Abstract
Corneal diseases cause severe visual impairment that necessitates corneal transplantation and frequently repetitive procedures due to graft rejection. We tested the hypothesis that exposure of donor corneas to recipient serum-derived factors during eye banking triggers a preoperative adaptation that is beneficial for postoperative tolerance. Donor corneas were incubated in a medium containing human serum (HS) obtained in each case from the prospective graft recipient in order to individually expose the donor cornea to the recipient's serum. All recipient serum-adapted corneas (RSACs) fulfilled the clinical criteria required by the national law and were transplanted successfully. The postoperative ophthalmological examination extended up to 8 years. All RSACs were tolerated by their recipients and did not cause postoperative complications and no rejection. Proteomic analysis of corneas cultivated in culture medium containing either fetal calf serum (FCS) that is routinely used for cornea banking or HS revealed different patterns of proteins. HS-cultured corneas showed a greater proteomic similarity with native human corneas than did the FCS-cultured corneas, indicating a differential nutrification of the cultured corneal tissue by HS-derived factors. The clinical results show for the first time that postoperative complications such as tissue intolerance and graft rejection might be managed if the corneal tissue is individually adapted to the recipient's serum trophic factors. This new donor tissue treatment procedure offers incontrovertible advantages and could be adapted for low-risk eyes as well as other transplantable tissues.
Introduction
Corneal transplantation is the oldest and most common form of solid tissue transplantation and was first initiated by Zirn (53). It is estimated that more than 20,000 corneas are grafted per year in the European Union (8,39,52). Corneal allotransplantation is highly successful in the short term, but less successful in the long term, with many grafts having to be repeated in recipients with high-risk conditions as a result of irreversible rejection, despite topical immunosuppression. The most frequent reasons for cornea transplantation are chemical injuries with secondary opacities, neovascularization and scarring, perforating injuries with corneal involvement, recurrent herpes keratitis, and progressive hereditary corneal dystrophies. In particular, when severe limbal stem cell destruction occurs together with persistent epithelial defects, they result in failure to repair the epithelium. Such severe damage to the corneal surface is usually an indication for new methods of surgical treatment like limbal stem cell transplantation (11,26) or corneal surface reconstruction using an amniotic membrane graft (33,46). Cultivation and expansion of corneal stem cells and their subsequent transplantation were considered a promising method for reconstructing superficial corneal defects (28,49). A suitable alternative to corneal cells has recently been found: autologous precultured oral epithelium cell transplantation (38). However, transplantation of cells or epithelium equivalents is only suitable for superficial defects (38), while deeper corneal damage requires penetrating transplantation of all corneal layers, including intact stroma and endothelium.
Various surgical approaches are available, and intense postsurgical medical care with immunosuppressive corticosteroids is provided to protect the donor tissue from immunological recognition and subsequent rejection and destruction by the recipient's allospecific immune cells, which would necessitate reoperation. However, reoperation increases the risk of new rejection, thus resulting in many cases of blindness. Advances in microsurgery (34), cornea banking (reviewed in 21), and immunosuppression have improved graft survival over the past few decades (8).
Cell-mediated immunological recognition (52) and subsequent graft rejection are the main causes of short-and long-term graft failure (9,48). Grafts placed into an avascular and noninflamed bed are termed “normal risk” keratoplasties, and the 2-year survival of these grafts is close to 90% (32,42,47). Placing a graft into a presensitized host with a history of graft rejection or a neovascularized or inflamed bed is termed a “high-risk” keratoplasty, and the 2-year survival of such grafts is less than 50% (47). Penetrating keratoplasty with donor material that has been cultured in medium containing fetal calf serum (FCS) is a frequent treatment for severe corneal diseases that are refractory to conservative treatment (1,10,13). On the other hand, FCS-free media have a lower efficacy for cell propagation and proliferation of various types of cells (44) in vitro and hence limited clinical applicability (19). Grafting of donor corneas after culturing at 37°C in FCS-containing medium over 4 weeks is a standardized procedure that offers certain advantages over the use of freshly retrieved corneas (6,12–15). One of the major advantages of cultured material is the time allocated for tissue matching, including histocompatibility antigen (HLA) typing and serology (7,20). In addition, the donor tissue can be nourished with trophic constituents provided by the culture medium and the FCS (36). Alternatives to FCS include human serum (HS), which has been proven suitable for culturing conjuctival cells (3), autologous corneal epithelium cells, and oral epithelium cells that can be regrafted in severe cases of Stevens–Johnson syndrome (37). However, postoperative graft maintenance, optical transparency, and acceptance within the recipient eye vary with both the severity of corneal changes in the recipient eye and the quality of the donor cornea during surgery. In addition, removal of bovine FCS from the culture medium is necessary because of the variability between different suppliers and the potential risk of infection (4).
