Abstract
Islet volume and endocrine pancreas architecture (islet size distribution) may be independent determinants of β-cell function. Furthermore, the accuracy of homeostatic model assessment (HOMA) indexes in predicting β-cell mass has never been assessed. Here we investigated the relationships between islet volume, islet density, and islet size distribution, estimated after pancreatic tissue digestion, with established indexes of β-cell function in humans. We included in this study 42 patients who were candidates for islet autotransplantation and had well-characterized glucose metabolism. Indexes of insulin secretion were calculated and compared with the islet volume, as a surrogate of β-cell mass, obtained after digestion of pancreas. Islet counting analysis showed considerable interindividual variation in islet density and size. Islet volume, but not density nor size, was the only independent determinant of β-cell function assessed by insulin HOMA β-cell. Islet volume was significantly reduced in the patients with overt hyperglycemia, but not in patients with impaired fasting glucose. Insulin HOMA β-cell predicted islet volume better than other measures of fasting insulin secretion. In conclusion, the present study documented a close direct relationship between indexes of β-cell function and islet volume in humans. The insulin HOMA β-cell provides a more reliable estimate of pancreatic islet volume than fasting glucose before islet isolation.
Keywords
Introduction
Homeostatic model assessment (HOMA) provides a quantitative estimate of β-cell function and insulin resistance using basal (i.e., fasting) glucose and insulin or C-peptide concentrations (19,38). The HOMA model is a proven robust clinical and epidemiological tool for describing different pathophysiological aspects of diabetes in humans (6,12,23,24,34,35). Over the years HOMA has become very popular with clinical physiologists, as documented by hundreds of publications (26). However, the accuracy of HOMA indexes in estimating β-cell mass in humans has not yet been determined. This is mainly due to the fact that we have no easy access to the human pancreas for measuring β-cell mass. Routine biopsy sampling of the pancreas is unrealistic, and pancreatic tissue from humans usually becomes available only at autopsy, when the pancreas may have already undergone extensive autolysis (9). Furthermore, measurements of glucose and insulin concentrations in steady state and reliable clinical information are often unavailable for autopsy cases.
All studies of correlation between β-cell mass and function in humans have been using the fractional β-cell area as a surrogate of β-cell mass (8,15,20–22). This approach is imprecise because there are individual differences in the pancreatic weight/volume (31,32,36), and the distribution of islet size and density within the pancreas is not homogeneous (39). An alternative surrogate of β-cell mass is the islet volume after enzymatic digestion of the pancreas for transplantation purposes. In fact, immediately after digestion and before purification islet volume can be measured by a standard counting procedure (27). The major difficulty in using this measure to study the correlation between β-cell mass and β-cell function is the fact that pancreas grafts become available at the time of brain death, a condition that does not allow any realistic evaluation of β-cell function and may itself affect the efficiency of islet isolation (10,13). Alternatively, pancreata from living donors are available in the case of patients undergoing autologous islet cell transplantation; however, this approach is used almost exclusively in patients undergoing pancreatectomy because of chronic pancreatitis (5), a clinical condition affecting islet mass (33), function (11), and isolation efficiency (18).
At the San Raffaele Scientific Institute in Milan some of these hurdles have been overcome. In fact, we started a clinical program for autologous islet cell transplantation (IAT) in a cohort of well-defined patients with pancreas disease other than chronic pancreatitis (3). Many of these patients have also been included in a prospective observational study to describe clinical features, risk factors, and etiopathogenetic aspects of T3cDM (a subset of diabetes mellitus that is associated with pancreatic disease) (2). This allows us to report the total islet volume obtained after digestion of the pancreas from individuals with well-characterized glucose metabolism, including fasting blood glucose, insulin/C-peptide measurements in steady state conditions, glycated hemoglobin, and anti-islet antibodies.
