Abstract
Restorative cell therapy concepts in neurodegenerative diseases are aimed at replacing lost neurons. Despite advances in research on pluripotent stem cells, fetal tissue from routine elective abortions is still regarded as the only safe cell source. Progenitor cells isolated from distinct first-trimester fetal CNS regions have already been used in clinical trials and will be used again in a new multicenter trial funded by the European Union (TRANSEURO). Bacterial contamination of human fetal tissue poses a potential risk of causing infections in the brain of the recipient. Thus, effective methods of microbial decontamination and validation of these methods are required prior to approval of a neurorestorative cell therapy trial. We have developed a protocol consisting of subsequent washing steps at different stages of tissue processing. Efficacy of microbial decontamination was assessed on rat embryonic tissue incubated with high concentrations of defined microbe solutions including representative bacterial and fungal species. Experimental microbial contamination was reduced by several log ranks. Subsequently, we have analyzed the spectrum of microbial contamination and the effect of subsequent washing steps on aborted human fetal tissue; 47.7% of the samples taken during human fetal tissue processing were positive for a microbial contamination, but after washing, no sample exhibited bacterial growth. Our data suggest that human fetal tissue for neural repair can carry microbes of various species, highlighting the need for decontamination procedures. The decontamination protocol described in this report has been shown to be effective as no microbes could be detected at the end of the procedure.
Keywords
Introduction
Fetal neural tissue transplantation has become a potential symptomatic treatment and disease management option for patients suffering from neurodegenerative diseases such as Parkinson's disease (PD) and Huntington's disease (HD). During the last two decades, several clinical trials on fetal tissue transplantation in PD and HD have been initiated. It has been shown that cells isolated from first trimester human fetuses have the capacity to survive transplantation, acquire a mature neuronal phenotype, and mediate a functional benefit (9,13,14,29,39). Since functional outcome was inconsistent between trials, new efforts like the EU-funded TRANSEURO trial (http://www.transeuro.org.uk) for neural transplantation in PD aim at optimizing the procedures and enrollment criteria in order to provide a better clinical outcome. Unlike trials in the past, neural transplantation of fetal tissue has to be performed in accordance with the guideline on good manufacturing practice (GMP), which applies to manufacturing of cell therapeutics and conventional drugs in Europe. Therefore, aspects of microbiological safety have to be investigated in detail.
Owing to the origin of fetal human tissue derived from elective low-pressure aspiration or medical abortions, a potential infection of the fetal human tissue with microbes from the female vaginal tract with subsequent potential infection of the host brain after transplantation has to be considered. The spectrum of microbes colonizing the female genital tract is broad with Lactobacillus species being predominant (7,10), and some of these organisms may have physiological protective and immune modulatory functions (41). Besides bacteria, yeasts of the Candida albicans species are part of the physiological vaginal flora but can also cause clinically apparent infections in most women during their reproductive lifetime (12,23).
While standards have been established for virological screening, including patient questionnaires and serological tests, published data describing methods for microbiological analysis and risk for contamination of fetal tissue are rarely available. Infections of the host were reported in individual cases in whole ganglionic eminence transplantation of HD patients (18,21), but it remains so far unclear whether this was due to the surgical procedure or due to contaminated fetal tissue. A single report on microbiological screening of human fetal tissue summarizes microbiological data of various stages of a human ganglionic eminence tissue preparation procedure (11). This investigation provides evidence for the possibility of microbes contaminating fetal tissue. However, the authors did not validate the applied washing procedure and methods of analysis, and the screening method was not suitable for large-scale systematic microbiological screening.
Strategies to remove such contaminations are required. Antibiotic treatment of fetal tissue prior to implantation has been applied in rare cases in past trials (20). However, it is unknown whether the period of antibiotic treatment prior to transplantation is sufficient for significant microbial reduction. In the case of tissue storage, antibiotic efficacy may be compromised due to low temperatures and slow microbial metabolism during tissue storage. In the present study, we developed a protocol based on subsequent washing steps to decontaminate human fetal tissue. Our analytic method is based on an established industrial system, which allows for semiautomated and highly sensitive detection of microbes (BacT/ALERT). It has been described for a variety of applications in blood product testing and pharmaceutical production (19,24) and has been tested for similar purposes in microbiological monitoring during umbilical cord blood recovery and storage (33). We established a model simulating the clinical protocol by artificially contaminating sterile fetal rat tissue with clinically relevant microbes at very high doses. We then designed a standardized washing method for microbial reduction of the contaminated tissues. By applying this method, we provide evidence for the first time that microbial burden can be reduced up to a factor of 106. Subsequently, we used the same washing and test methods to analyze clinically relevant human fetal tissue samples obtained for transplantation in patients with HD, showing that microbial contamination of fetal tissue present after abortion is no more detectable using our protocol.
