Abstract
Olfactory mucosa (OM)-derived olfactory ensheathing cells (OECs) are attractive candidates for autologous cell transplantation-based therapy of nervous system injury. However, defining the regenerative capacity of OM-derived OECs is impeded by the fact that cell cultures used for transplantation may contain significant amounts of contaminating trigeminal nerve Schwann cells that escape identification by sharing in vitro expression of OEC markers. The aim of the present study, therefore, was to quantify contaminating Schwann cells in OEC preparations and to develop a protocol for their specific depletion. Based on the observation that freshly dissociated, but not cultured, OECs and Schwann cells display differential expression of HNK-1 and p75NTR, magnet-activated cell sorting (MACS) was used to deplete myelinating (HNK-1-positive) and nonmyelinating (p75NTR-positive) Schwann cells from primary cell suspensions containing HNK-1-/p75NTR-negative OECs. Upregulation of p75NTR expression in OECs during culturing allowed their subsequent MACS-based separation from fibroblasts. Immunofluorescence analysis of freshly dissociated OM prior to MACS depletion revealed that 21% of the total and 56% of all CNPase-positive cells, representing both OECs and Schwann cells, expressed the Schwann cell antigens HNK-1 or p75NTR, indicating that freshly dissociated OM prior to culturing contained as many Schwann cells as OECs, while olfactory bulb (OB) primary cell suspensions revealed lower levels of Schwann cell contamination. Interestingly, neurite growth of neonatal rat dorsal root ganglion (DRG) neurons cocultured with OM-OECs, OB-OECs, and fibular nerve (FN) Schwann cells used as control was significantly higher in the presence of OECs than of Schwann cells. The first report on identification and specific depletion of Schwann cells from OEC preparations provides a solid basis for future efforts to fully define the regenerative potential of nasal mucosa OECs.
Introduction
Cell transplantation is considered a promising strategy for improving neural regeneration (9, 40, 61, 86). During the recent years, there has been growing interest in the olfactory mucosa (OM) as a cellular source of regeneration-promoting cells suitable for cell transplantation-based therapy of the injured nervous system (33, 34). OM is not only easily accessible in clinical practice and, therefore, suitable for autologous transplantation strategies, but it also harbors specialized glial cells with regenerative properties (53). In situ, olfactory ensheathing cells (OECs) are nonmyelinating and intimately associated with olfactory receptor neurons along their entire axonal projection to the olfactory bulb (OB) (17, 20, 62). Since OECs have been shown to stimulate neural regeneration in vivo after transplantation in a variety of different lesion paradigms (5, 70, 87), it was recommended to use them for fostering axonal regeneration and remyelination (7, 18, 22, 23, 66). However, the vast majority of experimental studies so far have been conducted with OB-derived OECs. A prerequisite for translating these data into clinical practice would be the demonstration that OECs derived from the OM in fact display the same regenerative properties (60).
The required comparative analysis of OM- and OB-derived OEC populations, however, is impeded by the fact that the OM is a highly complex structure that contains not only OECs but also myelinating Schwann cells from trigeminal afferents in addition to nonmyelinating cells associated with blood vessels in both localizations (51–53). In the absence of cell type-specific markers for cultured OECs and Schwann cells (65, 83), it is so far not possible to detect Schwann cells in, or to deplete them from, OEC preparations. Antibody-based purification protocols are usually based on cultured OECs and the neurotrophin receptor p75 (p75NTR) as a marker (39). Since p75NTR is also expressed by cultured Schwann cells, it is expected that they undergo copurification with OECs. The question of Schwann cell contamination is of particular relevance, since Schwann cells also have been shown to exert beneficial effects on regeneration (14, 15, 19, 41, 48, 73, 81) and because their effects may at least in part overlap with the effects of OECs. To exclude that the putative diverging characteristics of OM and OB cell preparations (3, 34, 67) arise from varying amounts of contaminating Schwann cells, clearly defined cell preparations are essential for in vitro and in vivo analysis.
