Abstract
A short course of anti-CD45RB leads to long-term islet allograft survival and donor-specific tolerance in approximately half of immunocompetent mice. We have previously demonstrated that anti-CD45RB antibody-mediated tolerance requires B-cells for cardiac allograft survival. We therefore asked whether B-cells were also required for anti-CD45RB antibody-mediated survival of islets. Unexpectedly, we found that nearly 100% of islet allografts survive long term in B-cell-deficient mice. Similarly, B-cell depletion by anti-CD22/cal augmented anti-CD45RB-mediated tolerance when administered pretransplant, although it had no effect on tolerance induction when administered posttransplant. Our results demonstrate that the role of B-cells in promoting tolerance with anti-CD45RB is graft specific, promoting tolerance in cardiac grafts but resisting tolerance in islet transplantation. These findings may help elucidate the varied action of B-cells in promoting tolerance versus rejection.
Introduction
Because T-cells are essential for allograft rejection, current immunosuppressive strategies primarily target T-cells. More recently, B-cell depletion has been used primarily to treat acute antibody-mediated rejection or to desensitize patients with preexisting donor-specific antibodies, but it is not currently used as part of standard induction or maintenance protocols. For example, rituximab, a humanized anti-cluster of differentiation 20 (CD20) monoclonal antibody, has been successfully used in ABO blood group-incompatible renal transplantation (15,29); it is often used as part of the treatment for acute humoral rejection (1,10,17) and, less frequently, to ameliorate chronic rejection (3,11).
However, there is increasing evidence in transplant models that B-cells play a role in cellular immunity. In one study, heart grafts survived longer in mice when B-cells were unable to present antigen (26), and recently B-cells have been found to promote T-cell memory (25). In nonhuman primates (NHPs), the addition of rituximab to anti-thymocyte globulin induction and a limited course of rapamycin prolonged islet allograft survival, in some cases for many years after rapamycin discontinuation (22). In an NHP heart transplant model, initial B-cell depletion as an adjunct to chronic cyclosporine administration not only prevented chronic allograft vasculopathy but also reduced episodes of acute cellular rejection (18). These studies suggest that B-cell depletion may be a critical factor in promoting tolerance. On the basis of these findings, we examined the effect of B-cell depletion on islet allograft survival in mice treated with anti-CD45RB (B isoform of CD45 receptor).
Materials and Methods
Mice
Wild-type C57BL/6 (B6, H-2b), B-cell-deficient C57BL/6 (μMT–/–B6, H-2b), BALB/c (H-2d), B6.CD45.1 (H-2b), and C3H (H-2k) mice were purchased from the Jackson Laboratory. Forkhead box P3 green fluorescent protein knock-in (Foxp3gfp.ki) mice were provided by Mohamed Oukka (University of Washington, Seattle, WA). All mice were housed under specific pathogen-free conditions in the animal facility of Massachusetts General Hospital. All mice were male and 6 to 10 weeks of age. All protocols detailed below were performed following the principles of laboratory animal care and approved by the Institutional Committee for Research Animal Care.
Islet Isolation and Transplantation
Diabetes was induced in C57BL/6 mice with streptozotocin [200 mg/kg intraperitoneally (IP); Sigma-Aldrich, St. Louis, MO, USA] and was defined as blood glucose levels > 300 mg/dl for at least 2 consecutive days, using Accu-chek glucose strips (Roche, Mannheim, Germany). Islets were isolated by collagenase digestion (Liberase RI, Roche) and then separated by discontinuous Euroficoll gradients (Cellgro, Manassas, VA, USA; densities: 1.11, 1.096, 1.066) from the pancreatic digest. Four hundred to 500 islets were transplanted into the renal subcapsular space of diabetic recipients. A functioning graft was defined as a nonfasting blood glucose level < 200 mg/dl, and rejection was diagnosed at blood glucose of > 200 mg/dl for at least 2 consecutive days. Mice were monitored at least twice per week by measuring blood glucose until the mice were sacrificed. Nephrectomy was performed to rule out recovery of native islet function in mice that remained normoglycemic after 100 days.
