Abstract
Pig islets demonstrate significantly lower insulin secretion after glucose stimulation than human islets (stimulation index of ~12 vs. 2 for glucose 1 and 15 mM, respectively) due to a major difference in β- and α-cell composition in islets (60% and 25% in humans and 90% and 8% in pigs, respectively). This leads to a lower rise in 3′,5′-cyclic adenosine monophosphate (cAMP) in pig β-cells. Since glucagon is the major hormonal effector of cAMP in β-cells, we modified pig islet structure in vivo to increase the proportion of α-cells per islet and to improve insulin secretion. Selected doses (0, 30, 50, 75, and 100 mg/kg) of streptozotocin (STZ) were intravenously injected in 32 young pigs to assess pancreatic (insulin and glucagon) hormone levels, islet remodeling (histomorphometry for α- and β-cell proportions), and insulin and glucagon secretion in isolated islets. Endocrine structure and hormonal content of pig islets were compared with those of human islets. The dose of STZ was significantly correlated with reductions in pancreatic insulin content (p < 0.05, r 2 = 0.77) and the proportion of β-cells (p < 0.05, r 2 = 0.88). A maximum of 50 mg/kg STZ was required for optimal structure remodeling, with an increased proportion of α-cells per islet (26% vs. 48% α-cells per islet for STZ <50 mg/kg vs. >75 mg/kg; p < 0.05) without β-cell dysfunction. Three months after STZ treatment (30/50 mg/kg STZ), pig islets were isolated and compared with isolated control islets (0 mg/kg STZ). Isolated islets from STZ-treated (30/50 mg/kg) pigs had a higher proportion of α-cells than those from control animals (32.0% vs. 9.6%, respectively, p < 0.05). After in vitro stimulation, isolated islets from STZ-treated pigs demonstrated significantly higher glucagon content (65.4 vs. 21.0 ng/ml, p < 0.05) and insulin release (144 μU/ml) than nontreated islets (59 μU/ml, p < 0.05), respectively. Low-dose STZ (<50 mg/kg) can modify the structure of pig islets in vivo and improve insulin secretion after isolation.
Keywords
Introduction
Numerous in vitro studies (6,7,12,29) describe major functional differences in insulin secretion by pig islets compared with human islets. Although the insulin content per islet is higher in pigs than in humans, glucose-stimulated secretion is 10-fold lower at 1 and 15 mM glucose (G1 and G15, respectively) in pig islets than in human islets (12,16). In vivo studies have confirmed this lower insulin secretion by pig islets in comparison with primate islets in response to glucose challenges (4). In preclinical and clinical allotransplantation studies, ~10,000 islets equivalent (IEQ/kg) of recipient's body weight is sufficient to achieve insulin independence in diabetic cynomolgus monkeys (25) and patients (17,36), while about 3,000 (autotransplantation) to 23,000 (allotransplantation) IEQ/kg of recipient's body weight is required to obtain normoglycemia in pigs (30,31). In preclinical xenotransplantation studies, 30,000–50,000 porcine IEQ/kg of recipient's body weight were required to cure diabetes in primates (3,18).
A possible way to increase pig insulin secretion is to use agents that trigger increased 3′,5′-cyclic adenosine monophosphate (cAMP) concentration within β-cells (forskolin, isobutylmethylxanthine, and glucagon) (1,7,12,23,35), which in turn increases β-cell insulin secretion (15,22,24). Under physiological conditions, the cAMP concentration in β-cells is increased by glucagon, which is secreted by α-cells that are closely associated with β-cells in pancreatic islets (19,34). Consequently, stimulation of α-cells improves insulin secretion. The increase in insulin secretion in response to glucagon is moreover used in clinical practice to determine the residual β-cell function in patients with diabetes (glucagon test realized in 10 min) (28). The physiological difference between insulin secretion from pig and human islets can be explained by a major difference in the cellular composition of islets between the two species. Cabrera et al. (2) compared the architecture of human islets versus those from pigs and found that human α-, β-, and δ-cells are scattered throughout islets (aligned along blood vessels with no particular order or arrangement). In contrast, porcine islets have α- and δ-cells situated along the periphery of the islet, while β-cells reside within the core of the islet. Furthermore, when comparing the proportions of β- and non-β-cells in human and porcine isolated islets, Dufrane et al. (9) showed that porcine islets have a higher ratio of β- to α-cells compared with human islets (90%/8% vs. 64%/26%, respectively) (20). These studies suggest a major structural difference between species that can affect insulin secretion (12,16).
Modifying pig islet structure to obtain a b/a proportion similar to that in humans would be one way to improve insulin secretion. To obtain a “human-like” α-cell proportion in pig islets, a 300% increase in α-cells is required with a simultaneous 30% decrease in β-cells. Dufrane et al. (11) demonstrated the plasticity of α-cells in a primate model following streptozotocin (STZ) treatment and β-cell destruction. After injection of 50 mg/kg STZ in nonhuman primates, a correlation was found between the increase of α-cell mass (89%) and the course of diabetes (>53 days). Functionally, glucagon serum levels also increased.