Materials and Methods
Human fetal tissue acquisition was accomplished within a multicenter clinical trial investigating the restorative capacity of fetal cell transplantation in HD [Multicentric Intracerebral Grafting in Huntington's Disease (MIG-HD); Clinicaltrials.gov identifier: NCT00190450]. Informed written consent of the women was obtained on the day of surgery [for ethical guidelines on the use of fetal tissue see report of the Network of European CNS Transplantation and Restoration NECTAR (3)]. Ethical approval for work on human fetal tissue was acquired from the ethics committee of the University of Freiburg, Medical Centre. In addition, experiments including rat tissue samples were approved by the animal research committee of the local authorities. All animal experiments were performed according to the animal protection directive of the European parliament (EU/2010/63).
Bacteria Growth Media, Agar Preparation, and BacT/ALERT Culture Bottles
Columbia blood agar contained Columbia blood agar basis 220 g (BD Bioscience, Franklin Lake, NJ, USA) in 5 ml Aqua VE (demineralized, distilled water, produced by an ultrafiltration osmosis facility provided by HERCO Wassertechnik, Freiberg am Neckar, Germany). Glucose yeast extract cysteine (GYEC) agar contained bacto agar, yeast extract (both from BD Bioscience), NaCl, pepton, glucose, meat extract, KOH 9.09% (all from Merck, Darmstadt, Germany), vitamin K1 (1%; Sigma Aldrich, St. Louis, MO, USA), hemin solution (0.05%), l-cysteine solution 3% (both from Serva, Heidelberg, Germany), and sheep blood (Fiebig Animalblood Products, Idstein, Germany). Hematin agar contained Columbia agar basis, 0.025% hemin solution, and sheep blood. For semiautomated evaluation, aerobic BacT/ALERT BPA bottles (bioMérieux, Marcy-l'Etoile, France; 40 ml supplemented tryptic soy broth in oxygen) and anaerobic BacT/ALERT BPN bottles (bioMérieux; atmosphere of CO2 in nitrogen) were used. The semiautomatic analysis of the BacT/ ALERT system is based on an indicator that reacts to CO2 produced by microbes. If no color change was detected by the system within up to 14 days of incubation, the sample was considered to be sterile.
Rat Tissue Acquisition and Experimental Contamination
Time-mated pregnant female Sprague–Dawley rats (Charles River Laboratories, Inc., Sulzfeld, Germany) were sacrificed between embryonic day E16 and E18, and all fetuses were removed from the uterus. Whole embryonic brains including upper spinal cord segments were dissected under sterile conditions. Tissue processing and further washing steps were performed according to the human tissue protocol. Thus, each fetal brain was kept separately in precooled 100 ml sterile Hanks' balanced salt solution (HBSS, Gibco, Grand Island, NY, USA) until bacterial contamination. Artificial microbe contamination was carried out using the following bacterial and fungal strains: Staphylococcus aureus, Lactobacillus jensenii, Pseudomonas aeruginosa (Leibniz Institute DSMZ -German Collection of Microorganisms and Cell Cultures, Braunschweig, Germany), Corynebacterium amycolatum, Bacillus subtilis, Candida albicans, and Clostridium sporogenes [American Type Culture Collection (ATCC), Manassas, VA, USA] (Table 1). All microbes were cultured on Columbia blood agar with the exception of Clostridium sporogenes, which was plated on GYEC agar. Microbes were cultivated under appropriate aerobic (6% CO2) or anaerobic conditions (GENbox anaer, bioMérieux) overnight at 37°C.
Microbes Used for Experimental Contamination of Rat Tissue

Microbes were selected in order to comprise key features of possible contaminants in neurorestorative cell therapies using fetal source tissue. The code in parentheses specifies the code of the used bacterial strains (DSM: acquired from Leibniz Institute DSMZ - German Collection of Microorganisms and Cell Cultures, Braunschweig, Germany; ATCC: American Type Culture Collection, Manassas, VA, USA).
For experimental tissue contamination, a microbiological load above levels expected in human samples was chosen. Appropriate microbial concentration was achieved by serial dilution of overnight cultures in phosphate-buffered saline (PBS; Gibco). Cell density was measured by the McFarland method (25). Defined volumes were plated as control onto agar for colony counts. These colony counts represented the real contamination load and were used for further calculations. Differences from the target value up to one logarithmic step were tolerated (for initial bacterial concentrations, see Table 2).
Quantification of Experimental Contamination in Rat Tissue

Efficiency of subsequent washing steps was validated on rodent tissue. The left column indicates the spectrum of microbes used for experimental contamination. The following column shows the number of microbes in the fluid used for artificial contamination, measured by an independent plating experiment. For samples A to C, the mean number of colonies is displayed. Minimum and maximum numbers are given in parentheses. All washing experiments were repeated three times. All numbers are related to the volume of 1 ml.
Ten milliliters of these microbe dilutions was added to the fetal brain samples, mixed gently, and incubated at room temperature for 2 h with gentle horizontal agitation to mimic transport of the human sample from the gynecologist to the cell therapy lab, following 4-h incubation at +2-8°C. As a control, 10 ml of sterile PBS was added to a bottle of 100 ml HBSS containing one fetal brain. All experiments were repeated three times for each microbe and concentration.