In the present study, we first established individualized, recipient-specific culture conditions that support cell proliferation of the cultured donor cornea in order to avoid graft rejection. To do this, we replaced the commonly used FCS with recipient serum in the culture medium, thus also improving its safety by removing the possibility of transmission of diseases such as bovine spongiform encephalitis. For the clinical part of this study, we cultured individual donor corneas in the serum of corresponding prospective recipients for 4 weeks. After evaluating the parameters required for transplantation, the corneas were then grafted according to a state-of-the-art procedure. We also examined the mechanisms underlying biological cornea adaptation to the recipient serum and explored the proteome profiles of human corneas cultured in either FCS- or HS-containing medium and compared them with the native cornea proteome profile. The new method reported here could help to establish a standardized procedure for tissue pretreatment before its transplantation that will minimize postoperative donor tissue rejection.
Materials and Methods
Selection of Recipients and Corneas
This study was a prospective, observational study including only high-risk eyes. Before culturing the corneas in recipient serum and subsequent transplantation in those patients whose serum was used for culture, the procedure of replacing the FCS with the recipient serum was approved by the institutional review board for human studies of the University of Münster. The study was conducted according to the principles expressed in the Helsinki Declaration. A detailed explanation of the procedures was given to each patient, who then signed a statement that he/she agreed to replacement of FCS in the cornea culture with his/her own serum and that he/ she understood the procedure and agreed to receive a cornea that was cultured in a medium containing his/her own serum instead of FCS. Inclusion in either group was decided by the patient. Afterwards, 150 ml of blood was taken from each patient's brachial vein according to routine medical methods and centrifuged. The prepared fresh serum was heat-inactivated to prevent complement activation (56°C 20 min) and divided into 5-ml aliquots and frozen at −80°C. One of the aliquots was used to perform serological, microbiological, and immunological testing according to the federal transplantation law concerning the extracorporeal use of blood products. The remaining aliquots were used to supplement the culture medium. Control corneas (n = 26) were cultured in basic medium containing 5% FCS according to routine clinical practice. No further concentrations of HS or FCS were tested. Written consent to participate in this study was obtained from all patients, in accordance with the tenets of the Declaration of Helsinki for research involving human material. High-risk criteria for including patients in the study were as follows (Tables 1 and 2): appearance of corneal vessels in two or more quadrants, deep stromal vascularization, history of graft rejection in the same eye, history of recurrent herpes keratitis, history of ocular inflammation or of chemical injury.
Patients Subjected to Fetal Calf Serum (FCS) Keratoplasty

No. re-KPL, total number of keratoplasties; ECD, endothelium cell density.
Patients Subjected to Recipient Serum-Adapted Cornea (RSAC)-Keratoplasty

Human donor eyes were obtained postmortem for transplantation according to scientifically, ethically, and legally established and certified hospital procedures for eye banking that conform to guidelines of the European Eye Bank Association. After superficial disinfection of the eyecup with a povidon-iodide preparation (Betaisadona, Mundipharma, Limburg, Germany), the corneas were trephinized with a 14- or 15-mm trephine including a 1.5-mm scleral ring (model G13, Geuder, Heidelberg, Germany) and cultured in 50-ml flasks (Falcon) containing conventional modified Eagle's medium (MEM; Gibco, Germany) supplemented with 5% HS from the prospective recipient. The medium was refreshed weekly. The transparency of the corneal tissue, density of the endothelium, and sterility of the media were documented weekly until the 4th week of culture. The endothelium cell density (ECD) was determined using a semiautomatic counting device attached to a microscope (Nidek, Düsseldorf, Germany).
Transplantation was performed under standardized penetrating keratoplasty procedures using state-of-the-art clinical practice. All surgery was performed by the same experienced surgeon in the same hospital. After trephination and grafting of the central donor cornea (6.0–7.5 mm), the peripheral corneal ring was used to histologically examine the integrity and maintenance of the tissue and thus compare corneas cultured in either FCS (Gibco, Germany) or HS. Histology included periodic acid Schiff (PAS)-Alcian blue, Masson's trichrome acetate, and hematoxylin–eosin staining (all from Merck Millipore, Darmstadt, Germany). Immunohistochemical staining was performed on cryostat sections of the tissue.
Grouping of patients was done in accordance with the clinical diagnosis and the history of keratoplasty and graft rejection in the same eye. Inclusion of patients into either group was done in accordance with their desire. It is therefore apparent that patient without history of rejection preferred to be grafted with the established method (Table 1) while patients with history of rejection preferred to be grafted with the new method (Table 2).
