Abstract
CTX0E03 is a human neural stem cell line previously reported to reduce sensory motor deficits in a middle cerebral artery occlusion (MCAo) model of stroke. The objective of this study was to investigate if CTX0E03 treatment promotes angiogenesis. As stroke leads to damage of the vasculature in the brain, angiogenesis may contribute to the functional recovery. To test this hypothesis, the angiogenic activity of CTX0E03 was assessed both in vitro and in vivo. In vitro, CTX0E03 expression of trophic and proangiogenic factors was determined by real-time RT-PCR, Western blot, and ELISA, and its angiogenic activity was investigated in well-established angiogenesis assays. In vivo, angiogenesis was investigated in naive mice and MCAo rat brain and was evaluated by immunohistochemistry (IHC) using Von Willebrand factor (VWF), a marker of blood vessel formation, and BrdU/CD31 double labeling in naive mice only. In vitro results showed that CTX0E03-conditioned medium and coculture significantly increased total tubule formation compared with controls (p = 0.002 and p = 0.0008, respectively). Furthermore, CTX0E03 cells were found to be in direct association with the tubules by ICC. In vivo CTX0E03-treated brains demonstrated a significant increase in areas occupied by VWF-positive microvessels compared with vehicle-treated naive mice (two-way ANOVA, Interaction p < 0.05, Treatment p < 0.0001, Time p < 0.0) and MCAo rat (p = 0.001 unpaired t test, Welch's correction). CTX0E03-treated naive mouse brains showed an increase in BrdU/CD31 colabeling. In conclusion, in vitro CTX0E03 cells express proangiogenic factors and may promote angiogenesis by both release of paracrine factors and direct physical interaction. Furthermore, in vivo CTX0E03-treated rodent brains exhibited a significant increase in microvessels at the site of implantation compared with vehicle-injected groups. Taken together these data suggest that CTX0E03 cell therapy may provide significant benefit to stroke patients through upregulation of angiogenesis in the ischemic brain.
Introduction
In the developed world, populations have an increasingly high risk of lifestyle and longevity diseases including cancer, atherosclerosis, diabetes, heart disease, and neurodegenerative conditions. Of these diseases, ischemic stroke is the third most common cause of death across the European Union and the US. The World Health Organization (WHO) estimates a likely increase from 1.1 million stroke events per year in 2000 to more than 1.5 million per year in 2025 solely due to demographic change (33). Currently, there are no clinical therapies available to treat ischemic damage or to restore loss of function in stroke patients beyond the use of tissue plasminogen activators in acute stroke (22). To overcome this growing clinical need, several studies have been undertaken to investigate the therapeutic potential of endogenous and transplanted stem cells in animal models of stroke (5,12,25). Stem cells have the capacity to respond actively to their environment, migrate to areas of injury, and secrete neuroprotective factors in addition to their potential for generating a variety of new functional cell types (6). These properties may confer therapeutic potential acutely and at later stages poststroke when conventional medical therapies would no longer be effective (4).
We have generated a clonal human neural stem cell line, CTX0E03, conditionally immortalized via a retroviral vector encoding the V-myc avian myelocytomatosis viral oncogene homolog mutated estrogen receptor (c-mycERTAM) fusion gene. The recombinant protein will be functional in the cell only in the presence of 4-hydroxytamoxifen (4-OHT), a minor metabolite of tamoxifen. Conditional growth of CTX0E03 allows continuous and stable cell expansion. CTX0E03 has been manufactured according to good manufacturing practice (GMP) to ensure reliable and reproducible stocks of cells for use in clinical applications (15). CTX0E03 is a multipotent neural stem cell with the capacity in vitro to differentiate into neurons, astrocytes, and oligodendrocytes and in vivo to ameliorate stable neurological deficits in a rodent model of cerebral ischemia (middle cerebral artery occlusion; MCAo) following transplantation into the putamen, adjacent to the infarcted region (26,29,31). The CTX0E03 cell line, delivered as the active ingredient via stereotactic injection into putamen, is currently in a Phase I clinical trial for patients with stable stroke disability in Scotland, the PISCES trial (NCT01151124, Clinicaltrials.gov).
Depending in part on the timing of cell implantation poststroke, the therapeutic effect of stem cell treatments has traditionally been attributed to neuronal and glial cell replacement in stroke models (20) and/or a more generalized reparative function of stem cells in these models (12). More specifically, recent brain ischemia studies have attributed the reduction of lesion size and recovery of behavioral functions to the angiogenic/vasculogenic, immunomodulatory, neuroprotective, neurogenic, and neurotrophic potential of stem cells (3,7,16). Studies performed in our labora tories have shown variable and often low long-term survival rates of implanted CTX0E03 in lesioned MCAo rats and there fore did not support the hypothesis that CTX0E03 therapeutic effects were merely attributed to cell replacement. However, the demonstration of neurogenesis and angiogenesis following CTX0E03 transplantation into the ischemic brain did suggest an alternative paracrine or bystander effect of the cell implants (31).
The regeneration of virtually all tissues is reliant upon angiogenesis; the fact that diffusion of nutrients, oxygen, and waste products is limited to a few hundred micrometers explains why inadequate angiogenesis leads to insufficient and reduced tissue repair (21). In the past years, many factors that play important roles in angiogenesis have been discovered and their critical and individual involvement in the angiogenic process extensively investigated including vascular endothelial growth factor A (VEGFA), basic fibroblast growth factor (bFGF), and epidermal growth factor (EGF) (8,10). VEGFA, an angiogenic growth factor, potent mitogen, and survival factor specific to endothelial cells (11,21), also demonstrates neuroprotective effects against ischemic injury (13,14,19,32,35). bFGF and EGF promote angiogenesis by a direct effect on endothelial cells and indirectly by the upregulation of endothelial cell VEGFA (18).
