Abstract
We recently showed that intraventricular transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) significantly attenuated posthemorrhagic hydrocephalus (PHH) and brain injury after severe intraventricular hemorrhage (IVH) in newborn rat pups. The purpose of this study was to optimize the timing of MSC transplantation for severe IVH. Severe IVH was induced by injecting 100 μl of blood into each ventricle of Sprague–Dawley rats on postnatal day 4 (P4). Human UCB-derived MSCs (1 × 105 cells in 10 μl of normal saline) were transplanted intraventricularly under stereotaxic guidance either early at P6 or late at P11. Serial brain MRIs and behavioral function tests, such as negative geotaxis and rotarod tests, were performed. At P32, brain tissue samples were obtained for histological and biochemical analyses. Intracerebroventricular transplantation of MSCs significantly attenuated the development of PHH, behavioral impairment, increased apoptosis and astrogliosis, reduced corpus callosum thickness and brain myelination, and upregulated inflammatory cytokines including interleukin (IL)-1α, IL-1β, IL-6, and tumor necrosis factor-α (TNF-α) at P6 but not at P11 after induction of severe IVH. Intracerebroventricular transplantation of human UCB-derived MSCs attenuated PHH and brain injury after severe IVH in newborn rats in a time-dependent manner. Significant neuroprotection was only demonstrated when administered early at 2 days after induction but not late at 7 days after induction of severe IVH.
Keywords
Introduction
Intraventricular hemorrhage (IVH) is a serious complication of preterm birth, and its risk and severity increases with earlier gestational age at birth (24,31). Severe IVH and ensuing posthemorrhagic hydrocephalus (PHH) still remain a major cause of mortality and neurological disorders in survivors (14,16,30). As there are no available effective treatments at present for this disease, the development of new therapeutic modalities is an urgent issue.
We recently reported that intraventricular transplantation of human mesenchymal stem cells (MSCs) significantly attenuated the development of PHH and brain damage after severe IVH in newborn rats (1). While our data suggest MSC transplantation as a novel therapeutic modality for severe IVH, the determination of optimal timing for stem cell transplantation is important for future clinical applications.
The pathogeneses of brain injury and PHH after severe IVH might result from inflammatory responses caused by blood contact and blood products within the subarachnoid space (12,20). The neuroprotective mechanism of MSCs in treating severe IVH may be primarily attributable to their anti-inflammatory effects rather than to their regenerative capabilities (1). In our previous study, MSC transplantation attenuated hyperoxic lung injury in newborn rats in a time-dependent manner, showing significant protection only in the early phase of inflammation (5). Together, these findings suggest that the therapeutic time window for transplantation might be narrow and may only occur during the early phase of inflammation.
In the present study, we tried to determine the optimal timing for transplantation of human umbilical cord blood (UCB)-derived MSCs for severe IVH.
Materials and Methods
Culture and Characterization of UCB-Derived MSCs
The Institutional Review Board of Samsung Medical Center and Medipost Co., Ltd., Seoul, Korea, approved this study. UCB was collected from umbilical veins after newborn infant delivery, with informed consent from mothers. In the present experiment, identical UCB-MSCs, derived from only one male newborn infant, were used to prevent donor variation. MSCs were isolated from human UCB and cultivated (4,36). UCB harvests were processed within 24 h of collection, with viability of more than 90%. Mononuclear cells were isolated by centrifugation in a Ficoll/Hypaque gradient (density 1.077 g/cm3; Sigma-Aldrich, Deisenhofen, Germany). The separated mononuclear cells were washed, suspended in α-minimum essential medium (α-MEM; Gibco BRL, Carlsbad, CA, USA), and seeded at a concentration of 5 × 105 cells/cm2 into culture flask. Colonies of spindle-shaped cells were formed. Cells were harvested after treatment with 0.25% (w/v) trypsin-EDTA (Gibco BRL) and were then reseeded for expansion. For cytometric analysis of cultured cell phenotypes, the cells were stained with anti-human CD14 FITC, CD34 FITC (BD Biosciences, San Jose, CA, USA), CD73 PE (BD Pharmingen, San Diego, CA, USA), and CD105 PE (Serotec, Oxford, UK) antibodies for 15 min at room temperature with 1:10 dilution. Cells were washed with PBS (BioWest, Nuaille, France) and fixed with 1% (v/v) paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA). FACSCaliber (BD Biosciences) was used for analysis, and the percentage of expressed cell surface antigen was calculated in 10,000 events of gated cells. The differentiation potential and karyotypic stability of human UCB-derived MSCs were confirmed up to 11 passages, as described in our previous report (15). In the present study, passage 6 cells were used, and there were 43 cumulative population doublings. These cells expressed CD105 (92.0%) and CD73 (98.6%) but not CD34 (0.5%), CD45 (0.9%), or CD14 (0.1%), as described in our previous report (7). Mononuclear cells (MNCs) of UBC were separated using a Ficoll/Hypaque solution (density 1.077 g/cm3; Sigma-Aldrich) maintained in α-MEM (Gibco BRL) supplemented with 15% fetal bovine serum (FBS; Invitrogen, La Jolla, CA, USA) and seeded at about 5 × 105 cells/cm2. Nonadherent cells were removed after 3 days, and cells were washed with PBS (Biowest, Kansas City, MO, USA). The medium was exchanged twice in a week. At 40–50% confluence, the cells were observed in colonies of fibroblast-like cells, harvested with 0.25% trypsin-EDTA (Hyclone, Logan, UT, USA), and seeded in a culture flask for expansion. Cells at the sixth passage were trypsinized, washed, and resuspended in PBS of about 1 × 105 cells. Cells were immunolabeled with the following mouse anti-human antibodies: CD14, CD34, CD45, CD73, and CD105 (BD, Becton Dickinson, San Jose, CA, USA). Mouse isotype antibodies served as the control (BD). The cells were then washed with PBS (Biowest) and fixed in 1% paraformaldehyde (Sigma-Aldrich). Data were tested by FACscan flow cytometer (BD) with a minimum of 10,000 events.
