (D) Levels of APN. Levels of APN are significantly decreased after allogeneic transplantation. One-way ANOVA (p < 0.001) with Holm–Šidák: *p < 0.01 versus synTx(LIP) and synTx(HIP); #p < 0.001 versus ctrl, alloTx(LIP), synTx(LIP), and synTx(HIP).
mRNA Expression in Mesenteric Resistance Arteries
Table 4 summarizes the relative expression of IDO, NO synthases, and investigated cytokines. Consistent with the increased IDO activity in the serum, IDO expression in the vessel wall was significantly elevated in allogeneic animals compared to syngeneic and control animals. In contrast, eNOS expression was significantly reduced in allogeneic compared to syngeneic animals, whereas iNOS expression did not significantly differ between the groups. The cytokines TNF-α, IFN-γ, IL-1α, IL-1β, IL-2, and IL-10 showed significantly higher expression after allogeneic BMT than after syngeneic BMT. The expression of IL-4 only tended to be higher after allogeneic BMT. IL-6 levels were only significantly elevated in HIP allogeneic animals and tended to be higher in LIP allogeneic animals. No differences could be observed for IL-11.
Relative Expression of IDO, NO Synthases, and Cytokines in Mesenteric Resistance Arteries
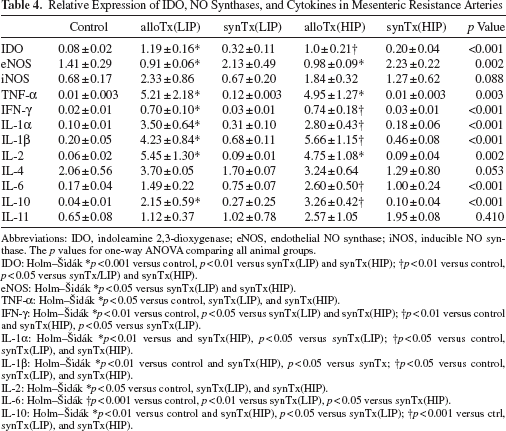
Abbreviations: IDO, indoleamine 2,3-dioxygenase; eNOS, endothelial NO synthase; iNOS, inducible NO synthase. The p values for one-way ANOVA comparing all animal groups.
IDO: Holm–Šidák *p < 0.001 versus control, p < 0.01 versus synTx(LIP) and synTx(HIP); †p < 0.01 versus control, p < 0.05 versus synTx/LIP) and synTx(HIP).
eNOS: Holm–Šidák *p < 0.05 versus synTx(LIP) and synTx(HIP).
TNF-α: Holm–Šidák *p < 0.05 versus control, synTx(LIP), and synTx(HIP).
IFN-γ: Holm–Šidák *p < 0.01 versus control, p < 0.05 versus synTx(LIP) and synTx(HIP); †p < 0.01 versus control and synTx(HIP), p < 0.05 versus synTx(LIP).
IL-1α: Holm–Šidák *p < 0.01 versus and synTx(HIP), p < 0.05 versus synTx(LIP); †p < 0.05 versus control, synTx(LIP), and synTx(HIP).
IL-1β: Holm–Šidák *p < 0.01 versus control and synTx(HIP), p < 0.05 versus synTx; †p < 0.05 versus control, synTx(LIP), and synTx(HIP).
IL-2: Holm–Šidák *p < 0.05 versus control, synTx(LIP), and synTx(HIP).
IL-6: Holm–Šidák †p < 0.001 versus control, p < 0.01 versus synTx(LIP), p < 0.05 versus synTx(HIP).
IL-10: Holm–Šidák *p < 0.01 versus control and synTx(HIP), p < 0.05 versus synTx(LIP); †p < 0.001 versus ctrl, synTx(LIP), and synTx(HIP).
Discussion
New Insights Into Vascular Function in GVHD
Recently. 4 , we could for the first time directly investigate vascular alterations of mesenteric resistance arteries in GVHD in an ex vivo setting. In our present study, we investigated functional vessel alterations after BMT conducted according to two different intensity protocols (Table 1). As objectified by the clinical GVHD score and the weight loss during the animal experiment, allogeneic animals of the HIP group developed more severe GVHD than animals of the LIP group (Fig 1). Interestingly, the HIP group matched the transplantation group of our previous study. For these animals, our present study confirmed endothelial dysfunction (Fig 2) and increased vessel compliance after allogeneic transplantation (Fig. 3A). New findings were slight changes in vessel geometry resulting in a reduced media/lumen ratio in terms of eccentric vessel remodeling (Table 3) and also significantly increased vessel stress compared to syngeneic and control animals (Fig. 3B). In contrast, allogeneically transplanted animals from the LIP group did not develop endothelial dysfunction (Fig 2) or increased vessel compliance (Fig. 3A) and only showed a trend toward eccentric vessel remodeling (Table 3). However, these animals also had increased vessel stress (Fig 3). Our present results therefore suggest that vessel alterations in acute GVHD correlate with the severity of GVHD. Increased vessel stress is already present at the early stages of GVHD. In progressive GVHD, vessels develop eccentric remodeling and, finally, increased vessel compliance and endothelial dysfunction. Since the main difference between both transplantation protocols consists in the higher number of transplanted splenocytes in the HIP group, which represent alloreactive cells in mice, our results further emphasize the hypothesis of a vascular GVHD.