Material and Methods
Patients
We reviewed the records of all patients who were candidates for IAT at the Pancreatic Unit of the Department of Surgery at the San Raffaele Scientific Institute (Milan, Italy) from November 2008 to September 2014. IAT was considered for the following groups of patients (3): (i) painful chronic pancreatitis, without any cephalic mass nor dilation of the main pancreatic duct; (ii) severe complications after pancreatic surgery [grade C pancreatic fistula, according to the definition of the International Study Group on Pancreatic Fistula (21)] requiring relaparotomy with complete pancreatectomy or left pancreatectomy; (iii) patients undergoing pancreaticoduodenectomy in whom pancreatic anastomosis was deemed at high risk of leakage (according to the judgement of the chief surgeon), based on a combination of narrow duct and soft and/or frail pancreatic texture (NCT01346098); (iv) patients undergoing extensive distal pancreatectomy for benign/borderline neoplasm of the pancreatic body neck. Forty-two out of 64 IAT candidates were analyzed in a prospective observational study to describe the clinical features of T3cDM (2) and were included in this study. Informed consent was obtained from patients. The study was approved by the Institutional Review Board of San Raffaele Scientific Institute, and the reported investigations were carried out in accordance with the principles of the Declaration of Helsinki as revised in 2008. The characteristics of study participants are reported in Table 1.
Baseline Characteristics of Study Patients, Stratified by Glucose Tolerance

Post hoc analysis:
NFG versus IFG; ¶NFG versus diabetes;
adenocarcinoma of the ampulla of Vater (n = 5), adenocarcinoma of the distal bile duct (n = 4), adenocarcinoma of the duodenum (n = 2), adenoma of the duodenum and papilla of Vater (n = 2), duodenal gangliocytic paraganglioma (n = 1);
serous cystoadenomas (n = 3), mucinous cystoadenomas (n = 2), intraductal papillary mucinous neoplasms (n = 1), solid pseudopapillary neoplasm (n = 1);
IFG versus diabetes.
Blood Biochemistry Prior to Pancreatic Surgery
Fasting blood samples were obtained during the preoperative workup [upon or just before hospital admission, median 8 days (IQR 1–42 days) before surgery]. Hemoglobin A1c (HbA1c; Bio-Rad Variant II HbA1c analyser; Bio-Rad Laboratories, Munich, Germany), fasting plasma glucose (FPG; glucose-oxidase method, Advia 2400; Siemens Diagnostics, Deerfield, IL, USA), fasting insulin (AIA-PACK IRI; Tosoh, Tokyo, Japan), and serum creatinine (kinetic alkaline picrate method, Advia 2400) were measured in all patients. Autoantibodies to glutamic acid decarboxylase (GADA), insulinoma-associated protein 2 (IA-2A), insulin (IAA), and zinc transporter 8 antigen (ZnT8A) were measured in all patients by radiobinding and immunoprecipitation assays. C-peptide (AIA-PACK C-Peptide; Tosoh) was measured in 31 out of 42 patients. Insulin resistance and β-cell function were estimated using the HOMA2 model (available from www.dtu.ox.ac.uk/homacalculator/) following the recommendations for its appropriate use (38). Glomerular filtration rate (eGFR) was estimated using the simplified Modification of Diet in Renal Disease (MDRD) formula (17). Study participants were defined as having diabetes if they had at least one fasting plasma glucose (FPG) ≥7.0 mmol/L or HbA1c ≥6.5% (48 mmol/mol), or they were prescribed diabetes medications. Study participants with a FPG between 5.6 mmol/L and 6.9 mmol/L were classified as having impaired fasting glucose (IFG). Study participants having FPG < 5.6 mmol/L were classified as having normal fasting glucose (NFG).
Islet Isolation and Purification
Islets were isolated and purified according to the automated method described by Ricordi, with local modifications (25). After digestion at 37°C in a modified Ricordi chamber and before purification, the total islet volume (mm3/g), islet number, and size distribution were measured for each participant. Total islet volume was calculated as previously described (27). Briefly, a sample islet preparation was stained with diphenylthiocarbazone (Sigma-Aldrich, St. Louis, MO, USA) to discriminate β-cells from exocrine tissue. Under light microscopy, the diameter of individual islet was directly measured. Next, islets were categorized according to their diameters within 50-μm increments, and the number of islets in each diameter category was multiplied by a related factor that converts the islet number and diameter category to islet equivalent (IEQ), which corresponded to the tissue volume of a perfectly spherical islet with a diameter of 150 μm (1,767,146 μm3) (27). Islet volume was expressed for each gram of pancreatic tissue digested (mm3/g). Islet density was calculated by dividing the islet number without the conversion in islet equivalent for the grams of digested pancreatic tissue (islet/g). Islet size was expressed as isolation index (islet volume expressed in IEQs per islet number), an indicator of the average size (e.g., an index of 1 indicates that the average islet size is 150 μm and vice versa).