They were positive for human leukocyte antigen (HLA)-AB (91.7%) but generally not for HLA-DR (0.4%). The cells also expressed pluripotency markers such as octamer-binding transcription factor 4 and stage-specific embryonic antigen 4, as described in our previous report (7). All cell markers were purchased from BD Biosciences except CD105 (Serotec).
Differentiation of UCB-Derived MSCs
With specific in vitro induction stimuli, human UCB-derived MSCs differentiated into various cell types, such as respiratory epithelium, osteoblasts, chondrocytes, and adipocytes (7,36). To induce osteogenic differentiation, cells were cultured in osteogenic medium for 3 weeks. The medium was composed of α-MEM supplemented with 10% FBS (Invitrogen), 0.1 μm dexamethasone (Sigma-Aldrich), 10 mm β-glycerol phosphate (Sigma-Aldrich), and 50 μm L-ascorbic acid 2-phosphate (Sigma-Aldrich). The medium was changed twice a week. Mineralized matrix was also stained with silver nitrate (Sigma-Aldrich) by the method of von Kossa. To induce chondrogenic differentiation, cells were treated with chondrogenic medium for 4 weeks. Medium was replaced twice a week. Chondrogenic medium consisted of high-glucose Dulbecco's modified Eagle medium (DMEM; Gibco BRL) supplemented with 100 nm dexamethasone (Sigma-Aldrich). The medium was changed twice a week. Mineralized matrix was also stained with silver, 50 μg/ml L-ascorbic acid (Sigma-Aldrich), 100 μg/ml sodium pyruvate (Sigma-Aldrich), 40 μg/ml L-proline (Sigma-Aldrich), 10 ng/ml transforming growth factor-β3 (TGF-β3; Sigma-Aldrich), 500 ng/ml bone morphogenic protein 6 (BMP-6; R&D Systems, Minneapolis, MN, USA), and 50 mg/ml ITS premix (1:100 dilution; BD Biosciences). For histologic evaluation, sections were stained with Safranin-O (Sigma-Aldrich). To induce adipogenic differentiation, cells were treated with adipogenic medium for 4 weeks. Medium changes were carried out twice a week. Adipogenic medium consisted of DMEM (Gibco BRL) supplemented with 10% FBS (HyClone), 0.5 mm 3-isobutyl-1-methylxanthine (IBMX; Sigma-Aldrich), 1 μm dexamethasone (Sigma-Aldrich), 0.2 mm indomethacin (Sigma-Aldrich), and 10 μm insulin (Sigma-Aldrich). The adipogenesis was evaluated with Oil red O stain (Sigma-Aldrich). To induce respiratory epithelial differentiation, cells were cultured in small airway growth medium (SAGM; Cambrex Bio Science Inc., Walkersville, MD, USA) for 5 days. The formation of respiratory epithelium was analyzed by measuring the level of surfactant protein C (SP-C; Chemicon, Temecula, CA, USA) using flow cytometry.
Animal Model
The experimental protocols described herein were reviewed and approved by the Animal Care and Use Committee of Samsung Biomedical Research Institute, Seoul, Korea. The animal experiments were also performed in accordance with Institutional and National Institutes of Health Guidelines for Laboratory Animal Care. Newborn Sprague–Dawley male rats (Orient Bio, Seoul, Korea) used for these experiments were reared with their mothers. Figure 1 shows details of the experimental schedule. The experiment began at P4 and continued through P32. As previously reported (1), IVH was induced through intracerebroventricular injection of 200 μl fresh maternal whole blood (100 μl into each ventricle) under stereotactic guidance (Digital Stereotaxic Instrument with Fine Drive; MyNeurolab, St. Louis, MO, USA) (coordinates, x = ±0.5, y = +1.0, z = +2.5 mm relative to bregma). The normal control (NC) group underwent a sham operation without intraventricular blood injection. After the procedure, rat pups were allowed to recover and were returned to their mothers.