In both groups postoperative care was done according to the guidelines used in the hospital and worked out by the section cornea of the German Ophthalmological Society (http://www.dog.org/dog-kornea). Briefly, if a herpetic keratitis was the cause for keratoplasty, oral acyclovir 5 × 400 mg/day or oral valacyclovir 3 × 1 g/day (both from 1APharma, Oberhaching, Germany) was administered over ≥6 weeks then acyclovir oral 2 × 400 mg/day or valacyclovir 3 × 0.5 g/day continued in individual cases up to 12 months. Systemic steroids were given at the day of surgery 1 mg/day kg body weight over 5 days, then reducing over 3 weeks, or in high-risk cases for longer. Immunosuppression with cyclosporin A or mycophenolate mofetil combined with oral steroids was introduced at the day of surgery over 6 months under slow reduction and in consultation with the internist to reach a serum level of 100–150 ng/ml in accordance with the ophthalmological society guidelines. In addition, prophylactic therapy was initiated to possible intraocular pressure (IOP) elevation. Locally, steroids were applied in the weeks after surgery. All treatments were performed in accordance with the national guidelines (www.dog.org/dog-kornea). Clinical signs of opacification and of transplant rejection were monitored at regular times after surgery according to good clinical practice.
Proteome Study Using Two-Dimensional Polyacrylamide Gel Electrophoresis
Proteins from the corneal powder were extracted under reducing conditions and analyzed with the aid of two-dimensional (2D) polyacrylamide gel electrophoresis (2D-PAGE). The denaturing and reducing lysis buffers contained 5 M urea, 2 M thiourea, 2% (w/v) 3-[(3-chloramidopropyl)-dimethylammonio]-1-1propanesulfate, 2% (w/v) N-decyl-N, n-dimethyl-3-ammonio-1-propanesulfonate (SB 3–10), 0.5% (v/v) IPG buffer, 10 mM dithiothreitol (DTT), and a cocktail of protease inhibitors (2 mM EDTA, 2 mM 1,10-phemanthroline, 40 μM E-64, and 2 mM Pefabloc SC; all from Sigma-Aldrich, Munich, Germany). Extracted proteins were separated by 2D-PAGE (Life Technologies, Carlsbad, CA, USA) using an immobilizing pH gradient from pH 3 to pH 10.
The solubilized samples (300 μl) were loaded onto 18-cm Immobiline DryStrips (Amersham Biosciences, Munich, Germany) with a pH range of 3–10 and covered with DryStrip Cover Fluid (Amersham Biosciences). After incubation for 12–18 h at 25°C, isoelectric focusing was performed using IPGphor System I (Amersham Biosciences). Proteins were focused in the first electrophoresis dimension for about 120 kV/h. The proteins were reduced and alkylated by incubating the immobilized pH gradient strips in 10 ml of reduction buffer containing 6 M urea, 50 mM Tris-HCl (pH 8.8), 30% (v/v) glycerol, 2% (v/v) sodium dodecyl sulfate (SDS), and 6.5 mM DTT at 25°C for 15 min under shaking conditions and then in 10 ml of alkylation buffer containing 6 M urea, 5 mM Tris-HCl (pH 8,8), 30% (v/v) glycerol, 2% (v/v) SDS, and 10 mM iodoacetamide (all from Sigma-Aldrich) at 25°C for 15 min, also under shaking conditions.
For the second electrophoretic dimension, the strips were transferred to 18 × 23.4 cm SDS-polyacrylamide gels (12.5%), covered with 0.5% agarose, and run in a Hoefer DALT tank (Amersham-Biosciences) for 6 h at about 15,000 W per gel at 20°C in running buffer containing 25 mM Tris, 192 mM glycine, and 0.1% (w/v) SDS. After fixation overnight, the gels were stained with Coomassie Brilliant Blue (Merck), scanned, and then stored in 5% (v/v) acetic acid at 4°C.
To prepare samples for mass spectrometry (MS), 2D gel spots of interest were excised and destained in 50:50 v/v 25 mM NH4HCO3/methanol (Chromasolv, Sigma-Aldrich). Gel spots were washed in high-purity water (Chromasolv), covered with acetonitrile (Sigma-Aldrich), and then dried in a vacuum concentrator. For in-gel tryptic digestion, 5–10 μl of trypsin solution (0.02 μg/μl, proteomics grade from porcine pancreas; Sigma-Aldrich) in 50 mM NH4HCO3 was added. After swelling, gel spots were covered with 50 mM NH4HCO3 and incubated at 37°C for 14 h. Peptide extraction was carried out using acetic acid/water/acetonitrile solutions at v/v/v 1/89/10, 1/49/50, and 1/19/80, successively, with vortexing for 30 min. The pooled extracts were dried in a SpeedVac (Thermo Fisher Scientific, Schwerte, Germany) and desalted into 3 μl of solvent (0.1% trifluoroacetic acid containing 80% acetonitrile) using micro ZipTips C18 (Millipore, Darmstadt, Germany), according to the manufacturer's protocol.