In this study, we have investigated the expression of the proangiogenic factors by CTX0E03 cells in vitro and further examined the angiogenic effect of CTX0E03 following transplantation in naive and ischemic MCAo rodent brain. We postulate that the functional recovery demonstrated in MCAo rats (26) may correlate with the angiogenic properties of CTX0E03.
Materials and Methods
CTX0E03 Cell Line
CTX0E03 cells (26) were cultured in a serum-free reduced modified medium (RMM) consisting of Dulbecco's modified Eagle's medium (DMEM)/F12 with high glucose (Invitrogen, Paisley, UK) containing 5 μg/ml insulin (Sigma, Poole, Dorset, UK), 100 μg/ml transferrin (Millipore, Watford, UK), 20 nM sodium selenite (Sigma), 60 ng/ml progesterone (Sigma), 16.2 μg/ml putrescine (Sigma), 0.03% human albumin solution (John Bell & Croyden, London, UK), and 2 mM glutamine (Invitrogen). Recombinant human bFGF (10 ng/ml), EGF (20 ng/ml) (PeproTech, London, UK), and 4-OHT (Sigma) were added as mitogens. CTX0E03 drug products, formulated for the in vivo studies, were prepared from frozen drug substance lots at passage levels P32–34 (15).
Real-Time RT-PCR Analysis
CTX0E03 total RNA was isolated using RNeasy (Qiagen, Manchester, UK), according to the manufacturer's protocol. Two micrograms of total RNA were reverse-transcribed into first-strand cDNA using random primer. Reverse transcription was performed with Superscript II reverse transcriptase (Invitrogen) for 1 h at 42°C, inactivated for 15 min at 70°C, and cooled to 4°C. cDNA (2.5 μl) were used in a PCR reaction containing 2× Roche master mix, 0.1 μg of human universal probe library (UPL, Roche, Welwyn Garden City, UK), and 0.4 μM primers. The cDNA was amplified using 35 PCR cycles in LC480 Roche. Serial dilution of a known amount of gDNA was used to produce a standard curve. The total copy number of the retrotranscribed total RNA of gene of interest was calculated using the standard curve. A panel of trophic factors, either directly correlated with angiogenesis (e.g., VEGFA) or originally discovered as neurogenic/neurotrophic growth factors, was selected for investigation. The primers used in the real-time RT-PCR are listed in Table 1.
List of Genes, Access Number, Universal Probe Library (UPL, Roche), and Primers

EGF, epidermal growth factor; dFGF, basic fibroblast growth factor; ANGPT1, angiopoietin 1; HIF-1α, hypoxia inducible factor-1α; TGF-β1, transforming growth factor-β1; VEGFA, vascular endothelial growth factor A.
Western Blot Analysis
Western blot analysis for angiopoietin 1 (ANGPT1) and VEGFA protein was carried out using CTX0E03 whole-cell lysate. To prepare the cell lysate, CTX0E03 cell monolayers in a T75 flask (Becton Dickinson & Co., Oxford, UK) were rinsed with cold phosphate-buffered saline (PBS; 4°C), lysed with 750 μl 1× SDS sample buffer and dithiothreitol (DTT) reducing agent (AMS Biotechnology, Abingdon, UK), and collected in a 1.5-ml centrifuge tube. Samples were triturated multiple times to lyse and shear the sample, then heated at 100°C for 2 min, and centrifuged. Cell lysates were loaded onto a 20% Tris-Tricine gel (PAGEgel Inc., San Diego, CA, USA), 20 μl per well. After electrophoresis, proteins were transferred onto a nitrocellulose membrane with 0.2 μm pore size by electroelution. Immunodetection was performed with a rabbit anti-ANGPT1 polyclonal (Abcam, Cambridge, UK; ab8451, dilution 1:250) or a mouse anti-VEGFA monoclonal antibody (Chemicon, Chandlers Ford, UK; mab3734 and SC73344, both used at 1:100). The proteins were detected using horseradish peroxidase-conjugated anti-rabbit-IgG (Cell Signaling Technology, Hitchin, UK; 1:2,500) or anti-mouse IgG (Pierce Biotechnology Inc., Loughborough, UK; 1:2,000) secondary antibodies, as appropriate. The nitrocellulose membrane was then processed using chemiluminescence detection reagents (Thermo Scientific, Hemel Hempstead, UK). The blots were then stripped and reprobed using anti-a-tubulin (Sigma, 1:1,000) to act as an internal loading level standard. Western blot images were captured using BioRad FluorS Imaging Unit (BioRad, Hemel Hempstead, UK). ANGPT1 has predicted molecular weights of approximately 57 kDa and approximately 75 kDa, representing highly glycosylated forms of the protein. VEGFA has a predicted band size of 24 and 45 kDa.