Experimental protocol.
After confirming severe IVH by brain magnetic resonance imaging (MRI) 1 day after IVH induction (P5), rat pups were randomly allocated into three experimental groups: IVH control group (IC, n = 16), IVH with early transplantation group (IE, n = 17), and IVH with late transplantation group (IL, n = 18). Human UCB-derived MSCs (1 × 105) in 10 μl of PBS (Welgene, Daegu, Korea) were administered at P6 or P11 in IE and IL, respectively. An equal volume of PBS was administered intraventricularly under stereotaxic guidance at P6 in IC (1). Follow-up brain MRIs (Bruker-Biospin, Fällanden, Switzerland) were performed 7 and 28 days after inducing IVH (P11 and P32). During MRIs and invasive procedures including IVH induction and cell transplantation, rat pups were anesthetized using 1.5% to 2% isoflurane (Hana Pharmaceutical Co. Ltd., Seoul, Republic of Korea) in oxygen-enriched air. Negative geotaxis tests and rotarod tests were performed 7, 14, 21, and 28 days after induction (P11, P18, P25, P32) and 26, 27, and 28 days after induction (P30 to 32), respectively.
The condition of animals was assessed and monitored twice per day, especially for 7 days after modeling. To minimize the suffering of rats, those with slower weight gain than normal rats were fed with supplementary artificial milk at least four times per day. A scoring system of humane endpoints composed of three categories was used, with categories of body weight growth, responsiveness, and appearance. Euthanasia was planned to be performed via deep pentobarbital (Entobar®; Hanlim Pharmaceutical Co., Seoul, Korea) anesthesia (60 mg/kg, intraperitoneal). However, no rats in the present study met the humane endpoints before sacrifice. To harvest organs, rat pups were sacrificed for group comparison under deep pentobarbital (Entobar®) anesthesia (60 mg/kg, intraperitoneal) at P32. For time course analysis of IVH-induced inflammatory cytokines, four to six additional normal and IVH control rats were sacrificed at P5, P6, P9, and P11. To evaluate MSC transplantation-induced growth factors, four additional normal, IVH control, and IVH animals with MSC transplantation performed at P6 were sacrificed at P6, P7, and P11 (right before and first and fifth day after MSC transplantation). Whole brain tissue was harvested after transcardiac perfusion with ice-cold PBS. For histologic analyses, brains were fixed with 4% paraformaldehyde (Biosesang Inc., Gyeonggi, Korea) overnight at room temperature and then embedded in paraffin wax (Merck, Darmstadt, Germany). For biochemical analyses, periventricular zone brain tissue including the corpus callosum was selectively obtained, snap-frozen in liquid nitrogen, and stored at −80°C. For time course analyses, whole brains were used.
Enzyme-Linked Immunosorbent Assay (ELISA)
Frozen samples of brain tissue from the periventricular zone were homogenized and centrifuged at 8,000 × g for 20 min at 4°C. The protein content of the supernatant was measured using the Bradford method with bovine serum albumin (Sigma-Aldrich) as a standard. Human- and rat-specific brain-derived neurotrophic factor (BDNF; #DBD00, Human BDNF Quantikine ELISA Kit; R&D Systems and #KA0330, BDNF Rat ELISA Kit; Abnova, Taipei, Taiwan) and hepatocyte growth factor (HGF; #DHG00, Human HGF Quantikine ELISA Kit; R&D Systems and #MHG00, Mouse/Rat HGF Quantikine ELISA Kit; R&D Systems) concentrations were also measured using the ELISA kit according to the manufacturer's protocol. Interleukin (IL)-1α, IL-1β, IL-6, and tumor necrosis factor-α (TNF-α) concentrations in tissue homogenates were measured using the Milliplex MAP ELISA Kit according to the manufacturer's protocol (Millipore, Billerica, MA, USA) (1).
In Vivo MRI and Assessment of Ventricle to Whole Brain Volume Ratio
Brain MRI was performed on neonatal rats to confirm severe IVH at P5 and to monitor subsequent PHH at P11 and P32 by an investigator blinded to the treatment groups. MRIs were performed using a 7.0-Tesla MRI system, as described previously (18). The ventricle to whole brain volume ratio was calculated for each pup. Two blinded examiners independently calculated volume ratios by manually outlining the ventricles and whole brains in 12 MRI slices using ParaVision software (version 2.0.2; Bruker BioSpin, Karlsruhe, Germany), as previously described (18). Volume estimates were then made according to Cavalieri's principal, and the ventricle to the whole brain volume ratio was calculated to determine the extent of PHH after severe IVH.
