Abstract
Delayed platelet engraftment is a well-known complication of umbilical cord blood transplantation (CBT). Megakaryocytes derived from cord blood (CB) in vitro are smaller than megakaryocytes derived from bone marrow (BM) in adults. A small megakaryocyte size might contribute to delayed megakaryocytic maturation. This study included 37 patients undergoing hematopoietic stem cell transplantation (HSCT) at Chang Gung Children's Hospital between July 2011 and June 2013. Blood samples were obtained at different times: preconditioning and post-HSCT days 56 and 97. To test whether platelet activation persists posttransplantation, two commonly used platelet activation marker antibodies, CD62P (P-selectin) and CD42b, were evaluated using whole blood flow cytometry, combining thiazole orange and anti-CD41a staining, to assess reticulated platelets. Serial peripheral blood (PB) samples were obtained posttransplantation from patients undergoing CBT (CBT group; n = 15) and mobilized peripheral blood transplantation (PBT group; n = 22). Platelet activation in the postengraftment samples was considerably higher in the PBT group than the CBT group. Moreover, immature platelet fractions (IPF) were higher in the CBT group. Our results emphasize the role of IPF for dynamic prediction of platelet engraftment in CBT.
Keywords
Introduction
Platelet production is the endpoint of megakaryopoiesis and the hallmark of the thrombopoietic potential of a megakaryocytic lineage. Mature megakaryocytes are characterized by a large cytoplasm and polyploid nucleus, with DNA content of up to 128N 1 . To gain new insights into delayed platelet recovery observed after cord blood transplantation (CBT), it was hypothesized that CB megakaryocytes do not have full thrombopoietic potential. To investigate the platelet response to agonist stimulation, platelet surface glycoproteins can be measured with flow cytometry in unfixed whole blood. Platelet activation and reticulated platelets were analyzed using a gating strategy based on ubiquitously expressed platelet membrane markers. Furthermore, the circulating immature platelet fraction (IPF) reflects real-time thrombopoiesis and correlates with platelet recovery in thrombocytopenia 2 .
In clinical settings, flow cytometry is increasingly being used to evaluate the activation state of circulating platelets and to monitor changes in platelet activation levels over time. In fact, several groups have reported using flow cytometry with different test protocols to characterize platelet activation caused by medical devices in human blood in vitro 3 .
Materials and Methods
Ethics Statement
The institutional review board (IRB) at Chang Gung Memorial Hospital approved the present study (IRB No. 103-5835D). Patients who had undergone successful hematopoietic stem cell transplantation (HSCT) at Chang Gung Children's Hospital between July 2011 and June 2013 were enrolled in the study after providing written informed consent according to the principles of the Declaration of Helsinki. We evaluated the specificity of our multicolor method combined with an antibody-based gating strategy to detect platelet surface and activation markers. We comprehensively studied the expression and kinetics for platelet activation in and the IPF for 15 patients undergoing CBT and 22 patients undergoing allogeneic peripheral blood progenitor cell transplantation (PBPCT).
Sample Collection
Blood samples were obtained at different time points: pretransplant (prior to the conditioning regimen) and post-HSCT days 56 and 97. Fresh heparinized venous blood from each donor was layered on a Ficoll-Plaque density gradient and centrifuged at 3,000 × g for 15 min at room temperature (RT). The peripheral blood mononuclear cell (PBMNC) layer was collected, washed, and resuspended in Hank's balanced salt solution (HBSS; Life Technologies, Grand Island, NY, USA). All preparations were >95% viable, as determined by trypan blue (Sigma-Aldrich, St. Louis, MO, USA) exclusion assay.
Platelet Activation
Platelet activation was determined by hematology analyzers and flow cytometry, and a fluorescent RNA-binding dye was used to quantify immature platelets. Further, platelet activation was determined by analyzing expression of CD62P (P-selectin) and CD42b [glycoprotein Ib (GpIb)] platelet surface markers with whole blood flow cytometry as previously described 4 .
Reticulated Platelets
Whole citrated blood was fixed overnight, incubated with and without RNase (Sigma-Aldrich), and stained with an anti-CD41a antibody (Becton Dickinson, Franklin Lakes, NJ, USA) and with thiazole orange (TO; AnaSpec, Freemont, CA, USA), which stains RNA. Two-color flow cytometry was used to detect the CD41a antigen on the surface of the platelets and their RNA content. The RNase-treated sample was used to correct for nonspecific staining of platelet granules by TO. The reticulated platelet (RP) percent was calculated by the following equation:
Immature Platelet Fraction
Immature platelet fraction (IPF) is a modern parameter that measures young reticulated platelets in the peripheral blood (PB), with a reference range from approximately 1% to 5% of the total platelet count. For quantification, reticulated platelets were expressed as an IPF using an XE-2100 blood cell counter with upgraded software (Sysmex, Kobe, Japan) 5 . The IPF was determined by staining platelets with an RNA dye, TO, and integrated as a quantitative parameter to evaluate the thrombopoietic state of the bone marrow6,7. After installation of this plug-in module onto the XE-2100, this method permitted automated sampling, a fixed incubation time with a polymethine RNA marker under a precise temperature control, adaptive cluster analysis, and automatic gating with a Sysmex proprietary algorithm. The IPF was reported as a percent of the total platelet count and provided a valid indication of the platelet production status.
