Abstract
Graft-versus-host-disease (GVHD) is a major complication of allogeneic hematopoietic stem cell transplantation (allo-HSCT), which is associated with high morbimortality and decreased patients' physical capacity. We evaluated the effects of an 11-week moderate-intensity exercise (treadmill) training program performed after allo-HSCT in a murine acute (aGVHD) and chronic GVHD model (cGVHD). Female mice (aged 8–12 weeks) were randomly assigned to the exercise or the control group. They completed a maximal treadmill test before allo-HSCT (with donor bone marrow cells and splenocytes) and after the 11-week period, during which we evaluated clinical severity scores and survival (Kaplan–Meier method). Before allo-HSCT and at days +21, +52 and +83 (upon sacrifice), we collected blood samples for immune cell reconstitution and cytokine analysis. The main results were that (i) in aGVHD, exercise improved maximal physical capacity over the 11-week period compared with pre-allo-HSCT conditions (p < 0.001 for the between-group comparison) and benefited total clinical score evolution (p = 0.05 for the group×time interaction effect), without altering immune reconstitution; (ii) in cGVHD, exercise training resulted in a lesser deterioration of physical capacity after 11 weeks (p = 0.023). Our results highlight the potential beneficial effects of exercise as coadjuvant intervention against GVHD, especially in the acute form of the disease.
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is widely used for the treatment of hematopoietic disorders, severe hematologic malignancies, or immunodeficiencies (7,10,11). Despite recent methodological advances (e.g., in conditioning regimens and donor selection), allo-HSCT is associated with high morbimortality (10), mainly due to a major complication, graft-versus-host disease (GVHD) (7). The high incidence (50–80%) and mortality risk of GVHD limits a broader use of allo-HSCT worldwide (7).
GVHD consists of a group of clinical and histological manifestations caused when the T-cells of the donor interact with tissue antigens in the host, whose immune system is suppressed (7). The disease is characterized by an exacerbated multiorgan inflammatory response that induces lesions in skin, liver, lungs, and gastrointestinal tract (34). This process is associated with decreased patients' functional capacity, as well as with impairments in domestic and vocational functions and in family and social interactions, all of which result in poor quality of life (22). The usual immunosuppressant treatment against GVHD has serious side effects (i.e., muscle toxicity and increased risk of infections), which further deteriorates the health status and physical capacity of patients (7,22). Two types of GVHD were traditionally distinguished based on the time of occurrence: if prior to day 100 after transplant it was considered acute, and if after that time it was classified as chronic (7). However, this original definition was revised by the U.S. National Institute of Health, and now the severity of clinical manifestations is the major criterion [for more information, see Filipovich et al. (8)].
There is strong evidence supporting a broad range of therapeutic properties associated with a nonpharmacological intervention: regular, moderate-intensity aerobic exercise [e.g., walking or brisk walking for at least 30 min/day in most days of the week (such as to accumulate ≥150 min/week)] (12). These include not only improved physical capacity and overall health status but also decreased systemic inflammation levels (26) and improved immune function (9). Additionally, exercise can ease some side effects of medical treatments (e.g., those caused by anticancer treatment) (30) and thus assist patients in returning to normal daily life activities. Nevertheless, no prior study has analyzed the effects of an exercise program on GVHD.
Based on the aforementioned potential exercise benefits and on the fact that allo-HSCT is the only effective therapeutic option for a variety of malignant and non malignant conditions, it is important to find ways to improve the clinical course of GVHD and the well-being and functionality of patients who suffer this condition, without adding negative side effects with the intervention. The aims of this study were twofold. First, we evaluated the impact of an exercise program on the physical capacity, survivorship, and clinical course (severity) evolution of GVHD (primary study outcomes) in acute and chronic murine models of GVHD. We also assessed in both models the effects of the exercise program in the immune reconstitution profile, inflammatory outline, histological and clinical state of organs, and muscle molecular adaptations to exercise (secondary outcomes). We hypothesized that the exercise intervention would result in an improved physical capacity and a better clinical outcome/survival rate with no harm to the immune function, at least in the least severe disease model (i.e., aGVHD).
Materials and Methods
Procedures were carried out according to European and Spanish laws and regulations (European convention ETS 123, about the use and protection of vertebrate mammals in experimentation and other scientific purposes and Spanish Law 32/2007, and R.D. 1201/2005 about the protection and use of animals in scientific research) and were approved by the Ethical Committee (Centro de Investigaciones Energéticas, Medioambientales y Tecnológicas, Madrid, Spain) for Animal Experimentation according to all external and internal biosafety and bioethics guidelines.