Statistical Analysis
Variables are summarized as mean ± standard deviation (SD) or median and interquartile range (IQR), according to their distribution. Variables were compared with one-way unpaired or paired Student's t-test, one-way ANOVA with post hoc Bonferroni multiple comparison calculation, Wilcoxon signed-rank test, Mann–Whitney U test, or Kruskal–Wallis test, as appropriate. Correlation analyses were carried out using linear or nonlinear regression functions. Decision criteria were the respective regression coefficients (r). All statistical analyses were performed using the SPSS 13.0 (IBM, Armonk, NY, USA).
Results
Islet Volume in Study Population
Twenty-nine out of 42 patients (group A) had pancreatic disease in the head of the pancreas and were originally candidates for a duodenopancreatectomy (DP). The healthy body/tail of the pancreas (65 ± 21 g of tissue) was removed because pancreatic anastomosis was impracticable at the time of DP because of technical difficulties/high risk of leakage (n = 19) or development of severe complications requiring relaparotomy with completion pancreatectomy [median 10 day (min 7–max 32 days) after DP] (n = 10). Among patients in group A, islet yield per tissue weight was 5.11 mm3/g (3.11–6.81–3,852 mm3/g). Eleven out of 42 patients (group B) had benign/borderline neoplasm in the pancreatic body neck. The healthy tail of the pancreas (45 ± 9 g of tissue) was obtained at the time of the extensive distal pancreatectomy (n = 9) or relaparotomy because of severe bleeding after enucleation with completion distal pancreatectomy (n = 2, 12, and 18 days after DP). In group B islet yield per tissue weight was 6.67 mm3/g (5.51–8.41–4,761 mm3/g) (p = 0.032 vs. group A). Finally, 2 out of 42 patients (group C) had chronic pancreatitis, and pancreatic tissue (80 ± 29 g) was obtained after subtotal pancreatectomy. In group C islet yield per tissue weight was 2.75 mm3/g (2.27–3.26 mm3/g) (p = 0.18 vs. group A; 0.036 vs. group B).
Glucose Metabolism
At the time of pancreatic disease diagnosis 6/42 (14%) of our patients already had diabetes (two sixths treated with lifestyle modifications and four sixths of new diagnosis), 8/42 (19%) had impaired fasting glucose (IFG), and 28/42 (67%) had normal fasting glucose (NFG). None of the patients had positive islet-specific autoantibody. As expected, fasting glucose was significantly higher in patients with overt diabetes or IFG compared to patients with NFG (p < 0.001) (Table 1). In contrast, insulin and C-peptide levels were similar in NFG, IFG, and diabetic patients. The islet volume was 5.57, 5.53, and 2.25 mm3/g in patients with NFG, IFG, and diabetes, respectively (p = 0.017) (Table 1), documenting approximately 60% β-cell deficit among patients with diabetes. Insulin HOMA β-cell function was 125%, 72%, and 70% for those with NFG, IFG, and diabetes, respectively (p = 0.092) (Table 1). The fasting insulin-to-glucose ratio and insulin HOMA insulin sensitivity were not different in the three groups (p = 0.31 and p = 0.38, respectively) (Table 1).
Predictors of Islet Volume
The association between islet volume and patients' characteristics, glucose control, and several validated indexes of β-cell function are shown (Table 2, Fig. 1). There was a significant linear relationship between islet volume and fasting blood glucose (r = −0.313, p = 0.043), fasting insulin (r = 0.469, p = 0.002), fasting insulin-to-glucose ratio (r = 0.595, p < 0.001), fasting C-peptide (r = 0.407, p = 0.025), insulin HOMA β-cell function (r = 0.664, p < 0.001), insulin HOMA insulin sensitivity (r = −0.423, p = 0.005), and C-peptide HOMA β-cell function (r = 0.365, p = 0.044). In contrast, no significant association was found between islet volume and age, weight, BMI, creatinine, eGFR, HbA1c, and the C-peptide HOMA insulin sensitivity. Furthermore, when patients with impaired fasting glucose and/or diabetes were excluded from the analyses, the association with fasting insulin, insulin-to-glucose ratio, insulin HOMA β-cell function, and insulin sensitivity remained statistically significant. The strongest predictor of islet volume was insulin HOMA β-cell function, with a stronger association among patients in group A (see Table 3).