Based on these results, we used this plasticity property of α-cells to improve pig insulin release by pancreatic tissue remodeling. Different doses of STZ were tested in vivo to modify the islet structure to obtain “human-like” proportions of α- and β-cells in pig islets (+300% α-cells) without affecting glucose tolerance. After determining the optimal STZ dose, we investigated the function of the modified isolated islets.
Materials and Methods
Human and Pig Pancreatic Donor Sources
Seven human pancreases were obtained from cerebral death donors (three females, four males) obtained from multiorgan donors through the Eurotransplant Network (Leiden, the Netherlands), in accordance with the ethics committee (protocol UCL-HIA-001, authorization 2001/79) following the principles of the Declaration of Helsinki of 2000 and the guidelines defined by Belgian authorities. The donors were 28–62 years old.
Thirty-two pancreases of young Landrace male pigs (12–15 weeks old, weighing 42.2–50 kg) obtained from Rattlerow Seghers (Lokeren, Belgium) were harvested.
Pig Pancreatic Tissue Remodeling
For STZ injection, five doses (0 mg/kg, n = 10; 30 mg/kg, n = 7; 50 mg/kg, n = 4; 75 mg/kg, n = 3; 100 mg/kg, n = 5) of filter-sterilized STZ (Sigma, Bornem, Belgium) were tested. STZ was solubilized in citrate buffer [25% (v/v) sodium citrate, 23% (v/v) citric acid, 52% (v/v) H2O, pH 4.5; all from Sigma-Aldrich, St. Louis, MO, USA] and filtered before injection in the external jugular (13).
To determine in vivo metabolic function, intravenous glucose tolerance tests (IVGTTs) were performed prior to and 3 months after STZ injection under general anesthesia induced by intramuscular injection of Zoletil (VIRBAC, Carros, France) at a dose of 6 mg/kg and maintained through intubation and inhalation of enflurane (0–1.5%; Abbott Laboratories, Queenborough, Kent, UK), nitrous oxide, and oxygen. A catheter (Terumo, Leuven, Belgium) was introduced into the external jugular vein, and a glucose (Mediatech Cellgro, Manassas, VA, USA) solution of 0.5 g/kg was injected. Blood samples were taken prior to and 1, 3, 5, 10, 20, 30, 60, and 90 min after glucose injection. Glycemia was measured from these samples (AccuChek, Roche, Brussels, Belgium), and serum was reserved for radioimmunoassay (RIA) quantifications (insulin).
Procurement of Pancreas
Human abdominal organs were perfused in situ with modified University of Wis-con sin (UW) solution (no hydroxyethyl starch and low K+/high Na+; Allopurinol, D+ raffinose pentahydrate, glutathione-free acid reduced, lactobionic acid-free acid, potassium phosphate monobasic, magnesium sulfate; all from Sigma-Aldrich) (8) (4°C). The warm ischemia time varied between 12 and 40 min. After removal, the pancreases were shipped in cold modified UW solution from the donor center to the isolation laboratory. The cold ischemia time varied from 2 to 10 h (10).
Young pigs underwent anesthesia as for measuring in vivo metabolic function. Pigs were subjected to laparotomy, the infrarenal aorta and vena cava were clamped, and the thoracic aorta was isolated and clamped. In situ intra-aortic perfusion was then carried out with a 4°C physiological solution (Baxter, Lessines, Belgium). During the in situ cold perfusion, the entire pancreas was completely dissected and freed. The pancreatic duct was revealed, and an 18-gauge catheter was inserted into it. The splenic and pancreaticoduodenal vessels were clamped, and the whole pancreas was harvested (head/tail for islet isolation and body for histomorphometry). The pancreas was then weighed and placed on ice to avoid any warm ischemia (8,10).
Pancreas Endocrine Structure Prior to Isolation
Insulin and Glucagon Hormonal Content.
For pancreatic hormonal content, insulin and glucagon were extracted from small samples of the pancreas collected from the three zones (head, body, tail) before being quantified by RIA.
For the extraction, ~1 g of each pancreas was cut into small fragments (~1–2 mm3) and 5 ml of a solution composed of 75.75% CH3CH2OH, 24.22% H2O, and 0.03% HCl (both from VWR, Leuven, Belgium) was added. The following steps were performed with the samples kept on ice. Samples were mixed (Ultra-Turrax T25, Janke & Kunkel IKA-Labortechnik, Staufen, Germany) for 20 s four times and 5 ml of a solution of 75% CH3CH2OH, 22% H2O, 3% HCl was added to the mixture. The samples were then sonicated for 20 s four times (Sonifier B12, Branson Sonic Power Company, Danbury, CT, USA) and centrifuged 10 min at 4°C at 450×g (Jouan CR422, Thermo Scientific, Breda, The Netherlands). The supernatant was removed and kept at −20°C. Two milliliters of a solution of 75% CH3CH2OH, 23.5% H2O, and 1.5% HCl was then added to the precipitate, and a second sonication was performed. The samples were then kept at −20°C overnight. After another centrifugation step for 10 min at 4°C at 450×g, a second supernatant was removed and kept at −20°C. Thereafter, 2 ml of a solution of 75% CH3CH2OH, 23.5% H2O, and 1.5% HCl was added to the precipitate; the mixture was sonicated and centrifuged for 10 min at 4°C at 450×g; and a third supernatant was removed. The three supernatants were then pooled and constituted the pancreatic extract.