Functional Behavioral Tests
Negative geotaxis tests were performed at P11, 18, 25, and 32 (1), while rotarod tests were performed consecutively at P30–32 to assess long-term effects on motor function by analyzing latency to fall, as described in our previous reports (1). Briefly, for a negative geotaxis test, pups were gently held for 3–5 s in a head downward position on a slanted slope. The time required for pups to rotate 180° to face uphill after release was recorded. For the rotarod test, the rotation speed of a treadmill (JEUNG DO Bio & Plant Co. Ltd., Seoul, Korea) was accelerated from 4 to 40 rpm over 100 s for a maximum of 3 min, and latency to fall was recorded. All animals were tested three times a day with a 15-min intertrial interval.
TUNEL Assay
Cell death in periventricular white matter was assessed with the immunofluorescent terminal deoxynycleotidyl-transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) technique (kit S7110 ApopTag; Chemicon), as described in our previous reports (1). Briefly, the brain slides were incubated with an equilibrium buffer in ApopTag kit for 5 min and incubated in reaction buffer with terminal deoxynucleotidyl transferase and 1 nmol of dUTP-digoxigenin for 60 min at 37°C, and then slides were treated by adding stop/wash buffer, and washed twice with PBS buffer. Anti-digoxigenin-FITC was added and reacted for 30 min at 37°C. After washing with PBS buffer, nuclei were counterstained with DAPI.
Immunohistochemistry
Immunohistochemical analysis of gliosis [neuronal-specific glial fibrillary acidic protein (GFAP), rabbit polyclonal, 1:1,000; Dako, Glostrup, Denmark], myelination [myelin basic protein (MBP), 1:500; Abcam, Cambridge, UK], and reactive microglia (ED-1, 1:200; Abcam, Cambridge, MA, USA) was performed on deparaffinized 4-μm-thick brain sections, as described in our previous reports (1). Briefly, brain slides were incubated with the diluted antibodies overnight at 4°C. Slides were washed three times with wash buffer (PBS/0.1% Triton X-100; Sigma-Aldrich). The brain slides were incubated with the diluted FITC-conjugated anti-mouse IgG (Invitrogen, Carlsbad, CA, USA). Then slides were washed three times with wash buffer (PBS/0.1% Triton X-100). Nuclei were stained with DAPI for counterstain.
Western Blot
Brains were homogenized in RIPA buffer (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Tissue lysates were adjusted for protein concentration, separated by SDS-PAGE, and transferred to nitrocellulose membranes. The membranes were incubated in blocking solution (3% bovine serum albumin in 0.1% Tween 20; Amresco, Solon, OH, USA) for 1 h and then treated with primary antibodies against caspase 3 (1:500) and GFAP (1:1,000) (Santa Cruz Biotechnology Inc.) overnight at 4°C. The membranes were then washed and incubated with appropriate horseradish peroxidase-conjugated secondary antibody (1:1,000; Dako UK). Band intensity was quantified with Quantity One software (Bio-Rad Laboratories, Richmond, CA, USA).
Measurement of Corpus Callosum Thickness
Paraffin sections (4 μm in thickness) were stained with hematoxylin and eosin (H&E) (Sigma-Aldrich). The thickness of the anterior corpus callosum was measured in the midline of coronal sections at the level of the medial septum area (+0.95 mm to −0.11/bregma), as previously reported (1).
Statistical Analyses
The primary end point was a continuous variable, with hydrocephalus defined as the volume ratio of dilated ventricles to whole brain at P32. Mean and standard deviation were based on our previous study (1). The estimated requirement for experimental subjects was 11 per group (power = 0.9 and type 1 error probability = 0.05). In consideration of mortality, more than 14 rat pups were assigned to each IVH-induced group. The data were expressed as means ± standard error of the mean. For time course variables, a univariate general linear model for repeated measures with Tukey's post hoc comparison was performed. For continuous variables, a statistical comparison between groups was performed by one-way analysis of variance (ANOVA) and Tukey's post hoc analysis. All of the data were analyzed using SPSS version 17.0 (IBM, Armonk, NY, USA). Values of p < 0.05 were considered statistically significant.
Results
Temporal Profile of Inflammatory Cytokine and Growth Factor Responses
Peak inflammatory responses are evidenced by markedly increased inflammatory cytokine levels in whole brain tissue, such as IL-1α, IL-1β, TNF-α, and IL-6. In time course experiments, peak inflammatory responses were observed 1 day after inducing severe IVH (P5). Values reached a plateau until P6, dramatically declined until P11, and then gradually decreased. Even with these dramatic decreases, significantly higher levels of inflammatory cytokines were observed in the IC group compared to the NC group until P32 (Fig. 2).