Immunotherapy
Recipient mice received 100 μg of anti-mouse CD45RB (Bio X Cell; West Lebanon, NH, USA) IP on days 0, 1, 3, 5, and 7 following transplantation. In vivo B-cell depletion was performed by injecting 160 mg/kg IP of anti-CD22/cal [CD22 antibody conjugated with calicheamicin (cal); Pfizer, Groton, CT, USA] on days 8 and 3 prior to transplantation or days 0 and 5 after transplantation.
Flow Cytometry and Adoptive Transfer
Single-cell suspensions were recovered from spleens and lymph nodes by passing through a 40-μm nylon mesh (BD Falcon, Franklin Lakes, NJ, USA). Erythrocytes were lysed with ammonium chloride buffer (Sigma-Aldrich) and collected cells were washed and counted using trypan blue exclusion staining (Lonza, Walkersville, MD, USA) with a hemocytometer (Fisher Scientific, Pittsburgh, PA, USA). One million cells were suspended in phosphate-buffered saline (PBS; American Bioanalytical, Natick, MA, USA) containing 0.1% azide (Sigma-Aldrich) and 2% fetal bovine serum (FBS; Hyclone, Logan, UT, USA) in 96-well plates (Fisher Scientific) with the following fluorochrome-tagged antibodies: CD3, CD4, CD19, B220 (B-cell isoform of CD45), CD45.1, CD45.2, and Foxp3. Antibodies were purchased from eBioscience (San Diego, CA, USA). Intracellular Foxp3 in lymphocytes was measured using Foxp3 Staining Kit (eBioscience). All samples were run on an Accuri flow cytometer (Accuri cytometers Inc., Ann Arbor, MI, USA) and analyzed using Flow Jo analysis software (Tree Star Inc., Ashland, OR, USA).
Cells were sorted on FACSAria (BD Biosciences, San Jose, CA, USA). A total of 5 × 106 CD4+Foxp3–GFP– T-cells were sorted from Foxp3gfp.ki mice and adoptively transferred to congenic CD45.2 (C57BL/6 background) recipients.
Statistical Analysis
Data were analyzed using GraphPad Prism (version 5, GraphPad Software, San Diego, CA, USA). Graft survival between experimental groups was compared using Kaplan-Meier survival curves and Wilcoxon statistics. Other differences between experimental groups were analyzed using the Student's t test. Statistics were not corrected for multiple comparisons. Values of p < 0.05 were considered statistically significant.
Results
Prolonged Islet Allograft Survival After Anti-CD45RB Treatment in B-Cell-Deficient Mice
Untreated wild-type B6 and μMT–/–B6 mice reject BALB/c islet allografts by day 20. Anti-CD45RB treatment in wild-type B6 mice significantly prolonged graft survival [median survival time (MST): untreated = 10 days vs. anti-CD45RB treated = 65 days] with 50% of grafts surviving greater than 100 days (Fig. 1A). Anti-CD45RB treatment and islet transplantation in μMT–/–B6 mice resulted in 10 of 11 grafts surviving > 100 days compared to only 50% in treated wild-type B6 mice (p < 0.05).

Anti-CD45RB-induced donor-specific tolerance was enhanced in B-cell-deficient recipients and splenocytes from tolerant recipients transfer allograft tolerance. (A) Anti-CD45RB [B isoform of cluster of differentiation 45 (CD45) receptor] treatment results in significant prolongation of BALB/c (H-2d) islet allograft survival in both normal C57BL/6 (H-2b) and B-cell-deficient μMT–/–B6 (H-2b) recipients compared to untreated mice (***p < 0.0001). B-cell-deficient μMT–/–B6 recipients show better graft survival compared to C57BL/6 mice with anti-CD45RB treatment (*p < 0.05). (B) Representative glucose level of wild-type (WT) B6 and μMT–/–B6 recipients bearing long-term functioning BALB/c islet allograft and third-party C3H donors, and second islet allograft from the BALB/c strain after nephrectomy without additional therapy is shown. Islets from the C3H mice are rejected rapidly, whereas islets from BALB/c mice survived indefinitely.