Association between islet volume and glucose control. Linear regression of islet volume (mm3 for gram of pancreas) and fasting blood glucose levels (FBG, mmol/L), fasting insulin (pmol/L), fasting C-peptide (nmol/L), insulin HOMA2-%B, insulin HOMA2-%S, and insulin-to-glucose ratio in 28 patients with NFG, 8 patients with IFG and/or IGT, and 6 patients with diabetes. R2 = coefficient of determination calculated by linear regression. Dotted lines: 95% confidence intervals for the mean predicted value.
Linear Regression of β-Cell Mass Estimated by the Islet Volume (mm3/g) and Baseline Characteristics of Study Patients Among Different Groups of Study Participants, Based on Glucose Tolerance

Linear Regression of β-Cell Mass Estimated by the Islet Volume (mm3/g) and Baseline Characteristics of Study Patients, Among Different Groups of Study Participants Based on Diagnosis

Relative Contribution of Islet Number and Islet Size to Islet Volume
Islet counting showed considerable interindividual variation in islet density (median 6,145, interquartile range 3,795–7,242, min 1,009, max 12,348 islet/g pancreas) and size, expressed as isolation index (median 0.50, interquartile range 0.39–0.71, min 0.24, max 1.54) (see Fig. 2). There was a significant negative correlation between islet density and islet size (Fig. 3A). The correlation was best described by an exponential decay function (r = −0.482, p = 0.001) (Fig. 3A). When patients were divided into tertiles based on islet density, the proportion of smaller islets was significantly higher in the highest islet density group than in the lowest islet density group (Fig. 3B). In the univariate analysis, the correlation between isolation index and the islet volume was much weaker than that between islet density and islet volume (Fig. 3C). After adjustment for age, sex, and BMI, islet density and isolation index were both significantly associated with islet volume, although the association was stronger for islet density than the isolation index (β = 0.97, p < 0.00001, and β = 0.693, p < 0.00001, respectively) (Table 4). This suggests that islet number is an important predictor of β-cell mass, while the contribution of islet size is relatively smaller.
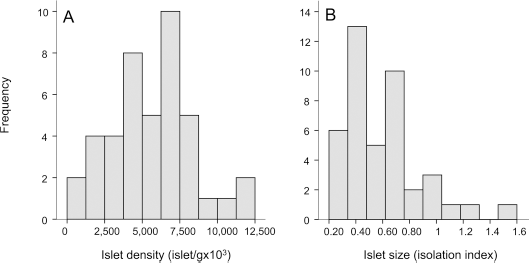
Interindividual variation in islet density and size. Frequency distribution of islet density (A) and isolation index (B) in study participants.

Correlation between islet density and islet size. (A) Relationship between islet density and isolation index. (B) Distribution of islet size among study participants classified into tertile according to islet density. Open bar, lowest tertile (< 4,510 islet/g); gray bar, middle tertile (4,510-6,801 islet/g); and closed bar, the highest tertile (>6,801 islet/g) of islet density. **p < 0.01. (C) Linear regression of islet volume (mm3 for gram of pancreas) and islet density and isolation index. R2 = coefficient of determination calculated by linear regression. Dotted lines: 95% confidence intervals for the mean predicted value.
Univariate and Multivariate Linear Regression Analyses of β-Cell Mass Estimated by the Islet Volume (mm3/g) and Age, Sex, BMI, Islet Density, and Isolation Index

Relationship Between HOMA Indexes and Islet Density and Size
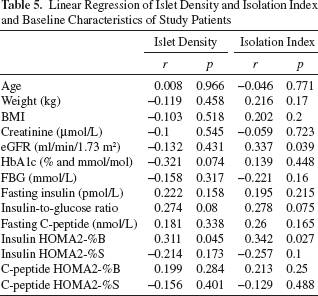
We studied the association between islet density and islet size with patient's characteristics, glucose control, and different indexes of β-cell function (Table 5). In the univariate analysis (Table 6), there was a significant correlation between insulin HOMA β cell and islet density (r = 0.311, p = 0.045) (Fig. 4), as well as islet size (r = 0.342, p = 0.027) (Fig. 4). After adjustment for age, sex, and BMI, islet density and islet size remained both equally significantly associated with insulin HOMA β-cell (β = 0.591, p < 0.001, and β = 0.537, p = 0.001, respectively) (Table 6). This suggests that both islet number and islet size are associated with β-cell function estimated by insulin HOMA β-cell. A significant negative correlation between insulin HOMA2-%S and islet density (r = −0.422, p = 0.012), as well as islet size (r = −0.372, p = 0.03) was also observed, but only after adjusting for age, sex, and BMI (Table 6).
