Abstract
The isolation and transplantation of porcine islets represent a future option for the treatment of type 1 diabetic patients. Stringent product release criteria and limited availability of transgenic and specific pathogen-free pigs will essentially require processing of explanted pig pancreata in specialized, possibly remote isolation facilities, whereby pancreata are exposed to cold ischemia due to prolonged tissue transit time. In the present study we investigated whether pancreas oxygenation can be efficiently combined with an antioxidant strategy utilizing intraductal L-glutamine administration. Pig pancreata were intraductally perfused after retrieval and after cold storage in oxygen-precharged perfluorohexyloctane utilizing University of Wisconsin solution supplemented with (n = 16) or without (n = 14) 5 mmol/L L-glutamine. After isolation purified islets were subjected to extensive quality assessment. Islet recovery postpurification was significantly higher in glutamine-treated pancreata (77.0 ± 3.3% vs. 60.3 ± 6.0%, p < 0.05). Glutamine administration increased intraislet content of reduced glutathione (117.8 ± 16.5 vs. 15.9 ± 2.8 ng/ng protein, p < 0.001) associated with increased islet recovery after culture (65.8 ± 12.1% vs. 40.3 ± 11.7%, p < 0.05), enhanced glucose stimulation index (1.82 ± 0.16 vs. 1.38 ± 0.10, p < 0.05), and improved posttransplant function in diabetic nude mice (p < 0.05). Furthermore, intraductally administered glutamine increased pig islet resistance toward reactive oxygen species, nitric oxide, and high-dose proinflammatory cytokines. The present study demonstrates that quality and function of pig islets exposed to warm and cold ischemia can significantly be improved using intraductal L-glutamine administration. As the efficiency of the intraductal route may be inferior compared to intravascular administration further studies should aim on assessment of L-glutamine as supplement for pancreas perfusion during organ procurement.
Introduction
Pancreatic islet transplantation for the treatment of type 1 diabetic patients has resulted in considerable clinical success over the past decade (27). However, the shortage of human pancreatic donors remains a challenge. In the search to find an alternative islet source, porcine islets represent a viable option for the future (5,12,31). This appears to be particularly realistic when genetically modified pigs are used as pancreas donors (32). Nevertheless, considering the stringent regulatory requirements for breeding of transgenic pigs in centralized biosecure facilities (26), the current opinion that isolated pig islets represent an unlimited xenogeneic tissue source (23) does not match with the estimated costs for future xenotransplantation (5). The limited availability of pancreata from genetically modified and specific pathogen-free pigs will necessitate that the islet isolation procedure be performed by highly experienced personnel in possibly distant-site isolation laboratories. However, organ transportation is associated with periods of cold ischemia, a significant determinant of subsequent islet yield and viability particularly in ischemia-sensitive pig pancreata (14).
It has been previously demonstrated in canine and human pancreata that tissue oxygenation during cold storage using hyperoxygenated oxygen carriers preserves the structural integrity of exocrine and endocrine pancreatic tissue during prolonged cold ischemia (17,22,30). Previous studies have clearly suggested that this strategy improves the isolation outcome also in ischemic pig pancreata if perfluorohexyloctane (F6H8), a lipophilic compound characterized by a relatively low specific density compared to other perfluorocarbons like perfluorodecalin, is used as an oxygen carrier for prolonged cold storage (9).
The most important contributors to ischemia-induced damage are intracellularly produced reactive oxygen species (ROS). Any condition, such as hypoxia or cold storage, that alters the mitochondrial membrane polarization and ATP production results in an elevated production of ROS (21,22).
While improved tissue oxygenation during cold storage aims to preserve mitochondrial metabolism and prevent excessive ROS production, increasing the capacity of antioxidant defense mechanisms in islets represents another strategy to reduce ischemic injury in cold-stored pancreata prior to islet isolation. An approach that is easily applicable during organ procurement was presented in previous studies in rat and human pancreata suggesting that the intraductal injection of L-glutamine (GLN) prior to cold storage or at the time of collagenase administration improves islet isolation outcome in ischemic pancreata (3,4).
The aim of the present prospective study was therefore to investigate the effect of pancreas oxygenation combined with GLN administration on the quality of pig islets isolated from adult pancreata after exposure to warm and cold ischemia.