Animals
We used C57BL/6J and BALB/C (recipient; female, 8–12 weeks) and C3H/SW and B10.D2 (donor; male, 12–18 weeks) mice bred at the Laboratory Animals Facility (registration no. ES280790000183) of the Centro de Investigaciones Energéticas, Medioambientales y Tecnológicas (Madrid, Spain) from breeding pairs originally obtained from The Jackson Laboratory (Bar Harbor, ME, USA). All mice were housed in euro-standard type IIL microisolator cages, maximum of five mice each, in controlled conditions (room temperature and humidity regulated at 20±2°C and 55±10%, respectively) with fluorescent lighting from 7:00 a.m. to 7:00 p.m., receiving food (Harlan Teklad Global Diets 2914; Harlan, Castellar, Spain) and water (50 mm filtered and UV irradiated) ad libitum. Mice were routinely screened for pathogens in accordance with FELASA (Federation of European Laboratory Animal Science Associations) recommendations.
Forty-eight female mice (age: 6–8 weeks, weight: 18–20 g) were the recipients of the transplant in order to induce GVHD; 20 were C57BL/6J (acute model of GVHD, aGVHD) and 28 were BALB/C (chronic model, cGVHD). Mice were randomly separated into the following two groups: exercise (n = 11 C57BL/6J and n = 15 BALB/C, submitted to an ~11-week treadmill exercise training after the transplant) or control (n = 9 C57BL/6J and n = 13 BALB/C, with movement confined to the cage space).
Study Protocol
Day 0 was considered the transplant day. The mouse manipulation was performed by the same researcher with the exception of the transplant and maximal treadmill tests that are described below (in order to minimize any subjective evaluation of the tests). The study protocol was identical for the two disease models and included three main phases: (i) preliminary phase, (ii) irradiation and transplant, and (iii) posttransplant phase, in which the exercise training intervention was performed (see Fig%. 1 for a schematic representation of the main study protocol).

Schematic representation of the study protocol. GVHD, graft-versus-host disease.
Preliminary Phase
This phase consisted of three parts: blood draw to obtain serum, mouse adaptation to treadmill, and the first maximal treadmill test.
On day –10 (i.e., healthy conditions), blood was drawn from each mouse (100 μl, tail vein) to obtain serum levels of several cytokines (see below). The serum was separated by two centrifugations of 10 min at 1,600 rpm each, and the samples were stored at −80°C until analysis.
The mouse adaptation to the treadmill was performed in three sessions (days –7, –6, and –5) so that the animals could learn how to run on the treadmill (Harvard Apparatus, Panlab, Barcelona, Spain). This adaptation period involved a gradual increase in running time and intensity, starting with just staying on the treadmill on the first day (inclination and speed of 0% grade and 0 cm/s, respectively, for 1 min, without applying electrical stimulation) and ending with 15 min at a low running intensity on the third day (25% grade and 15 cm/s speed, with an electrical stimulation of 0.2 mA, 1 Hz, 200 ms).
The first maximal treadmill test was performed after the last day of the mouse adaptation to the material (day –4). This test was performed to assess the maximal physical (“aerobic”) capacity of the mice (expressed as the total run distance) (14,19) and to determine training intensity from the maximal velocity reached in the last stage of the test (Vmax) (14). The test was performed after a warm-up period of 10 min at 10 cm/s. It started with an initial velocity of 5 cm/s, followed by increases of 3 cm/s every 2 min until extenuation. The treadmill inclination was kept constant at 25% grade during the test because this inclination allows attainment of maximal cardiovascular intensity while preventing injuries (14). Mice were defined as exhausted when they spent more than five continuous seconds on the electric grid and were incapable to continue running at the next faster speed (2).
