Abstract
Lung hypoplasia can be prevented in vitro by retinoic acid (RA). Recent evidence suggests that amniotic fluid stem (AFS) cells may integrate injured lungs and influence their recovery. We tested the hypothesis that AFS cells might improve lung growth and motility by paracrine mechanisms. Pregnant rats received either nitrofen or vehicle on E9.5. In vitro E13 embryonic lungs were cultured in the presence of culture medium alone or with RA, basophils, or AFS cells. In vivo green fluorescent protein-expressing (GFP+) rat AFS cells were transplanted in nitrofen-exposed rats on E10.5. E13 lung explants were cultured before analysis. The surface, the number of terminal buds, and the frequency of bronchial contractions were assessed. Protein gene product 9.5 (PGP 9.5) and α-actin protein levels were measured. The lung explants transplanted with AFS cells were stained for α-actin, PGP 9.5, and TTF-1. The levels of FGF-10, VEGFα, and TGF-β1 secreted by the AFS cells in the culture medium were measured. Comparison between groups was made by ANOVA. In vitro, the surface, the number of terminal buds, and the bronchial peristalsis were increased in nitrofen + AFS cell explants in comparison with nitrofen-exposed lungs. While nitrofen + RA lungs were similar to nitrofen + AFS ones, basophils did not normalize these measurements. PGP 9.5 protein was decreased in nitrofen lungs, but after adding AFS cells, the value was similar to controls. No differences were found in the expression of α-actin. In vivo, the surface, number of terminal buds, and peristalsis were similar to control after injection of AFS cells in nitrofenexposed rats. Colocalization with TTF-1-positive cells was found. The levels of FGF-10 and VEGFα were increased in nitrofen + AFS cell explants, while the levels of TGF-β1 were similar to controls. Lung growth, bronchial motility, and innervation were decreased in nitrofen explants and rescued by AFS cells both in vitro and in vivo, similarly to that observed before with RA. The AFS cell beneficial effect was probably related to paracrine action of growth factor secretion.
Keywords
Introduction
Severe pulmonary hypoplasia contributes significantly to the mortality and morbidity in newborns with congenital diaphragmatic hernia (CDH). Despite the improvements in neonatal resuscitation and intensive care, the mortality rate of CDH is still high, and most of the newer treatment modalities have replaced mortality for a higher morbidity. Long-term pulmonary sequelae are frequently described in survivors of CDH and depend on the severity of lung hypoplasia and the degree of lung injury (16,21,30,33,40,42,43).
Since the first description of nitrofen-induced diaphragmatic hernia in rodents by Iritani (17), this experimental model has been extensively studied and has become a widely accepted model that closely replicates many features of the human condition (15,22). Different treatment modalities have been tried in the experimental model of CDH in an attempt to modulate prenatally the natural course of the malformation. Among those, retinoic acid (RA) has been demonstrated to prevent pulmonary hypoplasia in vitro, to stimulate alveologenesis, and to accelerate alveolar cell proliferation in hypoplastic lungs (25–27,35). However, limitations on the clinical use of RA, which is known to be teratogenic (8,41), provide an opportunity to explore alternative procedures for the rescue of this devastating disease.
Stem cell-based therapies are promising new treatment approaches for a large number of diseases. Recently, it has been reported that amniotic fluid-derived stem (AFS) cells can be isolated from human and rodent amniotic fluid. AFS cells are broadly multipotent cells, able to differentiate into lineages belonging to all three embryonic germ layers (10). Moreover, they can also engraft in irradiated bone marrow and give rise to all hematopoietic lineages (13). Finally, they can functionally contribute to the regeneration of various tissues and organs when transplanted in models of diseases. Remarkable results have been obtained in injured kidneys, heart, and lungs (3,31,47). The latter have been explored both in models of diseases and during development, and AFS cells have shown the potential not only to engraft and differentiate in specialized pneumocytes but also to contribute and supplement endogenous lung repair mechanisms (7).
The aim of this study was to test the hypothesis that AFS cells could rescue lung growth and motility both in vitro and in vivo and to look further inside the mechanism of action of AFS cells.
Materials and Methods
Adult Sprague-Dawley female rats (Harlan Laboratories, Barcelona, Spain) were mated overnight. The finding of spermatozoids in the vaginal smear was considered as a mark of gestational day 0 (E0). Pregnant rats were then randomly divided into two groups. The animals in the experimental group received intragastrically 100 mg of nitrofen (Sigma-Aldrich, Madrid, Spain) dissolved in olive oil (Sigma-Aldrich) on E9.5 to induce fetal lung hypoplasia, whereas those in the control group received only vehicle.
In Vitro
On E13, the rats were sedated with isoflurane (Sigma-Aldrich) and killed by intracardiac injection of potassium chloride (Sigma-Aldrich). The embryos were recovered (Fig. 1), and the lung primordia were dissected free in Hanks buffer salt solution (HBSS; Gibco, Barcelona, Spain) under a dissecting microscope.