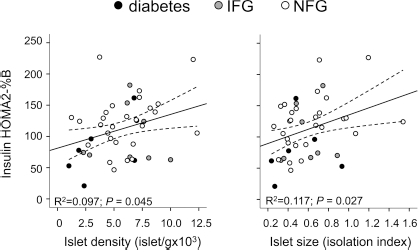
Relationship between HOMA indexes and islet density and size. Linear regression of insulin HOMA2-%B and islet density and isolation index in 28 patients with NFG, 8 patients with IFG and/or IGT, and 6 patients with diabetes. R2 = coefficient of determination calculated by linear regression. Dotted lines: 95% confidence.
Linear Regression of Islet Density and Isolation Index and Baseline Characteristics of Study Patients
Univariate and Multivariate Linear Regression Analyses of Different HOMA Indexes and Age, Sex, BMI, Islet Density, and Isolation Index
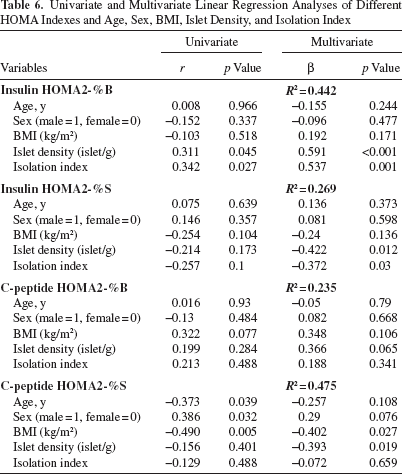
Regarding C-peptide HOMA indexes, islet density, but not islet size, was negatively correlated with HOMA2-%S after adjustment for age, sex, and BMI (r = −0.393, p = 0.019 and r = −0.072, p = 0.659, respectively) (Table 6), while no association was observed with HOMA2-%B.
Discussion
In 2008 at the Scientific Institute San Raffaele we started a clinical protocol in which autologous islet cell transplantation is performed at the time of pancreatic surgery in patients with pancreas disease other than chronic pancreatitis (2,4). This protocol has provided the unique opportunity to investigate whether the islet volume obtained after digestion of the patient's own pancreas is associated with the β-cell function and insulin sensitivity assessed by HOMA prior to pancreatic surgery (19,38). Our study shows that in humans: 1) islet volume is significantly associated with different measures of insulin secretion; 2) insulin HOMA β-cell is associated with islet volume better than other measures of fasting insulin secretion; 3) islet number contributes to islet volume more than islet size; 4) glucose control deteriorates with declining islet volume; and 5) both islet density and islet size are predictors of β-cell function as estimated by insulin HOMA β-cell. These findings are of significant interest because, unfortunately, all imaging techniques available have failed to quantify β-cell mass in vivo in humans with sufficient accuracy and specificity (1). Consequently, a reliable measure of insulin secretion may provide a practical alternative, (40) although, to be useful for clinical purposes, such a measure needs to allow for the repeated assessments of large numbers of patients and predict β-cell mass with accuracy. On the basis of our results, insulin HOMA β-cell may partially satisfy these requirements.
An association between insulin secretory responses to intravenous glucose and/or arginine administration and transplanted islet volume was previously reported in patients with type 1 diabetes after islet allotransplantation (14,30) and in patients with pancreatectomy for chronic pancreatitis after islet autotransplantation (37). The present study extends those results by providing for the first time direct evidence of a correlation between islet volume and measures of fasting insulin secretion in nontransplanted individuals with and without diabetes. Indexes of β-cell function calculated using insulin concentrations were more strongly associated with islet volume than those calculated using C-peptide. This may be because there was significant heterogeneity in eGFR among study participants and, consequently, in C-peptide clearance and basal levels.