Human Angiogenesis ELISA Assay
Human angiogenesis ELISA strips (Signosis, Sunnyvale, CA, USA) were utilized according to the manufacturer's instruction. Fresh RMM and 24-h conditioned CTX0E03 RMM were analyzed for eight angiogenesis cytokines: tumor necrosis factor-α (TNF-α), insulin-like growth factor 1 (IGF1), VEGFA, interleukin-6 (IL-6), bFGF, transforming growth factor-b1 (TGF-β1), EGF, and leptin. Individual wells of the strip, coated with each of the primary antibodies directed against the specific angiogenesis cytokines, were loaded with test samples. Absorbance was measured by a spectrophotometer at 450 nm. The concentrations of the angiogenesis cytokines were directly proportional to the color intensity of the test sample.
Evaluation of Angiogenesis Induced by CTX0E03-Conditioned Medium
Induction of angiogenesis/tubule formation was performed using Cultrex BME growth factor-reduced Matrigel (Trevigen, Abingdon, UK). Matrigel was thawed on ice to prevent premature polymerization; aliquots of 200 μl were plated into individual wells of 24-well tissue culture plates (Costar) and allowed to polymerize at 37°C for at least 60 min (17). Human umbilical vein endothelial cells (HUVECs; Lonza, Castleford, UK), 5 × 104, were directly plated from frozen cryovials and cultured for 24 h in CTX0E03-conditioned medium or RMM as control.
HUVECs and CTX0E03 Coculture In Vitro Angiogenesis Assay
HUVECs were grown with endothelial cell growth media (EGM-2; Lonza), detached with trypsin/EDTA (Lonza), and then resuspended in EGM-2. The formation of network structures was assessed using Matrigel basement membrane matrix, containing growth factors (BD Biosciences, Oxford, UK), thick gel method according to the manufacturer's instructions (150 μl/48 wells). HUVECs were seeded alone or in combination with CTX0E03 (2.25 × 104 HUVECs alone or plus 7.5 × 103 CTX0E03, 75/25% mixture) in Matrigel-precoated 48 wells containing EGM-2. Plates were incubated at 37°C and 5% CO2 overnight. The wells were photographed under a phase-contrast inverted microscope at 4× magnification. For each condition, total tubule length per field was measured using the Image Pro-Plus 7 software (Media Cybernetics, Rockville, MD, USA). Each condition was run in triplicate.
Evaluation of CTX0E03 Angiogenesis on AngioKit Plate
AngioKit (TCS CellWorks, Buckingham, UK) is an endothelial cell-specific human angiogenesis assay in which HUVECs are cocultured with human fibroblast cells in a specially designed medium. Growing cultures at the earliest stages of tubule formation are supplied in a 24-well plate format so that test compounds and tissue explants can be added to the cultures within individual wells. CTX0E03 cells (5 × 103) were added to individual wells of these cultures (×4). The effect of CTX0E03 coculture on tubule formation was measured after 11 days; medium changes were performed according to the manufacturer's instructions. VEGFA (2 ng/ml) was used in the assay as positive angiogenic stimulator control. Quantification of total tubule formation was measured by an ELISA colorimetric assay, using combined AngioKit ELISA/CD31 tubule staining kit (TCS CellWorks) performed according to the manufacturer's instructions.
Immunocytochemistry (ICC) Detection of CTX0E03 and HUVEC Cells
ICC was carried out on CTX0E03 and HUVEC cocultures fixed using 4% paraformaldehyde (PFA)/4% sucrose (Pioneer Chemicals Ltd., Colchester, UK) in PBS for 15 min. Anti-nestin (1:500) (Millipore) and anti-VWF (1:1000) (DAKO, Glostrup, Denmark) were used to detect CTX0E03 and HUVECs, respectively. Cell cultures were incubated with primary antibodies diluted in PBS containing 1% normal goat serum (Vector Laboratories, Peterborough, UK) overnight at room temperature. After rinsing, CTX0E03 and HUVEC cocultures were incubated for 1.5 h at room temperature with anti-mouse-conjugated Alexa Fluor 488 (Invitrogen, 1:2,000) for nestin and anti-rabbit-conjugated Alexa Fluor 568 (Invitrogen, 1:2,500) for VWF, respectively. CTX0E03 and HUVEC cell nuclei were then counterstained with Hoechst 33342 (Sigma, 1 μM) for 2 min. Representative images were captured using an Olympus IX70 fluorescent microscope (Olympus, Southend-on-Sea, UK).
Cluster of differentiation 31 (CD31) staining was performed using TCS Cellworks combined CD31 ELISA and histology staining kit according to the manufacturer's instruction. CD31 staining was visualized using BCIP/NBT, which produced dark blue indigo precipitating dye. Brightfield images of CD31 staining were captured using visible light microscopy.
Implantation of CTX0E03 Into Naive Mouse Brain
This study was conducted in accordance with a project license approved by the University of Surrey Ethical Review Committee and UK Home Office and complied with the UK Animals (Scientific Procedures) Act (1986). Adult male Balb-C mice (Harlan Shardlow, UK), approximately 25 g in weight, were group housed (light/dark) and fed ad libitum. Following acclimatization, mice were anesthetized with isoflurane (3%) in oxygen prior to being placed in a mouse stereotaxic frame (David Kopf Instruments, Tujunga, CA, USA). CTX0E03 cells, prepared at 50,000 cells/μl in hypothermosol (HTS; BioLife Solutions, Inc., Bothell, WA, USA) vehicle, were implanted into the right striatum using the stereotaxic coordinates: anterior +0.3 mm, lateral −2.0 mm, and ventral −4.0 mm. One deposit of 4.5 μl of CTX0E03 cells or vehicle (four per group) was implanted using a 27-gauge needle mounted on a delivery cannula over a period of 120 s followed by a 3-min rest period before withdrawing the delivery device. Mice did not receive immunosuppressive drug treatment.