Materials and Methods
Pancreas Procurement
Pancreata were retrieved from 40 female retired breeder pigs (≥24 months). All pigs were slaughtered in a local abattoir after mechanical induction of cerebral death prior to exsanguination. After a warm ischemia time of 30 min, cannulated splenic pancreatic segments were intraductally flushed with 1.25 ml/g of cold University of Wisconsin solution (UW; DuPont Pharmaceuticals, Bad Homburg, Germany) either plain or supplemented with 5 mmol/L GLN (Biochrom, Berlin, Germany). The GLN concentration was selected according to previous studies (3,4,8). During 3 h of cold storage pancreata were immersed either in UW or in oxygen-charged F6H8 (Novaliq GmbH, Heidelberg, Germany) as previously described (7). The different experimental groups are displayed in Table 1. Total cold ischemia time was defined as the interval between intraductal infusion of UW in the slaughterhouse and intraductal injection of the collagenase blend performed in the isolation facility.
Digestion Characteristics

Islet Isolation
After arrival at the isolation facility all pancreatic segments were intraductally distended with collagenase NB-1 (Serva Electrophoresis, Uetersen, Germany) at a concentration of 15 PZ-U [substrate 4-phenylazo-benzyloxycarbonyl-Pro-Leu-Gly-Pro-Arg (PZ) activity units] per gram trimmed pancreas weight supplemented with 1.1 DMC-U (substrate dimethylcasein activity units) per gram of neutral protease NB (Serva Electrophoresis). Enzymes were dissolved in a 50:50 mixture of cold UW and Hank's balanced salt solution (HBSS; Biochrom) intraductally injected at a ratio of 2.0 ml/g. In the experimental groups B and D, GLN perfusion was repeated by supplementing the enzyme solution with 5 mmol/L GLN. Isolation and purification were performed as previously described (9). Purified islet fractions were washed twice in HBSS supplemented with 10% newborn calf serum (Biochrom) and finally collected in 100 ml CMRL 1066 (PAA, Pasching, Austria) supplemented with 1 mmol/L GLN, 25 mmol/L HEPES (Biochrom), 10% porcine serum (Biochrom), 100 U/μg/ml penicillin–streptomycin (Biochrom), and 20 μg/ml ciprofloxacin (Bayer, Leverkusen, Germany). Islet culture was performed in untreated 10-cm Petri dishes (Greiner Bio-One, Frickenhausen, Germany) incubated in 5% CO2 atmosphere for 2 or 4 days at 37°C.
Islet Characterisation
Subsequent to pancreas digestion, purification and culture islet yield were determined in a standardized procedure converting islet number (IN) to islet equivalents (IEQ) with an average diameter of 150 μm (25). After culture, the percentage of islet recovery was calculated by comparing islet yield postculture with preculture. A fragmentation index was calculated by dividing IN by IEQ prior to and after purification.
Islet viability was measured utilizing the trypan blue (Sigma-Aldrich, Steinheim, Germany) exclusion assay to determine membrane integrity immediately after isolation, after culture, and after proinflammatory treatment (15). Islet overall survival after culture and after proinflammatory treatment was calculated by multiplying islet viability with islet recovery.
Two days after isolation in vitro function was assessed during static glucose incubation of duplicate samples of 20 hand-selected islets with an average diameter of 150-200 μm incubated for 120 min in bicarbonate-free CMRL 1066 supplemented with 2.0 or 20 mmol/L glucose (Sigma-Aldrich). After stimulation, islets were recovered and sonicated in distilled water for subsequent determination of intracellular insulin and intracellular total protein content. Intracellular and secreted insulin were measured utilizing an enzyme immunoassay specific for pig insulin (Mercodia, Uppsala, Sweden). Insulin release at different glucose concentrations was normalized to intracellular protein content determined by a method previously used by Lowry et al. (19) utilizing a commercially available kit (Bio-Rad, Munich, Germany). The glucose stimulation index was calculated by the insulin release at 20 mmol/L glucose divided by release at 2 mmol/L.
Islet posttransplant function was assessed in 35 6- to 8-week-old male NMRI nude mice (Harlan, Hanover, Germany) rendered diabetic by a single intravenous injection of 240 mg/kg streptozotocin (STZ; Sigma-Aldrich) 2 days prior to transplantation of 3,000 IEQ under the kidney capsule. Blood samples were taken from recipients' tail veins and analyzed utilizing a one-touch glucometer (Bayer). Pretransplant, the nonfasting serum glucose levels of all recipients exceeded 350 mg/dl. After transplantation, nonfasting serum glucose levels < 200 mg/dl were defined as normoglycemic, reflecting functional grafts. Thirty-two days posttransplant nephrectomy of graft-bearing kidneys was performed to demonstrate immediate return of hyperglycemia.
All animal studies were approved by the University ethics committees according to local ethical guidelines for animal management.