New Potential Mechanisms Involved in Vascular Dysfunction and GVHD
A main target of our study was to further elucidate possible mechanisms leading to the observed vessel alterations and particularly to endothelial dysfunction. Since GVHD is characterized by immense inflammatory processes, we further investigated factors of endothelial function also involved in inflammation, such as IDO, APN, and NO.
IDO has been known to be induced in different models of inflammation for over 30 years9,10. It is the first and rate-limiting enzyme of the kyn pathway, converting trp to kyn. IDO plays a remarkable role in immunomodulation in many inflammatory diseases and tumorigenesis11,12 and is predominantly activated by IFN-γ.13,14, but also by lipopolysaccharides. 15 , TNF-α. 16 , IL-6, and IL-1β 11 . As reviewed by Murakami et al. 11 , the immunosuppressive effect of IDO results from inhibited T cell proliferation by local depletion of trp, the apoptotic properties of trp metabolites such as 3-hydroxykynurenine (3-HK) and 3-hydroxyanthranilic acid (3-HAA) on T cells, and the ability of IDO to activate mature regulatory T cells (Tregs) and to convert naive T cells to Tregs. In animal models of GVHD, IDO activation is suggested to ameliorate disease severity and reduce mortality17–19. In GVHD in humans, kyn urine levels as a marker of IDO activity correlate with disease severity. 20 , and IDO is upregulated in gut GVHD in humans 21 . However, a high expression of IDO in intestinal mucosal mononuclear cells and a low expression in endothelial cells were associated with a favorable outcome in gut GVHD 22 . This finding suggests a specific endothelial effect of IDO, which was affirmed by actual research showing functional effects of endothelial IDO in inflammation23,24. Other studies reported vasoprotective effects of 3-HAA.25,26 and an apoptotic impact of 3-HK on endothelial cells 27 . These results suggest a paradoxical role of IDO in GVHD with favorable effects in immune cells by immunosuppressive effects and harmful actions in the endothelium by inducing apoptosis and microvascular dysfunction. In our study, we found a significantly elevated KTR as a marker of IDO activation in allogeneic animals with even significantly higher levels in the HIP than in the LIP group (Fig. 4C). Here IDO activity correlated with GVHD severity and the development of endothelial dysfunction. We could also show that IDO is expressed higher locally in the vessels of allogeneic animals than in those of syngeneic and control animals (Table 4). We concluded that, in our animal model, IDO activation may play an important role in vessel damage and endothelial dysfunction. We assumed that a shift from vasoprotective trp metabolites (3-HAA) toward metabolites with apoptotic effects in endothelial cells might be responsible for vascular alterations. This assumption could also explain the paradoxical effect of IDO in immune and endothelial cells on clinical outcome, further highlighting the importance of vascular function in GVHD. Further studies, for example, in IDO knockout animals, after a pharmacological blockade of IDO or in an IDO overexpression model, are required to further substantiate this hypothesis and the effects of trp metabolites on vascular function.
Another factor involved in endothelial function and inflammatory processes is APN. We and several other authors have shown that APN can induce vasodilation in small resistance arteries in rats and that hypoadiponectinemia is associated with endothelial dysfunction in a model of diabetes mellitus type 28,28–30. The reduced levels of APN observed in obesity and its comorbidities are usually explained with a low-grade inflammatory process in adipose tissue itself 31–33. In contrast, APN levels are reported to be elevated in classic chronic inflammatory processes, a paradox that is not yet well understood34,35. To our knowledge, only a single report is available showing that patients with chronic GVHD have significantly increased APN levels that correlate with disease severity 36 . This study provides the first data of APN in acute GVHD, showing a significantly reduced level after allogeneic BMT with even significantly lower levels in HIP than in LIP allogeneic animals (Fig. 4D). With its vasoprotective and anti-inflammatory properties in mind, we propose that hypoadiponectinemia is a possible additional factor in the development of endothelial dysfunction in acute GVHD.