In Vivo Labeling with 5-Bromo-2-deoxyuridine-5-monophosphate (BrdU)
BrdU, a thymidine analog incorporated into the DNA of dividing cells, was used to label proliferating cells. Naive mice were injected intraperitoneally 4 h prior to implantation (vehicle or CTX0E03) and once daily thereafter with BrdU (50 mg/kg, Sigma) until sacrifice at 24 h, 72 h, and 7 days.
Implantation of CTX0E03 in Ischemic Stroke Middle Cerebral Artery Occlusion (MCAo) Rat Brain
CTX0E03 cells, prepared at 50,000 cells/μl, were implanted into the right and left caudate putamen using the stereotaxic coordinates: anterior −1.3 mm, −1.8 mm; lateral ±3.5 mm, ±4.0 mm; and ventral −6.5 mm, −6.0 mm. Two deposits of 4.5 μl of CTX0E03 cells or vehicle (eight and four per group, respectively) were administered into the right and left brain hemispheres of male rats that had 3/4 weeks previously undergone MCAo (31), using the same delivery method as described above. Animals were treated with a combination of an anti-inflammatory, SoluMedrol (methylprednisilone 20 mg/kg daily SC; Pharmacia & Upjohn, Milton Keynes, UK), and immunosuppressant, Cyclosporin A-Sandoz in Cremaphore El (10 mg/kg SC; Sigma), on the day prior to and on the day of implantation and alternate days thereafter by the SC route.
Perfusion and Preparation of Brains for Histology
At 24 h, 72 h, and 7 days, post-CTX0E03 or vehicle-implanted mice were anesthetized with pentobarbitol (Animal Care, York, UK) prior to transcardial perfusion with 0.9% heparinized saline (CP Pharmaceuticals, Wrexham, UK) followed by cold 4% buffered paraformaldehyde fixative (Pioneer Chemicals Ltd.). Using the same procedure, rats were perfusion fixed at 7 days post-CTX0E03 or vehicle administration. Brains were removed from skulls and immersed in the fixative for a further 4 h. Whole mouse brains were processed to paraffin wax; rat brains were washed with 0.1 M PBS and stored in 30% sucrose solution at 4–8°C.
Histology and Immunohistochemistry
Rodent brains were cut in the transverse plane to obtain sections throughout the site of implantation. Five-micrometer paraffin-embedded mouse sections and 50-μm free-floating frozen rat sections were produced using a Leica sledge microtome (Leica, Milton Keynes, UK); a freezing stage was used for the latter. Brain sections with prominent injection tracts were selected for immunohistochemistry (IHC) investigation to determine CTX0E03 survival and host angiogenesis. Analysis of host angiogenesis was performed using fluorescence IHC using rabbit anti-VWF (Dako, 1:1,000) and mouse anti-smooth muscle actin (Abcam, 1:50). Dual-label fluorescence IHC using a cocktail of anti-BrdU (Abcam, 1:5) and anti-CD31 (Abcam, 1:50) antibodies was performed to quantify BrdU colocalization with vascular endothelial cells. CTX0E03 survival was investigated with the following antibodies: rabbit anti-human-specific nestin (Abcam, 1:10,000), mouse antihuman mitochondria (MIT) (Abcam, 1:100), or mouse antihuman nuclei (HuNuc) (Millipore, 1:400); these were used for single-label or dual-label staining in combination with anti-VWF or smooth muscle actin. Briefly, following deparaffinization, rehydration, and antigen retrieval, mouse brain sections were washed with PBS, and 10% normal goat serum was applied to prevent nonspecific binding. After blocking, the antibody cocktail was applied to test sections and a positive control section (with known CTX0E03 implant), which were incubated overnight at room temperature; a negative control section was incubated with PBS. Detection was performed following three washes with PBS and by incubating sections for 2 h in a cocktail of secondary antibodies, goat anti-mouse Alexa 488 (Invitrogen, 1:400), and goat anti-rabbit Alexa 568 (Invitrogen, 1:2,500). Finally, a Hoechst counterstain (Sigma, 1 μg/ml) was applied before sections were washed with PBS and mounted with Vectashield (Vector). Quantification of VWF staining was performed centered on the site of implantation histologically visible in the sections. The areas occupied by VWF-positive microvessels were measured using the Image-Pro Plus 7 software and divided by the total areas of the corresponding fields to obtain the density of the microvessels in proximity to the injection site. Operators were blinded to treatments during evaluation of the VWF area measurements. Quantification of BrdU/CD31 colocalization was performed centered on the site of implantation. Cell counts of BrdU, CD31, and BrdU/CD31 dual-positive cells were collected. CTX0E03 cell survival was quantified by counting total numbers of nestin-positive CTX0E03 cells per section. CTX0E03 cell counts were collected from a minimum of eight sections spanning approximately 50 μm through the injection site. Representative fluorescent images of sections stained with a cocktail of nestin and smooth muscle actin antibodies were captured to demonstrate the tight association of CTX0E03 cells with host blood vessels in mouse brain 72 h post-CTX0E03 implantation.
Free-floating rat brain sections were stained using a similar protocol with the omission of the pretreatments required for paraffin-embedded tissue. VWF staining was quantified by measuring the density of microvessels per field, as previously described.