Transplantation
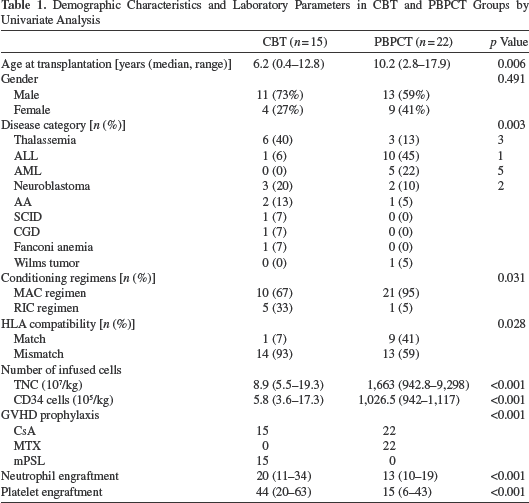
All patients underwent a comprehensive pretransplantation assessment for infection risks and organ function. The demographic characteristics and laboratory parameters are shown in Table 1. Patients received different regimens based on their disease and disease status. The standard regimen for graft-versus-host disease (GVHD) prophylaxis in PBPCT was a combination of cyclosporine A (CsA) (Novartis, Basel, Switzerland) and short-term methotrexate (Ebewe, Unterach, Austria). In CBT, CsA and Depo-Medrol (methylprednisolone acetate, USP) were administered to prevent GVHD. In patients undergoing a matched unrelated donor HSCT, anti-thymocyte globulin (Thymoglobulin; Genzyme, Neu Isenburg, Germany) was used as part of GVHD prophylaxis.
Demographic Characteristics and Laboratory Parameters in CBT and PBPCT Groups by Univariate Analysis
Engraftment
Stable graft function regarding platelets was defined as a platelet count of >20 × 10 9 /L for 7 days without platelet transfusion. For neutrophils, stable graft function was defined as a neutrophil count of >0.5 × 10 9 /L for 3 consecutive days. Graft failure was defined as no neutrophil engraftment on day 42 posttransplantation.
Statistical Analysis
Survival outcomes were evaluated for patients with no graft failure. The demographic factors and laboratory data between the CBT and PBPCT groups were compared by the Mann–Whitney U-test for numerical variables and by the Fisher's exact test for categorical variables. Values of p < 0.05 were considered statistically significant. Descriptive statistics were performed with SPSS (15.0.1 version; IBM, Armonk, NY, USA).
Results
A total of 37 children were prospectively enrolled in the study. Patients were divided into two groups: 15 patients underwent CBT and 22 patients received PBPCT (24 males and 13 females; age range, 0.4–17.9 years; median age, 9.3 years). Patients who underwent stem cell transplantation had the following conditions: thalassemia major (9 patients); acute lymphoblastic leukaemia (11 patients); acute myeloid leukaemia (5 patients); neuroblastoma (5 patients); severe aplastic anemia (3 patients); severe combined immunodeficiency (1 patient); chronic granulomatous disease (1 patient); Fanconi anemia (1 patient); and Wilms’ tumor (1 patient).
Peripheral blood specimens were obtained after transplantation of whole blood samples. Compared with the PB groups, recipients of CB group were younger and were more likely to have thalassemia. The median time to engraftment in the CB group was 20 days for neutrophils (range 11–34 days) and 44 days for platelets (range 20–63 days). The median time to platelet engraftment in the CB group was 44 days, compared with 15 days in the PB group that had a 29-day improvement. The cumulative incidence of platelet engraftment of patients in the CB group by day +97 was 93% (95% CI, 79–99%). Platelets in the postengraftment samples were significantly higher in the PB group than in the CB group. No major bleeding complications were observed in either of the groups.