Human insulin-specific and glucagon RIA kits (Millipore, Billerica, MA, USA) were used to determine the hormonal content in human and pig pancreatic extracts as well as in isolated islets. A cross reaction of 100% between both human and porcine insulin and human and porcine glucagon was described by the RIA kit manufacturer. The insulin and glucagon concentrations in pancreatic extracts were measured by following the instructions of the manufacturer. Briefly, 100 μl of hydrated 125I-insulin or 125I-glucagon and 100 μl of specific antibodies were added to 100 μl of pancreatic extract diluted in the dilution buffer provided by the manufacturer to 1:500, 1:1,000, 1:3,000, and 1:9,000; 100 μl of pure sera; 100 μl of media (1:20); and 100 μl of modified islet extract (1:200). After incubation overnight at room temperature, 1 ml of precipitating reagent was added, followed by incubation for 20 min at 4°C. The samples were then centrifuged for 20 min at 2,000–3,000 × g at 4°C. The supernatants were immediately removed, and insulin and glucagon contents were determined by a Wallac Wizard 1470 Automatic gamma counter (GMI, Inc., Ramsey, MN, USA) for 1 min.
Immunohistochemistry
The cellular composition of untreated human and pig islets was determined by immunohistochemistry and histomorphometry. After overnight fixation in formol (VWR) at room temperature, the small samples were embedded in paraffin and cut in 5-μm sections. These sections were then deparaffinized, rehydrated, and washed in 0.05 mol/L Tris-HCl-buffered solution (TBS, pH 7.4; Sigma-Aldrich and VWR). After inactivation of endogenous peroxidase by a 30-min incubation in 0.3% H2O2 (Sigma-Aldrich), the sections were incubated with 10% normal goat serum (Biochrom, Berlin, Germany) in TBS for 30 min. The sections were then incubated overnight at 4°C with glucagon rabbit polyclonal antibody (BD Biosciences, Erembodegem, Belgium) diluted at 1:100. After washes in TBS, the slides were incubated for 30 min with anti-rabbit-IgG (1:500; Prosan, Merelbeke, Belgium), and the antibodies were detected by EnVision anti-rabbit system (Dako A/S, Glostrup, Denmark) for 1 h at room temperature. The peroxidase activity was revealed by immersion of sections for 10 min in a solution of 3,3′-diaminobenzidine hydrochloride (3,3′-diaminobenzidine, 50 mg/100 ml at pH 7.4; Fluka, Buchs, Switzerland), supplemented with 0.01% H2O2. The glucagon staining was followed by the detection of the β-cells by alkaline phosphatase anti-alkaline phosphatase system (Promega, Madison, WI, USA). After washing, nonspecific sites were again inhibited by a 30-min incubation with 10% normal goat serum in TBS. The slides were thereafter incubated overnight with mouse insulin monoclonal antibody (Abcam, Cambridge, UK) diluted at 1:800. After washes in TBS, the slides were incubated for 30 min with anti-mouse–IgG–biotin conjugate (Abcam) and then with streptavidin–peroxidase conjugate at a dilution of 1:500 (Roche Diagnostics, Mannheim, Germany), and the reaction was revealed by the kit fast red/naphtol AS MX tablet sets (Sigma-Aldrich).
After washes, the slides were counterstained with Mayer's hemalum (Millipore) and mounted with a hydro-soluble mounting (Dako, Carpenteria, CA, USA).
Histomorphometry Analysis
The cellular a/b proportion and geometric distribution (core/periphery) in islets from human and pig pancreases were quantified. Based on islet size, three groups of five islets each (50–99, 100–149, and 150–200 μm) were compared in control and modified young pig versus human pancreas.
The number of small β-cell clusters (≤3 insulinimmunoreactive cells) in the total area of the slide was also assessed. The number of these clusters was manually counted through the entire slide. The area of the slide was measured with ImageJ 1.43i software after capture of slide pictures with a camera (NIKON Coolscan 5000, accessory FH-G1, Brussels, Belgium) and calibrated with a stage micrometer to correspond to the number of pixels shown on the image analyzer.
Islet Structure After Isolation
Islet Isolation
Human islets were isolated (n = 4 pancreas) using a simplified previously described method (10). Briefly, after being digested with enzyme (Liberase HI, Roche/Boehringer Mannheim, Brussels, Belgium; 0.5 mg/ml) dissolved in modified UW solution, each pancreas was cut into approximately 3 × 4-cm pieces and digested by a dynamic incubation at 37°C. The pieces were placed in a sterile 1-L Nalgene jar (Leuven, Belgium) that was closed and agitated by hand from time to time for ~45 min. The digest was then filtered in a 500-μm filter (Negofiltres, Ecuelles, Belgium) and purified in molecular biology grade Ficoll (densities 1.096, 1.080, 1.070, and 1.060) (Mediatech Cellgro, Manassas, VA, USA). The islets were hand-picked, washed, and suspended in Ham-F10 + 10% human serum (Cambrex, East Rutherford, NJ, USA).
Pig islet structure and functionality were assessed for selected doses of STZ (30 and 50 mg/kg inducing an appropriate a/b islet remodeling) and compared with control pig islets (0 mg/kg STZ).