Human Fetal Tissue Collection
Human fetal tissue from the first trimester of pregnancy was obtained from routine elective low-pressure aspiration abortions as part of the ongoing MIG-HD neurotransplantation trial. Fetal tissue was collected in precooled 100 ml sterile HBSS (storage fluid) immediately after surgery and stored at +2-8°C for a maximum duration of 6 h, including transport to the cell therapy lab (Fig. 1).
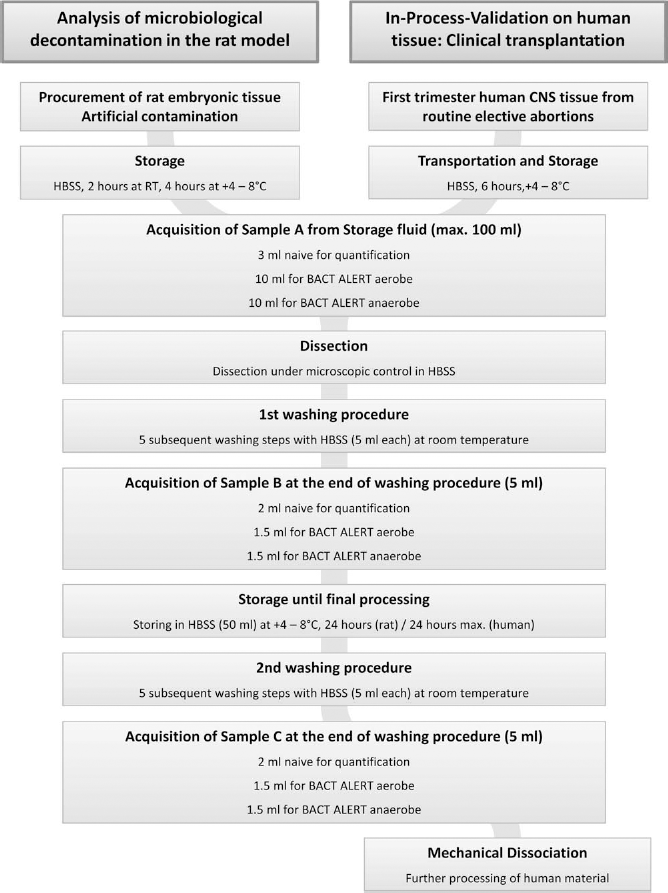
Flow chart for tissue processing and acquisition of samples. The intervals as stated in the chart represent the maximum intervals as accepted for the clinical trial protocol. Samples A and B were taken at the day of arrival. Sample A was taken from the transportation fluid, while sample B represented the level of contamination after five steps of washing of the dissociated tissue. After overnight storage, tissue pieces received five further washing steps, and sample C was taken prior to final tissue processing. HBSS, Hanks' balanced salt solution.
Tissue Processing and Microbe Depletion in Rat and Human Tissue
To characterize microbial decontamination of rat tissue and to perform in-process validation on human material, three supernatant samples were taken for microbiological analysis at three sampling time points (A-C) of tissue processing (Fig. 1).
The first sample (sample A) was directly taken from the storage medium and consisted of a 3-ml naive sample and a set of single aerobic and anaerobic BacT/ALERT bottles filled with 10 ml transportation fluid each. The dissected tissue was then washed five times for 3 min in 5 ml sterile HBSS with gentle agitation at room temperature. The second sample (B) was collected from the final washing step solution (5 ml). A 2-ml naive sample was used for colony quantification and 1.5 ml each for aerobic and anaerobic incubation in BacT/ALERT bottles. The dissected tissue was then stored in 50 ml sterile HBSS at +2–8°C for 24 h in the rat study and for a maximum of 24 h in human tissue samples. Following tissue storage, the sequential washing procedure consisting of five washes with 5 ml sterile HBSS was repeated, and the washing solution from the last washing step was fully collected for the third sample (sample C) and split into three parts as done before for sample B.
Microbiological Analysis
For colony quantification in contamination assays, 10 μl and 100 μl (depending on the expected colony count) of naive samples taken at each step were inoculated on two agar plates. Columbia blood agar or GYEC agar was used for aerobic or anaerobic overnight incubation at 37°C, depending on the species chosen for contamination.
Afterward, the initial number of microbes/ml was calculated, and the values from the two samples were averaged. In a number of cases, bacterial growth was only detected in BacT/ALERT bottles, but not on agar plates inoculated with naive samples. Therefore, absolute quantification was not possible, since the BacT/ALERT system does not allow quantification of colony-forming units (CFUs). In these cases, the minimum number of positive microbes was estimated in relation to the volume plated on blood agar plates: after plating of 100 μl and detection of bacterial growth, this would correspond to 1 CFU/100 μl and thus 10 CFU/ml. In cases of detection of bacterial growth in BacT/ALTERT bottles but not on agar plates, the number of CFU was therefore estimated to be 10 CFU/ml.