As already stated in our recent study. 4 , NO produced by eNOS and iNOS also plays an important role in endothelial function and inflammation. We assumed that reduced eNOS and increased iNOS expression could contribute to endothelial dysfunction and apoptosis. In fact, we could confirm our former results, showing that eNOS—which provides low doses of NO essential for endothelial function. 37 —is significantly downregulated in both allogeneic animal groups in comparison to syngeneic animals. A higher expression level of iNOS in allogeneic animals producing NO in cytotoxic concentrations. 38 could not be verified in mesenteric arteries (Table 4). However, we measured the expression of NO synthases, but not NO levels, directly. Such measurements are methodically extremely complex, so that elevated NO levels, as described in GVHD.39,40, cannot be reliably excluded in mesenteric arteries. Thus, dysregulation of NO homeostasis with reduced expression of eNOS is a further possible mechanism linking GVHD to endothelial dysfunction.
Cytokine Pattern in Mesenteric Arteries After Allogeneic BMT
A further aim of our study was to characterize inflammatory processes in the vessel walls by analyzing the expression of the different cytokines involved in GVHD. According to the established pathophysiological model of acute GVHD, an immense release of different cytokines—often referred to as a “cytokine storm”—is essential to trigger and coordinate the attack of graft T cells on host tissue1,41. To our knowledge, we now report here for the first time an expression pattern of several relevant cytokines in vessels of acute GVHD. We could find significantly elevated expression levels for IL-1α, IL-1β, IL-2, IL-6, TNF-α, and IFN-γ in allogeneic animals of both the HIP and LIP groups (Table 4), which resembles the pathophysiological model of GVHD. Besides their function in coordinating the immune response, many cytokines are also assumed to have a direct effect on the endothelium. For TNF-α and IFN-γ, some studies have shown that they can induce endothelial cell apoptosis on their own or sensitize the apoptotic effects of other factors42,43. Also, IL-1β and IL-6 seem to cause endothelial dysfunction44,45. However, not only proinflammatory Th1 cytokines are involved in the pathophysiology of acute GVHD, but anti-inflammatory Th2 cytokines such as IL-4 and IL-10 also play an important role46,47. IL-4 is known to be upregulated in GVHD. 48 and may beneficially influence the severity of GVHD49,50. In contrast, other studies have suggested a harmful role of IL-4 in GVHD51,52. In our study, IL-4 tended to be expressed higher in allogeneically transplanted animals, but this finding slightly missed the significance level. Data exist that IL-4 has proinflammatory and proatherogenic effects in vascular tissues by decreasing NO bioavailability through nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activation. 53 , which may also contribute to endothelial dysfunction. IL-10 is generally considered to have anti-inflammatory effects. 54 , and a high production of IL-10 before transplantation correlates with subsequent low incidence of GVHD 55 . However, IL-10 administration had a detrimental outcome in GVHD. 56 , and elevated IL-10 levels were associated with a poor prognosis in GVHD, possibly due to functional immunodeficiency 57 . Accordingly, other studies have shown that administration of IL-10 might reduce GVHD 58 . In our study, we found that IL-10 is significantly overexpressed after allogeneic BMT in both transplantation groups, which is in line with elevated IL-10 levels in severe GVHD. A beneficial effect on endothelial function has been described for IL-10.59,60, which acts vasoprotectively.61,62 and regulates the endothelial barrier function63,64. However, these favorable effects of IL-10 are not noticeable, at least not in HIP allogeneic animals that show endothelial dysfunction and increased vessel compliance. Whether IL-10 expression contributes to intact endothelial function in LIP allogeneic animals remains unclear. For IL-11, a cytokine with pleiotropic effects in multiple tissues. 65 , no significant differences were found after BMT.
The expression pattern of the different cytokines present in small vessel arteries resembles that of typical GVHD known from other tissues. This fact further emphasizes the hypothesis of a self-contained form of vascular GVHD. Additionally, TNF-α, IFN-γ, IL-1β, IL-6, and IL-4 can directly or indirectly mediate endothelial cell apoptosis or endothelial dysfunction and pose another mechanism of vascular damage.
Conclusions
Our present study could confirm vascular injury after allogeneic BMT and show that vascular alterations develop in correlation with the severity of GVHD. Thereby, we are the first to show an association of APN and IDO with the observed vascular changes. Since APN and different trp metabolites are known to have vascular effects, it is important to further investigate whether these factors are also causative for the development of vascular GVHD. The presented cytokine expression pattern from mesenteric arteries in acute GVHD is unique and resembles that of typical GVHD from other tissues. This finding strongly supports the hypothesis of a self- contained vascular form of GVHD. In conclusion, our study has generated new topics for future research targeting the pathophysiology and the treatment of vascular GVHD. These results may allow a better understanding of GVHD to ameliorate vascular damage and avoid potential systemic GVHD.
Footnotes
Acknowledgments
The study was supported by a grant from the “Regensburger Forschungsförderung in der Medizin” (ReForM A) to Peter M. Schmid. We thank Gabriela Pietrzyk for her excellent technical assistance. The authors declare no conflicts of interest.