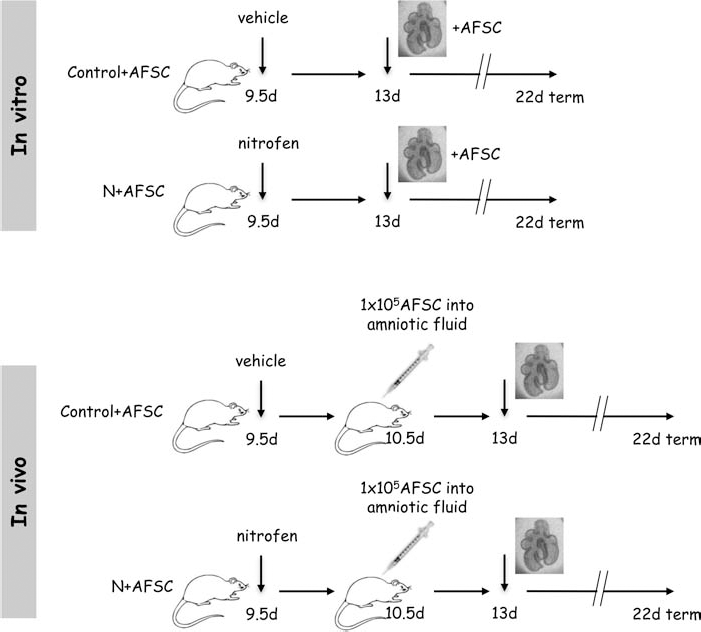
Experimental schedule. In the in vitro model, pregnant rats received intragastrically either nitrofen (N) or vehicle on E9.5. On E13, the embryos were recovered, and the lung primordia harvested. In the in vivo model, pregnant rats received intragastrically either nitrofen or vehicle on E9.5. A laparotomy was performed on E10.5, and 1×105–1×106 green fluorescent protein-positive rat amniotic fluid-derived stem cells (GFP+ rat AFS cells; AFSCs) were injected into the amniotic fluid of both groups. The embryos were recovered on E13, and the lung primordia were dissected free.
In Vivo
On E10.5, a laparotomy was performed, and after exposure of the uterus, 1×105–1×106 green fluorescent protein-expressing (GFP+) rat AFS cells, previously trypsinized (Gibco), were injected into the amniotic fluid of nitrofen-exposed and control rats. On E13, as described above, the rats were sacrificed, the embryos were recovered, and the lung primordia were freed carefully from the heart and esophagus (Fig. 1). The Animal Care Committee approved all the animal experiments (License Number: 32-06).
Collection and Characterization of the Rat AFS Cells
Samples of rat amniotic fluid were collected from transgenic GFP-positive pregnant Sprague-Dawley rats (Charles River Laboratories, Margate, UK) on E16, and GFP-rAFS cells were isolated and cultured, as previously described (4,10,13).
GFP-rAFS cell characterization was carried out with the following primary antibodies: anti-octamer binding transcription factor 3/4 (Oct 3/4; mouse monoclonal, Santa Cruz Biotechnology, Santa Cruz CA, USA; 1:50), anti-cluster of differentiation 45 (CD45; rabbit polyclonal, Abcam, London, UK; 1:100), anti-CD34 (rabbit polyclonal, Abcam; 1:150), anti-pan-cytokeratin (mouse monoclonal, Abcam; 1:300), anti-vimentin (mouse monoclonal, Abcam; 1:1,000), and anti-α smooth muscle actin (αSMA; mouse monoclonal, Abcam; 1:100). Cells were seeded on a cover glass (Gibco) at 2,000 cells/cm2 and were rinsed in phosphate-buffered saline (PBS; Gibco) and fixed in 4% paraformaldehyde (Gibco) for 20 min. Cells were permeabilized with a 0.5% Triton X-100 (Gibco) solution, rinsed in PBS with 3% bovine serum albumin (BSA; Gibco) to block nonspecific binding, and incubated with primary antibodies for 1 h at room temperature. After being washed with PBS, the samples were incubated at room temperature for 1 h with fluorochrome-labeled anti-mouse and anti-rabbit secondary antibodies (Alexa Fluor 594; 1:150; Molecular Probes, Invitrogen, London, UK). After further washing with PBS, the specimens were then mounted in fluorescent medium with 4′,6-diamidino-2-phenylindole (DAPI; 1.5 μg/ml; Sigma-Aldrich) on a polylysine slide (ThermoScientific, London, UK) and observed under an epifluorescence microscope (ZEISS Axiophot, Cambridge, UK). Cell counts were blindly evaluated for each stained slide over three low-magnification fields (10×) and reported in the chart as percentage.