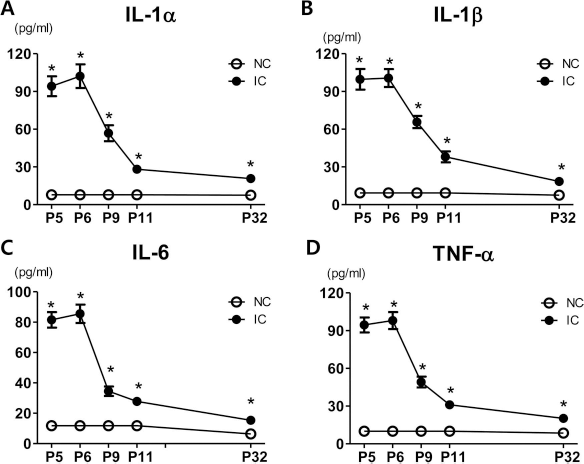
Temporal profiles of inflammatory cytokines. Interleukin (IL)-1α, IL-1β, tumor necrosis factor-α (TNF-α), and IL-6 levels measured with ELISA at P (postnatal day) 5, 6, 9, 11, and 32 in tissue homogenate of whole brain. NC, normal control; IC, IVH control. *p < 0.05 versus NC.
Human- or rat-specific neurotropic factors, including BDNF and HGF, were also assessed in whole brain tissue at P7 and P11, which is 1 day and 5 days after human MSC transplantation, respectively. Human BDNF and HGF were only detected in the MSC transplantation group (IE) at P7, not in the NC or IC groups, but they were not detected at P11 (Fig. 3). Rat BDNF and HGF levels were significantly increased at P7 in the IC group (3 days after IVH induction) compared to the NC group. These values normalized at P11 (7 days after IVH induction). These increases were significantly augmented in the human MSC transplantation group (BDNF, p < 0.05 vs. IC at P7 and P11; HGF, p < 0.05 vs. IC at P7 and p < 0.05 vs. NC at P11).

Temporal profiles of human- and rat-specific brain-derived neurotrophic factor (BDNF) and hepatocyte growth factor (HGF). Levels of BDNF and HGF were measured with ELISA right before MSC transplantation and at 1 day and 5 days after MSC transplantation (P6, P7, and P11, respectively) in tissue homogenate of periventricular brain region. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) on the second day after IVH induction (P6). *p < 0.05 versus NC, #p < 0.05 versus IC.
Serial Brain MRI
Figure 4 illustrates representative serial brain MRIs of each group at 1, 7, and 28 days postinduction of severe IVH (P5, P11, and P32, respectively). One day after inducing severe IVH (P5), the ventricular dilatation severity, presented as a ratio of ventricle to whole brain volume, did not differ between IC, IE, and IL groups (Fig. 4B). Progressive ventriculomegaly, observed in the IC group on follow-up brain MRIs at P11 and P32, was significantly attenuated only in the IE group but not in the IL group.
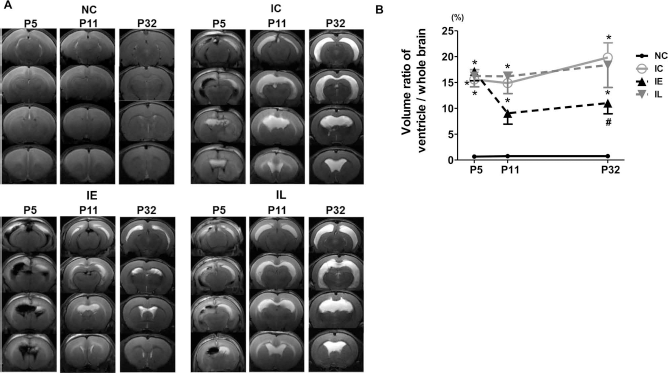
Early mesenchymal stem cell transplantation attenuated ventricular dilatation and its progression after severe intraventricular hemorrhage (IVH). (A) Representative serial brain MRIs from each group 1, 7, and 28 days after inducing IVH (P5, P11, and P32, respectively). (B) Ventricle-to-whole brain volume ratio as measured by MRI. Data are expressed as mean ± SEM. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) on the second day after IVH induction (P6); IL, IVH with transplantation of human UCB-derived MSCs on the seventh day after IVH induction (P11). *p < 0.05 versus NC, #p < 0.05 versus IC.
Functional Behavior Tests
To evaluate sensorimotor function, negative geotaxis and rotarod tests were performed. In the IC group, the negative geotaxis test displayed significant functional impairment compared to the NC group at P11, P25, and P32. This impaired performance was significantly improved only in the IE group but not in the IL group (Fig. 5A). The rotarod test was performed at P30, P31, and P32. Although initial rotarod test results at P30 were not significantly different between study groups, the NC group displayed an appropriate learning curve with longer latency to fall at P31 and P32 (Fig. 5B). In contrast, the IC group had significantly shorter latency to fall at P31 and P32 compared to the NC group. This impaired function significantly improved over time only in the IE group but not in the IL group. In other words, function improved after early transplantation at P6 but not after late transplantation at P11 (duration in seconds at P32: 194 ± 25, 78 ± 6, 128 ± 24, and 91 ± 12 in the NC, IC, IE, and IL groups, respectively; IC vs. IE, p < 0.05) (Fig. 5B).