Islets from young pigs treated with selected doses of STZ (0, 30, and 50 mg/kg; n = 3 for each dose) were isolated following a method previously described by Dufrane et al. (8). Briefly, after being completely dissected, the pancreases were digested by a modified static digestion method: infusion of the pancreas with Liberase DL research grade (Roche/Boehringer Mannheim; 0.43 mg/ml) dissolved in modified UW solution; placement in a sterile 1-L Nalgene jar for digestion by static incubation at 37°C for 30-50 min; filtration with a 500-μm filter; purification in molecular biology grade Ficoll (densities 1.090, 1.060, and 1.010); and washing and suspension of islets in Ham-F10 (Gibco) + 10% newborn calf serum (Merck-Eurolab, Overijse, Belgium).
Histology
The endocrine cellular composition of isolated human and pig islets was determined by immunohistochemistry. After overnight fixation in formol at room temperature, aliquots of islet preparations were embedded in paraffin and cut in 5-μm sections. Insulin and glucagon staining was performed as already described (see Immunohistochemistry section).
α- and β-cell proportions and localizations inside pig and human islets were quantified and compared between isolated islets (50-99, 100-149, and 150-200 μm) from control and STZ-treated (30 and 50 mg/kg) pigs.
In Vitro Islet Functionality
After overnight incubation at 37°C (after islet isolation), 5% CO2/95% air in Roswell Park Memorial Institute (RMPI) medium (Life Technologies, Gent, Belgium) containing 10% heat-inactivated fetal calf serum (VWR), 100 IU/ml penicillin, 100 μg/ml streptomycin (Life Technologies), and 5 mmol/L glucose, islet function was assessed by 24-h incubation of 100 islets in 2 ml RPMI at 5 mmol/L glucose, 15 mmol/L glucose, and 15 mmol/L glucose + 1 μmol/L forskolin (Calbiochem-Behring, San Diego, CA, USA) added from a 1 mmol/L stock solution in dimethyl sulfoxide (VWR). Three replicates per concentration were performed. Media were thereafter recovered for insulin and glucagon quantification, and islets were transferred to acid-ethanol for extraction and quantification of hormones by RIA.
Statistical Analysis
Values are presented as means±standard deviations (except where otherwise specified). One-sample Kolmogorov-Smirnov testing was used to assess the normal distribution of values. The statistical significance of differences between experimental groups was tested by one-way analysis of variance (ANOVA) with a Bonferroni's post hoc test. The statistical tests were carried out with Systat version 8.0 (Chicago, IL, USA). Differences were considered to be significant at p < 0.05.
Results
STZ doses at 30, 50, 75, and 100 mg/kg did not modify the IVGTT curves compared with those from control animals (0 mg/kg STZ) (Table 1). In contrast, additional pancreatectomized pigs and pigs treated with 150 mg/kg STZ showed a significantly higher area under the curve for IVGTT curves, with a slower decreasing glucose phase: 1,833.6±858.9 mg/dl/min (pancreatectomized, 150 mg/kg STZ pigs) versus 2,086.7±1,011.4 mg/dl/min (30–100 mg/kg STZ-treated pigs) and 2,062.6±564.3 mg/dl/min (control animals) [data previously reported by Dufrane et al. (13)].
Blood Glucose Levels (mg/dl) During IVGTT [Before Glucose Injection (Baseline), 1 (Peak), and 90 min After Glucose Injection]

IVGTT, intravenous glucose tolerance test; STZ, streptozotocin.
Pancreas Structure Prior to Isolation
Insulin and Glucagon Content.
Control pig and human pancreatic insulin content were not significantly different, and a correlation was found between pancreatic insulin content and STZ dose (r2 = 0.77, p < 0.05) (Fig. 1A, Table 2). By linear regression, a theoretical 30% reduction of the insulin content would be obtained with a STZ dose between 30 and 50 mg/kg as revealed by the histomorphometry analysis. STZ doses of 75 and 100 mg/kg induced a significant reduction of the pancreatic insulin content compared with control and human pancreases (p < 0.05) (Fig. 1A) and were inadequate for tissue remodeling.
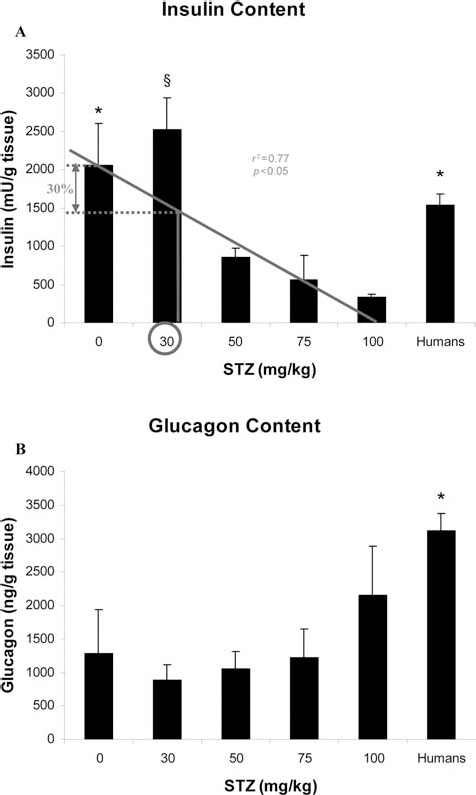
Pancreatic hormonal content. (A) A correlation was found between the dose of STZ and the pancreatic insulin content (r2 = 0.77; p < 0.05). By extrapolation, a theoretical reduction of 30% in the pancreatic insulin content would be obtained with a dose of 30 mg/kg STZ. Native pig and human pancreas demonstrated a higher insulin content than pigs treated with 75 and 100 mg/kg STZ (*p < 0.05). A dose of 30 mg/kg STZ showed a higher insulin content than treatment with 100 mg/kg (§p < 0.05). (B) Pancreatic glucagon content in function of the dose of STZ. Note a higher glucagon content in human pancreas than in native islets as well as in the pancreas of pigs treated with 30, 50 and 75 mg/kg STZ (*p < 0.05). STZ, streptozotocin.