For human samples, in order to allow detection of a broad spectrum of microbes from the vaginal flora, naive specimens were inoculated on various agar plates [Columbia blood agar, Chocolate agar (hematin), GYEC agar]. For each specimen, two agar plates were inoculated with 330 μl of naive sample fluid. Human samples were inoculated into BacT/ALERT bottles as described above for rat samples. BacT/ALERT bottles were incubated at 37°C for up to 14 days. If BacT/ALERT indicated bacterial growth, samples were drawn from the bottles and subcultivated for further identification. Aerobe bottles were subcultivated aerobically on Columbia blood and hematin agar, while anaerobe bottles were subcultivated on Columbia blood (aerobically) and GYEC agar (anaerobically). For identification, two automated test methods were applied. The Vitek 2 System (bioMérieux) identifies microbes based on an array of biochemical tests and performs a fluorescence-based readout. Alternatively, matrixassisted laser desorption ionization time-of-flight analysis (MALDI-TOF, Microflex LT, Bruker, Fremont, MA, USA) was used. Both methods have become clinical routine for microbiological identification and susceptibility testing (6,40). All microbiological differentiation raw data were reviewed and interpreted by a clinical microbiologist.
Results
Microbial Decontamination by Subsequent Washing Steps in Rat Tissue
To determine the efficacy of the washing procedure and to verify the sensitivity of the analytic system, brain structures from E16 to E18 rat fetuses were contaminated with a set of microbes modeling several properties of typical bacteria and fungi one would expect to find on human samples (Table 1). Three samples were taken for microbiological analysis at three different time points of tissue processing (sample A from the transportation fluid, sample B after washing steps, sample C after storage and final washing steps). For each of the samples A–C, two agar plates and a pair of aerobe and anaerobe BacT/ALERT bottles were inoculated. Tissue processing was performed three times for each microbe, thus giving information on bacterial growth from six agar plates and three pairs of aerobe and anaerobe BacT/ALERT bottles for each of the samples A–C.
For all microbes, contamination was strongly reduced between sample A (transportation fluid) and sample B (taken after the first washing procedure). For most microbes, only a minor further reduction was seen in sample C (after 24-h storage and second washing procedure) (Table 2, Fig. 2).
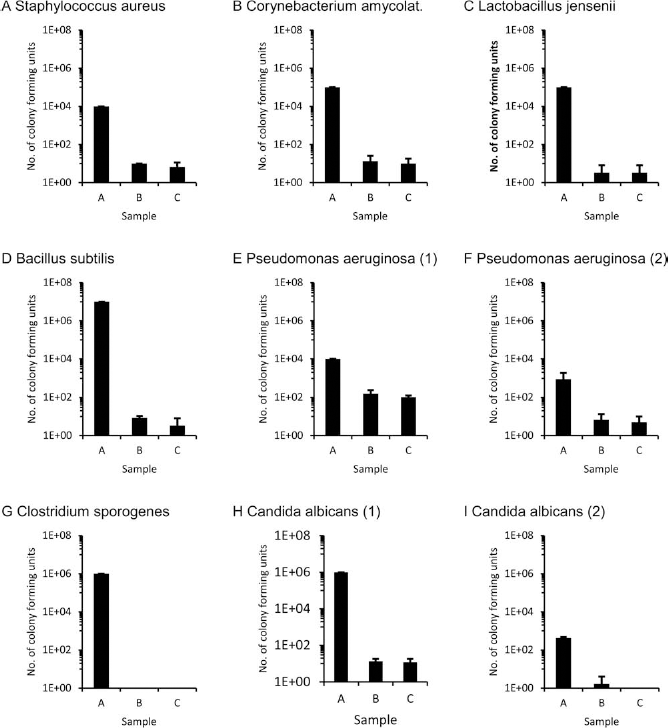
Microbial decontamination in the rat model by subsequent washing steps. Tissue was first artificially contaminated with defined fluids (for details, see Table 2). For some of the microbes, different concentrations were used (E/F; H/I). Sample A was taken from the initial storage fluid after 6 h of incubation, while sample B was taken after tissue dissection and five washing steps, and sample C was taken after 24 h and five additional washing steps. The number of colony-forming units is provided on the y-axis in a logarithmic scaling. The error bars represent the standard error of mean. Microbial contamination was strongly reduced by at least two log ranks for all microbes.
For Staphylococcus aureus, an initial contamination of 104 CFU/ml was measured in sample A. Subsequent washing steps strongly reduced bacterial contamination, and none of the agar plates for samples B and C showed bacterial growth. BacT/ALERT bottles still indicated some bacterial growth in all three repeats of sample B and for two repeats in sample C (Fig. 2A).
A reduction of more than three log ranks was obtained for Corynebacterium amycolatum from sample A (105 CFU/ml) to sample B (13.3 CFU/ml). In samples B and C, one of the agar plates showed bacterial growth, and in both samples, two BacT/ALERT bottles indicated bacterial growth (Fig. 2B).