Early mesenchymal stem cell transplantation improved impaired sensorimotor function after severe IVH. Sensorimotor functional outcomes on negative geotaxis test (A) and rotarod test (B). Data are expressed as mean ± SEM. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) on the second day after IVH induction (P6); IL, IVH with transplantation of human UCB-derived MSCs on the seventh day after IVH induction (P11). *p < 0.05 versus NC, #p < 0.05 versus IC, †p < 0.05 versus IL.
Cell Death, Reactive Gliosis, and Myelination
To determine the extent of cell death after severe IVH, the number of TUNEL-positive cells was assessed by immunohistochemistry and caspase 3 expression using Western blot analysis in the periventricular brain tissue of animals at P32 (Fig. 6A and D). The number of TUNEL-positive cells and protein expression of caspase 3 was significantly increased in the IC group (p < 0.05 vs. NC). These increases were significantly attenuated in the IE group (p < 0.05 vs. IC) but not in the IL group (Fig. 6B, D, and E). Reactive gliosis in periventricular brain tissue was assessed using the optical density of GFAP-positive cells via immunohistochemistry. Protein expression of GFAP by Western blot assay was significantly increased in the IC group (p < 0.05 vs. NC). These increases were significantly attenuated in the IE group (p < 0.05 vs. IC) but not in the IL group (Fig. 6C, D, and F). Myelination in the periventricular area was also assessed by MBP immunostaining at P32. The optical density of MBP, indicative of myelination extent, was significantly reduced in the IC group compared to the NC group. This impaired myelination was significantly improved only in the IE group but not in the IL group (Fig. 7A,B).

Early mesenchymal stem cell transplantation ameliorates cell death and reactive gliosis induced by severe IVH. (A) Representative immunofluorescence photomicrographs of periventricular area with staining for terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL, green; scale bars: 100 μm), glial fibrillary acidic protein (GFAP, red; scale bars: 25 μm), and DAPI (blue). (B) Average number of TUNEL-positive cells per high-power field (HPF) and (C) average optical density of GFAP staining in periventricular area. Caspase 3 and GFAP protein expression indicative of cell death and reactive gliosis in whole brains of P32 rats. (D) Representative Western blots of caspase 3 and GFAP, and (E and F) densitometric histograms. Data are expressed as mean ± SEM. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) on the second day after IVH induction (P6); IL, IVH with transplantation of human UCB-derived MSCs on the seventh day after IVH induction (P11). *p < 0.05 versus NC, #p < 0.05 versus IC, †p < 0.05 versus IL.

Early mesenchymal stem cell transplantation attenuates delayed myelination and corpus callosum thinning after severe IVH. (A) Representative immunofluorescence photomicrographs of periventricular area with staining for myelin basic protein (MBP, green; scale bars: 25 μm) and DAPI (blue) in periventricular area. (B) Average density of MBP staining in periventricular area in each group. (C) Representative optical microscopic photographs of corpus callosum stained with hematoxylin and eosin (original magnification: 100×; scale bars: 100 μm). (D) Average corpus callosum thickness in each group. Data are expressed as mean ± SEM. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSC) on the second day after IVH induction (P6); IL, IVH with transplantation of human UCB-derived MSCs on the seventh day after IVH induction (P11). *p < 0.05 versus NC, #p < 0.05 versus IC, †p < 0.05 versus IL.
Corpus Callosum Thickness
As ventricular dilatation progressed, the anterior corpus callosum (measured in the midline of coronal sections at the level of the medial septum) was significantly thinner in the IC group at P32 than in the NC group (Fig. 7C). This compression of the periventricular corpus callosum induced by PHH after severe IVH was significantly improved only in the IE group but not in the IL group (Fig. 7D).
Inflammatory Cytokines in Periventricular Brain
Levels of inflammatory cytokines, such as IL-1α, IL-1β, IL-6, and TNF-α, in periventricular brain tissue homogenates at P7 and P32 were significantly higher in the IC group compared to the NC group (Fig. 8). Compared to the IC group at P7, significant downregulation of inflammatory cytokines was observed after MSC transplantation at P6 in the IE group. At P32, the IVH-induced increase in inflammatory cytokines was consistently reduced in the IE group but not in the IL group.