The aim of the present study, therefore, was to develop a protocol for the generation of Schwann cell-free OEC preparations from OM and OB suitable for comparative in vitro and in vivo analysis and to visualize Schwann cells for calculating their numbers in OM and OB preparations. Since OECs and Schwann cells display differential antigenic expression in situ but not in vitro (10, 57), both the immunocytochemical analysis and the purification protocol were largely based on freshly dissociated tissue rather than on cultured cells as previously done. Prompted by the observation that adult canine myelinating and nonmyelinating Schwann cells, but not OECs, in situ express the human natural killer-1 (HNK-1) epitope and p75NTR, respectively (10), we used specific antibodies against both antigens and freshly prepared OM and OB cell suspensions for specific depletion of Schwann cells. Following upregulation of p75NTR expression in OECs during culturing (10, 21, 85), anti-p75NTR antibodies and magnet-activated cell sorting (MACS) were used for final purification of OECs to generate Schwann cell-free cultures.
We show here that primary cell suspensions of the adult canine OM and OB contained substantial and differing numbers of cells with a Schwann cell-like antigenic expression profile. This is an exciting finding relevant for the retrospective analysis of in vivo transplantation studies. Moreover, the newly established two-step purification protocol efficiently eliminated Schwann cell contaminants yielding highly purified preparations of OM- and OB-OECs. The observation that OM-OECs stimulated neurite growth in vitro to a higher extent than Schwann cell controls from fibular nerve underscores the relevance of the OM as a source of regeneration-promoting cells suitable for clinical application.
Materials and Methods
Antibodies
Monoclonal antibodies included mouse anti-human-p75NTR antibodies (clone HB8737; American Tissue Culture Collection; 42,49) used as hybridoma supernatants (1:5), mouse anti-CD57/HNK-1 antibodies (1:500; C6680, Sigma-Aldrich, St. Louis, MO, USA) (10), mouse anti-2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) antibodies (1:500; Chemicon, Billerica, MA, USA) (28, 57), and rabbit anti-neuronal class III β-tubulin antibodies (1:1,000; Covance, Emeryville, CA, USA).
Experimental Animals
Tissues were obtained from adult male (n = 6) and female (n = 13) dogs (Beagle, n = 15; French bulldog, Labrador retriever, Pyrenean shepherd, and mongrel, each n = 1) with a mean age of 2 years that did not suffer from diseases affecting the nervous and respiratory system, as determined by clinical and histopathological examination. Dorsal root ganglia (DRG) were obtained from neonatal rats (n = 8, Sprague–Dawley; Hannover Medical School). All animals were treated according to the legal and ethical requirements of the University of Veterinary Medicine Hannover.
Tissue Isolation and Single-Cell Dissociation
Tissue was collected under sterile conditions at necropsy within a postmortem interval of less than 2 h (42). For isolation of olfactory mucosa (OM) and olfactory bulb (OB), heads were skinned and transected sagittally along the nasal septum as described previously (10). OB was separated from the brain by frontal sectioning with a scalpel, and OM was extracted from the caudal regions of the nasal septum and conchae (11). Fibular nerve (FN) segments were excised directly distal to the sciatic nerve bifurcation. Tissue was sequentially rinsed in ProntoVet® disinfectant (B. Braun Melsungen AG, Melsungen, Germany) and phosphate-buffered saline without calcium and magnesium (PBS-) containing penicillin/streptomycin (1%; PAA, Marburg, Germany) for 5 and 15 min, respectively. OM was cut into pieces of about 1 mm3 in size using a scalpel; meninges were removed carefully, and the outer layers of the OB were isolated under binocular control using fine forceps and a scalpel and dissected as described (10, 42). After removal of the FN, epineural sheath teased nerve preparations were prepared using fine forceps (84) and transferred to Petri dishes (10 cm; Nunc, Roskilde, Denmark). Nerve preparations were maintained in complete medium consisting of Dulbecco's modified Eagle's medium (DMEM; Life Technologies GmbH, Darmstadt, Germany) supplemented with fetal calf serum (FCS, 10%; PAA, Marburg, Germany), penicillin/streptomycin (1%; PAA), and sodium pyruvate (1%; PAA) for 5–7 days under standard conditions (5% CO2, 37°C).