Reduced glutathione (GSH) content in isolated islets was determined in aliquots of 2,000 IEQ cultured overnight in GLN-free CMRL 1066 but otherwise supplemented as described above. Intraislet GSH was determined utilizing a commercially available kit (Biocat, Heidelberg, Germany) and colorimetrically measured at 405 nm. GSH was normalized to intracellular protein content measured as described above.
Treatment with Proinflammatory and Oxidative Biomolecules
After isolation five aliquots of 10,000 IEQ each were suspended in 10 ml GLN-free CMRL 1066 but otherwise supplemented as described above. Aliquots were supplemented with either 25 μmol/L hydrogenperoxide (H2O2; Sigma-Aldrich), 25 μmol/L DETA-NONOate (DETA; Alexis, Grueningen, Germany), or a cytokine cocktail of interleukin-1β (IL-1β; Cell Concepts, Umkirch, Germany), tumor necrosis factor-α (TNF-α; Cell Concepts) and interferon-γ (IFN-γ; Cell Concepts). The concentration of each cytokine was adjusted to 1,000 U/ml. The incubation time using H2O2 or DETA was 48 h while islets treated with cytokines were incubated for 96 h. Control islets were incubated under identical conditions without (w/o) adding inflammatory mediators.
Data Analysis
All statistical analysws were performed utilizing Prism 6.0f for MacIntosh (GraphPad, La Jolla, CA, USA). Comparisons of data were carried out by Mann–Whitney test and Fisher's exact test. Repeated measurements at different time points or related samples were analyzed using the Wilcoxon signed-rank test. Differences are considered significant at p < 0.05. Values of p > 0.05 are termed nonsignificant (NS). For clarity, results are expressed as mean ± standard error of the mean (SEM) rather than the correct nonparametric measures of median and quartiles.
Results
Islet Isolation Outcome
Digestion time, percentage of undigested tissue, and amount of packed tissue volume did not vary between the experimental groups (Table 1).
As shown in Table 2, pancreata preserved without GLN and without F6H8 (group A) had a significantly lower total islet yield and islet yield per gram trimmed pancreatic tissue (p < 0.05) when compared to pancreata preserved without GLN, but with F6H8 (group C). As the fragmentation index of these islets was extremely high compared to the other experimental groups, purification was not performed in group A. Supplementation of 5 mmol/L GLN significantly increased islet yield per gram prepurification when pancreata were stored in UW (A vs. B, p < 0.05), but had no effect when storage in F6H8 was applied (C vs. D, NS). In contrast, islet yield counted postpurification was significantly higher in F6H8-oxygenated pancreata when GLN was supplemented (C vs. D, p < 0.05). When GLN-supplemented pancreata were compared it was found that storage in precharged F6H8 had a significantly positive effect on total islet yield pre- and postpurification (B vs. D, p < 0.001) as well as on islet viability (p < 0.05). Islet fragmentation and purity were not different between experimental groups when compared postpurification (Table 2).
Isolation Outcome

n.d., not determined;
p< 0.001 versus D,
p< 0.05 versus A,
p< 0.01 versus A,
p< 0.05 versus D.
Islet Characterization
A comparison of the intraislet GSH content was performed only in islets isolated from F6H8-oxygenated pancreata. The intraductal infusion of 5 mmol/L GLN increased GSH from 15.9 ± 2.8 ng/ng to 117.8 ± 16.5 ng/ng protein when islets were isolated from glutamine-infused organs (C vs. D, p < 0.001) (Fig. 1).

GSH content in isolated pig islets. GSH (ng/ng protein) was determined in aliquots of 2,000 IEQ isolated from pancreata exposed to 30 min of warm ischemia and subsequently perfused with plain UW (group C, open circles, n = 8) or with UW supplemented with 5 mmol/L GLN (group D, black circles, n = 6) prior to and after 3 h of cold storage in oxygenated F6H8. Before measurement, islet aliquots were cultured overnight in GLN-free culture medium. Mann–Whitney test revealed ***p < 0.001 as indicated.
Postculture quality assessment (Table 3) demonstrated an improvement in terms of islet recovery and islet viability after 2 days of culture at 37°C when GLN-supplemented pancreata were stored in F6H8 instead of UW (B vs. D, p < 0.05). Comparing only F6H8-stored pancreata, it was noted that significantly more pig islets could be recovered after injecting 5 mmol/L of GLN (C vs. D, p < 0.05). The postculture overall survival of islets revealed a synergistic effect between GLN supplementation and oxygenation as combined in group D (p < 0.05 vs. B and C). This synergism was also observed when in vitro function of islets was assessed. The stimulatory capacity determined as stimulation index was highest when pancreas GLN supplementation and oxygenation was combined (p < 0.01 vs. B, p < 0.05 vs. C). The intracellular insulin content was not significantly altered by GLN administration or F6H8 storage (Table 3).
