Abstract
The last three decades have documented preclinical and clinical data supporting the use of acupuncture in relieving symptoms of many diseases, including allergies, infections, and neurological disorders. The advent of electroacupuncture has not only modernized the practice of acupuncture, but also has improved its efficacy, especially for producing analgesic-like effects. Although the mechanism of action of acupuncture-induced analgesia remains largely unknown, several lines of investigation have implicated modulation of pain processes via brain opioid signaling and neuroimmunoregulatory pathways. Here, we review key findings demonstrating the efficacy and underlying mechanisms of acupuncture-induced analgesia. In particular, we discuss potent analgesic effects of acupuncture via neural pain processes through inhibition of microglial activation. The safe and effective use of acupuncture stands as a nonpharmacological alternative for induction of analgesia, which has direct clinical applications, especially for pain-related diseases.
Introduction
Acupuncture, which has been practiced in Eastern countries for thousands of years, has become recognized as a potential therapy in Western countries. Acupuncture involves the insertion and manipulation of microneedles into specific body sites, so-called acupoints, on the meridian. This process is believed to elicit profound psychophysical responses resulting in improved blood flow throughout the body (39,65). Due to the heavy workload of manual acupuncture, a novel electroacupuncture (EA) was invented to apply this treatment method to more patients. EA is a modified technique of acupuncture that utilizes electrical stimulation. A number of clinical studies have indicated that acupuncture and EA are effective for the management and treatment of immune-related diseases, including allergic disorders, infections, autoimmune diseases, and immunodeficiency syndromes (2,39), but further well-controlled randomized studies are needed (33). Accumulating evidence suggests that acupuncture modulates both specific and nonspecific immune functions (12,15). In parallel, recent studies have implicated acupuncture in the alteration of pain processes (53). The coupling of these two systems likely occurs via common signaling molecules (i.e., opioid peptides) (62). In this regard, we surmise that opioid activation leads to the processing of opioid peptides from their precursor, proenkephalin, and the simultaneous release of antibacterial peptides contained within the precursor as well. Thus, the central nervous system (CNS) pain circuits may be coupled to immune enhancement. Interestingly, acupuncture elicited increased signals bilaterally in the region of the primary and secondary somatosensory cortices in human brain, as determined by magnetic resonance imaging (22).
Current Mechanisms for Acupuncture-Induced Analgesia: Endogenous Opioids and Descending Inhibitory System
Acupuncture or EA facilitates the release of certain neurotransmitters, especially opioids, in the CNS and activates either the sympathetic or parasympathetic nervous systems (24,50,59). Such activation of certain neurotransmitters after acupuncture or EA then elicits profound psychophysical responses including potent analgesia, regulation of visceral functions, and immune modulation (24,50,59). Indeed, a number of brain imaging studies in animals and humans have shown that EA treatment activates the hypothalamus, which is a primary center for neuroendocrine-immune modulation and also regulates activities of the autonomic nervous system (52). We previously showed that EA-induced enhancement of natural killer (NK) cell activity was abolished by lesion of the lateral hypothalamic area in normal rats (11). In addition, the amount of β-endorphin, mainly released from the hypothalamus, was significantly increased in the spleen as well as in the brain by EA treatment and coincided with an increase of interferon-γ (IFN-γ) levels and NK cell activity levels, which were abolished by pretreatment with naloxone, a general opioid antagonist (74). Taken together, these findings support the notion that the activation of the hypothalamus and the release of endogenous opioid peptides represent a likely mechanistic pathway of an acupuncture-induced immune response and pain relief.