Recapitulative Table: Human Islets Versus Native Pig Islets Versus Modified Islets
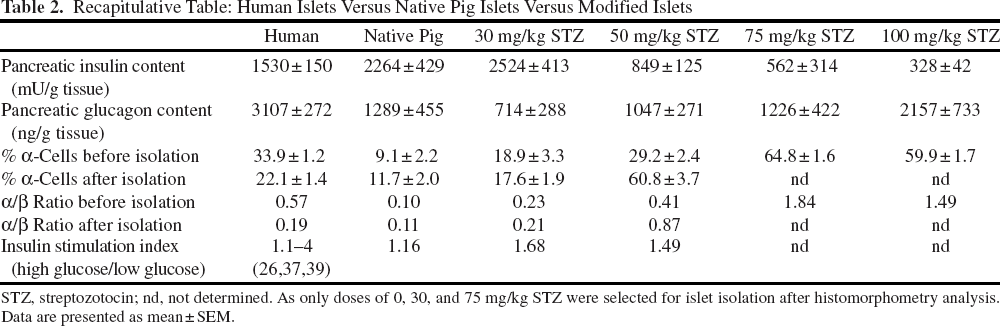
STZ, streptozotocin; nd, not determined. As only doses of 0, 30, and 75 mg/kg STZ were selected for islet isolation after histomorphometry analysis. Data are presented as mean±SEM.
The glucagon content was significantly higher in human pancreases in comparison with those from control and treated pigs (30, 50, and 75 mg/kg STZ) (p < 0.05) (Fig. 1B, Table 2) at 3 months after STZ treatment. In contrast, the pancreatic glucagon content was not significantly different in pigs treated with 100 mg/kg STZ and in humans.
Histomorphometry Analysis
Streptozotocin treatment of native pigs was performed in view to improve the proportion of α-cells per islets to obtain a “human-like” structure (+300% of α-cells). The percentage of α-cells per islets before isolation gained after treatment in comparison with native islets is summarized in Table 3A.
Percentage of α-Cells per Islet Before Isolation Gained After STZ Treatment in Comparison With Native Islets
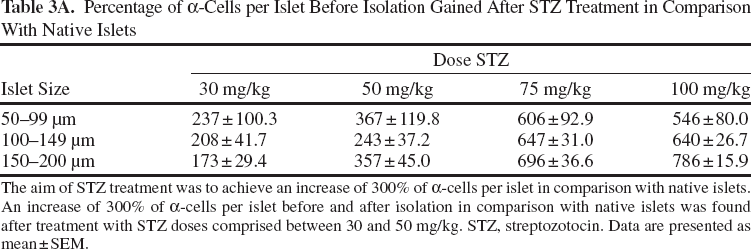
The aim of STZ treatment was to achieve an increase of 300% of α-cells per islet in comparison with native islets. An increase of 300% of α-cells per islet before and after isolation in comparison with native islets was found after treatment with STZ doses comprised between 30 and 50 mg/kg. STZ, streptozotocin. Data are presented as mean±SEM.
In human islets from untreated pancreases, the mean percentage of α-cells was 34% (Fig. 2A, Table 2). In pigs, a linear correlation (r2 = 0.88; p < 0.05) was found between the STZ dose and the α-cell proportion per islet (Fig. 2A). Following a linear regression, a 300% increase in the proportion of α-cells (similar to human islets) would theoretically be obtained with a STZ dose of 50 mg/kg (Fig. 2A).

α-Cell proportion and content before isolation. (A) The proportion of α-cells increased with the dose of STZ (r2 = 0.88; p < 0.05). Native islets presented a lower α-cell proportion than other groups (p < 0.001). Doses of 75–100 mg/kg induced a significant higher proportion of α-cells/islet than 30/50 mg/kg and human (p < 0.001). Treatment of pigs with 30–50 mg/kg STZ is the only dose allowing a similar α-cell proportion than in humans in islets of 50–150 μm. A higher proportion of α-cells in large islets (150–200 μm) was found in human in comparison to 30–50 mg/kg STZ-treated pigs (#p < 0.05). Pictures of islets from native and STZ-treated pigs versus human islets. β-Cells are stained in brown and α-cells in pink. Note the increasing proportion of α-cells after STZ treatment. (B) The number of α-cells per islet was lower in native islets in comparison to other groups (*p < 0.05). Pigs treated with doses of 30 and 50 mg/kg demonstrated a lower number of α-cells per islet than pigs treated with 75 or 100 mg/kg STZ (p < 0.01). Islets from 30–50 mg/kg STZ-treated pigs showed a lower number of α-cells per islet of 50–100/150–200 μm in comparison with human islets (§p < 0.001). STZ, streptozotocin.