Organ Culture
In vitro
The lung explants were divided into eight study groups: control (n = 8), nitrofen (n = 12), control + RA (n = 8), nitrofen + RA (n = 12), control + basophils (n = 8), nitrofen + basophils (n = 12), control + AFS cells (n = 8), and nitrofen + AFS cells (n = 12).
Control and nitrofen-exposed lung explants were cultured on translucent membrane inserts (polyester membrane transwell-clear inserts, pore size: 3 μm; Corning, Madrid, Spain) for 72 h at 37°C in 5% CO2. Culture medium [Dulbecco's modified Eagle's medium (D-MEM)/F12 (Gibco, Barcelona, Spain) with 10% fetal bovine serum (FBS; Gibco), 100 IU/ml penicillin, and 100 μg/ml streptomycin (Gibco)] was added until the lungs were lying on the air–medium surface and changed every 24 h.
When lung explants were cocultured in the presence of RA, exogenous RA (all-trans-retinoic acid, 1 μM; Sigma, Madrid, Spain) was diluted in ethanol and added daily to the culture medium at a final concentration of 0.4% (25).
When basophils needed to be added, the day before recovering the embryos, rat RBL 2H3 cells (a Wistar rat basophilic leukemia cell line; Harlan Laboratories) were trypsinized, and 5,000 cells/μm2 were cultured on culture dishes (12-well culture dish; Corning) in culture medium.
When lung explants were cocultured in the presence of AFS cells, the day before harvesting the embryos, rat AFS cells were trypsinized, and 5,000 cells/cm2 were cultured on 12-well culture dishes in minimum essential medium with a modification (α-MEM) containing 15% embryonic stem cell-fetal bovine serum (ES-FBS), 1% glutamine, 1% penicillin/streptomycin (Gibco) supplemented with 18% Chang B and 2% Chang C (Irvine Scientific, Santa Ana, CA, USA) at 37°C with 5% CO2 atmosphere (10). Before placing the inserts on the culture dishes, the medium was removed and replaced with culture medium.
In vivo
Control (n = 8) and nitrofen-exposed (n = 12) lung explants were cultured on translucent membrane inserts (polyester membrane transwell-clear inserts, Corning) for 72 h at 37°C in 5% CO2. Culture medium was added until the lungs were lying on the air–medium surface and changed every 24 h.
Lung Morphometry
Cultured lungs were photographed daily on an inverted microscope. The digitalized images of the last culture day were analyzed with the assistance of image processing software (Image Pro-Plus, version 5.0, Media Cybernetics, Washington, DC, USA). The outline of the lung explant was contoured on the PC screen with the cursor, and the resulting surface was integrated by the software. In the same images, the terminal lung buds were counted on each explant.
Motility
At 72 h of culture, airway peristaltic contractions were recorded for 10 min under an inverted microscope with LAS-AF software (Leica TCS SP5, Barcelona, Spain), and their frequency was expressed in number per minute.
Immunoblotting
To quantify the levels of protein gene product 9.5 (PGP 9.5) and α-actin proteins, five lungs from control, nitrofen, nitrofen + RA, and nitrofen + AFS cell groups were pooled and homogenized in cell disruption buffer (Protein Isolation System Paris, Ambion, Madrid, Spain). The protein content was assessed using a protein assay kit (BCA Protein Assay Kit, Pierce, Madrid, Spain). Western blotting was performed with 18% sodium dodecyl sulfate (SDS)-polyacrylamide gel (Bio-Rad, Madrid, Spain). Anti-PGP 9.5 (1:6,000; rabbit polyclonal PGP 9.5; DakoCytomation, Glostrup, Denmark) and α-actin (1:6,000; mouse monoclonal α-actin, Sigma) antibodies were used, and the values were normalized to polyclonal antibody anti-Cu/Zn cytosolic superoxide dismutase (SOD; Abnova, Madrid, Spain).
Immunohistochemistry
After detaching the in vivo group nitrofen-exposed cultured lungs from the membranes, they were fixed in 4% paraformaldehyde overnight and subsequently rinsed in PBS. Nonspecific binding was blocked by washing them in PBS with 1% BSA before incubating them with the primary antibodies overnight. To evaluate the AFS cell differentiation toward the neuronal, muscle, and epithelial lineages, three stainings were performed.
In the first, a mouse monoclonal antibody against protein gene product 9.5 (PGP 9.5; 1:500; Acris antibodies, Barcelona, Spain) was used to stain the neural tissue. In the second, smooth muscle was identified with a mouse monoclonal anti-α-actin fluorochrome-labeled antibody [anti-actin, α-smooth muscle-cyanine 3 (Cy3) antibody; 1:1,000: Sigma-Aldrich, St. Louis, MO, USA]. In the third, the epithelial cells were stained with monoclonal mouse anti-thyroid transcription factor (TTF-1; DakoCytomation).