Neural Pathways and the Neuroimmune Effects Activated by Acupuncture in Neuropathic Pain
Recent clinical studies have shown the effectiveness of EA on various neuropathic pain diseases, such as neuropathic pain of malignancy, diabetic neuropathy, phantom limb pain, and below-level central neuropathic pain (1,4,6,17,58,75). Although multiple reviews have speculated about the analgesic mechanisms of acupuncture or EA, no systematic study has been published on the effects of acupuncture or EA on neuropathic pain, and the mechanism underlying pain modulation remains unclear. Interruption of sensory rather than motor nerve fibers may be responsible for the influence of chemical or surgical denervation on the acupuncture analgesic response because motor paralysis does not influence acupuncture's action (66). So far, the neurotransmitter pathways implicated in EA pain modulation include spinal endogenous opioidergic, adrenergic, serotonergic, cholinergic, and GABAergic systems (26,31,32,34–36,55,56), but further studies are warranted to clarify the interactions between these different neurotransmitters following EA (21).
The nervous system, particularly somatic sensory nerve fiber stimulation, may mediate acupuncture's analgesic actions. This speculation is consistent with the clinical observations of many traditional Chinese medicine (TCM) practitioners who describe their patients as experiencing the sensation of De Qi, a burning sensation accompanied by fullness or heaviness in the extremity or trunk where acupuncture is applied (47). This documented involvement of the nervous system in acupuncture-related pain effects suggests that the pathways associated with acupuncture are interwoven with pain pathways. Of note, sections of the spinal dorsal columns failed to affect acupuncture stimulation-induced inhibition of nociceptive responses in thalamic neurons; in contrast, convergence of impulses originating from pain sites and acupoints occur in the spinal dorsal horn and medial thalamus (such as the nucleus parafascicularis), where consolidation of two kinds of impulses takes place (49). Moreover, the acupuncture-induced increase in the pain threshold to noxious heating was similar in chronically dorsal chordotomized and intact animals (20). The pain threshold after nerve injury is reduced by the appearance of sensitized nociceptive neurons in the peripheral nervous system (PNS) (68). To this end, special attention may need to be given to the intracellular energy source, adenosine 5'-triphosphate (ATP), which is released by neuronal and nonneuronal cells (37). Indeed, ATP is an important neurotransmitter that communicates sensory information in dorsal root ganglion (DRG) neurons associated with neuropathic pain (7). ATP can activate cation-permeable ion channels (P2X receptors) and G-protein-coupled receptors (P2Y receptors) on the cell surface, especially the subtype P2X3 (5). Altogether, acupuncture or EA may stimulate different types of afferent nerve fibers to produce an analgesic effect in vivo, and ATP may play a key role in transmission of pain signals in neuropathic pain.
P2X Receptor in Midbrain Periaqueductal Gray (Pag) and the Acupuncture-Induced Analgesia
PAG is a crucial site for the modulatory system involving integration of somatic and autonomic responses to nociceptive and other stressful stimuli (54). Pain thresholds are decreased, while P2X3 receptor expression is upregulated in the lateral PAG when neuropathic pain occurs (72). EA treatments enhance pain thresholds and increase P2X3 receptor immunoreactivity in the PAG in rats with experimentally induced neuropathic pain (72). Conversely, the downregulated P2X3 receptor expression in the PAG with antisense oligodeoxynucleotide for P2X3 gene significantly attenuates the antinociceptive effect of EA (72). These findings demonstrate that PAG is a major brain structure involved in EA pain modulation.
P2X3 Receptors in Rat Dorsal Root Ganglion Neurons and the Acupuncture-Induced Analgesia
EA treatment can increase the mechanical withdrawal threshold and thermal withdrawal latency values and decrease the expression of P2X3 receptors in DRG neurons in chronic constriction injury (CCI) rats. In tandem, EA treatment attenuates the ATP and ATP-evoked currents. Accordingly, EA may induce analgesic effects by decreasing expression of P2X3 receptors and inhibiting their activity in DRG neurons of CCI rats. There is a comparable analgesic effect between rats with contralateral EA and those with ipsilateral EA (67).