Irradiation and Transplant
On –1 and 0 days, recipients mice received lethal total body irradiation (300 kV, 10 mA; Philips MG-324, Hamburg, Germany) twice within a 24-h interval between them to minimize gastrointestinal toxicity (dose per day: 5.5 Gy for C57BL/6J and 4.75 Gy for BALB/C). To conduct the transplant, donor bone marrow cells were prepared by flushing femurs and tibias, and splenocytes by passing spleens through a 100-μm steel mesh (Cell Strainer; BD Falcon, San Agustín de Guadalix, Madrid, Spain) with 1× phosphate buffered saline (PBS; Sigma-Aldrich Química, S.L., Tres Cantos, Madrid, Spain), 2% fetal bovine serum (FBS; Gibco, Invitrogen, Madrid, Spain), and 0.5 M ethylenediaminetetraacetic acid (EDTA; Sigma-Aldrich). Subsequently, recipients were transplanted intravenously through the tail with a single inoculum: C57BL/6J mice received 20 × 106 splenocytes and 10 × 106 bone marrow cells, and BALB/C mice received 10 × 106 splenocytes and 10 × 106 bone marrow cells. The allo-HSCT models used in this study were MHC-matched, miHA-mismatched models: (i) C3H/ SW (H-2b) à C57BL/6J (H-2b) and (ii) B10.D2 (H-2d) à BALB/C (H-2d) (29). The first of them, an aGVHD model, depends primarily on CD8 T-cells, and the second one is a chronic sclerodermatous model of GVHD that depends on CD4 T-cells (31). These two models represent mild forms of the disease (29).
Posttransplant Phase
This phase included the exercise training intervention and the second maximal treadmill test; the evaluation of survival and clinical evolution of GVHD; the sampling of blood and serum; and the sacrifice.
The ~11-week exercise training program started 2 days after allo-HSCT (day +2) and consisted of treadmill training on 5 days/week (Monday to Friday, total of planned sessions = 57). Exercise duration, treadmill speed, and inclination were gradually increased over the program (beginning with duration = 25 min, speed = 35% of the Vmax, and inclination = 0% grade, and ending with duration = 60 min, speed = 70% of the Vmax, and inclination = 25% grade). All exercise sessions took place between 7:15 a.m. and 12:00 a.m. With the aim of maintaining the running abilities in the control groups, but without inducing the training adaptations that we expected to occur in the exercise groups, the former performed three weekly treadmill sessions of very short duration (5 min) and low intensity (speed = 5–8 cm/s, inclination = 25% grade) (25). All mice performed the aforementioned maximal treadmill test again after the training program (day +80).
Survival and clinical parameters indicative of GVHD were evaluated daily. On days +21 and +52, 200 μl of blood was drawn from the tail vein to analyze the cellular immune reconstitution and cytokine levels. Finally, on day +83, mice were sacrificed with Avertina (0.2%, 0.15 μl/g; Sigma-Aldrich), and when a surgical plane of anesthesia was reached, whole blood was extracted (axillary vein) for the cytokine analyses. Then we removed skeletal muscle tissue (from the posterior limbs), which was immediately frozen with liquid nitrogen and stored at −80°C until analysis, disease target tissues (liver, intestine, skin), which were preserved in formalin, and spleens to analyze immune cell reconstitution.
Measurement of Study Outcomes
Primary Outcomes
Physical capacity was determined as the total distance covered by mice (14) in the two maximal treadmill tests. Survival data were plotted by the Kaplan–Meier method, whereas for evaluation of the clinical course (severity) evolution of GVHD, we used the score system described elsewhere (6). Animals were individually scored every day on a score from 0 to 2 for five clinical parameters (percentage of weight loss, posture, fur texture, skin integrity, and activity) as follows (6): percentage of weight loss, score of 0, 1, or 2 for weight loss <10%, >10%, and <25% or >25%, respectively; posture, 0, 1, or 2 for normal, hunching noted only, and rest or severe hunching impairing movement; fur texture, 0, 1, or 2 for normal, mild to moderate ruffling, or severe ruffling/poor grooming; skin integrity, 0, 1, or 2 for normal, scaling of paws/tail, or obvious areas of denuded skin; and activity, 0, 1, or 2 for normal, mild to moderately decreased, or stationary unless stimulated. A clinical GVHD score (0–10) was generated daily by summation of the five individual scores (5).