In all experiments, the anti-GFP rabbit IgG fraction (Molecular Probes) unveiled the injected AFS cells. After being washed with PBS, the samples were incubated at room temperature for 1 h with fluorochrome-labeled anti-rabbit and anti-mouse secondary antibodies (Alexa Fluor 488 and 568; 1:500; Molecular Probes). After further washing with PBS, the specimens were then mounted in glycerol on a glass slide (both Gibco). As a control, the primary antibody was omitted, with no staining above background as the expected result.
Confocal Microscopy
Fluorescent images of the nerves, smooth muscle, epithelial cells, and the injected AFS cells in the double-stained whole mount preparations were obtained using a confocal laser scanning microscope with LAS-AF software (Leica TCS SP5). The fluorescent markers were detected by a krypton/argon laser with excitation wavelengths of 488 nm for Alexa 488, 568 nm for Alexa 568, and 561 nm for Cy3. The whole mounts were optically sectioned by scanning at increasing depths of focus in steps of 5 μm. After double staining, the green and red images were captured separately, colorized, and merged to show a composite image. The specific staining of neural structures by anti-PGP 9.5 antibody, of smooth muscle by anti-a-actin antibody, and of epithelial cells by anti-TTF-1 antibody and the percentage of colocalization were assessed in each lung explant.
Total RNA Extraction and Retrotranscription
Trypsinized AFS cells harvested from the culture setting of 8 control lungs cocultured with AFS cells and 12 nitrofen-exposed explants cocultured with AFS cells were suspended in sterile RNase-free 1.5-ml microcentrifuge tubes (Eppendorf, Stevenage, UK) and centrifuged (Centrifuge 5415D, Eppendorf) for 1 min to pellet the cells. After pouring off the supernatant, 1 ml of TRIzol reagent (Life Technologies, Madrid, Spain) was added to the tubes. Cells were lysed by pipetting. The homogenate was then centrifuged at 12, 000×g for 10 min at 4°C. The homogenate was then transferred in a sterile microcentrifuge tube. The samples were incubated for 5 min at room temperature. Chloroform (0.2 ml; Sigma-Aldrich, London, UK) was added to each tube. Samples were shaken vigorously by hand for 15 s, incubated at room temperature for 5 min, then centrifuged for 15 min at 12, 000×g at 4°C. The upper aqueous phase was transferred to a fresh tube. Isopropyl alcohol (0.5 ml; Sigma-Aldrich, UK) was added to precipitate RNA. Samples were incubated at room temperature for 5 min and centrifuged at 12, 000×g for 10 min at 4°C. Supernatant was discarded, and pellet was washed with 1 ml of 75% ethanol. The sample was mixed by vortexing and centrifuged at 7, 500×g for 5 min at 4°C. Supernatant was then removed, and pellet was left to air-dry for 5-10 min. The pellet was dissolved in RNase-free water or 0.5% SDS (Sigma-Aldrich, UK) by passing the solution through a pipette tip and incubating for 10 min at 55-60°C. RNA concentration and purity of the sample were assessed by pipetting the diluted RNA sample in to a clean cuvette, and absorbance was read at 260 and 280 nm (PR4100; Bio-Rad, Hemel Hempstead, UK). In order to determine the purity of the RNA sample, the A260/A280 ratio was calculated. We analyzed only samples with ratios ranging between 1.6 and 2, which represent a good RNA extraction. The first-strand cDNA was synthesized using 1 μg of total RNA in a total volume of 8 μl of diethylpyrocarbonate (DEPC; Bio-Rad, UK) water. After treatment with 1 μl of DNase, the mix was heated at 65°C for 10 min, and 1 μl of oligo(dT) plus 1 μl of dNTPs mix (10 mM) were added. The solution was then heated for 5 min at 65°C, then cooled at 4°C, and added with 4 μl of 5× First Strand Buffer, 2 μl of 0.1 M DTT, and 1 μl of RNaseOUT; after a preheating at 42°C for 2 min, 1 μl of Taq polymerase (Invitrogen) was added, and the solution was left for 50 min at 42°C and then for 15 min at 70°C. Lastly, 1 μl of RNase H was added, and the solution was incubated for 20 min at 37°C. The generated cDNA was diluted 1:5 in DEPC H2O and stored at −20°C.
Primer Design and Real-Time PCR
Real-time PCR oligonucleotide primers (Table 1) were manually designed for each of the genes to ensure maximal efficiency and sensitivity, according to the following parameters: primer length, melting temperature, and avoidance of the formation of self- and hetero- dimers, hairpins, and self-complementarity. These properties were verified using two different internet-based interfaces: Primer-3 (http://primer3.wi.mit.edu/) and Oligonucleotide Properties Calculator (http://www.basic.northwestern.edu/biotools/OligoCalc.html). When possible, “GC clamps” were placed at the 3′ end of each primer to minimize breathing between primer and template DNA, which can promote mispriming and decrease efficiency. Primers were designed such that amplicon sizes ranged between 50 and 250 bp. Melting curve analysis was always performed at the end of each PCR assay to control the specificity; definite reactions should result in a single melting peak corresponding to the PCR product being amplified. Real-time PCR was performed using the default thermocycler program for all genes: 3 min of preincubation at 94°C followed by 50 cycles for 30 s at 94°C, 30 s at 60°C, and 45 s at 72°C. Individual real-time PCR reactions were carried out in 30-μl volumes in a 96-well plate (Applied Biosystems, London, UK) containing 8 μl of DEPC water, 1 μl of sense and antisense primers (10 μM), and 15 μl of SYBR Green with ROX (Qiagen, Crawley, UK) plus 5 μl of sample. At the end of each reaction, cycle threshold (Ct) was manually set up at the level that reflected the best kinetic PCR parameters, and melting curves were acquired and analyzed.