Early mesenchymal stem cell transplantation attenuates brain inflammation after severe IVH. Inflammatory cytokine (IL-1α, IL-β, IL-6, and TNF-α) concentrations in brain homogenate from periventricular area were measured with ELISA at P7 (A) and at P32 (B). Data are expressed as mean ± SEM. NC, normal control rats; IC, IVH control rats; IE, IVH with transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) on the second day after IVH induction (P6); IL, IVH with transplantation of human UCB-derived MSCs on the seventh day after IVH induction (P11). *p < 0.05 versus NC, #p < 0.05 versus IC, †p < 0.05 versus IL.
Discussion
MSCs have recently emerged as a new potential therapeutic agent for neuroprotection due to their multipotent anti-inflammatory, antioxidative, antiapoptotic, and angiogenic activities (22,28,29). Among various sources of MSCs, including bone marrow, adipose tissue, and UCB (17), UCB is considered the most promising because of its obtainability, lack of ethical issues, high proliferation capacity (17,36), and low immunogenicity (25) related to a deficit of MHC class II molecule expression. The latter characteristic allows high tolerance, even with xenotransplantation in immunocompetent, wild-type rats. We previously showed that the development of PHH and brain damage after severe IVH was significantly attenuated with intraventricular transplantation of human UCB-derived MSCs in newborn rats. Donor MSCs migrated to the damaged periventricular area after IVH induction, shown using in vivo brain MRI (1). In addition to its protective effects against disorders such as bronchopulmonary dysplasia (BPD) (5,6), acute respiratory distress syndrome (19), and neonatal stroke, the safety and feasibility of MSC transplantation have been demonstrated in a phase I clinical trial of MSC transplantation for BPD in very premature infants (4). These findings strongly support the therapeutic potential of human UCB-derived MSCs as a novel therapeutic modality for severe IVH in premature infants, an intractable disease currently without any available effective treatment.
In the present study, the therapeutic neuroprotective efficacy of human UCB-derived MSCs after severe IVH was time dependent. This was evidenced by significant attenuation of PHH and brain damage markers such as TUNEL-positive cells, astroglial reactions, inflammatory responses, and improved behavioral tests, only when administered early at 2 days (P6) but not late at 7 days (P11) after induction of severe IVH. These findings suggest that the therapeutic time window of transplantation might be short, only during early days after severe IVH. An inadvertent delay in administering MSCs outside of the therapeutic window might obscure or nullify its therapeutic effectiveness.
Acute and chronic PHH after severe IVH might occur via different mechanisms, including mechanical obstruction by blood, iron-induced free radical damage and inflammatory cytokines, and the deposition of extracellular matrix proteins and fibrosis (12,20,27,34). In the present study, attenuation of acute PHH was observed on brain MRIs performed at P11 for the group with early transplantation at P6 but not in the group with late transplantation at P11. In previous studies of BPD (5), we showed that the antifibrotic effects of transplanted MSCs are primarily mediated by downmodulation of acute inflammatory responses before structural changes such as fibrosis occur. Overall, these findings suggest that early transplantation is critical for achieving optimal anti-inflammatory and antifibrotic effects and thus for preventing PHH and brain damage after severe IVH.
In the time course experiments of the present study, inflammatory responses decreased dramatically 2 days after severe IVH was induced. In concordance with our data, Hallevi et al. reported that a brisk inflammatory response developed around day 2, lasted 5 days, and then subsided in patients with IVH (10). In the present study, MSCs were effective in preventing PHH and attenuating brain injury only when given during the early host inflammatory response, but not when given during the late stabilization period of inflammation after severe IVH. MSCs were most effective when given during acute inflammation, but their therapeutic efficacy was less apparent when given after disease stabilization. This might be due to low levels of inflammatory cytokines in host tissue that are insufficient to elicit the anti-inflammatory effects of MSCs. This phenomenon was also observed in other disease models, including mouse graft-versus-host disease and experimental autoimmune encephalomyelitis models (35,37). These findings suggest that severity of host tissue inflammation plays a critical role in MSC-mediated tissue repair (8). Our data indicated no protective effects with MSC transplantation during the late stabilization phase of inflammation after IVH induction, which might be attributable to low levels of inflammatory cytokines in the host tissue that are insufficient to elicit the anti-inflammatory effects of MSCs. Taken together, the findings suggest that the therapeutic efficacy of MSCs can be enhanced or limited by a damaged host tissue microenvironment. Thus, the determination of optimal timing of MSC administration is very important for successful clinical translations.
Along with increased inflammatory cytokines, levels of host neurotrophic factors, such as rat BDNF and HGF (11,13,32), also increased early after IVH induction in this experiment. These increases were augmented by human BDNF and HGF secreted by transplanted human MSCs. The results suggest that self-defense initiated in host tissue early after injury might be insufficient. The trophic action of transplanted MSCs via neurotrophic factor secretion is an important mechanism for neuroprotection in this animal model.