Secondary Outcomes
Serum samples were used for quantifying the levels of the following cytokines, according to the manufacturer's instructions (BioPlex Pro, Mouse Cytokine Standard Group I 23-Plex Laboratories Inc., Hercules, CA, USA): interleukin-2 (IL-2), IL-4, IL-6, IL-17a, interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α). The absolute numbers of leucocytes in peripheral blood (PB) were counted with an AC.T Cell Counter (Beckman Coulter Spain S.A., Madrid, Spain). Blood and spleen immune reconstitution analyses were performed with flow cytometry using the following antibodies: cluster of differentiation 3 (CD3; T lymphocytes), B220 (B lymphocytes), and macrophage-1 antigen (mac-1; granules). These antibodies and 7-amino-actinomycin D (7AAD) were from BD Bioscience (San Agustín de Guadalix, Madrid, Spain). For histological analysis, samples of the aforementioned organs were fixed in 7% formaldehyde (Sigma-Aldrich) solution and embedded in paraffin. For these tissues, 5-μm-thick sections were stained with hematoxylin and eosin (Sigma-Aldrich) for histological examination. Cumulative histopathology scores were calculated following a mouse GVHD pathological scoring system: 0 as normal, 0.5 as focal and rare, 1.0 as focal and mild, 2.0 as diffuse and mild, 3.0 as diffuse and moderate, and 4.0 as diffuse and severe (38). Scores were added to provide a total score for each specimen. Two independent pathologists analyzed slides in a blinded fashion to assess the intensity. Citrate synthase activity, a classical marker of muscle aerobic adaptation and upregulated mitochondrial biogenesis (27), was spectrophotometrically determined in skeletal muscle homogenates at 30°C in the presence of 0.1% Triton X-100 (Sigma-Aldrich), following the formation of 5-thio-2-nitrobenzoic acid (TNB) at 412 nm as previously described (33). Samples of skeletal muscle homogenates (40 and 20 μg) were also used to perform semiquantitative analysis of p70 S6 kinase and phospho-p70 S6 kinase protein levels by immunoblotting (Western blot; see Fig. 2 for an example). Sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE; Bio-Rad, Alcobendas, Madrid, Spain) was performed on a 7.5% separation gel. Resolved proteins were transferred to a polyvinylidene difluoride (PVDF) membrane (GE Healthcare, Alcobendas, Madrid, Spain). Blots were blocked and incubated with the following primary antibodies: rabbit anti-p70 S6 kinase and rabbit anti-phospho-p70 S6 kinase (Thr389) (Cell Signalling Technology Inc., Izasa, Barcelona, Spain). Immunodetection of primary antibodies was carried out with peroxidase-conjugated goat anti-rabbit antibodies (GE Healthcare). The signal was detected with Novex ECL HRP Chemiluminescent Substrate Reagent Kit (Life Technologies S.A., Alcobendas, Spain). Band densities were evaluated by densitometric scanning (Image J software, NIH, Bethesda, MD, USA). To verify that the total protein amount loaded in each lane was the same, α-tubulin was immunodetected with mouse anti-α-tubulin (Sigma-Aldrich, Madrid, Spain) coupled to peroxidase-conjugated goat anti-mouse antibody (GE Healthcare). We calculated the ratio of phospho-p70 S6 kinase/p70 S6 kinase, which is a marker of muscle anabolism (24).

Phospho-p70 S6 kinase (Thr389) and p70 S6 kinase expression levels in skeletal muscle homogenates of exercised (EX) and control (CT) mice of acute (aGVHD) and chronic disease model (cGVHD). α-Tubulin was used as a loading control.
Statistical Analyses
Data are shown as means±SD (or as median and interquartile range for those variables that were analyzed with nonparametric tests—see below) and the level of statistical significance was set at 0.05. Statistical analyses were performed using SPSS (v. 18.0, Chicago, IL, USA) and Stata (v. 11.2, College Station, TX, USA). The Shapiro–Wilk test was performed to check normality of the data. Between-group comparisons were performed within each model of GVHD as explained below.
Primary Outcomes
We compared the change in physical capacity (posttraining minus pretransplant) with the Mann–Whitney U test. Between-group comparisons of the survival data plotted by the Kaplan–Meier were analyzed with the log-rank test. We used a two-factor (group, time) analysis of variance (ANOVA) with repeated measures to assess the effects of the exercise intervention on the GVHD score.
Secondary Outcomes
We applied the two-factor ANOVA with repeated measures that is described above to assess the effects of the exercise intervention on blood inflammatory profile and immune cell reconstitution. The Mann–Whitney U test was used for between-group comparisons of those variables collected upon animals' sacrifice (day +83), that is, spleen immune reconstitution, histological affectation of organs, and muscle molecular markers of exercise adaptation (citrate synthase activity and ratio of phospho-p70 S6 kinase/p70 S6 kinase).
Results
We explored the effects of a supervised physical activity program in different models of GVHD in mice. We used miHA-driven GVHD models, which recapitulate many aspects of clinical allogeneic hematopoietic transplants and represent mild forms of the disease (29).