For Lactobacillus jensenii, bacterial growth was reduced by four log ranks from sample A to samples B and C. In sample B, bacterial growth was found on one agar plate, while all BacT/ALERT bottles were negative. In sample C, no agar plates but one of the BacT/ALERT bottles indicated bacterial growth (Fig. 2C).
For Bacillus subtilis, bacterial growth was reduced by six log ranks from sample A to B. While colony counts higher than 107 CFU/m (confluent growth) were detected on all agar plates in sample A, only a single colony was found on one of the agar plates in sample B, and agar plates were negative in all cases in sample C. BacT/ALERT bottles gave a positive signal in all repeats in sample B and in one of the three repeats in sample C (Fig. 2D).
Contamination with Pseudomonas aeruginosa was reduced from 104 CFU/ml in sample A to 153.3 CFU/ml in sample B and 103.3 CFU/ml in sample C. Along with growth detection in BacT/ALERT bottles, cultures on agar plates exhibited colony growth in all repeats and reduction of the microbial burden was less pronounced compared to the other microbes analyzed in this study (Fig. 2E, F). Since Pseudomonas aeruginosa is known to cause severe infections in immunodeficient patients, we performed a further decontamination experiment using a reduced initial bacterial concentration (1.2 × 103) giving rise to a reduced bacterial growth in sample A (907 CFU/ml). After subsequent washing, bacterial growth was found in BacT/ALERT bottles in two cases for both samples B and C. Growth of one colony was counted on an agar plate in a single repeat of sample C.
For Clostridium sporogenes, microbial decontamination was very effective: whereas sample A contained 107 CFU/ ml, there was no bacterial growth on agar plates or in BacT/ ALERT bottles in samples B and C (Fig. 2G).
Candida albicans was selected to represent a fungal infection. Contamination was strongly reduced from sample A (106 CFU/ml) to samples B (13.3 CFU/ml) and C (8.6 CFU/ml). Colony growth on agar plates was detected in one run for sample B and in two runs for sample C, and all BacT/ALERT bottles for samples B and C indicated growth of Candida. Since fungal infections represent a severe threat to immunodeficient patients, we further analyzed whether complete removal could be obtained in rat tissue when lower and more realistic initial contaminations were applied. Thus, after an initial inoculation with 1.0 × 103 CFU/ml, 420 CFU/ml were measured in sample A. In sample B, growth of Candida albicans was observed on one of the agar plates, whereas all agar plates and BacT/ALERT bottles in sample C were negative, indicating complete removal of Candida albicans contamination (Fig. 2H, I).
In the majority of cases, bacterial growth was detected in either BacT/ALERT bottles only or in parallel in BacT/ ALERT bottles and on agar plates. Growth on agar plates, but not in BacT/ALERT bottles, was only found for a single repeat of contamination with Candida albicans, Lactobacillus jensenii, and Corynebacterium amycolatum.
Microbial Decontamination in Human Tissue
In total, 44 human fetal tissue samples were analyzed for microbiological monitoring during tissue processing for clinical applications. Possible contaminations were assessed using inoculation of naive fluid on agar and in aerobic and anaerobic BacT/ALERT bottles, similar to the procedures described for rat tissue.
In samples taken from the transportation fluid after arrival at our clinic (sample A), a contamination was detected in 21/44 samples (47.7%). Microbial contamination was never observed during or after the washing procedures in samples B or C. Within our analytic setting, anaerobic BacT/ALERT bottles were most sensitive compared to aerobic BacT/ALERT bottles or inoculation of agar plates (Fig. 3A). A positive signal in anaerobic BacT/ ALERT bottles was observed in 16 cases. Aerobic BacT/ ALERT bottles returned a positive signal in 15 cases, while inoculation on Colombia blood agar resulted in detection of contamination in seven cases (15.9%). Microbial growth was never detected in naive specimen in cases where the corresponding BacT/ALERT samples were negative, but naive medium plating added to the microbial spectrum in a few cases (see below).

Characterization of microbial contamination in 44 human tissue samples. (A) Overview on all positive samples analyzed during in-process validation of human tissue (comprising samples A–C). (B) Representation of the spectrum of microbial contamination in sample A. Microbial contamination was never detected in samples B and C. Abbreviations: Sp., Species; m., maltophilia.
Microbial Spectrum Identified in Human Naive Fluid and BacT/ALERT Samples
Microbiological differentiation revealed the spectrum displayed in Figure 3B. The most common microbes detected in transportation fluid were part of the Lactobacillus species (n = 9). Other microbes belonged to the genus Staphylococcus [n = 5; Staphylococcus hominis (n = 1); Staphylococcus epidermidis (n = 1); unspecified (n = 3)] or Streptococcus species [n = 4; Streptococcus anginosus (n = 2) and Streptococcus mitis (n = 1); unspecified (n = 1)]. Microbes that were found in rare cases comprised of Corynebacterium species, Bacillus species Stenotrophomonas maltophilia and Gardnerella vaginalis (n = 2 each). Microbes that were found in single cases were Proprionibacterium, Prevotella bivia, or belonged to the Pseudomonas species. In the majority of cases where microbial growth was detected on naive medium, the strains were also identified in BacT/ALERT bottles. However, in two cases, naive medium revealed a contamination with Lactobacillus that was not detected by the BacT/ALERT system at the same time.