Control pig pancreatic islets showed a significantly lower α-cell proportion than human islets for each islet size (p < 0.001) (Fig. 2A). However, pig islets treated with 30 or 50 mg/kg STZ had a significant increase in the α-cell proportion in comparison with control pig islets (p < 0.001), with an α-cell proportion (24%) similar to human islets (Fig. 2A). Nevertheless, in large islets (150–200 μm), a significantly lower α-cell proportion was found after treatment with 30 and 50 mg/kg STZ (Fig. 2A) in comparison with human islets (p < 0.05). Doses of 75 and 100 mg/kg STZ induced an excessive α-cell increase (p < 0.001) (for each islet size) in comparison with human islets (Fig. 2A).
Native pig (0 mg/kg STZ) pancreatic islets showed a significantly lower number of α-cells per islet than human and STZ-treated pigs for each islet size (p < 0.05) (Fig. 2B).
A significantly lower number of α-cells per islet was found for islets of 50–100 μm and 150–200 μm from pigs treated with 30 and 50 mg/kg STZ than from humans (p < 0.001) (Fig. 2B). However, the number of α-cells per islet of 100–150 μm was not significantly different in pigs treated with 30 and 50 mg/kg STZ and humans (Fig. 2B).
The number of α-cells observed in large islets of pigs treated with 75 or 100 mg/kg STZ was not significantly different than human islets (islets >100 μm) (Fig. 2B).
No change of the cellular distribution within islets (core/periphery) was observed.
No significant increase of β-cell clusters (≤3 insulinimmunoreactive cells) was found between control and treated pig pancreases.
Islet Structure After Isolation
The percentage of α-cells per islet after isolation gained after treatment in comparison with native islets is summarized in Tables 2 and 3B.
Only islets from pigs treated with 30 or 50 mg/kg STZ were investigated following pancreatic structural (histomorphometry/hormonal content) modification. Control pig and human pancreases were used as controls and objectives for tissue remodeling, respectively (Table 2).
Control Pig Versus Human Islets
After islet isolation, a significantly lower proportion of α-cells was found in control pig islets in comparison with human islets (p < 0.05) (Fig. 3A).

α-Cell proportion and content after isolation. (A) Native islets showed a lower α-cell proportion than 30–50 mg/kg STZ-treated islets and humans (p < 0.05). Pigs treated with 50 mg/kg STZ showed a significant higher α-cell proportion in isolated islets of 50–100 μm in comparison with human islets (*p < 0.01). A significant higher proportion of α-cells was found in 30–50 mg/kg STZ-treated islets of 100–150 μm in comparison with human islets (**p < 0.01). Large islets (150– 200 μm) of pigs treated with 50 mg/kg STZ demonstrated a higher α-cell proportion than other groups (#p < 0.001). (B) A lower number of α-cells was found in native islets of 50–100/150– 200 μm in comparison with human (*p < 0.05). Islets from pigs treated with 30 mg/kg STZ showed a higher number of α-cells than human islets of 50–100 μm (**p < 0.001) and native pig islets of 100–150 μm (§p < 0.05). Islets from 50 mg/kg STZ-treated pigs demonstrated a higher number of α-cells per islets than other groups (p < 0.05). STZ, streptozotocin.
Native pig islets of 50–100 and 150–200 μm also demonstrated a lower α-cell number than human islets (p < 0.05) (Fig. 3B). However, no significant difference was observed for islets of 100–150 μm.
STZ (30 mg/kg) Versus Human Islets
Pigs treated with 30 mg/kg STZ had an α-cell proportion not significantly different than humans for islets of 50–100 and 150–200 μm. However, a higher proportion of α-cells per islet (100– 150 μm) was found in pigs treated with 30 mg/kg STZ (p < 0.01) (Fig. 3A).
A significantly higher number of α-cells per islet of 50–150 μm was also found in pigs treated with 30 mg/kg in comparison with human islets (p < 0.05) (Fig. 3 B).
STZ (50 mg/kg) Versus Human Islets.
Pigs treated with 50 mg/kg STZ presented a higher α-cell proportion and number in comparison with humans for each islet size (p < 0.05) (Fig. 3A, B).
Control Versus STZ (30 and 50 mg/kg) Islets
A significantly lower α-cell proportion was found in control islets in comparison with 30 and 50 mg/kg STZ-treated pigs (p < 0.05) (Fig. 3A).
Control islets presented a lower number of α-cells per islet of 100–150 μm in comparison with islets from pigs treated with 30 mg/kg STZ (p < 0.05). A lower α-cell number was found in control islets of each size in comparison with islets isolated from pigs treated with 50 mg/ kg STZ (p < 0.05) (Fig. 3B).
After islet isolation, a significantly higher proportion of α-cells was located in the core of islets from pigs treated with 30 mg/kg STZ than from untreated pigs (p < 0.05) (data not shown).