Exercise and aGVHD
In the C3H/SW → C57BL/6J model, the induction of GVHD depends mainly on donor CD8 T-cells.
Primary Outcomes
We observed a significant improvement in the physical capacity after training in the exercise group versus a decrement in their controls (p < 0.001 for the between-group comparison) (Fig. 3). Although no significant differences were found between the two groups (Fig. 4), there was a trend toward higher survival in the exercise group (Fig. 4A). All mice in the exercised group were alive at day +83, versus 77.8% of controls (p = 0.107) [note that the C3H/SW à C57BL/6J model has minimal lethality (29)]. Looking at disease clinical course/severity (Fig. 5), in addition to the better survival rate, exercise intervention induced a significant beneficial effect in aGVHD (i.e., lower severity scores over time in the exercised mice), as indicated by a significant group × time interaction effect for the total severity score (p = 0.05; F = 3.159) (Fig. 5A). This result shows that the mice that followed the exercise program presented a significantly better overall clinical evolution over time.
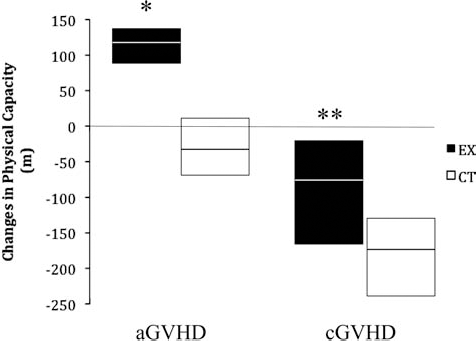
Change in physical capacity expressed as the difference in the total distance (meters) covered by mice in the maximal treadmill test performed posttransplant (day +83) minus the total distance (meters) in the maximal treadmill test performed pretransplant (day –4). Data are expressed as median and interquartile range. aGVHD, acute model of GVHD; cGVHD, chronic model of GVHD; CT, control group; EX, exercise group. *p < 0.001 for the between-group comparison, **p = 0.023 for the between-group comparison.
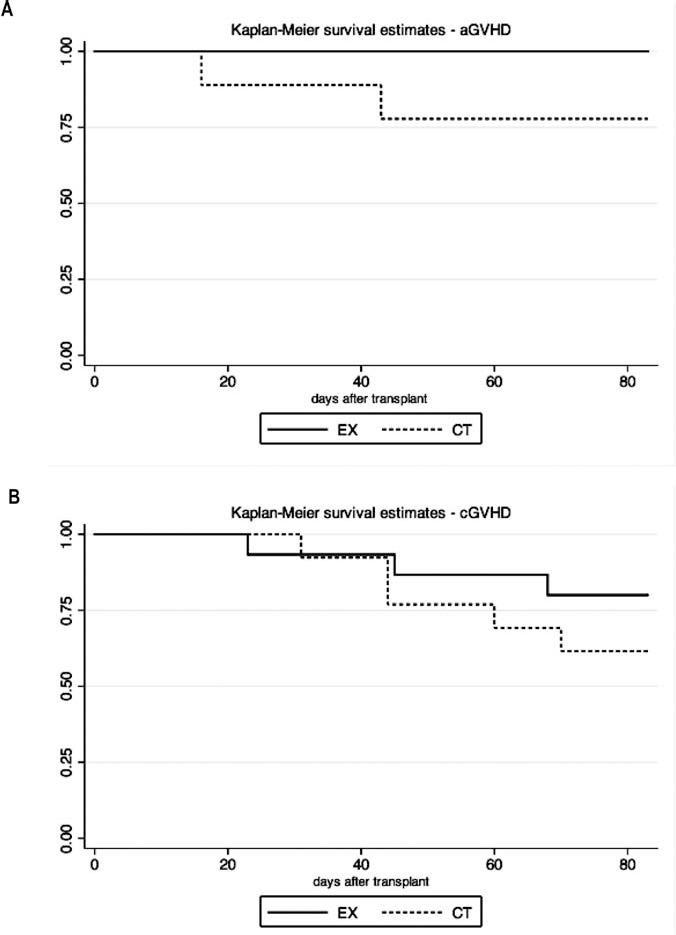
Survival estimates in the acute (A) and chronic models (B) of GVHD. aGVHD, acute model of GVHD; cGVHD, chronic model of GVHD; CT, control group; EX, exercise group. The p values for the between-group comparisons were 0.107 (aGVHD) and 0.302 (cGVHD).