By inoculating agar plates with naive HBSS sample fluid, we aimed at increasing the spectrum identified with our microbiological method and, at the same time, allowing for microbe quantification. In this case, contrary to the rodent tissue-washing experiment, only in a few instances was microbe growth detected on culture plates inoculated with naive medium from sample A. Most microbes grown from naive medium belonged to the Lactobacillus species (n = 6). Mean colony numbers for Lactobacillus were 33.3 CFU/ml (ranging from 1 to 100 CFU/ml). Staphylococcus hominis (6 CFU/ml) and Staphylococcus epidermidis (1 CFU/ml; found in combination with Lactobacillus) were found in a single case each in naive medium samples. Both species grew also in BacT/ALERT bottles at the same time.
Discussion
To date, more than 350 PD patients (2) and approximately 100 HD patients have received primary fetal tissue grafts. While so far no contamination has been notified in reports of fetal tissue transplantation in PD, microbial complications in HD transplantation have been published in single cases. Reporting the outcome of seven transplanted patients (18), medical action was required in three cases due to positive microbiological findings linked with the transplantation procedure. One patient was treated preventively with piperacillin for 2 weeks after identification of Alcaligenes dentrificans in the transportation medium. Another grafted patient developed fever within 36 h after surgery. A donor tissue culture showed growth of Streptococcus viridans and Candida albicans, and the patient was therefore treated for 2 weeks with fluconazole orally and vancomycin intravenously. A third patient had a superficial wound infection after surgery, and methicillin-sensitive Staphylococcus epidermidis was identified in cultures taken from the wound. This patient was treated with vancomycin for 2 weeks. The authors did not give specific information on any remaining clinical worsening due to potential infections. However, the report did not suggest that the infections had any lasting impact on the patient or the graft.
In another report on two patients who had received transplants produced from the fetal whole ganglionic eminence (WGE), one of the patients developed pyrexia, neck stiffness, and reduced levels of consciousness after withdrawal of immunosuppression 1 year after grafting. Specimens taken from the cerebrospinal fluid were sterile, but a lymphocytosis was detected in the liquor. Brain magnetic resonance imaging (MRI) revealed an edema around transplant tracks. An unspecified broad spectrum antibiotic treatment including tuberculostatics was administered. Finally, the patient was reported to have recovered clinically to baseline levels (32). However, a similar clinical report on another HD patient who had received fetal grafts within another trial also reveals the possibility that functional worsening, and the picture of a cerebral inflammation might resemble clinical signs of a graft rejection (22).
One report of a single case of bone flap infection in a series on three HD patients who received fetal lateral ganglionic eminence (LGE) grafts did not provide further details on the microbes found (21). Microbiological testing included cultivation of adjacent fetal tissue pieces, but further details on methods were not given. The authors claimed that all microbiological tests were negative.
Owing to the lack of systematic analysis in many trials, it is unclear whether clinical infections in grafted patients were associated with the surgical procedure, with a potential contamination of the fetal tissue during abortion or due to microbial contamination during tissue processing. It is thus surprising that, so far, only a single group has published data from a systematic assessment of fetal tissue contamination for cell transplantation (11). The system of analysis described in this report was considered as prospective validation rather than as in-process control. Therefore, the applied methods consume a large amount of fetal tissue, which cannot be transplanted and relies on a high amount of manual work during microbiological testing.
In contrast, the system proposed in the present report is intended to overcome these shortcomings: The direct inoculation of BacT/ALERT bottles should allow for a highly sensitive detection of microbes, while naive medium is inoculated on special agar plates, which should facilitate the detection of a broader microbial spectrum. Since our system analyzes a significant portion of transportation fluid as well as the washing fluid from subsequent washing procedures, no valuable transplantable tissue needs to be used.
Microbial Spectrum Found in Human Fetal Tissue Samples for Cell Transplantation
The severity of a potential infection of the host due to a microbial contamination of the graft will depend on the amount of microorganisms and also on pathologic features of the infecting species. We identified a broad spectrum of microbes with different virulence factors. Some of the bacterial species detected are of particular relevance due to their known capacity to form cerebral abscesses.
The most frequently identified microbes in our human tissue samples belonged to Lactobacillus (n = 9), Staphylococcus (n = 5), Streptococcus species (n = 4), and Gardnerella species (n = 3). While Lactobacillus and Gardnerella species are not known to be commonly associated with cerebral infections, certain Staphylococcus and Streptococcus strains may cause CNS infections (1).