Aside from attenuating PHH and abnormal histology, improvement in functional sensorimotor outcome is another priority for future clinical use of MSCs for severe IVH in premature infants. In the present study (1), the negative geotaxis test was significantly improved only after early transplantation at P6 but not after late transplantation at P11 from the initial test performed at P11 until follow-up tests performed at P25 and P32. This improvement coincided with significantly attenuated PHH on brain MRI done at P11 and P32 only with early transplantation at P6 but not with late transplantation at P11. PHH itself may cause brain damage through a combination of inflammation, progressive ventriculomegaly disrupting periventricular fibers, and increased intracranial pressure with resultant decreased cerebral perfusion pressure (9,34). This contributes to periventricular white matter injury and demyelination, as evidenced by corpus callosum thinning and reduced MBP expression (1).
In the present study, significant improvement in corpus callosum thinning and reduced MBP expression was observed only with early transplantation but not with late transplantation. Prevention of PHH development after severe IVH with early transplantation might be responsible, at least in part, for the improved behavioral tests, corpus callosum thinning, and demyelination. The improved rotarod tests performed at P31 and P32 only with early transplantation imply that the protective effects of early transplantation might persist into human adolescence. These findings suggest that the therapeutic time window of transplantation for behavioral improvements might be limited to the early fulminant phase of inflammation after severe IVH. In this study, the negative geotaxis and rotarod tests were repeated weekly for 4 weeks and daily for 3 days, respectively. This repetition may induce some compensation in behavioral function (23), and the rotarod test results in the present study showed different day-today learning progress between groups.
In the present study, only early transplantation at P6 but not late transplantation at P11 significantly attenuated the increased levels of TUNEL- and GFAP-positive cells after severe IVH. These findings suggest that the antiapoptotic and antiastroglial effects of MSCs might be partially mediated by their anti-inflammatory effects. Because the fate of cells and resultant brain injury might be determined early after severe IVH, MSC transplantation during the early fulminant phase of inflammation after severe IVH is optimal for maximum antiapoptotic and antiastroglial effects. In another neonatal brain injury model of hypoxic ischemic encephalopathy (HIE), brain-activated microglial cells became numerous and peaked at 7 days after ischemia (3). In IVH, however, inflammatory cytokines peaked at 2 days after modeling and then rapidly decreased. Compared to HIE, IVH seems to have an earlier and shorter acute inflammatory phase. Thus, earlier transplantation might be required to induce the appropriate therapeutic effects.
Clinically, severe IVH primarily occurs in immature brains. The ensuing PHH usually requires surgical intervention at around term. We have already shown the therapeutic efficacy of intraventricular MSC transplantation at 2 days (P6) after inducing severe IVH at P4 during the acute peak phase of inflammation (1). In the present study, we investigated whether the therapeutic time frame of MSC transplantation could be extended to 7 days (P11) after inducing severe IVH during the subacute subsiding phase of inflammation. The development of rat brains at birth, P7, P12–13, and P70 is comparable to human brains at ~24–26 weeks of gestation, 32 weeks of gestation, term newborn, and during adolescence, respectively (2,26,27,33,34). In another study (18), we observed that intraventricular MSC transplantation with the same dose (1 × 105 cells) given at P10 significantly attenuated MCAO-induced severe brain injury. PHH after severe IVH had not yet developed at P11, and the extent of ventriculomegaly was comparable to that of P5 in this study.
Clinically, germinal matrix hemorrhage and the ensuing IVH in premature infants usually occurs within 3 days after birth (21). Because time is a critical element for successful clinical trials, constant vigilance is needed for early detection of IVH. Administration of MSCs as soon as possible after detecting severe IVH may be required for maximal neuroprotection in future clinical applications. Further meticulous studies using multiple additional doses of MSCs transplanted at various time points are required as the next step.
In summary, intraventricular transplantation of human UCB-derived MSCs reduced brain injury in a time-dependent manner by attenuating PHH progress, reducing impaired behavioral function, increasing TUNEL- and GFAP-positive cells, increasing inflammatory cytokines, reducing corpus callosum thickness, and reducing MBP expression. Protection was demonstrated only with early transplantation at 2 days (P6) but not with late transplantation at 7 days (P11) after inducing severe IVH. These findings are expected to have important implications for future clinical translations in the determination of optimal timing for MSC transplantation in treating IVH in premature infants.
Footnotes
Acknowledgments
We are grateful to Ye Rim Park and Yoo Jin Chang for their animal modeling and technical assistance. This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2014R1A1A3051220); by a grant from the Korean Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (A110445, HR14C0008); by a Samsung Biomedical Research Institute grant (SMX1150481); and by an IN SUNG Foundation of Medical Research grant (CB28021). Won Soon Park and Yun Sil Chang have issued or filed patents about composition for treating intraventricular hemorrhage in preterm infants comprising mesenchymal stem cells. Soo Jin Choi is in the paid employment of MEDIPOST Co., Ltd.