Daily clinical evolution over time (total and individual severity scores of GVHD) in the acute (A) and chronic disease models (B). Data are expressed as mean±SD. CT, control group; EX, exercise group. *p = 0.05 for the interaction (group×time) effect in aGVHD total clinical score (no other significant interaction effect was found, including all individual scores).
Secondary Outcomes
We quantified the levels of IL-2, IL-4, IL-6, IL-17a, IFN-γ, and TNF-α cytokines in the peripheral blood of transplanted mice, and found no exercise training effect in the profile of circulating cytokines (i.e., no significant groupxtime interaction effect with the ANOVA test) (Table 1). Similarly, no significant group×time interaction effect was noted for the kinetics of immune cell reconstitution profile (Table 2), and we found no between-group differences in the spleen cells at day +83 (data not shown). The absence of a significant interaction indicates that both control and exercise groups showed a similar response throughout the study time (~11 weeks) in terms of immune cell reconstitution and inflammatory cytokine production. As for histological analyses of target organs (intestine, skin, and liver), no significant differences were observed among surviving mice of each group at the end of the experiment (Table 3). Results of muscle markers are shown in Figure 6. We detected higher levels of citrate synthase activity in the exercise group (p = 0.03). No significant between-group differences were observed in the ratio of phospho-p70 S6 kinase/p70 S6 kinase, indicating no differences in muscle anabolic/catabolic balance.
Inflammatory Profile Over Time in the Acute (aGVHD) and Chronic Model of GVHD (cGVHD)
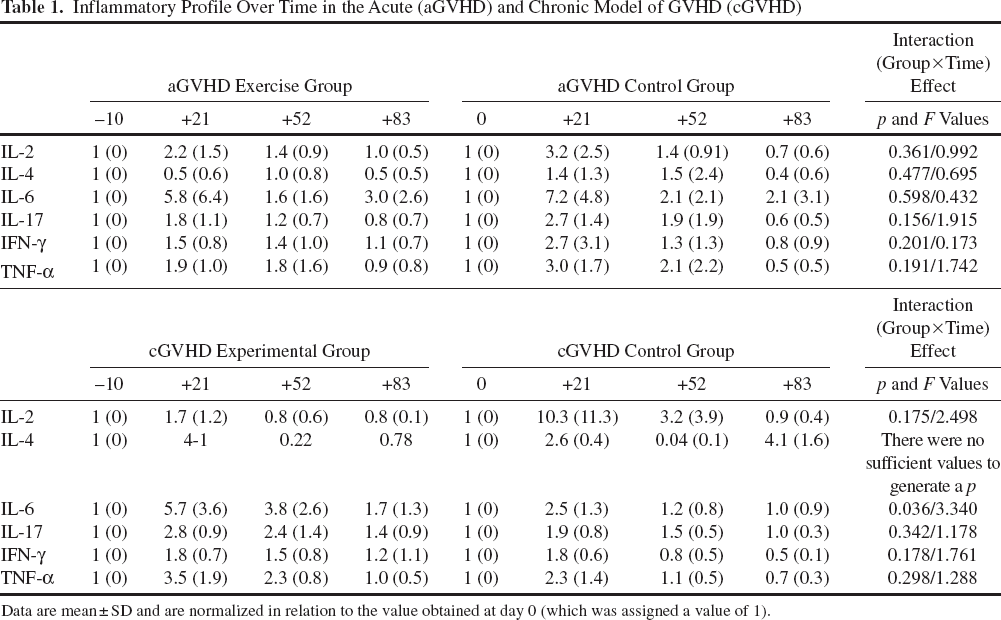
Data are mean ± SD and are normalized in relation to the value obtained at day 0 (which was assigned a value of 1).
Blood Immune Reconstitution Profile in the Acute (aGVHD) and Chronic Model of GVHD (cGVHD)

Data are mean ± SD and units are number of cells/ìl.
Histological Scores of GVHD in the Target Organs of Mice Surviving the Complete Experiment Time

Data are median (min, max).
In summary, in the aGVHD model, exercise training induced significant benefits in terms of physical capacity and clinical score, without interfering in the immune reconstitution of the recipients.