Donor-specific tolerance was confirmed in the μMT–/–B6 recipient by removal of the long-term surviving islet allograft via nephrectomy and then transplanting C3H islets under the contralateral kidney. Euglycemia was maintained for less than 14 days, but a third islet transplant from a BALB/c donor to the same kidney was again accepted indefinitely (Fig. 1B). Tolerant wild-type recipients demonstrate the same capacity to accept a second graft from the same donor without additional antibody therapy. These data suggest that the absence of B-cells improves the ability of anti-CD45RB treatment to induce tolerance in a mouse islet allograft model.
Lymphocytes From Tolerant B-Cell-Deficient Mice Are Able to Transfer Tolerance
We next asked whether we could adoptively transfer tolerance using splenocytes from long-term survival (>100 days) μMT–/–B6 recipients. A total of 2 × 106 splenocytes from either tolerant μMT–/–B6 long-term survival (LTS) or B6 LTS recipients were able to prolong graft survival in transplanted, untreated wild-type recipients (Fig. 2). Consistent with Figure 1 showing that B-cells are not necessary for (and may actually inhibit) tolerance, purified B-cells isolated from tolerant B6 did not prolong graft survival. These data suggest that a tolerogenic population, perhaps regulatory T-cells, developed in both tolerant μMT–/–B6 and B6 recipients after anti-CD45RB treatment.

Lymphocytes from tolerant mice are able to transfer tolerance. A total of 2 × 06 splenocytes (SPCs) from recipients with long-term grafts were purified and adoptively transferred to secondary C57BL/6 recipients receiving BALB/c islets without antibody treatment. Recipients not receiving adoptive transfer or recipients receiving adoptive transfer of long-term survival (LTS) B-cells demonstrated no graft survival prolongation. In contrast, adoptive transfer of splenocytes from LTS μMT–/–B6 or LTS C57BL/6 prolonged islet allograft acceptance, *p < 0.05. SPC LTS B6 versus no cell transfer, ***p < 0.001.
Anti-CD22/cal Antibody Treatment Results in Significant B-Cell Depletion
To confirm that the absence of B-cells prolongs islet allograft survival, rather than additional immune deficiency in μMT–/–B6, we performed selective B-cell depletion by anti-CD22/cal coinjection (5,9,12). Two injections (160 mg/kg, 5 days apart) of anti-CD22/cal mAb completely depleted B-cells from the spleen and lymph nodes (LNs) as early as day 6 after therapy, and B-cells did not reemerge until day 30 (Fig. 3A and B).
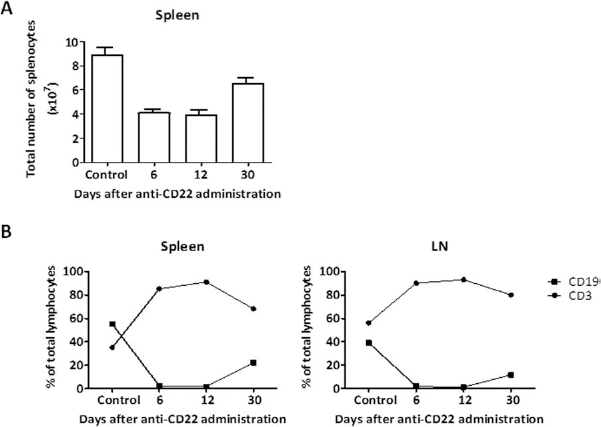
Effect of anti-CD22/cal mAb on the absolute and relative numbers of lymphocytes in spleens and lymph nodes (LNs). Splenocytes and LN cells were harvested at three different time points after anti-CD22/cal (CD22 antibody conjugated with calicheamicin) mAb therapy. (A) Absolute numbers of the splenocytes were counted using trypan blue exclusion on a hemocytometer. (B) Flow cytometric analyses were gated on live cells, and analysis was performed by using anti-CD3 and anti-CD19 mAbs in spleens and LNs. Each time point represents three to five animals.