For peptide mapping, a MALDI micro MX instrument (Waters, Manchester, UK) was employed in reflectron mode and 1 μl of the desalted peptides was spotted. α-Cyano-4-hydroxycinnamic acid (Waters) was used as a matrix. For peptide sequencing and match verification, Q-TOF Premier (Waters) was employed with either manual nano-MS/MS or coupled to nanoliquid chromatography (ultraperformance liquid chromatography, UPLC; Waters). The liquid junction source for manual analysis was homemade (30) and employed the stage of the online source. Chromatography was performed on a 1.7-μm BEH 130 100 μm x 100 mm C18 nanoAcquity UPLC column (Waters). The gradient went from 3% solvent A (0.1% formic acid, 5% acetonitrile) to 50% solvent B (0.1% formic acid, 5% water) in 30 min. Data-dependent MS/MS was recorded by preference on doubly and triply charged peptides.
Spectra were analyzed using Mascot software (Matrix Science, London, UK) in-house and ProteinLynx Global SERVER (Waters) with the SwissProt public database (Swiss Bioinformatics Institute SIB, Lausanne, Switzerland). Both general and species-refined searches were performed. Modifications (methionine oxidation, carbamidomethylation, N-acetylation, methylation of aspartic and glutamic acids) were only considered with secure matches to provide additional information. All assignments were reevaluated manually employing tools such as ProteinProspector (UCLA; Los Angeles, CA, USA) and BLAST (SIB).
Different keratin isoforms (e.g., cytokeratins 1, 2, 9, and 10) were found in abundance likely as contaminants in some spots. Distinct proteins were categorized according to function and cellular or acellular localization, as reported in the current bibliography. In cases where it was apparent that the same spot had been analyzed on gels loaded with either probe (e.g., native cornea, FCS-cultured cornea, or HS-cultured cornea), the spots were given the same spot ID.
Immunohistochemistry
Proteins of interest were examined in frozen corneal sections obtained from native, FCS-cultured, or HS-cultured corneas. Standard immunohistochemistry protocols were used to detect following proteins, which exhibited regulated expression under alternative culture protocols: vimentin, keratin, collagen, β-actin, galectin, and apolipoprotein A1 (APO-A1). The following antibodies were used as primary antibodies: monoclonal antihuman vimentin (Sigma, dilution 1:200), monoclonal antihuman pan keratin (Abcam, Cambridge, UK; dilution 1:50), mouse antihuman collagen VI (Chemicon, Darmstadt, Germany; dilution 1:100), monoclonal anti-human β-actin (dilution 1:500), antihuman galectin-3 (Santa Cruz, Heidelberg, Germany; dilution 1:300), and monoclonal antihuman APO-A1 (Acris, Herford, Germany; dilution 1:80). The secondary antibodies were directed against the respective first ones and were stained with either the cyanine dyes Cy2 or Cy3 to allow examination with fluorescence (Sigma-Aldrich). Control sections were only incubated with the second antibody (i.e., the primary antibody staining step was missed). Staining of the cell nuclei was performed with the nuclear stain DAPI (Hoechst Chemicals, Frankfurt, Germany).
Statistical Analysis
To compare between endothelial cell densities, statistical significance (p < 0.05) was determined using unpaired t tests in Prism software (GraphPad, La Jolla, CA, USA). For determining probability of rejection between the two groups of grafts, Fisher's exact test (p < 0.01) was applied.
Results
Keratoplasty with Recipient Serum-Adapted Corneas RSACs
To establish whether human serum is suitable to replace FCS in the culture medium, all corneas retrieved for transplantation were inspected weekly and examined microscopically for an intact endothelium carpet (Fig. 1A, B) and optical transparency. The culture medium was then exchanged, and the new medium was supplemented with an aliquot of either FCS or the recipient's serum. The endothelium was uniformly visible in both culture media (Fig. 1A, B), exhibiting no intercellular gaps when viewed at higher magnification (Fig. 1C, D). At the end of the 4th week, each cultured cornea was finally evaluated, and when considered suitable, it was released for grafting. The ECDs of the control corneas (n = 26) incubated in FCS-containing medium before and after incubation were 2,495 ± 363/mm2 and 2,461 ± 313/mm2, respectively (Fig. 1E). In the HS-supplemented group, the ECDs before and after incubation were 2,565 ± 478/mm2 and 2,675 ± 431/mm2, respectively (Fig. 1E). No significant endothelial cell loss occurred under either condition (p > 0.2) (Fig. 1F). Histological staining revealed no abnormalities in the appearance of any of the layers and basement membranes (not shown). The stroma structure appeared unchanged, and the epithelium was intact (Fig. 2). Neither cell abnormalities nor structural defects were observed, thus documenting that HS and FCS are equally suitable for supplying nutrients to and preserving human corneas in culture. No perioperative complications were reported in any of the cases, regardless of the serum used during corneal culture.