To identify conditions promoting both tissue dissociation and preservation of p75NTR and HNK-1 antigenicity, OB and OM (n = 4) were treated with different enzyme cocktails for 45 min at 37°C (Table 1). Applied enzymes were from Sigma-Aldrich (Taufkirchen, Germany) and included trypsin (T-8003), hyaluronidase type IV (H-3884), and collagenase type IV (C-5138) and type XI (C-9407), while Accutase® was from PAA (Marburg, Germany). Teased nerve preparations were incubated with trypsin (T-8003), hyaluronidase type IV (H-3884), and collagenase type XI (C-9407; 0.5% each; all from Sigma-Aldrich, Taufkirchen, Germany) for 45 min at 37°C. Single-cell dissociation of enzyme-treated tissue was done in the presence of DNase I (0.05%; Roche Diagnostics, Mannheim, Germany) using a fire-polished Pasteur pipette (57).
Single-Cell Dissociation of Adult Canine OM and OB and Preservation of Antigenicity

(A–E) Human natural killer-1 (HNK-1) or p75NTR depends upon the dissociation enzymes used. Despite efficient tissue digestion, use of trypsin completely (A, 0.5%) or partially (B, 0.25%) abolished p75NTR immunoreactivity, while cocktails consisting of hyaluronidase IV and collagenase type XI preserved antigenicity but did not allow aggregate-free dissociation (C). The mixture of hyaluronidase IV, collagenase IV, and XI together efficiently degraded the extracellular matrix and preserved antigenicity (D). Accutase® only promoted partial single cell dissociation and reduced anti-HNK-1 as well as anti-p75NTR immunolabeling (E). Solution C and D were for treatment of OB and OM, respectively. n.d., not done.
Establishment of Schwann Cell-Free Cultures of Purified Adult Canine Olfactory Ensheathing Cells
Generation of highly purified, Schwann cell-free olfactory ensheathing cell (OEC) preparations was done using a novel two-step protocol (Fig. 1) that included the antibody-mediated depletion of Schwann cells from freshly dissociated tissue (OM, OB) (Fig. 1, I) followed by the separation of OECs from fibroblasts (Fig. 1, IV) after 5–7 days in vitro. Myelinating and nonmyelinating Schwann cells were removed immediately from primary cell suspensions (Fig. 1, I) by magnet-activated cell sorting (MACS; Miltenyi Biotec, Bergisch Gladbach, Germany) and anti-HNK-1 (OM) and anti-p75NTR antibodies (OM, OB), respectively (10, 42, 85) (Fig. 1). Briefly, cells were pelleted by centrifugation and resuspended in ice-cold, degassed PBS- with ethylenediamine tetraacetic acid (EDTA, 2 μM; Carl Roth GmbH & Co. KG, Karlsruhe, Germany) and bovine serum albumin (BSA fraction V, 0.5%; Sigma-Aldrich, Munich, Germany) (PEB) containing anti-HNK-1 (1:500) and anti-p75NTR (1:5; hybridoma supernatant) antibodies on ice for 20 min with gentle agitation (10). After washing with PEB, goat anti-mouse IgG MicroBeads were applied (1:5; Miltenyi Biotec, Bergisch-Gladbach, Germany) for 20 min on ice. Cells were then washed with PEB and passed over a MiniMACS column (Miltenyi Biotec, Bergisch-Gladbach, Germany) (Fig. 1). PEB used for washing the column was collected as the nonbound fraction (Fig. 1, II), while the bound fraction contained HNK-1- and p75NTR-positive Schwann cells that were eluted with PEB and a syringe following the column's dislodgement from the magnet (Fig. 1, III). To control MACS efficiency, myelinating and nonmyelinating Schwann cells targeted by anti-HNK-1 and anti-p75NTR antibodies were visualized in the nonbound (Fig. 1, II) and bound fractions (Fig. 1, III) by incubation with immunofluorescent secondary antibodies replacing the IgG MicroBeads, and the number of immunopositive cells was determined (see below). Moreover, bound and nonbound fractions were seeded in complete medium supplemented with human basic fibroblast growth factor-2 (FGF-2, 20 ng/ml; Peprotec, Tebu, Frankfurt, Germany) onto poly-l-lysine-coated (PLL, 100 μg/ml; P1274, Sigma-Aldrich, Taufkirchen, Germany) 24-well microtiter plates and culture flasks (Nunc, Roskilde, Denmark), respectively, and maintained under standard conditions for 5–7 days with medium change every 3 days.