STZ (30 vs. 50 mg/kg) Islets
Large islets (150–200 μm) from pigs treated with 50 mg/kg STZ demonstrated a higher α-cell proportion than those from pigs treated with 30 mg/kg STZ (p < 0.001) (Fig. 3A). A higher number of α-cells for each islet size was found in islets from pigs treated with 50 mg/kg STZ in comparison with those from pigs treated with 30 mg/kg STZ (p < 0.05) (Fig. 3B).
Pigs treated with 30 mg/kg showed a higher proportion of α-cells in the periphery of islets of 100–150 μm compared with pigs treated with 50 mg/kg STZ (p < 0.05) (data not shown).
In Vitro Islet Functionality
Islets from pigs treated with 30 and 50 mg/kg STZ were isolated, and their functions were tested in vitro by incubation with different glucose concentrations and compared to control islets (0 mg/kg).
Significantly higher glucagon secretion was observed after G5 and G15 stimulations of islets from pigs treated with 50 mg/kg STZ in comparison to 0 mg/kg (2.5 and 3.0 times higher, respectively) and 30 mg/kg (2.0 and 1.6 times higher, respectively) (p < 0.01) (Fig. 4).

Islet glucagon release and content after glucose stimulation. Islets from pigs treated with 50 mg/kg STZ demonstrated a higher glucagon release after G5 and G15 stimulation than native and 30 mg/kg treated islets (*p < 0.01). A higher glucagon content was also found after G5 and G15 stimulation in 50 mg/kg treated islets than native and 30 mg (§p < 0.05). STZ, streptozotocin.
The glucagon content was 4.4 times higher in islets from pigs treated with 30 mg/kg in comparison to control islets at G5 and G15 stimulations. A significantly higher glucagon content was extracted from 50 mg/kg treated islets than control islets at G5 (7.9 times higher; p < 0.05) and at G15 stimulation (10.4 times higher; p < 0.05) (Fig. 4).
Percentage of α-Cells per Islet After Isolation Gained After STZ Treatment in Comparison With Native Islets

The aim of STZ treatment was to achieve an increase of 300% of α-cells per islet in comparison with native islets. An increase of 300% of α-cells per islet before and after isolation in comparison with native islets was found after treatment with STZ doses comprised between 30 and 50 mg/kg. STZ, streptozotocin. Data are presented as mean±SEM.
No effect of forskolin on glucagon secretion or content was observed in both control and treated pig islets (data not shown).
Islets from pigs treated with 30 mg/kg STZ secreted higher levels of insulin after G5 and G15 stimulations than islets from control pigs (2.1 and 2.1 times, respectively) and 50 mg/kg treated islets (1.6 and 1.5 times, respectively) (p < 0.005) (Fig. 5).

Islet insulin release and content after glucose stimulation. Islets from pigs treated with 30 mg/kg STZ demonstrated a higher insulin secretion after G5 and G15 stimulations (**p < 0.005). Islets from pigs treated with 30 mg/kg STZ also demonstrated higher insulin content after G5 stimulation (*p < 0.05). STZ, streptozotocin.
A STZ dose of 50 mg/kg increased the islet insulin secretion 1.3 and 1.4 times in comparison with the control at G5 and G15 stimulations, respectively (Fig. 5).
Islets from pigs treated with 30 mg/kg STZ contained 3.3 times more insulin than control islets at G5 stimulation (p < 0.05). These islets also presented an increase in the insulin content of 2.5 times in comparison with control islets at G15 stimulation (Fig. 5).
Pigs treated with 50 mg/kg STZ had islets with an insulin content 1.5 times higher than the control at G5 stimulation and 1.4 times higher after G15 stimulation (Fig. 5).
Forskolin had no effect on insulin content in control and treated pig islets. However, a twofold to fourfold increase in insulin secretion was found after G15 + forskolin stimulation in comparison with G5 and G15 in control and treated islets (p < 0.05) (data not shown).
Discussion
Glucose homeostasis requires complex control of insulin secretion from β-cells. A major problem related to the use of porcine islets in pig-to-human xenotransplantation is their poor insulin release in response to glucose challenge. An increased proportion of α-cells within pig islets could (i) increase the glucagon secretion, (ii) increase cAMP concentration inside β-cells, and thereby (iii) induce insulin secretion in response to glucose stimulation.
To investigate this hypothesis, a pig model compatible with preclinical and clinical applications is needed.
To obtain efficacious pancreas remodeling, a young pig model was chosen due to the endocrine pancreatic immaturity. Porcine islets are still maturing within islets between 5 and 24 weeks after birth with a difference in the frequency and destruction of endocrine cells, therefore presenting α- and β-cell plasticity during this period (21). Additionally, there is the possibility of endocrine cell mass growth and remodeling due to the high number of immature endocrine cells (both a and b). Moreover, islets from young pigs may have better cell survival and cellular renewal in comparison with those from adult pigs. These young pigs also show a low sensitivity to STZ and demonstrate no abnormal β-cell function after low-dose STZ treatment. In our study, the reduction of the β-cell proportion by 30% after treatment with 30 and 50 mg/kg STZ did not affect glucose uptake as revealed by a normal IVGTT, in contrast with pigs treated with 150 mg/kg STZ or pigs that underwent pancreatectomy. No sign of diabetes was apparent after destruction of 30% of the β-cell mass, whereas glucose intolerance and diabetes occurred with β-cell reductions of 50% and 90%, respectively. In comparison with rats and primates, which become diabetic after injection of 50 mg/kg STZ, a dose of 150 mg/kg is needed to induce irreversible hyperglycemia in pigs. Pigs are protected against STZ due to a low expression of the glucose transporter Glut-2 on the β-cell membrane (13) through which STZ enters in β-cells, which explains why low doses of STZ have no impact on glucose metabolism in pigs. Moreover, young pigs also possess a lower sensitivity to STZ due to their higher metabolism compared with adult pigs. The use of young pigs also offers an advantage in term of sterile breeding and pancreas procurement (with controlled warm and short cold ischemia times) in contrast to adult pigs.