Biomicroscopic appearance of the corneal endothelium after 4 weeks of culture. (A, C) In fetal calf serum (FCS)-supplemented medium at lower (A) and higher magnification (C). (B, D) Human serum (HS)-supplemented endothelium at lower (B) and higher (D) magnification. The cells are arranged regularly in endothelium from both media, without gaps in the layers. Scale bar: 20 μm. (E) Histogram showing the mean central cornea endothelium cell density (ECD) before and after culture in the two types of serum. (F) Endothelium cell loss (ECL) after culturing human corneas in either FCS- or HS-supplemented medium shows no significant differences between the two media.

Four typical high-risk eyes that have rejected grafts and opacified (A, D, G, J). These eyes received RSACs and were rephotographed at 6 months (B, E, H, K) and 12 months (C, F, I, L) after grafting. All recipient serum-adapted corneas (RSACs) showed great transparency with no signs of postoperative complications.
We proceeded to examine all transplants over the postoperative period (up to 8 years in some cases) and evaluated the clinical appearance of HS-supplemented compared to FCS-supplemented transplants. All HS-supplemented transplants exhibited transparency in the postsurgical period (Fig. 2), and both groups (HS- and FCS-supplemented transplants) were treated topically with tear substitutes and steroids, according to standardized protocols. The patients who received recipient serum-adapted corneas (RSACs) were classified into two categories. One subgroup consisted of high-risk eyes in patients with multiple keratoplasties (≥2) as a result of high-risk conditions and a history of rejections in the same eye of all previous grafts (n = 16). Of these presensitized host corneas, three patients had received three grafts in the past and rejected them, while one patient had already rejected eight previous grafts. The second subgroup consisted of high-risk keratoplasties with a history of no rejections or rejection of one graft (n = 10). The decision to perform the first grafting with biologically adapted material was based on the clinical history and the severity of the corneal changes with increased risk of rejecting conventional corneal grafts.
The individual diagnoses that allowed a classification as high-risk are listed in Tables 1 and 2. It appears that no cornea in either high-risk subgroup was rejected up to 84 months postoperatively. The mean follow-up time was 64 months (range 48–96 months). Figure 3 shows the preoperative and long-term postoperative situs of the eye that had a history of eight rejections and received an RSAC in March 2002. Although no postoperative complications have occurred over the postoperative period of more than 9 years, the blood vessels have yet to penetrate the central graft (examined at 1, 5, and 9 years after surgery, respectively) (Fig. 3B–D). The central graft is still opaque (Fig. 3D) due to inappropriate reepithelization caused most likely by circumferential stem cell insufficiency of the limbus stem cell population.

Morphological follow-up of a high-risk eye that had received multiple transplantations (i.e., eight) before it received an RSAC. (A) Image before RSAC. (B) Image at 12 months after RSAC transplantation showing a clear cornea that was tolerated over this postgraft period. The visual acuity remained stable at 1/20. (C) Image 5 years after grafting, showing a stable graft and the limbus blood vessels restricted to the graft edge, not passing into the graft. (D) At 7 years after grafting, no vessels have penetrated the graft, which still exhibits stable morphology. The central cornea is not transparent, most likely due to failure of limbal stem cells to reepithelialize the cornea. No episodes of rejection were monitored. Scale bar: 2 mm.
Seven of 26 eyes that had undergone FCS-supplemented keratoplasty showed postsurgical signs of rejection (Table 1), even though they were treated with clinically standardized protocols of immunosuppression. All episodes of rejection in the postsurgical time of examination were properly treated and were reversible. No signs of transplant rejection were observed in the 26 eyes included in the HS-supplemented keratoplasty group (Table 2). The probability for rejecting grafts in the HS-supplemented group is therefore significantly lower than in the FCS-supplemented group (Fisher's exact test, p < 0.01). The Kaplan–Meier plot of Figure 4 shows the survival time of the grafts in the two groups. Thus far, the data show that preincubation of the corneal tissue in recipient serum-containing medium reduces the probability of rejection and enhances the tolerance of the grafted tissue. Taken together, the data emphasize the suitability of the individualized procedure for minimizing graft rejection.
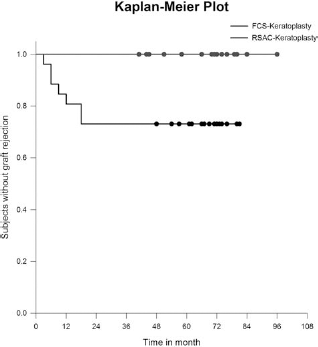
Kaplan–Meier plot showing the survival of grafts in both groups during the period observed.