Two-step purification of olfactory ensheathing cells (OECs) from olfactory mucosa (OM) and olfactory bulb (OB) using sequential specific depletion of Schwann cells (II) and subsequent positive selection of OECs (V). Primary cell suspensions (I) were incubated with anti-p75NTR (OM, OB) and anti-human natural killer-1 (HNK-1) antibodies (OM) and IgG MicroBeads for separation of nonmyelinating (p75NTR-positive) and myelinating Schwann cells (HNK-1-positive). The nonbound fraction consisting of OECs and fibroblasts (II) was maintained in vitro for 5–7 days, while the bound fraction enriched in nonmyelinating and myelinating Schwann cells (III) was discarded. After upregulation of p75NTR in cultured OECs (IV), cells underwent positive selection using anti-p75NTR antibodies and magnet-activated cell sorting (MACS) for separation of OECs (V) from fibroblasts (VI). M-SCs, myelinating Schwann cells; NM-SCs, nonmyelinating Schwann cells; OECs, olfactory ensheathing cells; Fbs, fibroblasts.
Purification of OECs was done from the nonbound fractions of OB and OM (Fig. 1, II) following upregulation of p75NTR expression in vitro during 5–7 days (10, 85) (Fig. 1, IV). Cells were detached from the substratum using a short incubation (45 s) with Accutase® and incubated sequentially with anti-p75NTR antibodies (1:5; hybridoma supernatant) and goat anti-mouse IgG MicroBeads (1:5; Miltenyi Biotec, Bergisch-Gladbach, Germany) in ice-cold PEB (10 min, 4°C) as described above. Control experiments verified that short-term incubation with Accutase® did not interfere with p75NTR antigenicity. After passage over the column, the bound fraction (Fig. 1, V) was seeded on PLL-coated flasks (100 μg/ml) in complete medium and expanded in the presence of FGF-2 (20 ng/ml), while the nonbound fraction consisting of fibroblasts (Fig. 1, VI) was discarded.
Purification of Adult Canine Fibular Nerve Schwann Cells
Dissociated fibular nerve (FN) Schwann cells were maintained on PLL-coated flasks (100 μg/ml) in complete medium for 5–7 days. Purification was done by MACS (Miltenyi Biotec, Bergisch-Gladbach, Germany) using anti-p75NTR antibodies as previously established (10, 57, 78). Briefly, cells were detached with Accutase® and incubated with anti-p75NTR antibodies (1:5; hybridoma supernatant) and goat anti-mouse IgG MicroBeads (1:5; Miltenyi Biotec, Bergisch-Gladbach, Germany) in ice-cold PEB (10 min, 4°C) prior to passage over the MiniMACS column (Miltenyi Biotec, Bergisch-Gladbach, Germany). Expansion of FN Schwann cells was done under the same conditions as OECs (see above).
Establishment of Dorsal Root Ganglion Explant-Glial Cell Cocultures
Neonatal rat dorsal root ganglia (DRGs) were isolated under binocular control and divided in two pieces using a scalpel. For establishment of cocultures, explants were transferred to PLL-coated (100 μg/ml) 96-well microtiter plates (Nunc, Roskilde, Denmark) in 20 μl of DMEM/ F12 (Life Technologies GmbH, Darmstadt, Germany) containing additives according to Giulian and Baker (27) and BSA (0.25%; Sigma-Aldrich, Munich, Germany) (78). Human β-nerve growth factor (β-NGF, 20 ng/ml; Peprotec, Tebu, Frankfurt, Germany) was added to promote DRG neuron survival (47). After a short incubation period (10 min) under standard conditions, canine OM-OECs, OB-OECs, or FN-Schwann cells were seeded at a density of 20,000 cells/well in 80 μl of DMEM/F12 (see above) containing additives, BSA (0.25%; Sigma-Aldrich), and β-NGF (20 ng/ml). Cocultures were maintained under standard conditions for 36 h.