Primer Templates

rβ2M, rat β2-microglobulin; rFGF-10, rat fibroblast growth factor-10; rVEGFα, rat vascular endothelial growth factor α; rTGF-β1, rat transforming growth factor-β1.
We used a relative quantification method to measure gene expression by relating the PCR signal of the target transcript in a treatment group to control AFS cells of comparable passage number cultured for 72 h at 37°C in 5% CO2 in culture medium. In this work, the 2–ΔΔCt method of relative quantification was adapted to estimate copy numbers in our target genes. The ΔΔCt calculation for the relative quantification of target was used as follows: ΔΔCt = (Ct, target gene - Ct, 2-microglobulin)x - (Ct, target gene -Ct, 2-microglobulin)y, where x = unknown sample and y = AFS cell control. After validation of the method, results for each sample were expressed in N-fold changes in x target gene copies, normalized to 2-microglobulin relative to the copy number of the target gene in AFS cell control, according to the following equation: amount of target = 2- ΔΔCt. A minimum of two experiments were carried out for each gene and sample. At each experiment, each individual sample was run in duplicate wells, and the Ct of each well was recorded at the end of the reaction. The average and standard deviation (SD) of the three Cts were calculated, and results for each sample were expressed as the N-fold copy number of a given gene relative to AFS cell control as expressed by calculating the geometric mean between the two experiments.
Statistical Methods
The results were expressed as percentages or as means ± SD, and the groups were compared by ANOVA tests with a threshold of significance at p < 0.05. Tukey's range test was used as post hoc test.
Results
Thirty adult Sprague-Dawley pregnant rats were randomized to receive nitrofen or vehicle at E9.5. All animals survived the procedure and were sacrificed at E13. Fetuses were collected, and a total of 80 embryonic lungs were isolated under a dissecting microscope and cultured under defined conditions (see Materials and Methods) (Fig. 1). An additional culture set was dedicated to observe 20 lungs derived from control and nitrofenexposed fetuses, which received, at E10.5, an in utero transplantation of AFS cells (Fig. 1). This procedure was, however, associated with a high mortality, and only 15% of embryos survived. We continued the injections of AFS cells in utero until we obtained 20 lungs (12 nitrofen-exposed lungs and 8 control lungs) to study. Similarly to the first group, animals were sacrificed at E13, and the harvested lungs underwent in vitro culture for 72 h.
GFP-rAFS cells expressed the marker Oct 3/4. CD34, CD45, and pan-cytokeratin were absent, while the mesenchymal cell marker vimentin and the smooth muscle marker αSMA were expressed in the cells (Fig. 2).

Immunophenotyping of GFP-rAFS cells. More than 90% of the GFP-rAFS cells expressed vimentin (D), while octamer binding transcription factor 3/4 (Oct 3/4) (A) and a smooth muscle actin (αSMA) (F) were expressed to a lesser extent. Cluster of differentiation 45 (CD45) (B), CD34 (C), and pan-cytokeratin (E) were absent.
Embryonic lungs appeared dramatically different among the groups. In particular, as described before (25), the embryonic lungs of fetuses that were exposed to nitrofen showed a marked hypoplasia, which appeared to be not influenced by basophil coculture (Fig. 3). On the contrary, it appears that embryonic lungs that were exposed to nitrofen but cultured in the presence of AFS cells or RA did not differ from the ones derived from normal control animals (Fig. 3A). Remarkably, this was also the case for the nitrofen-exposed lungs transplanted in utero with AFS at E10.5 and harvested at E13 (Fig. 3A).

Images, growth parameters, and peristalsis of lung explants. (A) Photographs of embryonic lung explants taken at the beginning of the culture, at 24 h, 48 h, and 72 h. Nitrofen-exposed embryonic lungs were markedly hypoplastic (second column) in comparison with controls, and they were not influenced by their coculture with basophils (BAS; fifth column). In contrast, AFSCs rescued growth of nitrofen-exposed lung explants both when cocultured with lungs (third column) and when injected in the amniotic fluid (sixth column). A similar effect was obtained by adding retinoic acid (RA) to the medium (fourth column). Scale bar: 0.5 mm. Growth parameters (B, C) and peristalsis (D) analyzed in lung explants after 72 h of culture. The lung surface (B), the number of terminal buds (C), and the number of contractions per minute (D) were decreased in nitrofen-exposed lungs and restored to control values in the presence of RA or AFS cells. This was also the case of the nitrofen-exposed lungs transplanted with AFS at E10.5. #p < 0.05 versus control. The presence of basophils did not influence the growth or functional movement of the embryonic lungs, which remained impaired.