Bacteria belonging to the Staphylococcus strains are facultative pathogens, which are known for their involvement in cerebral abscess formation (1,5). Beside Staphylococcus hominis and Staphylococcus epidermidis, which were found in the present analysis, Staphylococcus aureus is one of the most critical pathogens in the CNS, causing abscess formation and local wound infections, especially after neurosurgical interventions. Staphylococcus aureus was not found in our samples, but might contaminate donor tissue at each surgical step involved (e.g., abortion of pregnancy, tissue transplantation).
Streptococcus bacteria also play a role in brain abscess formation: Streptococcus pyogenes and Streptococci of the anginosus group are known to be more virulent and are regularly present. Nonetheless, all named Streptococcus species can be found in the urogenital tract and could potentially contribute to intracerebral abscess formation (1,5).
Microbes found in rare cases (n = 1–2 in our study) may also cause CNS infections. Prevotella strains have been described in cases of brain abscesses with sinusoidal or dental origin (4,8,34). Proprionibacterium is another pathogen associated with surgical infections. The clinical course often consists of a chronic, sometimes unapparent cerebritis with or without abscess formation. In rare cases, the infection can become clinically relevant for the first time as late as years after infection (27,30). Pseudomonas species are known for causing wound infections but are less common in brain abscess formation. However, there are rare cases of encephalitis with abscess formation due to this microbe (16,38). In two cases in the present study, bacteria belonging to the Stenotrophomonas strain have been found in the transportation fluid. Although neurosurgery and previous carbapeneme treatment are discussed as potential risk factors, reports on Stenotrophomonas being the cause of the disease are extremely rare (42). The remaining species identified, such as Corynebacteria and Bacilli, are facultative pathogens, are less virulent, and play only a minor role in cerebral infections.
Taken together, the present study reveals the broad spectrum of potential microbe strains found in first trimester abortions, which could infect the host brain. Beside microbes typical for the vaginal flora, species that usually colonize the skin and the oral cavity have been detected. In contrast with previous reports, we did not detect fungal microbes in the transportation fluid. However, due to past reports of potential contamination with Candida albicans (11,18), the possibility of fungal contamination in the transportation fluid should not be excluded.
Microbial Reduction in Rat and Human Tissue
In the contamination experiments, we infected rodent tissue with a defined microbe suspension at a known concentration in order to investigate the washing efficiency for various bacteria. Following 6 h of storage, intended to simulate the transportation time passing between abortion and dissection of brain tissue, in most cases colonies were growing confluent. Therefore, CFU numbers had to be estimated and were set to the highest colony density, which could still be separated in plating experiments. Therefore, estimated numbers for the transportation fluid (sample A) are probably still lower than real CFU numbers.
The washing steps following dissection of tissue resulted in a microbial reduction of several log ranks. The lowest washing efficiency was measured for Pseudomonas aeruginosa with a reduction of only approximately two log ranks. The second washing procedure resulted in further microbial reduction for samples B and C for most microbes. A complete removal was only found for Corynebacterium amycolatum and Candida albicans.
In contrast, human fluid samples did not give rise to bacterial growth in B and C samples, conflicting with the remaining contamination detected in rodent tissue samples. Several considerations might explain these differences. Microbes used in experiments on rodent tissue were defined strains with certain recognized characteristics, such as expression of specific adherence molecules or resistance to certain treatments. These lab strains do not necessarily resemble features of wild-type microbes, which are adapted to distinct natural niches, for example, vaginal or skin flora. In this respect, tissue adherence of microbes may have to be considered as well with easy reduction of nonadherent microbes by replacing the contaminated transportation fluid, while tissue-adherent microbes would remain attached to the tissue. In this scenario, microbes may not be detected by our system, and further washing procedures would not contribute to a relevant cleaning of tissue.
Differences between human and rodent studies may also be explained by the amount of initial microbial contaminations. While rodent samples were contaminated with microbe concentrations ranging from 103 up to 7.0 × 106 CFU/ml, maximum colony counts in human samples mostly were below 100 CFU/ml. Microbiological quantification in low ranges may be hindered by the sensitivity of the methods. In many cases, not a single colony was detected on agar plates inoculated with naive medium, but BacT/ALERT bottles still revealed bacterial growth. In rodent contamination experiments, the sample volume on agar plates was 100 μl. Thus, at least one microbe per 100 μl or 10 CFU per ml needed to be estimated as minimum concentration. For the BacT/ALERT system, the higher amount of fluid used (10 ml, respectively, 2.5 ml) allows identification of a contamination of down to 0.1 CFU/ml.
Advantages and Limitations of the Applied Test Method
Our testing system is composed of two different elements: the cultivation of naive medium on various agar plates suitable for bacterial and fungal growth, as well as the direct inoculation into BacT/ALERT bottles. When designing the system of microbiological testing, eubacterial genome amplification was also considered but not performed due to lack of improved microbe specification and sensitivity (26,36).