Muscle citrate synthase activity (A) and ratio of phospho-p70 S6 kinase/p70 S6 kinase (B) at sacrifice (day +83). Data are expressed as median and interquartile range. aGVHD, acute model of GVHD; cGVHD, chronic model of GVHD; CT, control group; EX, exercise group. *p = 0.03 for the between-group comparison, **p = 0.008 for the between-group comparison. No significant p value was found for between-group comparisons of the ratio of phospho-p70 S6 kinase/p70 S6 kinase (p = 0.478 and p = 0.211 for aGVHD and cGVHD, respectively).
Exercise and cGVHD
In the B10.D2 → BALB/C model, the induction of GVHD depends mainly on donor CD4 T-cells. Pathological and clinical findings reflect the effects of inflammatory cytokine mediators produced by the activated donor T lymphocytes.
Primary Outcomes
The mice that followed the exercise training had a significantly lower decrement in physical capacity compared to their controls (p = 0.023 for the between-group comparison) (Fig. 3). No significant between-group difference was found in survival (Fig. 4B), with 80% survival in the exercise group versus 61.5% in controls (p = 0.302 for the between-group comparison). Regarding disease clinical course/severity, the exercise intervention induced no significant group×interaction effect for total or individual severity scores in the cGVHD model (Fig. 5B).
Secondary Outcomes
We found no exercise effect in the profile of circulating cytokines, except for IL-6 (Table 1). Similarly to the aGVHD model, no significant group×time interaction effect was noted for the immune cell reconstitution profile (Table 2) or for the numbers and subtypes of splenocytes at the end of the experiment (data not shown) in the cGVHD model. We found no differences in the histological scores of GVHD in the target organs of mice surviving the complete experiment time (Table 3). Finally, the levels of citrate synthase were significantly higher in the exercise group (p = 0.008), with no significant differences in the ratio of phospho-p70 S6 kinase/p70 S6 kinase (Fig. 6).
In summary, in the cGVHD model, exercise training attenuated the deterioration of physical capacity without improving the clinical course of the disease and without interfering in the immune reconstitution of the recipients.
Discussion
We analyzed the effects of regular exercise in a very aggressive and debilitating disease as is GVHD, using both clinical and biological markers of disease evolution and exercise adaptations. We set up models that address important aspects of human GVHD such as the role of donor CD8 and CD4 T lymphocytes separately, as well as the different features of the acute and chronic forms of the disease. An aerobic, moderate-intensity exercise program that mimics the widely accepted public health recommendations for physical activity in humans, that is, accumulating at least 30 min of such type of exercise (e.g., brisk walking) in most days of the week (Monday to Friday in our model) (12), was well tolerated by the diseased mice and had positive effects on both their physical capacity and clinical outcome in the acute, CD8-driven GVHD model. In addition, exercise had no negative impact on the kinetics of immune cell reconstitution and on the production of cytokines in either model, and attenuated the deterioration of physical capacity in the chronic GVHD model. GVHD is an extremely aggressive disease and is associated with a substantial morbimortality; as such, it seriously compromises patients' physical capacity, leading to functional limitations in daily living activities (e.g., climbing stairs, walking, household tasks) and to very poor quality of life (22). Thus, our result supporting a potential therapeutic value of exercise in debilitated and immunocompromised recipients of allogeneic transplants, contributing to an improvement (or an attenuated decrement) in their physical capacity according to the severity of the disease, is of clinical relevance. Strategies aiming at improving physical capacity in patients with GVHD, or at least attenuating functional deterioration, can make a difference in the patient's daily life.
Physical capacity, especially if determined as the maximal aerobic capacity as we did here, is a strong, independent health indicator and mortality predictor in healthy and diseased people (18,23,32). It is noteworthy that in the aGVHD model, exercised mice were able to increase their physical capacity after 11 weeks compared with healthy (i.e., pretransplant) conditions, despite suffering this aggressive disease for this long period of time; in contrast, their controls showed the opposite pattern. In the cGVHD, which is the more aggressive of these two models, the exercise group had decreased substantially less in their functional capacity after the program, compared with the control group. Despite the energy-consuming, catabolic nature of this disorder, the exercise intervention was able to induce beneficial molecular adaptations at the muscle level: increase in aerobic enzyme (citrate synthase) activity in the exercise group, with no further catabolic effects, that is, no between-group differences in either model for a marker of muscle anabolic/catabolic balance (i.e., ratio of phospho-p70 s6 kinase/p70 S6 kinase) (24).