To confirm this quality aspect, both lung surface and number of terminal buds were blindly measured. Similarly to what has been reported before (25), both surface (nitrofen 0.3 ± 0.12 mm2 vs. control 0.56 ± 0.17 mm2, p < 0.005) (Fig. 3B) and number of terminal buds (nitrofen 41 ± 9 vs. control 63 ± 8, p < 0.005) (Fig. 3C) were significantly decreased in nitrofen-exposed lung explants in comparison with the controls, and the values did not normalize after being cocultured with basophils (surface: nitrofen + basophils 0.34 ± 0.19 mm2 vs. control 0.56 ± 0.17 mm2, p < 0.005; buds: nitrofen + basophils 36 ± 4 vs. control 63 ± 8, p < 0.005) (Fig. 3B, C). In contrast, both parameters showed normalization in these lungs cocultured in the presence of AFS cells (surface: nitrofen + AFS cells 0.50 ± 0.08 mm2 vs. control 0.56 ± 0.17 mm2, p = 0.17; buds: nitrofen + AFS cells 60 ± 11 vs. control 63 ± 8, p = 0.42) (Fig. 3B, C) or when RA (surface: nitrofen + RA 0.50 ± 0.07 mm2 vs. control 0.56 ± 0.17 mm2, p = 0.18; buds: nitrofen + RA 61 ± 6 vs. control 63 ± 8, p = 0.47) was added to the culture medium (Fig. 3B, C). The latter were confirmed also when AFS cells were transplanted in utero (surface: in vivo nitrofen + AFS cells 0.48 ± 0.08 mm2 vs. control 0.56 ± 0.17 mm2, p = 0.19; buds: in vivo nitrofen + AFS cells 56 ± 14 vs. control 63 ± 8, p = 0.17) (Fig. 3B, C).
Importantly, no changes were noted when basophils, AFS cells, or RA was added to control lungs (data not shown).
Among the various functional parameters that could be evaluated, peristalsis may play a pivotal role in the normal development of embryonic lungs (34). After video recording, samples were examined blindly and showed that rescue of normal development by AFS cells is also evident by the normalization of peristaltic movement. In fact, while bronchial peristalsis was significantly decreased in nitrofenexposed explants in comparison with controls (nitrofen 0.5 ± 0.1 contractions/min vs. control 0.9 ± 0.1 contractions/min, p < 0.005) (Fig. 3D), in nitrofen + AFS cells (0.9 ± 0.2 contractions/min vs. control 0.9 ± 0.1 contractions/min, p = 0.69) (Fig. 3D) as well as in nitrofen + RA (0.8 ± 0.2 contractions/min vs. control 0.9 ± 0.1 contractions/min, p = 0.77) (Fig. 3D) lungs, it was similar to normal controls.
Interestingly, the presence of basophils did not influence the functional movement of the embryonic lungs, which remained impaired (nitrofen + basophils 0.6 ± 0.07 contractions/min vs. control 0.9 ± 0.1 contractions/min, p < 0.005; Fig. 3D). In contrast, the transplantation in utero of AFS cells resulted in the restoration of regular peristalsis in the nitrofen-exposed lungs (Fig. 3D). Normal controls in medium did not differ from the RA- or AFS-exposed cells (data not shown).
It has been suggested that both airway smooth muscle and neural tissue contribute to normal airway differentiation and branching from the early stages of development. To see whether the growth of neural tissue and smooth muscle was normal in lung explants, PGP 9.5 and α-actin protein levels were assessed. PGP 9.5 is a pan-neuronal protein that reveals neurons and their processes from the onset of lung development. The PGP 9.5 protein signals were seen at 20–25 kDa. Similarly to what has been demonstrated before (29), the PGP 9.5 protein levels were significantly decreased in the nitrofen-exposed explants when compared to controls, and the values did not normalize in the presence of basophils (Fig. 4A). In contrast, PGP 9.5 expression was rescued to normal values when the nitrofen-exposed explants were cocultured with AFS cells (Fig. 4A). This is similar to what is obtained when RA is administered (Fig. 4A), as demonstrated before (29). α-Actin, on the other hand, did not differ among the groups (Fig. 4B).

PGP 9.5 and α-actin levels normalized to SOD. (A) Protein gene product 9.5 (PGP 9.5) protein level normalized to superoxide dismutase (SOD) in Control, N, N + RA, N + AFSC groups. The expression of PGP 9.5 was decreased in nitrofen-exposed explants (#p < 0.05 vs. control). The expression normalized after adding RA to the medium or coculturing the nitrofen-exposed lungs with AFS cells. (B) α-Actin protein expression normalized to SOD. No differences were seen in the levels of the protein in the four study groups.