Statistical Analysis
In vitro angiogenic results were analyzed using the t test. In vivo angiogenic results were analyzed by two-way ANOVA or unpaired t tests with Welch's correction applied when necessary. A value of p < 0.05 was considered as significant. Mean ± SD are shown. Analyses were performed with Prism 4 software (GraphPad, San Diego, CA, USA).
Results
CTX0E03 Proangiogenic Factor Analysis by Real-Time PCR, Western Blot, and ELISA
In order to elucidate the proangiogenic potential of CTX0E03, we performed absolute quantitative RT-PCR, Western blot, and ELISA analysis on CTX0E03 cells. Absolute quantification of ANGPT1, ANGPT2, EGF, bFGF, hypoxia inducible factor-1α (HIF-1α), TGF-β1, and VEGFA gene copy numbers was measured, and all tested trophic factor genes were found to be highly expressed in CTX0E03 (Fig. 1A). The protein expression of ANGPT1 and VEGFA was also validated by Western blot analysis on CTX0E03 cell lysates (Fig. 1B, C, respectively). Furthermore, CTX0E03-conditioned medium, analyzed by ELISA, showed greater optical density readings compared with the control-tested angiogenesis cytokines (TNF-α, VEGFA, IL-6, bFGF, TGF-β1, EGF, and leptin), with the exception of IGF1, which cross-reacted with the unconditioned RMM (Fig. 1D).
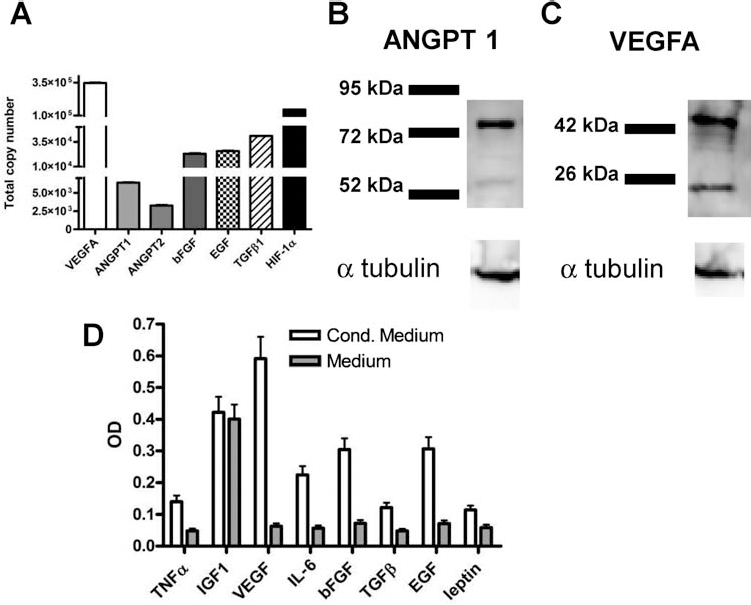
In vitro CTX0E03 characterization of proangiogenic and trophic factors. (A) CTX0E03 real-time RT-PCR absolute quantification of gene copy number of vascular endothelial growth factor A (VEGFA), angiopoietin 1 (ANGPT1), ANGPT2, basic fibroblast growth factor (bFGF), epidermal growth factor (EGF), transforming growth factor-β1 (TGF-β1), hypoxia inducible factor-1α (HIF-1α) performed on 0.250 ng/reaction of CTX0E03 retrotranscribed total RNA. (B, C) Western blot analysis for ANGPT1 (B), and VEGFA (C) proteins in CTX0E03 whole-cell lysate. (D) Human angiogenesis ELISA strip optical density read out performed on CTX0E03-conditioned and unconditioned medium. TNF-α, tumor necrosis factor-a; IGF1, insulin-like growth factor 1; IL-6, interleukin-6.
CTX0E03 Effects on In Vitro Tubule Formation on Matrigel
Matrigel tube formation assays are an established model for studying in vitro angiogenic capacity of endothelial-like cells. CTX0E03-conditioned medium significantly enhanced HUVEC total tubule formation on Matrigel compared with control HUVECs cultured in unconditioned RMM (Fig. 2A–C).
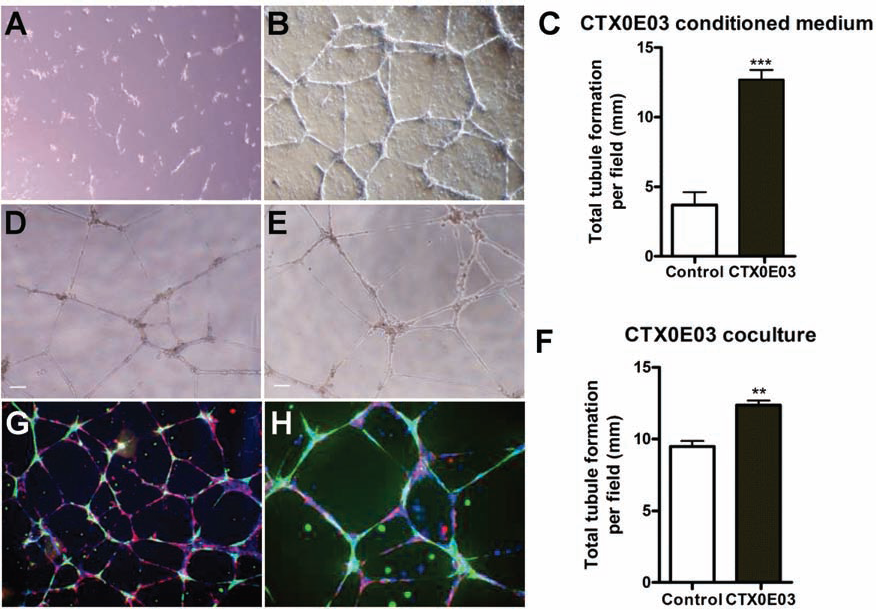
In vitro angiogenic biological activity of CTX0E03 analyzed with Matrigel angiogenesis assay. (A, B) Representative images (scale bar: 50 μm) showing the effect of (A) unconditioned reduced modified medium (RMM) and (B) CTX0E03 24-h conditioned medium on human umbilical vein endothelial cell (HUVEC) Matrigel tubule formation. (C) Graph showing total tubule length on Matrigel. CTX0E03-conditioned wells showed a significant increase in tubule length (t test, p = 0.002). Representative images of tubule formation of HUVECs alone (D), in coculture with CTX0E03 (E) seeded on Matrigel precoated wells. (F) Graph showing total tubule length. HUVEC/CTX0E03 coculture wells showed a significant increase in tubule length (t test, p = 0.0008). (G, H) Immunocytochemistry (ICC) analysis of HUVEC/CTX0E03 coculture wells; HUVECs were stained with Von Willebrand factor (VWF; red) and CTX0E03 with human-specific nestin (green).