Most microbes from human fluid samples were detected in BacT/ALERT bottles (n = 21), while only seven naive samples gave rise to bacterial growth. In addition, our experiment on rodent tissue showed that, even in cases without bacterial growth on agar plates, BacT/ALERT bottles frequently gave a positive signal, suggesting better survival conditions for microbes in BacT/ALERT bottles compared to agar plating. Overall, the BacT/ALERT system has been validated as a system for sterile tests in general biopharmaceutical manufacturing for various applications (19). Another advantage of the BacT/ALERT system is that it represents a closed system, thereby reducing the risk of an environmental contamination. Therefore, this system does not require a dedicated cleanroom for diagnostic procedures. Our data suggest that for identification of bacterial contamination of fetal tissue samples used for transplantation, the BacT/ALERT system is superior to classical plating of small volumes on agar plates. Nonetheless, we still think that naive media inoculation on various agar plates is essential for two reasons. First, slowly growing microbes might be suppressed by faster dividing organisms if only one fluid medium (BacT/ALERT medium) would be used. Second, some organisms might not be able to grow in Bact/ALERT bottles but could specifically grow on agar media used for naive medium plating. And indeed, in two of the seven positive cases in the human study, cultivation of the naive medium on agar plates added to the microbial spectrum.
Practical Considerations
The proposed system of applying three different samples (A, B, and C) has now become part of our clinical protocols. At the day of tissue arrival, samples A and B are assessed. The tissue is then stored overnight with transplantation planned for the coming day. One day after arrival of tissue and prior to transplantation, preliminary results for samples A and B are obtained and can thus be taken into consideration within our standard release procedures. A positive sample A will not prevent transplantation of tissue, but the microbial spectrum of sample A is part of our quality assessment and may help to choose an antibiotic treatment in case of clinical signs of infection after transplantation. So far, we have not yet had a positive B sample when applying the described tissue processing system. In case of a positive result for this sample prior to transplantation, the graft would not be released for transplantation. The C sample is taken at the end of the last washing procedure prior to transplantation. Results are therefore not available prior to transplantation. A positive result in case of a contamination in any of the C specimen is not expected prior to 24 to 48 h after transplantation but would then initiate further discussions on the use of antibiotic treatment according to our standard operation procedures.
Although we judge the risk of a clinically relevant cerebral infection due to cell transplantation as very low, we propose the use of prophylactic antibiotics. Antibiotic prophylaxis is not uncommon in the field of neurosurgery and has among others been discussed for the prevention of intraventricular catheter-associated infections. However, although many trials have been designed as randomized, placebo-controlled trials, it remains controversial whether antibiotic prophylaxis is effective for the prevention of cerebral infections after implantation of extraneous material (31,37). Proposed regimens consist of the administration of various antibiotics, including vancomycin, cephalosporine, and others [a comprehensive analysis of available data is provided in Ratilal et al. (31)]. Most antibiotic regimens are usually administrated for the first 24 h after surgery, beginning with an intraoperative single shot. For fetal progenitor cell therapy in HD, various regimens have been reported, including intravenous piperacillin and vancomycin plus oral fluconazole for 3 days (18) or cefotaxime and metronidazole for 48 h postoperatively (35). In PD fetal progenitor cell transplantation, antibiotic treatment has been reported in few trials. Perioperative antibiotic prophylaxis involved treatment with unspecified broad spectrum antibiotics (17,18,20,28) and in one case with piperacillin, ceftazidime, and fluconazole for 5 days beginning immediately prior to surgery (15). In a placebo-controlled transplantation study in PD, patients received undisclosed broad spectrum antibiotics perioperatively (28).
Data from the present study suggests that there is a higher risk for a bacterial contamination due to fetal tissue processing, whereas the risk for fungal infections appears to be lower. In our clinic, peri- and postoperative intravenous administration of cefuroxime is now the standard antibiotic treatment, with 1.5 g given as a single shot intraoperatively followed by 1.5 g three times a day for the following 3 days, whereas an antifungal drug is not administered routinely. Although cefuroxime is capable of penetrating brain tissue, ventricular infections would not be treated sufficiently. In order to target the whole range of microbes expected according to our analysis, broad spectrum antibiotics like imipeneme are required.
Conclusions
Safety of fetal cell transplantation depends, to an important part, on microbiological aspects. We designed and tested a protocol for microbiological testing and decontamination. Subsequent washing seems to be sufficient to remove microbes from human tissue samples. Since results from experimental rodent embryonic tissue contamination suggest an important impact of the initial microbe concentration, prevention of initial growth and rapid reduction of the initial microbe concentration should be aimed at. Detection of a relevant microbial contamination within the narrow time window prior to transplantation remains difficult. While contamination of the transportation fluid is not an exclusion criterion for tissue transplantation, detection of contamination after washing steps would be judged critical. Peri- and postoperative antibiotic prophylaxis is justified to further reduce the risk of brain infection in fetal neural transplantation.
Footnotes
Acknowledgments
We thank Jessica Beckert for technical assistance. This work was funded by grants of the 7th framework program of the European Union (TRANSEURO) and by the German research foundation (DFG). The authors declare no conflicts of interest.