When we analyzed the nitrofen-exposed cultured lungs after in utero AFS cell transplantation, confocal microscopy revealed that GFP+ AFS cells were diffusely present on the bronchial tree, but interestingly, they did not colocalize with α-actin- or PGP 9.5-positive cells (Fig. 5A, B). In contrast, 50–70% of GFP+ AFS cells colocalized with TTF-1-positive cells (Fig. 5C, D). TTF-1 is a marker for alveolar epithelial cells type II (AECs-II), and it has been found to be essential for branching and for lung surfactant protein A and B production by the AECs-II. We postulated, however, that differentiation was not essential for the therapeutic effect, since the lungs were rescued also when not exposed in direct contact to AFS cells. In this setting, the AFS cells were lying attached on the bottom of the well, and no GFP+ cells were seen floating in the medium or on the membrane close to the lung explant. To evaluate whether the beneficial effect of stem cells could be mediated by paracrine mechanisms through secretion of growth factors (9), we evaluated the levels of three growth factors involved in lung morphogenesis in the culture medium of nitrofen-exposed lungs cocultured with AFS cells and compared to those of a control. Fibroblast growth factor 10 (FGF-10), which has a pivotal role in modulating embryonic lung branching morphogenesis and cytodifferentiation, and vascular endothelial growth factor a (VEGFα), which may play an important role in the angiogenesis, were significantly higher expressed in the medium of nitrofen-exposed lung explants cocultured with AFS cells (Fig. 6A, B). In contrast, transforming growth factor β1 (TGFβ1) mRNA expression, whichi is a negative regulator of lung epithelial proliferation and differentiation, was similar to controls (Fig. 6C).

Confocal projections of nitrofen-exposed lung cultured after in utero AFS cell transplantation. GFP+ AFS cells were diffusely present on the bronchial tree, but they did not colocalize with α-actin-positive (A) or PGP 9.5-positive (B) cells. Of GFP+ AFS cells, 50–70% colocalized with the alveolar epithelial thyroid transcription factor-1 (TTF-1)-positive cells (C, D). Scale bars: 250 μm (A, C); 125 μm (B, D).
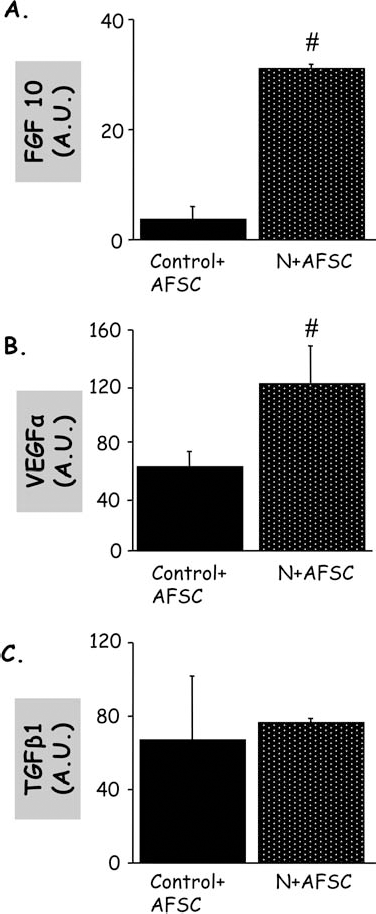
mRNA of factors secreted into media of lung explant cocultures. Fibroblast growth factor 10 (FGF-10) (A), vascular endothelial growth factor α (VEGFα) (B), and transforming growth factor-β1 (TGF-β1) (C) mRNA expression in the medium of nitrofen-exposed lung explants cocultured with AFS cells compared with a control (AU, arbitrary unit). While the levels of FGF-10 and VEGFα, which are respectively a promoter of branching morphogenesis and a promoter of vasculogenesis, had increased, the values of TGF-β1, a negative regulator of lung epithelial proliferation and differentiation, were similar to controls.
Discussion
Congenital diaphragmatic hernia is a malformation still causing mortality in newborns mainly because of severe respiratory failure secondary to pulmonary hypoplasia. This condition can be reproduced in pup rats by administering the herbicide nitrofen (2,4-dichlorophenyl-p-nitrophenyl ether) to the pregnant mothers. In this model, lung hypoplasia is usually found in all pups and diaphragmatic hernia in most of them (17). These lungs can be easily cultured in vitro, and this has been very useful to investigate lung development, which can be generally influenced by altering the composition of the medium (18,34,38).