The Role of p38 Mapk Signaling Pathway in Acupuncture-Induced Analgesia
The p38 mitogen-activated protein kinase (MAPK) signal transduction pathway is typically activated by cellular stress and proinflammatory cytokines and plays a critical role in inflammatory responses (25). Systematic or intrathecal administration of a p38 MAPK inhibitor has been shown to effectively alleviate inflammation and arthritis (13). EA-induced analgesia and anti-inflammatory effects are associated with the inhibition of spinal p38 MAPK activation (43). In addition, pretreatment with EA has prophylactic analgesic effects on rats suffering from visceral pain by suppressing the local and spinal p38 MAPK activity (27). EA can also significantly attenuate (complete Freund's adjuvant) CFA-induced inflammatory pain, which is regulated by the p38 MAPK/ATF-2/VR-1 pathway, but not by the p38 MAPK/ATF-2/cyclooxygenase-2 (COX-2) pathway in the persistent phase of inflammatory pain (16).
Acupuncture also inhibited the expression of inflammatory mediators (9). The analgesic effect of acupuncture may be partly mediated by inhibiting reactive oxygen species (ROS)-induced microglial activation and inflammatory responses and provide the possibility that acupuncture can be used effectively as a nonpharmacological intervention for spinal cord injury (SCI)-induced chronic neuropathic pain in patients (9).
Taken together, purinergic signaling and inflammatory responses appear to be an important local communication system in acupuncture- and EA-induced pain modulation (5). In neuropathic pain, ATP released after tissue injury can activate primary sensory afferent neurons via purinergic receptors, including P2X receptors in nerve injury models (19). Acupuncture activates some CNS structures, such as PAG and nucleus raphe magnus (NRM), contributing to descending inhibitory modulation (46), and deactivates multiple limbic areas, contributing to modulation of emotion associated with pain, such as the insular and anterior cingulate cortex (ACC) (18,40,57). That acupuncture or EA effectively modulates central homeostasis and produces analgesia in the treatment of patients diagnosed with neuropathic pain supports the notion that acupuncture or EA regulates the balance of “Yin” and “Yang” in the ancient meridian.
Acupuncture Mediates Neural Pain Through Inhibition of Microglial Activation
SCI can dramatically increase the activation of microglia in the lumbar dorsal horn (77). Furthermore, microglial activation contributes to neuronal hyper responsiveness, which coincides with alterations in behavioral pain thresholds (76). After SCI, microglial cells are actively induced in ongoing pain phenomenology (77), most notably in the case of peripheral injury, microglial activation seems to be associated with the induction phase of pain (23).
Increasing evidence reveals that spinal cord glia (microglia and astrocytes) are involved in the onset and progression of an inflammatory response and neuropathic pain (14,38,48,61,63,70). In addition, recent studies found that spinal glia exhibit an intimate relationship with EA analgesia (29) and that acupuncture inhibits microglial activation (30). As the primary immune cells of the CNS, microglia are capable of secreting proinflammatory and neurotoxic mediators upon activation, as well as initiating rapid proliferation, creating a positive feedback loop, and resulting in increased ROS levels (51). Moreover, EA alleviated oxidative damage in the hippocampal CA1 region by preventing microglial activation through a yet unidentified neuroprotective mechanism (8).
EA stimulation of ipsilateral “Huantiao” (GB30) (Fig. 1) and “Yanglingquan” (GB34) (Fig. 2) significantly suppressed CFA-induced nociceptive behavioral hypersensitivity and spinal microglial activation (60). Furthermore, the combination of EA with minocycline, a microglial inhibitor, significantly enhanced the inhibitory effects of EA on allodynia and hyperalgesia (63). Similarly, acupuncture attenuated the upregulation of macrophage-1 antigen (MAC-1), a marker of microglial activation, and reduced the elevated expression of COX-2 and inducible nitric oxide synthase (iNOS) in a 1-methyl-4-phenyl-1,2,3,6-tetrahydro-pyridine (MPTP)-induced Parkinson's disease (PD) model. Accordingly, acupuncture could be used as a neuroprotective intervention for the purpose of inhibiting microglial activation and inflammatory events in PD (28,29). Interestingly, the combination of a low dose of propentofylline and EA dramatically suppressed mechanical allodynia, suggesting a synergetic analgesia between EA and propentofylline, possibly via interruption of inflammatory activity between microglia and astrocytes in the spinal cord (42). As for the mechanism of these observed phenomena, it has been postulated (Fig. 3) that EA might affect glial cells directly or indirectly via opioid receptors (21). The blockade of activation of spinal glia in the spinal dorsal horn may cause the synergetic analgesic interaction (44).