Survival is a classic endpoint when studying GVHD. We did not anticipate that exercise by itself would have a curative effect in such an aggressive, life-threatening disease. Although not statistically significant, we observed a trend toward a superior survival in the exercise groups (especially in aGVHD). This finding is in agreement with the better clinical evolution (lower severity scores) of the exercised mice suffering aGVHD compared with their controls. Though the molecular mechanisms linking improved physical capacity and better tolerance to GVHD remain to be elucidated, we can speculate that the trend toward better survival rates in the exercise groups is associated with their higher physical capacity. The muscle activity of citrate synthase, a key mitochondrial enzyme for aerobic energy provision and a marker of upregulated mitochondrial biogenesis (27), increased with exercise training in both disease models. A shift toward more oxidative (or “aerobic”) muscle phenotypes could have a beneficial influence on health status and disease resistance. Classic studies in rats by Koch and Britton's group showed that improved oxidative pathways in mitochondria may be a common factor linking physical fitness and decreased disease risk (16,36).
Previous findings indicate that exercise can mitigate inflammatory levels in patients with chronic conditions such as cardiovascular disease (1,4,9). The clinical improvement we observed in the exercise group could not be linked to a systemic anti-inflammatory effect of exercise. However, we cannot rule out the possibility that production of inflammatory mediators at the target organs was different between groups. With regards to this, although the pathology score of target organs was not different between groups, it must be kept in mind that the histological analysis was performed with the mice that survived the total duration of the study (i.e., 12 weeks) (Fig. 1), and exercised mice tended to live longer. On the other hand, exercise can mobilize cells with the immunophenotype of mesenchymal stem cells (MSCs), as we have previously reported in two independent studies (21,28). The MSCs play a role as immunomodulators, and they have been used as a therapy for GVHD (20). We have not studied MSCs in the present work, but it is attractive to speculate that exercise-induced mobilization of these cells may have contributed to the better outcome of the trained mice.
Exercise did not negatively affect the kinetics of immune reconstitution in the context of severely debilitated and immunocompromised mice receiving lethal irradiation and transplantation. This finding suggests that the exercise would not negatively affect the graft-versus-tumor or the graft-versus-infection effect of the transplant. Further experiments using models adapted for these two situations (i.e., tumor or infectious agents) will elucidate these clinically important questions. Although the beneficial effects of moderate-intensity exercise on the immune function of healthy individuals are well established (35), there is more controversy regarding its effects in immunocompromised people, with both positive effects (15) and no benefit but also no deleterious effect, like here, being previously reported (3,13,17). We analyzed the effects of an exercise program in animals that did not receive pharmacological treatment. Future investigation should focus on the combined effect of exercise and the usual pharmacological (immunosuppressive) treatment in GVHD. Importantly, however, such drug treatment has multiple toxicities further aggravating the muscular and functional capacity of patients (37), which provides additional support for the rationale of including exercise as a coadjuvant therapy.
In conclusion, our results highlight the potential beneficial effects of an exercise program, especially concerning physical capacity and clinical evolution in the acute, less severe model. Exercise might be a nonpharmacological way of improving the clinical course and quality of life of GVHD patients by increasing their physical capacity and allowing a more positive clinical evolution of the disease without any harm to a fragile immune function. Since the number of patients receiving transplants from unrelated donors will likely grow considerably in the next years, the incidence of GVHD will consequently increase (7). Thus, new approaches aiming at improving the well-being of patients with GVHD and alleviating disease complications such as impaired physical capacity are urgently needed.
Footnotes
Acknowledgments
This study was funded by Fondo de Investigaciones Sanitarias (FIS, ref. #PS09/00194). We acknowledge Nieves Lozano and Miguel Fernández Navas for their technical assistance in analysis of histological damage to tissues and muscle markers, respectively. C.F.-L. performed most experiments, all exercise interventions, and was a significant manuscript writer. A.G.-M. and J.M.-P. performed significant work in mouse handling and experiments. L.S.-M. was a significant manuscript writer and performed most statistical analyses. I.C. and F.C. performed histological analyses. G.M. performed significant work in data (cytokine) analyses. M.M. performed all skeletal muscle analyses. A.L. was a significant manuscript writer, main provider of reagents and materials from grants, and performed statistical analysis together with L.S.-M. M.R. conceived and designed the research, contributed with materials/instruments, and significantly assisted in manuscript writing. All authors thoroughly proofread the paper and gave their approval to submit the final version. The authors declare no conflict of interest.