Using this setting, we have demonstrated for the first time that AFS cells are able to rescue both in vitro and in vivo hypoplastic nitrofen-exposed embryonic lungs. This is comparable to the rescue obtained when hypoplastic nitrofen-exposed embryonic lungs were cultured in the presence of exogenous RA (25,27,28,35). The mechanism of action has not been fully elucidated, but it has been demonstrated that nitrofen inhibits retinaldehyde dehydrogenase 2 (RALDH-2), which catalyzes the final step in RA production (24). Therefore, the increase in lung branching morphogenesis in nitrofen-induced hypoplastic lungs exposed to exogenous RA is consistent with the hypothesis that the decrease of RALDH-2 activity can be partially countered by the increase of substrate (2).
In our experimental conditions, RA, which was used as positive control, restored the hypoplastic lungs to the size and the number of terminal buds of the control lungs. The frequency of peristaltic waves, which was decreased in nitrofen-exposed lungs, normalized after adding RA to the medium. While it is interesting to explore the different pathways that could be involved in the hypoplastic lungs and their rescue, RA has the limitations of not being suitable for use in the clinic (8,41), and alternative therapeutic options are needed.
It has been recently demonstrated that the pluripotent AFS cells (10) are able to integrate into developing as well as injured lung tissue, influencing its recovery from injury, and differentiate into lung epithelial lineages (7). In the present study, we found that hypoplastic nitrofenexposed lung explants cocultured with AFS cells were restored to the size and the number of terminal buds of the control lungs. Moreover, the frequency of peristaltic waves, which was decreased in nitrofen-exposed lungs, as expected (20), normalized after adding AFS cells to the medium. The improvement of airway peristalsis obtained with AFS cells as well as with RA was not related to smooth muscle hypertrophy (19,29), because the expression of smooth muscle-specific marker α-actin was not increased in AFS cells or RA treated groups. In contrast, the levels of PGP 9.5, a pan-neuronal marker that unveils ganglionic precursors since early phases of development (39), were decreased in nitrofen-exposed lungs, and they normalized after adding the AFS cells or RA to the medium.
In order to mimic a possible therapeutic approach, we subsequently tested whether AFS cells could be therapeutic in nitrofen-exposed pregnant rats. Indeed, lung growth and motility, evaluated after harvesting and culturing the embryonic lungs, could be rescued when AFS cells were injected in pregnant rats 1 day after the administration of the nitrofen. This could be observed in spite of a high rate of embryonic mortality, 15% being the overall survival. To determine whether AFS cells were able to differentiate into neural, muscle, or epithelial lineages after integration, we analyzed the colocalization of GFP+ AFSCs with PGP 9.5+ or α-actin+ or TTF-1+ cells. We found that AFS cells colocalized with TTF-1+ cells.
Despite the minimal indication of engraftment, the main therapeutic effect produced by AFS cells seems to be mediated by paracrine mechanisms elicited through trophic mediators (5,9,14,32,36,37), as evident by the coculture experiments. Among the various factors that could be driving this effect, FGF-10, VEGFα, and TGF-β1 were evaluated in this study. FGF-10 is essential for lung branching morphogenesis, having a central role in inducing the spatial coordinates for patterning the epithelial tubules. TGF-β1 opposes these effects: its signaling is thought to prevent local budding and to maintain proximal airways in an unbranched form by suppressing epithelial cell proliferation and by promoting synthesis of extracellular matrix components around airways. VEGFα is a strong promoter of angiogenesis, and its signaling is responsible for the differentiation of embryonic mesenchymal cells into endothelial cells; the interaction between the epithelium and mesenchyme contributes to lung neovascularization, which is crucial in normal lung development (6,11,23,45,46,48). Interestingly, we found that the levels of FGF-10 and VEGFα secreted by AFS cells were increased in comparison with controls when nitrofen-exposed lung explants were cocultured with AFS cells. These findings are consistent with the demonstration that the temporospatial pattern of FGF-10 expression is severely disrupted in the presence of nitrofen-induced lung hypoplasia, whereas exogenous FGF-10 can rescue hypoplastic lungs in culture (1). This is also consistent with the fact that it has recently been shown that also RA has a major role in early lung morphogenesis by inducing FGF-10 expression within the mesoderm subjacent to the site of origin of the laryngotracheal groove (12). It is possible that AFS cells are capable of producing high levels of FGF-10 in response to the damaged lungs to which they are exposed (44).
In conclusion, we have demonstrated for the first time that AFS cells can rescue nitrofen-induced hypoplastic lungs in coculture. It is possible that, as previously demonstrated in other model of disease, AFS cells might have a therapeutic role in CDH, particularly in babies with severe hypoplastic lungs. This effect could be also obtained during gestation, since AFS cells transplanted in utero in the same animal model produced a similar effect.
Footnotes
Acknowledgments
Supported in part by FIS (06/0486) and FMM Grants and by the Spanish Health Institute Carlos III (Grant N. RD08/0072: Maternal, Child Health and Development Network). P.D.C. is supported by the Great Ormond Street Hospital Charity, London UK. The authors declare no conflict of interest.