Postculture Islet Quality Assessment

n.d., not determined;
p < 0.05 versus B,
p < 0.01 versus B,
p < 0.05 versus C.
In vivo function after transplantation beneath the kidney capsule of diabetic nude mice was assessed only in pig islets isolated from F6H8-oxygenated pancreata (groups C and D). In both experimental groups an immediate restoration of euglycemia was observed followed by a gradual increase in blood glucose that was significantly higher when islets were isolated from pancreata not infused with GLN (Fig. 2). After 4 out of 14 recipients of islets in group C had to be euthanized because of hyperglycemia- related poor physical conditions, the blood glucose levels of the non-GLN-treated group approximated the lower blood sugar in mice transplanted with islets from glutamine-infused pancreata. Nevertheless, the average blood glucose calculated from day 2 until day 32 was significantly higher in group C recipients compared to mice in the GLN-treated group (191 ± 40 vs. 95 ± 21 mg/dl, p < 0.05). Altogether, 8 out of 14 (57.1%) and 18 out of 21 (85.7%) diabetic recipients were cured by islets from noninfused or glutamine-infused pig pancreata (C vs. D, p < 0.05), respectively.
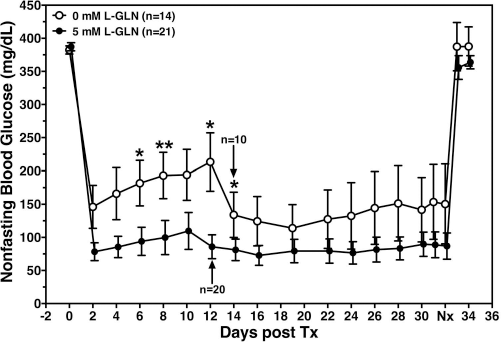
Posttransplant function of adult pig islets isolated from ischemic pancreata. Pancreata were exposed to 30 min of warm ischemia and subsequently perfused with plain UW (group C, open circles, n = 14) or with UW supplemented with 5 mmol/L GLN (group D, black circles, n = 21) prior to and after 3 h of cold storage in oxygenated F6H8. Aliquots of 3,000 IEQ were transplanted beneath the kidney capsule of STZ-treated diabetic NMRI nude mice after 2 days of culture at 37°C. Nephrectomy (Nx) of graft-bearing kidneys was performed on day 32 as indicated. Arrows indicate remaining number of recipients. Data are calculated from six individual islet preparations in each experimental group. Mann–Whitney test revealed *p < 0.05 and **p < 0.01 for 0 versus 5 mmol/L of GLN supplementation as indicated.
Treatment with Proinflammatory and Oxidative Biomolecules
Resistance toward proinflammatory treatment was assessed only in pig islets isolated from F6H8-oxygenated pancreata (groups C and D). Exposure of isolated pig islets to different inflammatory mediators, starting immediately after isolation, revealed that hydrogen peroxide severely affects integrity of islets from non-GLN-infused pancreata (group C) even when used in a concentration of 25 μmol/L. However, the detrimental effect on islet recovery (39.6 ± 11.3 vs. 75.3 ± 7.1%, p < 0.05) and viability (61.2 ± 11.6 vs. 89.6 ± 3.4%, p < 0.05) was significantly ameliorated when islets had been isolated from glutamine-treated pancreata (group D, Table 4). A less distinct impact was observed when the nitric oxide-generating compound DETA or a cytokine cocktail of high-dose IL-1β, TNF-α plus IFN-γ was used for proinflammatory treatment (Table 4). While no significant differences were found between groups C and D regarding islet recovery after DETA (68.2 ± 9.4 vs. 81.3 ± 6.8%) or cytokine incubation (77.5 ± 8.0 vs. 83.4 ± 6.1%), a significantly protective effect of glutamine infusion could be detected with respect to islet viability after DETA (85.1 ± 3.7 vs. 93.7 ± 2.0%, p < 0.05) or cytokine treatment (92.6 ± 1.4 vs. 97.2 ± 1.7%, p < 0.05).
Islet Survival After Proinflammatory Treatment

p <0.05 versus D.