The selected STZ dose must reprogram pig pancreatic tissue to obtain a human-like α-cell proportion. It was previously demonstrated that after STZ treatment, the glucagon content increased in hamsters (14) and the number of α-cells increased in monkeys (11). In the latter study, an ~89% increase in glucagon cell mass after long-term diabetes (>53 days) was observed in treated primates. This was associated with an exponential increase in glucagon content in the pancreas. In our current study, a linear relationship was obtained between the dose and the proportion of pig pancreatic α- and β-cells. When the dose of STZ was increased, a significant rise in the number of α-cells per islet occurred, which was correlated with an increase in the glucagon content in the pancreas. Although a lower proportion of α-cells per islet was found in pig treated with 100 mg/kg STZ in comparison to 75 mg/kg, a higher pancreatic glucagon content was found for pigs treated with doses of 100 mg/kg STZ. This can be explained by a higher cellular glucagon content in comparison to 75 mg/kg, as described previously in a primate model (11). The increases in α-cell proportion and α-cells per islet were confirmed after pancreas digestion and islet isolation. These results indicated that STZ doses between 30 and 50 mg/kg allow a reproducible remodeling of porcine islets with a ~30% decrease of β-cells and a ~300% increase in α-cells, leading to a “human-like structure” of 60% β-cells and 30% α-cells in pig islets. In addition, no sign of β-cell regeneration (from duct or exocrine tissue) was found, with no significant difference of β-cell clusters between control and treated pig pancreases. However, the origin of the growing number of α-cells still needs to be determined. α-Cells could be derived from pancreatic stem or progenitor cells that reside within pancreatic ducts and can differentiate and migrate to develop new islets during both organogenesis and regeneration (27,33) or from progenitor cells located in islets (32). Following STZ-induced damage to β-cells, the progenitor cells could be reprogrammed towards the α-cell lineage, in contrast to the α- and β-cell transdifferentiation described in the literature (5,38). Finally, as the general shape of pig islets was similar prior to and after STZ treatment, destruction of β-cells could allow α-cells to replicate and take the space initially occupied by β-cells. Thus, the remodeling of pig islets structure into “human-like” islets should induce physiological consequences by improving glucagon and insulin release. In addition, modification of the α-/β-cell ratio after STZ treatment did not affect the cell distribution within islets. The cytoarchitecture of pancreatic pig islets should therefore not have an impact on islets function.
Although pig islet remodeling by STZ is confirmed by histology and hormonal content, the improvement of pig β-cell function must be confirmed by an increased glucagon concentration. After 30 and 50 mg/kg STZ, increased secretion of glucagon (1.6–3.0 times higher) was obtained from isolated pig islets. The effect of glucagon on the improvement of insulin release was previously studied in vitro by Bertuzzi et al. (1) and Davalli et al. (7) on adult pig islets. An increased stimulation index of 30% was observed after acute exposure to 1 or 10 μM glucagon (7). Moreover, islets cultured in the presence of 10 μM glucagon demonstrated a basal insulin release twofold higher in comparison with a control (7). In our study, islet remodeling with low doses of STZ induced increased glucagon content and secretion, which resulted in a glucagon concentration in isolated islets in culture of about 10−4–10−5 μM. This secretion acted as a cAMP triggering agent and induced an increase of insulin release (1.29–2.13 times higher). The secretion of insulin also increased after a G15 stimulation in comparison with G5, with a G15/G5 ratio (stimulation index) at 1.16 for control islets compared with 1.68 and 1.49 for STZ at 30 and 50 mg/kg STZ-treated islets, respectively. This represents a stimulation index increased by 45% and 28% for 30 and 50 mg/kg, respectively, in comparison to control islets.
In conclusion, a dose of 30 and 50 mg/kg STZ can modify the structure of pig islets in vivo, inducing an increase in the proportion and the number of α-cells per islet as well as a decrease in the proportion and the number of β-cells per islet to obtain an α-/β-cell ratio similar to that found in human islets. This remodeling induces an increase in hormonal content and release, thus improving the function of porcine islets.
We are currently investigating the potential of modified “human-like” pig islets in a primate model.
Footnotes
Acknowledgments
We are grateful to P. Y. Adnet and E. Legrand for technical assistance, M. Nenquin for providing media, and P. Segers for editorial help. This work was supported by a European grant titled Xenome (UE LSHB-CT-2006-037377) and by the national fund “Fonds national de la recherche scientifique” (FRS-FNRS). Sophie Vériter is an aspirant (doctoral candidate) of FRS-FNRS. The authors declare no conflict of interest.