Proteome Profiling of the Corneas
We sought to examine the potential mechanisms underlying the observed tissue adaptation when cultured in either serum. When human native corneal tissue with no clinical signs of pathology and normal history was subjected to 2D-PAGE, 120 protein spots were visible after Coomassie Brilliant Blue staining (Fig. 5A). Of these, 42 protein spots, which were stained reproducibly with similar intensity in the three gels, were submitted to proteomic analysis (Table 3). As expected, most of the proteins are isoforms of collagens, keratin, vimentin, APO-A1, β-actin, and transforming growth factor-β (TGF-β) fragments.

Two-dimensional gel electrophoresis of a native cornea and corneas cultured in either FCS- or HS-supplemented medium. Protein spots stained with Coomassie Brilliant Blue were numbered consecutively and excised for mass spectrometry. The images show that fewer proteins were labeled in the FCS-cultured cornea than in the HS-cultured cornea, which exhibits a pattern that is quite similar to that of native cornea.
Proteins in the Human Cornea

(A) Native cornea, (B) cultured in FCS-containing media, (C) cultured in human serum. For spot numbers, see Figure 5.
In contrast to the numerous proteins identified in the native cornea, 60 protein spots were stainable with Coomassie Brilliant Blue in gels after separation of FCS-cultured corneas, and 10 proteins were reproducibly stained in all gels and excised (Fig. 5B). In the corneas cultured in HS-supplemented medium, 60 protein spots appeared in the Coomassie Brilliant Blue staining and 24 spots were reproducibly stained in all gels, and excised (Fig. 5C). When compared with the native cornea, the gel image showed that both cultured corneal tissues differed from the native one. On the other hand, the profile of HS-cultured corneas apparently differs from that of FCS-cultured corneas. Verified proteins are listed in Table 3.
Staining of Proteins in Corneal Sections
Certain proteins with different expressions on 2D-PAGE were selected to immunostain corneal sections of native, FCS-cultured, or HS-cultured corneas. Vimentin is a pronounced structural protein and was highly expressed within all layers of the native cornea (Fig. 6A). It was drastically downregulated in the stroma of FCS-cultured corneas (Fig. 6B) but well preserved in the HS-cultured corneas (Fig. 6C). Some keratin isoforms were stained in the gels with native or HS-cultured corneas. The antibody used labeled all corneal keratins and detected a high expression of keratin within the epithelium of native (Fig. 6D), FCS-cultured (Fig. 6E), and HS-cultured corneas (Fig. 6F). In addition, strong keratin staining was observed within the endothelium of HS-cultured corneas (Fig. 6F), while the endothelium of native and FCS-cultured corneas remained virtually unstained (Fig. 6D, E). β-Actin showed a strong staining within the epithelium and endothelium of native (Fig. 6G) and FCS-cultured corneas (Fig. 6H); however, in the HS-cultured cornea, the protein showed weaker fluorescence within the epithelium and superficial stroma (Fig. 6I). Collagen VI was strongly stained in the epithelium and endothelium of both native (Fig. 6J) and FCS-cultured corneas (Fig. 6K) but was restricted to the endothelium in the HS-cultured corneas (Fig. 6L). Galectin was stained diffusely in the native corneal stroma (Fig. 6M) and similarly weakly stained in the HS-cultured corneas (Fig. 6O), but was strongly up regulated in the epithelium and endothelium of FCS-cultured corneas (Fig. 6N). There was strong APO-A1 fluorescence within the epithelium and endothelium of native and HS-cultured corneas, which was downregulated in the FCS-cultured cornea (not shown). The IgF-κ chain was uniformily stained throughout all cornea layers and in all samples (not shown). It appears from these additional data obtained from immunostained sections that HS- and FCS-cultured corneas exhibit different patterns of protein staining, as might be expected from the results of their respective proteome analyses.

Immunohistochemical examination of the proteins in corneal sections of native (left column), FCS-cultured (middle column), and HS-cultured (right column) corneas. Depicted are the proteins vimentin (Vim) (A–C), keratin (Ker) (D–F), β-actin (b-act) (G–I), collagen VI (Col) (J–L), and galectin (Gal) (M–O), which showed differential expression between the probes. A noteworthy finding is that culturing in FCS- and HS-supplemented media results in differential patterns of protein staining, as might be expected from the proteome profiling. Abbreviations: ep, epithelium; en, endothelium; st, stroma. The blue stain is nuclear DAPI stain. Scale bar: 200 μm.
Discussion
We have demonstrated that culturing donor corneas in a medium containing prospective recipient serum is a suitable method for preserving and transplanting corneas that has a high tolerance and low probability for rejection. Proteomic analysis of corneas cultured with either FCS- or HS-supplemented medium revealed that FCS reduces the number of stainable proteins compared to HS. On the other hand, HS-cultured corneas exhibited a proteome profile close to the native one. The assessment of the expression of selected corneal proteins using immunohistochemistry confirmed that the staining patterns in HS-cultured corneas were more similar to the native cornea than in FCS-cultured corneas. Taken together, these data demonstrate the suitability of RSACs for clinical transplantation, particularly in the worst cases of severe corneal disease.