Culturing CTX0E03 cells together with HUVECs significantly improved the networking-forming capacity. CTX0E03 was able to interact with HUVECs and form complex networks, composed of nodes connected by several thin branches (Fig. 2D, E). Total tubule length was measured, and CTX0E03/HUVEC cocultures showed a significant increase in total tubule length compared with control samples (Fig. 2F). Interconnections established between HUVEC/CTX0E03 cells were analyzed by fluorescence immunocytochemistry (Fig. 2G, H). CTX0E03 and HUVEC were labeled with nestin (green) and VWF (red) antibodies, respectively. Analysis by fluorescence microscopy revealed that CTX0E03 cells were positioned both at the crosspoint as well as along the tubules.
CTX0E03 Effects on In Vitro Tubule Formation on AngioKit Assay
AngioKit colorimetric CD31 ELISA assay demonstrated that in CTX0E03-treated wells, there was a significant (p < 0.001) increase in total tubule formation compared with the control. Plates were subsequently stained for detailed analysis of tubule morphology (Fig. 3A–E). Overlaid images, acquired from the same field, showed that CD31 staining did not colocalize with human nestin (specifically expressed by CTX0E03) and indicated that CD31 marker expression was restricted to HUVECs (Fig. 3F–H).
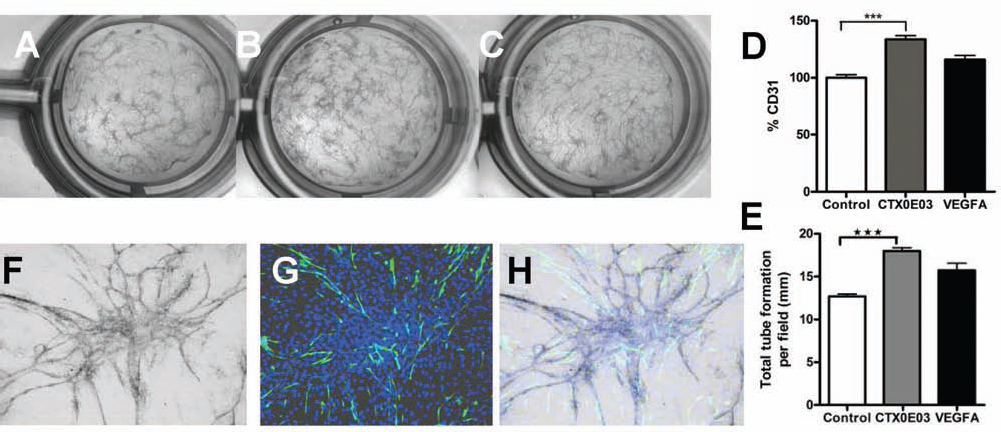
In vitro angiogenic biological activity of CTX0E03 analyzed on AngioKit. Representative images of AngioKit 24-well plate, (A) control well untreated, (B) cocultured with 5 × 103 CTX0E03 cells, (C) or treated with VEGF (2 ng/ml). (D) CD31 ELISA quantification measured by a colorimetric assay and expressed as percentage compared with control. Cluster of differentiation 31 (CD31) values from CTX0E03 cocultured samples were significantly greater than controls (t test, p < 0.001). (E) Graph showing total tubule length. HUVEC/CTX0E03 coculture wells showed a significant increase in tubule length (t test, p = 0.0017) compared with control. (F–H) Representative images showing CD31 (HUVECs) and nestin (CTX0E03) costaining. CD31 reactivity was detected in HUVECs only. (F) CD31 BCIP/NBT staining, (G) nestin ICC staining, and (H) overlay of nestin and CD31 images.
CTX0E03-conditioned medium was not tested using the AngioKit cultures due to the fact that FBS and growth factors, contained in the specially designed medium, would interfere with the final read out. For this reason, we restricted the analysis of the CTX0E03-conditioned medium to the Matrigel angiogenesis assay in which all the reagents used, both medium and Matrigel, did not contain any growth factors. We could, therefore, conclude that any increase in total tubule formation was a direct effect of the growth factors released in to the medium by CTX0E03.