B-Cell Depletion Pretransplant but Not Posttransplant Prolongs Graft Survival
Anti-CD22/cal alone prolonged graft survival, whether administered before or after islet transplantation (Fig. 4). As in μMT–/–B6 mice, depletion by anti-CD22/cal treatment on days −8 and −3 prior to transplant in combination with anti-CD45RB administration resulted in seven of eight mice maintaining allograft function long term (Fig. 4B), as compared to 50% in mice receiving anti-CD45RB alone. However, when we treated recipient mice with anti-CD22/cal on days 0 and 5, we saw no increase in long-term graft survival induced by anti-CD45RB alone (Fig. 4A).

Treatment of anti-CD22/cal mAb prior to transplant augments islet allograft survival with anti-CD45RB treatment. BALB/c islets were transplanted into diabetic C57BL/6 recipients. Median survival time (MST) for no treatment = 10 days. (A) Anti-CD45RB plus anti-CD22/cal treatment pretransplant (days −8 and −3) resulted in prolongation of islet allograft survival (*p < 0.05 vs. anti-CD45RB alone), whereas (B) anti-CD45RB plus anti-CD22/cal treatment posttransplant (days 0 and 5) did not promote graft survival (MST = 27 days). Anti-CD22/cal treatment alone pretransplant resulted in MST = 25 days and posttransplant = 16.5 days.
Regulatory T-Cells Increase Following Treatment with Anti-CD45RB
We have previously demonstrated that anti-CD45RB treatment generates Tregs, which can confer antigen-specific graft survival upon adoptive transfer (8). To distinguish whether anti-CD45RB antibody expands the existing Foxp3+ T-cell population versus whether anti-CD45RB stimulates the conversion of Foxp3– cells to Foxp3+ in vivo, CD4+Foxp3–GFP– cells were sorted from Foxp3–GFP knock-in mice (19) and adoptively transferred to naive congenic CD45.1 mice. We can distinguish adoptively transferred cells from host cells using the anti-CD45.2 antibody. Recipients were either left untreated or treated with the standard course of anti-CD45RB. On day 14, spleen and lymph node were examined for GFP expression, which demonstrates the conversion of Foxp3–cells to Foxp3+ (Fig. 5A). Treated recipients demonstrated significantly elevated levels of Foxp3–GFP related to untreated recipients. From this we conclude that anti-CD45RB is able to stimulate the conversion of CD4+Foxp3+ T-cells from Foxp3– cells.

Percentage of Foxp3+ T-cells does not increase upon B-cell depletion. (A) CD4+Foxp3–GFP– T-cells underwent fluorescence-activated cell sorting (FACS) from naive Forkhead box P3 green fluorescent protein knock-in (Foxp3gfp.ki) mice and adoptively transferred to congenic CD45.1 recipients. Half of the recipients were treated with anti-CD45RB. Fourteen days after the first antibody dose, the spleen and lymph node were examined by FACS analysis for Foxp3–GFP induction, only in the CD45.2+ (donor cell) population. Bar graph represents fold increase in Foxp3–GFP expression relative to untreated animals, p < 0.05. Data are from two independent experiments, two animals per group. (B) C57BL/6 recipients were grafted and treated either with anti-CD45RB alone, with anti-CD22/cal alone, or with anti-CD45RB plus anti-CD22/cal. Top, depletion of B-cells in grafted animals or depletion of B-cells in grafted animals in combination with anti-CD45RB did not increase the percentage of Tregs. Each group is representative of two to three animals. Bottom, a representative dot plot.
Furthermore, to evaluate the impact of B-cell depletion on anti-CD45RB-mediated Treg expansion, Foxp3 expression on CD4+ T-cells from spleen was examined in animals with functioning grafts 50 days after transplantation and treatment with anti-CD22/cal, anti-CD45RB, or both antibodies. As expected, anti-CD45RB treatment increased the percentage of Tregs in transplanted mice, but the addition of anti-CD22/cal treatment did not further increase the percentage of Tregs. Anti-CD22/cal treatment alone also had no effect on Treg percentages (Fig. 5B).