Although our data support the use of insulin HOMA β-cell for the assessment of islet volume in humans, it is important to acknowledge the potential limitations of our study. First, the study was not designed to fully test β-cell function in the pretransplant period and insulin and C-peptide measurements after stimulation (e.g., Mixed Meal Test). It should be mentioned that stimulated measures of insulin secretion, as reported by other authors after islet autotransplantation (29,37), exhibit stronger correlation coefficients with measures of islet volume than what is being reported for HOMA-β in our study, suggesting that indices of β-cell function derived from fasting measures remain surrogates for the more robust measures derived, for example, from glucose potentiation of arginine-induced insulin secretion. Second, all the patients included in our study had pancreatic disease and were candidates for islet autotransplantation, introducing a potential bias for the efficiency of pancreas tissue digestion. Moreover, the measure of β-cell mass after the isolation procedure could be partially underestimated because of the damage to cells, which certainly occurs during the isolation procedure. Third, the current calculation method tends to overestimate true islet volume (7). Furthermore, islet volume was assessed for the islets in the body and/or tail and not in the whole pancreas. In addition, islet volume includes endocrine cells other than β cells and, consequently, is only a surrogate of β-cell mass. Finally, it is also important to stress that any association between functional indexes of insulin secretion and islet volume is valid exclusively in the absence of concomitant diabetes treatment.
Despite these limitations, the results of our study are important for understanding the relationship between β-cell function and β-cell mass. Both islet number and islet size determine β-cell mass, but their relative contribution in humans remains largely undefined. The present study suggests that islet number rather than islet size is a major determinant of total islet volume, even if both are equally significantly associated with insulin HOMA β-cells. A considerable variation in islet number and size among patients included in our study was evident, but the underlying mechanism for the interindividual difference is unclear. Increased islet size might be a compensatory response to decreased islet number, a finding consistent with the significant negative correlation between islet density and islet size observed in the present study. In agreement with our results, Kou et al. (15) have recently reported in 72 adults with no history of diabetes that islet density is more strongly correlated with β-cell mass than islet size. Moreover, Le Bacquer et al. (16) have reported that islet density is decreased in nondiabetic adults who are carriers of the type 2 diabetes susceptibility gene TCF7L2 rs7903146 (T/T) compared with noncarriers, while islet size was rather increased. All together these data suggest that islet number or density may be genetically determined, and diabetes may develop when the compensatory increase in islet size is insufficient to compensate their reduced number.
In our study a 60% islet volume reduction was evident in patients with diabetes, a deficit already reported using the fractional β-cell area as a surrogate of β-cell mass (9,20,21). Although islet volume was significantly reduced in patients with overt hyperglycemia, there was no reduction in patients with IFG. In line with this finding a very small reduction in β-cell area was also reported in patients with IFG undergoing surgery for underlying pancreatic disorders (especially chronic pancreatitis) (21). These findings are in contrast with those reported by Butler and colleagues showing a ~40% deficit in β-cell area in individuals with IFG (9,28). It is important to emphasize that in our study only patients with underlying pancreatic or abdominal disorders requiring pancreatic surgery were included, whereas Butler and colleagues studied a group of patients more likely to have (pre-) type 2 diabetes. In line with this, the mean BMI of the patients in the IFG group studied by Butler and colleagues was ~37 kg/m2, whereas our patients with IFG had a mean BMI of 26.9 kg/m2. It is therefore likely that the mechanisms underlying the β-cell destruction in the patients included in this study may be different from those in obese patients and insulin resistance, and therefore, the findings of our study should not be readily generalized to all patients with type 2 diabetes.
In conclusion, our study documented an association between indexes of β-cell function and islet volume in humans in vivo. Insulin HOMA β-cell may provide a reasonable estimate of pancreatic islet volume, whereas other indexes appears to be less suitable in this regard. Insulin HOMA β-cell may be useful to assess the β-cell loss in longitudinal studies and to estimate residual islet volume in individual patients before islet isolation.
Footnotes
Acknowledgments
This study was supported by the Italian Minister of Health (Ricerca Finalizzata RF-2009-1483387), Ministry of Education, University and Research (PRIN 2008, Prot. 2008AFA7LC), EU (HEALTH-F5-2009-241883-BetaCellTherapy), and Associazione Italiana per la Ricerca sul Cancro (AIRC, bando 5 × 1,000 N_12182 and Progetto IGN_11783). We thank the nursing personnel of the Department of Surgery (San Raffaele Scientific Institute) for their invaluable help in caring for the patients included in this study. R.N., R.F., and A.M. performed islet isolation. M.S. and R.B. critically reviewed and edited the manuscript and contributed to the discussion. R.N., G.P., and L.P. developed the concept, designed the experiments, wrote the manuscript, promoted the study, and researched data. Dr. Lorenzo Piemonti is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for data integrity and accuracy of data analysis. The authors declare no conflicts of interest.