Angiogenic Effect of CTX0E03 Following Implantation Into Naive Mouse Brain
The angiogenic effect of CTX0E03 in naive mouse brain was quantified by measuring the density of VWF-positive microvessels at the site of implantation (Fig. 4). In the CTX0E03-treated brains, the percentage area covered by VWF per field ranged from 3.32 to 5.61, with a mean value of 4.13 ± 1.15 at 72 h, and from 5.08 to 11.10, with a mean value of 8.33 ± 2.18 at 7 days, respectively. In contrast, vehicle-treated brains were considerably lower and ranged from 0.38 to 1.41, with a mean value of 1.07 ± 0.42 at 72 h, and from 1.10 to 2.83, with a mean value of 1.94 ± 0.81 at 7 days, respectively (Fig. 4B). At 7 days postimplantation, both CTX0E03 and vehicle-treated groups demonstrated an increase in VWF expression compared to earlier time points; however, blood vessel formation was significantly more prominent in the CTX0E03-treated brains compared with those treated with vehicle (two-way ANOVA, Interaction p < 0.05, Treatment p < 0.0001, Time p < 0.01). A dense network of branched blood vessels was observed closely associated with cell implantation sites in brains treated with CTX0E03 (Fig. 4A). In vivo BrdU labeling was used to investigate new blood vessel formation. Sections from brains collected at 24 h, 72 h, and 7 days post vehicle or CTX0E03 treatment were subjected to BrdU/CD31 dual-label IHC (Fig. 4H, I). Cell counts of CD31, BrdU, and CD31/BrdU dual-positive cells were recorded (data not shown). At 24 and 72 h postimplantation, BrdU-CD31 double-positive cells were only present in the CTX0E03 group, representing 62.5 ± 18.30% and 83.33 ± 16.67% of the CD31-positive cells, respectively. At 7 days, 73.70 ± 10.53% of the CD31-positive cells were found dual labeled in CTX0E03 brains compared to 22.22 ± 16.68% in the control group. These data indicate that CTX0E03 treatment enhanced the formation of neovessels by inducing CD31-positive cell division. In conclusion, CTX0E03 treatment demonstrated a significant angiogenic effect compared with vehicle in naive mouse brain at 72 h and 7 days following implantation. Colocalization of implanted CTX0E03 cells and host microvasculature was further demonstrated by duallabel fluorescence IHC using anti-human-specific nestin and anti-smooth muscle actin. These brains exhibited the presence of a possible “neurovascular niche” (24) in which CTX0E03 interacted in tight association with host blood vessels (Fig. 4E–G).

In vivo angiogenic investigation in naive mice. (A) IHC analysis of implanted CTX0E03 cells (green, detected with human mitochondria) and host VWF expression (red). Scale bar: 10 μm, 72 h postimplantation. (B) VWF quantification measured as percentage area occupied by VWF immunoreactive microvessels at 72 and 7 days postimplantation in CTX0E03 and vehicle-injected samples, respectively (two-way ANOVA, Interaction p < 0.05, Treatment p < 0.0001, Time p < 0.01). (C) Representative images of CTX0E03 survival (red, detected with human-specific nestin) and Hoechst nuclear counterstain (blue), at 4 h, 24 h, 72 h, and 7 days postimplantation. Scale bar: 50 μm. (D) CTX0E03 quantification expressed as total number of cells detected per field. For each brain, an average of eight sections were analyzed spanning ±50 μm from the injection site. (E) CTX0E03 cells (red, detected with human-specific nestin) were found in close association with blood vessels (green, detected with smooth muscle actin); (F, G) magnification of the selected squared area of image (E). Scale bar: 50 μm. (H, I) Representative images of CD31 (red)/BrdU (green) dual-label IHC showing vehicle (H) and CTX0E03 (I) brains at 7 days posttreatment. Scale bar: 10 μm.
CTX0E03 cell survival was investigated up to 7 days postimplantation (Fig. 4C). The incidence and total number of surviving CTX0E03 cells were both reduced over time (Fig. 4D); no CTX0E03 cells were found in any of the vehicle-treated brains.
Angiogenic Effects of CTX0E03 Following Implantation Into Ischemic MCAo Rat Brain
Quantification of host VWF expression following CTX0E03 implantation was also performed in an MCAo rat stroke lesion model. Host VWF expression was detected in the contralateral hemispheres of MCAo-treated rat brains (Fig. 5A, B). In CTX0E03-treated brains, the percentage of the area covered by VWF ranged from 3.45 to 16.98, with a mean value of 9.24 ± 4.26. In contrast, the host VWF area percentage in the vehicle treated was considerably lower and ranged from 0.22 to 2.08, with a mean value of 0.81 ± 0.74 (Fig. 5C). At 7 days postimplantation, VWF expression was more prominent in CTX0E03-treated rats compared with vehicle-treated groups, (p = 0.001 unpaired t test, Welch's correction) (Fig. 5D). These data reflect the angiogenic effect of CTX0E03 in a second rodent model and are comparable with those generated in naive mouse brain.

In vivo angiogenic investigation in a rat MCAo model. Host VWF (red) expression analysis performed by immunohistochemistry (IHC) on sections with prominent implantation sites. (A) CTX0E03 (green)-treated brain and (B) vehicle-treated and Hoechst nuclear counterstain (blue) at 7 days postimplantation. Scale bar: 50 μm. (C) Magnification of the selected squared area of image (A). Scale bar: 10 μm. (D) VWF expression levels were presented as percentage area occupied by VWF immunoreactive microvessels at 7 days postimplantation in CTX0E03 and vehicle-injected samples (unpaired t test, Welch's correction, p = 0.001). MCAo, middle cerebral artery occlusion.