Corneal grafting is the oldest and most common method of tissue grafting to replace diseased tissue with a transparent donor cornea. It has progressed over the past few decades due to improved eye-banking methods, better suturing materials, the ever-improving optical quality of microscopes, and more efficient postoperative suppression of inflammation. About half of the corneas grafted are cultured in media containing FCS (31,36), while serum-free media have also been tested (5,19). We have demonstrated in 26 clinical cases classified as high-risk keratoplasties that transplanted corneas cultured in the prospective recipient serum prior to surgery were tolerated and not rejected over the years. The new finding of this study is the significantly improved clinical outcome in high-risk keratoplasty, with a reduced graft rejection rate compared to keratoplasties using conventional grafts propagated in FCS-containing medium. However, more cases in randomized, double-blind, prospective studies will be essential to analyze the mechanisms underlying the beneficial effects of RSACs.
A marked sign of healthy cultured corneas is the density of the unilayered endothelium cells. A serum-free culture medium that was designed to improve endothelial cell survival, cell membrane integrity, and keratocyte density was developed (4). Long-term storage of corneas in an endothelial serum-free medium appears to be superior to conventional cultures with respect to changes in the ECD (19). However, most cornea banks use FCS-supplemented (13,14). Since one of the goals of preparing corneas for grafting is to remove FCS from the culture medium, thus increasing the risk of infection, recipient-derived serum appears to be a possible step toward this goal. As we have shown, the ECD in HS-cultured corneas is comparable to that in FCS-cultured corneas. In addition, no significant endothelial cell loss was observed in RSACs compared to FCS-cultured corneas. These data probably reflect a metabolic adaptation of the cultured corneal tissue, and hence a higher probability of tolerance after grafting. Although the mechanisms underlying this adaptation remain to be analyzed at the cell-signaling level, the successful clinical application of this method promises to remove FCS from corneal cultures in the future.
The positive effects of autologous serum have been tested in a randomized double-blind trial that compared topical autologous serum with hyaluronic acid (45). Those authors investigated the healing of epithelial defects after vitrectomy or cataract surgery and found that epithelial healing was faster in the autologous serum-treated group than in the hyaluronic acid-treated (45). This validation of the growth-promoting effects of autologous serum could be applied to reduce the risk of complications in sight-affecting epithelial defects. In a further prospective randomized study, Autologous serum eye drops after laser-assisted in situ keratomileusis improved prolongation of the tear but was associated with a reduction in the Rose Bengal staining score compared to artificial tears (40). These findings are confirmed by the present finding that HS provides advantages over FCS in the preservation of corneal tissue for keratoplasty.
High-risk keratopathies are defined by severe destruction of the cornea, pathological vascularization, dense/opacity, and the tendency to reject donor tissue. The worst cases are those in which multiple grafts have been rejected. Among the techniques applied to date for severe keratopathies, amnion membrane grafting (27,46) has been the most promising, especially in cases of heavy chemical burns. The amnionic membrane facilitates corneal reepithelialization underneath the graft, inhibits fibrosis through downregulation of TGF-β, reduces inflammation and neovascularization, and decreases the risk of postoperative infection due to its antimicrobial properties (35). However, this method is not applicable in cases with total limbal stem cell insufficiency (11). The alternatively used limbus stem cell transplantation is restricted only to unilateral diseases, such as alkali burns, because it requires a healthy limbus in the contralateral eye (30). Heterologous transplantations from donor eyes result in limited improvement, due to the high incidence of neovascularization and/or rejection (46,50,51), therefore requiring immunosuppression. However, deep neovascularizations with stromal opacification require a perforating keratoplasty with exchange of all corneal layers. An improvement can be achieved with HLA-matched heterologous limbus tissue, which reduces the risk of rejection (46). However, a valid evaluation of this method demands a long-term follow-up of these cases. The method of pretreating donor corneas in a culture medium that contains the recipient's serum is, to our knowledge, the first corneal tissue bioadaptation procedure to be applied to cases of high-risk keratoplasty. One advantage over the use of FCS might be the biological adaptation of corneal cells to prospective recipient-derived HS, thus diminishing FCS-derived side effects (Fig. 7). Apparently normal structure of all layers of the cornea suggests that HS exerts no destructive side effects on the corneal cells and basement membranes. The native cornea-like expression of proteins also supports the view that HS is suitable for nourishing corneal cells by providing the factors necessary for its survival. As summarized in Figure 7, such factors might confer upon the transplanted tissue the “recipient-specific” expression of surface antigens, making it less vulnerable to immunological rejection following transplantation.