GB30 anatomy. GB30 is located at the junction of the middle and lateral third of the distance between the highest point of great trochanter and the hiatus of the sacrum.

GB34 anatomy. GB34 is located at the depression anterior and inferior to the head of the fibula.
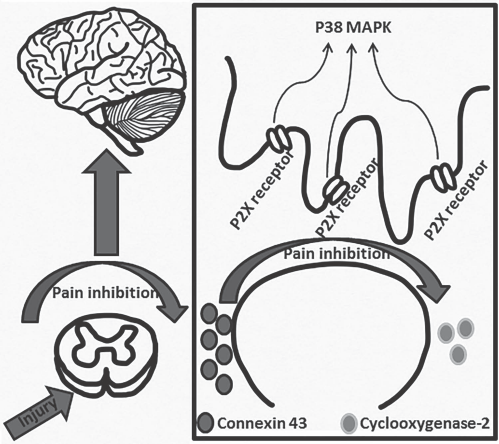
Acupuncture-induced analgesia. Proposed mechanism of acupuncture-mediated immune and pain interaction through suppression of microglial activation. Pain is initiated in the spinal cord following injury, sending pain signals to the brain. Inhibition by acupuncture involves immunoregulatory signals by blocking P2X receptors, thereby suppressing p38 MAPK activation, and accompanied by increased connexin-43 levels with decreased cyclooxygenase-2 levels.
These data provide evidence of the involvement of spinal microglial cells in the antinociceptive effects of EA. The analgesic effects of EA might be associated with its counterregulation of spinal glial activation, thereby offering a potential strategy for the treatment of arthritis (78). In addition to preventing microglial activation and attenuating oxidative stress, EA pretreatment may stand as a neuroprotective therapy.
Advancing the Concept of Acupuncture-Induced Analgesia via Microglia Inhibition
Microglia and astrocytes in the spinal dorsal horn are involved in the induction and maintenance of pathological pain (41). The inhibition of microglia attenuated inflammation-induced mechanical allodynia in rats (38) and enhanced EA analgesia in rats with inflammation-associated pain (64). Accumulating evidence has documented that acupuncture affects neuropathic pain following peripheral nerve injury. Microglial activation is a key cell death process for nerve injury (3,55). In SCI, microglial activation plays a crucial role in ongoing pain phenomenology (77). Acupuncture has shown promise in inhibiting microglial and astrocytic proliferation (69,71,73) coupled with improved functional recovery after SCI in rats (10). Additionally, acupuncture exerts a remarkable analgesic effect on SCI by also inhibiting production of microglial cells through attenuation of p38MAPK and ERK activation.
In summary, this review article summarizes preclinical and clinical evidence demonstrating that acupuncture is capable of producing analgesia in neuropathic pain by suppressing microglial activation (45). Further studies are warranted to elucidate the mechanism of action underlying this acupuncture or EA-induced pain modulation, as well as the optimal condition to produce and maintain such analgesic effects. A safe, effective acupuncture or EA treatment, with an in-depth understanding of its mode of action, will facilitate its successful translation into the clinic as an alternative to pharmacological induction of analgesia.
Footnotes
Acknowledgments
This work is supported by the Natural Science Foundation of Fujian Province (Grant No. 2014J01353); the project about the effect of acupuncture is supported by the National Natural Science Foundation of China (Grant No. 81273672). C.V.B. is funded by USF Department of Neurosurgery and Brain Repair (NIH 1R01NS071956-01A1), Department of Defense (W81XWH-11-1-0634), and the James and Esther King Biomedical Research Foundation (1KG01-33966). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors declare no conflicts of interest.