Intraductal glutamine infusion also significantly increased pig islet overall survival after H2O2 (25.8 ± 10.7 vs. 66.8 ± 5.5%, p < 0.05) and DETA treatment (58.3 ± 8.3 vs. 75.7 ± 5.6%, p < 0.05), but not after cytokine incubation (71.6 ± 7.2 vs. 80.8 ± 5.4%), which most likely reflects the cytotoxicity of ROS or nitric oxide-generating compounds used in the present study.
Discussion
Previous studies in ischemic rat, pig, and human pancreata have demonstrated that storage in oxygenated F6H8 improves islet isolation outcome when compared to storage in oxygenated perfluorodecalin (7,9,10). The data of the present study indicate that the protective effect of F6H8 is even larger when compared to cold storage in UW solution. The characteristics of F6H8 significantly improve oxygenation of cold-stored pancreatic tissue, stimulating the ATP synthesis in acinar and islet tissue as demonstrated by intrapancreatic pO2 determination and 31P-nuclear magnetic resonance spectroscopy in a porcine split lobe model (9). Continuation of mitochondrial ATP synthesis appears to be essential to keep the ROS production in the mitochondria within a physiological range. In spite of constant ROS generation, mitochondria remain functional as long as the mitochondrial antioxidant defense mechanism composed of manganese-dependent superoxide dismutase and glutathione peroxidase can cope with the amount of ROS generated (20). The extent of ROS production is particularly relevant for pancreatic β-cells, which are highly sensitive to the mitochondrial generation of superoxide anion, hydroxyl radical and hydrogen peroxide as they have a specifically low expression of antioxidant enzymes in comparison with other organs (13,18,29).
The present data suggest that islet isolation outcome in UW-stored pig pancreata exposed to 30 min of warm ischemia and 3 h of cold storage is significantly increased when 5 mmol/L of GLN is intraductally injected immediately after retrieval and at the time of collagenase administration. This is in agreement with previous findings in GLN-perfused rat and human pancreata (3,4) indicating that GLN seems to enhance islet survival during enzymatic pancreas digestion, which represents a hypoxic and proinflammatory environment (1,6,11). The protective effect of GLN is also present when pig pancreata are oxygenated during cold storage in F6H8. The improved survival of pig islets after significant ischemia not only concerns islet yield but also islet potency as reflected by increased graft survival after transplantation into diabetic nude mice. Consideration of purified islet yield, enhanced glucose stimulation index, and the increased porcine islet survival after 2 days of culture at 37°C suggest a synergism between GLN administration and effective pancreas oxygenation by means of F6H8.
The beneficial effects of intraductal GLN injection on islet quality seems to be associated with the increased formation of intraislet GSH (3,4). The primary step of mitochondrial GSH synthesis is the ligation of glutamate and cysteine, which is preceeded by the deamination of glutamine to form glutamate (2). As shown in human islets, increased GSH levels are inversely correlated with the formation of malondialdehyde, a marker of lipid peroxidation (3). In accordance, our culture experiments, performed in the absence of exogenous GLN, indicated that higher intraislet GSH levels are associated with an increased resistance toward oxidative biomolecules such as hydrogen peroxide and nitric oxide.
The induction of the heat shock response is another protective mechanism that has to be discussed in the context of GLN administration. As shown in cardiac, intestinal, and islet tissue from rats and in human leucocytes, GLN induces the expression of heat shock protein 70 and heme oxygenase-1, which improved cell survival in an adverse or even lethal environment (16,24,28). As heme oxygenase-1 is a heat shock protein only in rodents it needs further evaluation whether this defense mechanism is of importance in islets isolated from large mammalians.
In summary, the present study demonstrates that the quality of pig islets exposed to warm and cold ischemia can significantly be improved using administration of 5 mmol/L GLN via the pancreatic duct. As the efficiency of the intraductal route may be inferior compared to intravascular administration further studies should assess whether GLN can be used as supplement for pancreas perfusion during organ procurement.
Footnotes
Acknowledgments
This work was supported in part by a grant of the German Ministry for Economy and Technology (BMWi). The Oxford Islet Transplant Research Group is partly funded by a generous grant from the Diabetes Wellness and Research Foundation (DWRF) U.K. Daniel Brandhorst and Heide Brandhorst participated in research design, in performance of research, in data analysis, and in the writing of the paper. Bastian Theisinger and Berhard Guenther participated in the writing of the article and contributed the perfluorohexyloctane. Paul R. Johnson participated in the writing of the paper. Bastian Theisinger and Bernhard Guenther are employed by Novaliq GmbH as indicated. The authors declare no conflicts of interest.