Discussion
In the present study, we have characterized the in vivo and in vitro angiogenic potential of a conditionally immortalized human neural progenitor cell line, CTX0E03. This cell line has a long-term culture capability, exhibiting a normal karyotype, which allows a consistent and homogenous source of cells to test extensively in preclinical models and ultimately to treat a large patient population on demand. CTX0E03 has been shown to reverse the functional deficits associated with stroke disability when administered several weeks after the stroke event in relevant preclinical models (26,29,31). As stroke leads to damage of the vasculature in the brain, angiogenesis may contribute to the functional recovery of the ischemic region with increased collateral circulation (2,28). CTX0E03 cells were also found to be able to increase blood flow on the lesioned side in MCAo-lesioned rats (9). Given the beneficial effects of CTX0E03 treatment in the MCAo stroke model, it is possible that CTX0E03 may exert its mode of action via modulation of an angiogenic mechanism. To test this hypothesis, we characterized the angiogenic potential of CTX0E03 in vitro and in vivo.
In vitro, we characterized CTX0E03 by real-time PCR, Western blot, and ELISA. The expression of several well-known angiogenic factors (VEGFA, EGF, bFGF, ANGPT1, TGFβ1, HIF-1α and ANGPT2) was confirmed in CTX0E03 cultures under standard proliferation conditions. We focused our attention on VEGFA production, which was confirmed by both Western blot and ELISA. CTX0E03 proangiogenic potential was assessed furthermore in well-established in vitro angiogenesis assays, using Matrigel and AngioKit. Total tubule formation was measured in wells treated with the addition of CTX0E03. In both cases, CTX0E03-treated wells showed an increase in total tubule length formation compared with untreated controls. Furthermore, CTX0E03-conditioned medium induced HUVEC tube formation in the Matrigel assay. All these findings support a biological angiogenic activity of CTX0E03. Interestingly, CTX0E03 cells appeared to perform a dual role in the promotion of angiogenesis in vitro: first, by the release of paracrine factors, as indicated in the Matrigel-conditioned medium assay and, second, by direct physical cell interaction, as shown in the CTX0E03-treated wells in Matrigel and AngioKit assays.
To determine the angiogenic potential of CTX0E03 in vivo, an MCAo model of stroke in both rats and naive mice was used. Postimplantation IHC analysis using an antibody raised against VWF, an endothelial cell marker of blood vessel formation, demonstrated a clear increase in new host blood vessel formation in CTX0E03-treated brains in both rodent models. In vivo BrdU labeling in naive mouse brain confirmed enhanced and accelerated neovessel formation following CTX0E03 treatment. CTX0E03 cells were found in precise association with vessels suggesting the establishment of a “neurovascular niche.” Within this type of niche, neural progenitor cells lie in close proximity to endothelial cells and can induce angiogenesis and at the same time neurogenesis; furthermore, endothelial cells can secrete soluble factors that regulate neuronal differentiation (1,24,27,34). It is pertinent to suggest that CTX0E03 may support this effect by the local delivery of growth factors. The present data demonstrate that CTX0E03 neural stem cells can interact with vascular endothelial cells specifically in areas of vascular remodeling, sprouting, and angiogenesis and ultimately aid repair.
Additionally, we determined time-related CTX0E03 cell survival in naive mice following intracerebral implantation. Our data confirmed a substantial and rapid reduction of the implanted CTX0E03 cells by day 7 (Fig. 4C, D). In MCAo rats, a greater and more extended CTX0E03 cell survival was observed at day 7 (30), but still a substantial reduction was evident at a later time point of investigation (26,31). The mechanisms involved in implanted CTX0E03 loss over time are not yet fully understood. However, our current investigations focus on the role of apoptosis on CTX0E03 cells following implantation. Our findings do confer with those of others who also report the loss of implanted cells and short-term survival following implantation (1,24,27,34). Also, these data further support the theory that stem cells promote recovery by providing trophic factors to the injured tissue and brain, fostering both neurogenesis and angiogenesis and not merely by a cell replacement effect (23).
Conclusion
In summary, we have demonstrated the angiogenic potential of CTX0E03 cells by their ability to release several proangiogenic factors, their close association with vascular cells in vitro and in vivo (in the neurovascular niche), and the significant promotion of new blood vessel formation in rodent brain following CTX0E03 implantation. The lack of long-term CTX0E03 survival in vivo further supports a paracrine effect. Taken together, these data support the hypothesis that CTX0E03 aids in functional recovery in stroke animals by promoting angiogenesis and further demonstrate the potential of CTX0E03, human neural stem cell line, as a therapeutic agent for stroke and other conditions.
Footnotes
Acknowledgments
We acknowledge Dr. Randolph Corteling, Susan Hines, Cathy Oliveira, and Julie Heward for helping in the preparation of CTX0E03 for in vivo studies and Dr. Erik Miljan for his general support and helpful discussion. Authors' contribution: C.H. (angiogenesis in vivo assessment and analysis, immunohistochemistry, imagine acquisition, writing of the manuscript); L.S. (angiogenesis in vitro assessment, molecular biology testing, data analysis, writing of manuscript); R.P.S. (surgery, cell implantation, data analysis); E.T. (surgery, cell implantation); S.R. (histology support); J.D.S. (conception of study, financial support). All authors are employees, stock, and/or stock option holders in ReNeuron Ltd or its parent company.