We found that platelet activation was significantly higher in the PB group, compared with the CB group on post-HSCT day 56 (12.83 ± 2.38 × 10 9 /L vs. 3.46 ± 3.00 × 10 9 /L, respectively; p < 0.01) and day 97 (11.38 ± 1.94 × 10 9 /L vs. 5.24 ± 2.44 × 10 9 /L, respectively; p < 0.05) (Fig. 1). In contrast, patients in the CB group had a higher IPF than the PB group on post-HSCT day 56 [4.32% (range 2.42–6.22%) vs. 2.83% (range 1.33–4.33%), respectively] and post-HSCT day 97 [5.57% (range 3.88–7.26%) vs. 2.70% (range 0.96–4.44%), respectively] (Fig. 2). As shown in Figure 2, the IPF increased with time post-CBT, but did not in the PBT group.

Platelet activation was monitored at different time points in both groups. Statistical analysis was performed by ANOVA with Tukey's post hoc analysis; p < 0.05, p < 0.01, when compared with basal or as indicated.

The substantial increase in IPF preceded platelet engraftment on post-CBT days 56 and 97. However, the difference was not statistically significant.
Discussion
Compared with recovery after PBPCT, the likelihood of recovery of the neutrophil count and the platelet count was significantly lower in the first month after CBT 8 . CB transplants engrafted slower and were associated with less grades 2–4 acute GVHD but similar chronic GVHD, relapse, and transplant-related mortality9,10. Platelet regeneration represents an important and separate element in the engraftment process for HSCs. In our study, we have found that platelet activation is increased with PBPCT. Moreover, platelet activation was lower in patients undergoing CBT, which was partly explained by delayed platelet engraftment. Consequently, platelet activation tests may be worth exploring as a method to predict the speed of platelet engraftment.
Automated flow cytometry with blood cell counters allows for reliable quantification of reticulated platelets that are expressed as the IPF. As for the immature population that comprises the IPF reported with routine complete blood counts, there are several components including CD62P and CD42b. CD62P is a component of the α-granule membrane of resting platelets that is expressed only on platelet surfaces during and after platelet degranulation and secretion. CD42b serves as a receptor for von Willebrand factor, which participates in coagulation; it interacts with the extracellular matrix and adhesion molecules. Further, its expression and activation seem to be regulated by vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF). CD42b reacts with GpIb on megakaryocytes and platelets. However, CD62P is considered the gold standard marker of platelet activation11,12.
IPF counting may provide a relevant measure of thrombopoietic activity, leading to early evaluation of marrow function and allowing monitoring of platelet regeneration13–17. They are larger than senescent platelets, contain residual RNA that confers a reticulated appearance, and are hemostatically more active because they express more glycoprotein Ib and IIb/IIIa receptors 18 . IPF determination may be more sensitive and specific than the mean platelet volume for evaluating platelet reactivity10,19,20. To examine whether determining the IPF could solve this dilemma, we assessed the IPF as a predictor of the timing of platelet recovery in recipients of allogeneic HSCs. The IPF increased during the early posttransplant period in all engrafted patients undergoing CBT that might reflect the explosive increase in platelet production at the beginning of platelet engraftment as shown in Figure 2. No such precedence was evident after PBPCT, although this finding may be attributable to more rapid platelet recovery following PBPCT.
The present study had several limitations. The sample size included in this study was small. A small sample size, given that the disease indications for CBT and PBPCT are substantially different, might account for the differences observed. Future studies would benefit from the use of large, clinically heterogeneous samples, as HSCT procedures, conditioning regimens, and GVHD prophylaxis significantly differed between the two stem cell sources. Although delayed maturation of CB-derived megakaryocytes may be a possible cause of this observation, other contributing factors might exist such as a megakaryocyte deficiency and megakaryocyte progenitors for efficient platelet production from the CB source, compared with PB, and cannot be excluded based on the data demonstrated in this study.
Conclusion
Considering all the above-mentioned limitations, our results demonstrated that the attainment of platelet maturation in CB-derived megakaryocytes is delayed after CBT. Our study highlights the value of measuring the IPF for prediction of platelet engraftment in CBT. In contrast to PBPCT, IPF is not a precise predictor of time to platelet engraftment.
Footnotes
Acknowledgments
This work was financially supported by a research grant (CMRPG4A0033) from Chang Gung Memorial Hospital and the research grant KMRP405 for the Intractable Diseases from the Ministry of Health and Welfare of Taiwan. The authors would like to thank Ms. Wan-Chen Lin, B.S., for her technical assistance with flow cytometry, platelet assays, and data assimilation. Y.-C. Wen, T.-Y. Chang, and P.-K. Tsay performed the research and analyzed the data; T.-H. Jaing and S.-H. Chen participated in the diagnosis, management, and recruitment of patients; T.-H. Jaing and J.-L. Huang designed the research, obtained funding, analyzed and interpreted the data, and wrote the paper. All authors read and approved the final version of the manuscript. The authors declare no conflicts of interest.