Discussion
Fifty percent of immunocompetent mice are resistant to tolerance induction by a short course of anti-CD45RB treatment. However, less than 10% of μMT–/–B6 mice, which lack mature B-cells, reject their islet allografts when treated by anti-CD45RB. Interestingly, when using a model of antibody-mediated B-cell depletion, we find that B-cells contribute to tolerance resistance only if present at the time of transplant. When B-cell depletion begins posttransplant, even if complete by day 6 posttransplant, graft survival is the same as when mice undergo no B-cell depletion. These and other data suggest that recipient B-cells, possibly acting as critical antigen-presenting cells (APCs), play a key role in initiating the cellular immune response against islet allografts but are less important in resisting tolerance as early as 1 week posttransplant (6,14,27,32). Late B-cell depletion may be ineffective at augmenting anti-CD45RB-induced tolerance because the B-cells play an initiating role in islet rejection and tolerance.
In contrast to our findings with islets, we previously reported that tolerance induced by anti-CD45RB treatment in heterotopic heart transplantation was dependent on the presence of recipient B-cells, rather than improving in the absence of B-cells (7). It has long been recognized that the rate of graft acceptance varies by organ (13,16,33), and gene array studies in humans suggest that the mechanism of tolerance varies between liver and kidney transplant recipients, despite the fact that similar drugs are used (4,23,24,28). These differences have been attributed to variations in passenger leukocyte or antigen-presenting cell loads, microbial exposure, and endothelial cell activity (2,30,31), but the mechanisms are not well elucidated. In particular, it has been noted that islet rejection requires far fewer CD8+ T-cells and less CD4+T-cell help than heart rejection, even though the CD8+T-cells infiltrate cardiac allografts faster than they infiltrate islet allografts (16). If the lack of T-cell dependency reflects a requirement for B-cell activity in islet rejection, but not heart rejection, this could explain why removing B-cells improves tolerance in islet transplantation, both in the presence or in the absence of anti-CD45RB treatment, but not in heart transplantation.
We have also observed differences in the anatomic pattern of early sensitization between islets and vascularized heart grafts in a T-cell receptor transgenic model of rejection (21). Following islet transplantation under the kidney capsule, T-cell activation was seen mainly in the draining lymph nodes, whereas heart allografts stimulated T-cells diffusely in lymph nodes throughout the body, as well as the spleen. Vascularized heart grafts may result in antigen dissemination to the spleen, a major B-cell repository, generating tolerogenic effects, whereas antigen presentation by B-cells in the draining lymph nodes may generate primarily immunogenic effects. As there is no systemic or tolerogenic B-cell effect following islet transplantation, B-cell depletion improves tolerance induction in islet transplantation, but has the opposite effect in heart transplantation. Indeed, mouse heart allograft recipients experience higher rates of tolerance after anti-CD45RB treatment than islet allograft recipients do (7,20). Experiments are under way to determine whether anti-CD45RB treatment of mice undergoing simultaneous transplantation of a heart allograft with islets provides islet allograft protection and changes the B-cell effects on tolerance to islets.
In summary, we find that B-cell depletion increases tolerance to islet allografts following anti-CD45RB treatment, but only if performed prior to transplant. Although anti-CD45RB-mediated tolerance to islets appears to depend on Tregs, B-cell depletion is not associated with increased proportions of Tregs in this model. As we only examined animals with functioning grafts, it is possible that the proportion of Tregs in recipients with rejected grafts is even lower than in those with functioning grafts at day 50. B-cells may play an early role in activating effector T-cells, rendering them relatively resistant to Treg suppression, or promote rejection in ways other than increasing Treg numbers.
Footnotes
Acknowledgments
This work was supported in part by NIH grants RO1AI057851-05 (J.F.M.), k01Dk079207 (J.I.K.), 5T32AI7529 (K.M.L.), and 5k08Dk094965-02 (H.Y.). The authors declare no conflict of interest.