A simplified cellular model to summarize the putative biological adaptation mechanisms operating in cultured corneal cells. Various cytokines, IgFs, serum proteins, hormones, and trophic factors, which are different between FCS and HS, confer signals to the cells. These signals might be mediated by membrane receptors, internalization into the cytoplasm, or activation of nuclear receptors. As a consequence, mRNAs inferred by the recipient serum are transcribed that encode for structural or cell surface proteins.
However, the protein(s) responsible for the biological adaptation remain to be determined, although several factors are probably involved. It is well known that corneal cells proliferate in organ culture (4,19). This proliferation involves all cellular layers of the cornea including endothelial cells, keratocytes, and epithelial cells, which are probably dividing as they do in situ. Evidence for such cell division is the morphological appearance of the epithelium, which resembles freshly removed tissue. Keratocytes also proliferate in culture, and the endothelium does not show gaps or larger defects, as revealed by microscopy.
Increasing our knowledge of the biochemistry of normal and cultured corneas is essential to understanding the increased tolerance of RSACs. In recent studies, 141 proteins were identified in the normal human cornea using different tissue-treatment and protein separation protocols (2,23,24). The list of these proteins is available at the cornea protein database, which provides a summary of proteins identified in five different 2D gels using different pH gradients and extraction protocols (23,24). In the present study, one protocol was used for protein separation because the main aim was to compare native corneas with two different culture-medium-derived corneas, and most of them were excisable and identifiable. The FCS-cultured cornea showed exceptionally few of the proteins that are usually represented in the typical cornea. The reasons for the downregulation of numerous proteins in FCS-supplemented media remain to be established. On the other hand, HC-cultured corneas appeared closer to the native corneas, with several proteins showing similar expressions in both the native and HC-cultured corneas. Some of the spots have not yet been identified due to the low protein content of the spot after in-gel trypsinization. We are performing an ongoing investigation of further eluation protocols to identify these proteins.
Additional immunohistochemistry confirmed that there were some differences in the staining patterns of some of the typical cornea proteins between FCS- and HS-cultured corneas. Thus, the “biological adaptation” hypothesized in this work is probably a multifactorial process. The factors contributing to this adaptation are different proteins subserving multiple cellular functions.
The great success of corneal grafting compared to vascularized organs has led to the concept of an “immunological privilege,” which has also been observed in the anterior chamber (reviewed in 5,7,8,20,25,39,41). However, corneal grafts can induce an allograft reaction-rejection, perhaps due to the reaction of antigen-presenting cells in the cornea. In particular, severe cases of corneal pathology with ulcers, neovascularization, and insufficient populations of stem cells result in a higher frequency of intolerance and rejection. The immune privilege of corneal allografts is a phenomenon that is determined by different parameters, such as the (1) appearance of Langerhans cells, (2) expression of Fas ligands on the epithelium and endothelium, and (3) induction of immune deviation of the systemic immune response (39), might be altered in corneas cultured with the host serum. The first line of evidence for such biological modification could be the reduction in the numbers of Langerhans cells. It is likely that the immunogenic properties of the graft change in order to reduce interference between the graft and the native tissue, with induction and expression of self-destructive immune responses, which culminate in rejection.
The therapeutic options for high-risk eyes are currently limited. Methods reported to be associated with increased graft tolerance that are not yet clinically applicable include immunological protection with a blocking antibody, which has been observed in rabbits (6). However, all antibodies from donor strains might cause adverse effects, as shown in corneal mice grafts (18). Immunomodulation of T-cells within the cornea is an alternative approach in experimental animals, promising to prevent activation of these cells (16). In addition, oral immunization with cultured corneal epithelial and endothelial cells from donor mice strains appears to increase the survival of corneal grafts (17). In clinical practice, prolonged graft survival can usually be achieved by early detection of immunological responses and intense treatment with corticosteroids (reviewed in 22). However, cases that have a high risk of graft failure and repeated cornea grafting can only be managed with systemic cyclosporine, a treatment that has moderate beneficial effects (43). It therefore seems reasonable to explore new strategies for better graft acceptance by treating the grafts themselves, in order to adapt proliferating donor tissue to conditions containing host components. To date, the approach of cultivating corneas in host serum promises to become an alternative and clinically testable method with direct therapeutic relevance for high-risk keratoplasties. This method might also be extrapolated for use in low-risk keratoplasties if clinical evaluation of the eye raises suspicion of a high probability of rejection. Finally, all solid tissues that are considered allotransplantable, such as skin, cartilage, and muscle, can be pretreated by incubation in the recipient's serum to minimize the rejection probability.
Footnotes
Acknowledgment
The authors declare no conflict of interest.