Immunocytochemistry
Detection of intracellular (CNPase) and cell surface antigens (p75NTR, HNK-1) was carried out using fixed and viable cells, respectively. For visualization of CNPase, known to be expressed by both Schwann cells and OECs in situ (57), primary cell suspensions from the OM and OB (Fig. 1, I) were fixed immediately after seeding in 96-well microtiter plates (Nunc, Roskilde, Denmark) with paraformaldehyde (PFA, 4%) and permeabilized using Triton X-100 (0.25%) diluted in PBS- (PBST) containing BSA (3%; Sigma-Aldrich) for 15 and 5 min at room temperature (RT), respectively. To reduce nonspecific binding, cells were treated with normal goat serum (5%) diluted in PBST containing 3% BSA (Sigma-Aldrich) for 15 min at RT. Incubation of primary and secondary goat anti-mouse antibodies coupled to cyanine 3 (Cy3; 1:200; Jackson ImmunoResearch Laboratories, Dianova, Hamburg, Germany) was performed for 2 h each at RT, and nuclei were stained using bisbenzimide H 33258 (0.01% in aqua bidest) for 5 min at RT.
For cell surface staining of HNK-1 and p75NTR associated in situ with myelinating (OM) and nonmyelinating Schwann cells (OM, OB), respectively (10), viable cells of the primary cell suspensions (OM, OB) (Fig. 1, I) and the nonbound (Fig. 1, II) and bound fractions (Fig. 1, III) were immunostained in suspension, centrifuged to 96-well microtiter plates (Nunc), and fixed with PFA (4%). Prior to MACS separation, primary OM and OB cells (Fig. 1, I) were sequentially incubated with primary anti-p75NTR (OM, OB) and anti-HNK-1 (OM) antibodies and secondary IgG1- (OM, OB; 1:100; Alexa Fluor 555, Invitrogen Molecular Probes, Karlsruhe, Germany) and IgM-specific (OM; 1:100; Alexa Fluor 488, Invitrogen Molecular Probes, Karlsruhe, Germany) antibodies. Cells were then centrifuged onto PLL-coated (100 μg/ml) 96-well plates, fixed at RT with PFA (4%; 15 min), and stained with bisbenzimide H 33258 (5 min, RT; 42). To visualize Schwann cells in the bound and nonbound fractions after MACS, thereby verifying separation efficiency, cells of both fractions were incubated with the respective immunofluorescent secondary antibodies (see above). Displacement of the IgG MicroBeads by immunofluorescent antibodies visualized both subpopulations in the bound and nonbound fractions.
DRG explant-glial cell cocultures were immunostained for p75NTR prior to fixation with PFA (4%) essentially as described above, followed by labeling of fixed and permeabilized cell cultures (see above) with anti-class III β-tubulin antibodies (1:1,000).
Statistical Analysis
For determination of Schwann cell numbers in primary OM and OB cell suspensions and in the bound and nonbound fractions after MACS, immunopositive cells were counted in five different areas per well at 20x magnification using an inverted microscope (Olympus IX-70, Olympus Optical Co. GmbH, Hamburg, Germany). Labeled cells (HNK-1, p75NTR) were related either to the total number of bisbenzimide-stained cells or to the number of CNPase-positive cells determined in parallel cultures. Data obtained from 19 independent experiments in triplicates were subjected to statistical analysis using SPSS for windows (version 16, SPSS Inc., Chicago, IL, USA) and expressed as mean ± standard deviation.
Neurite growth of DRG neurons cocultured with OM-OECs, OB-OECs, and FN Schwann cells was measured using analySIS 3.1 software package (Soft Imaging System, Münster, Germany) by determining the total area covered by class III β-tubulin-positive processes. For measuring neurite length, the explant area was divided into eight parts, and the length of the longest neurite in each part was determined and summated. Statistical analysis was done using SPSS for windows (see above) employing the Mann–Whitney U test. Data represent the median, minimum, and maximum from four independent experiments. Statistical significance was designated as p < 0.05.
Illustrations
Cell culture images were acquired with an inverted fluorescence microscope (Olympus IX-70, Olympus Optical Co. GmbH, Hamburg, Germany), and microphotographs were taken using the PM-30 photo system (Olympus) and a color reversal film (Ektachrome 400). Slides were scanned with a DiMAGE Scan Multi Pro scanner (Konika Minolta Photo Imaging Europe GmbH, Unterföhring, Germany), and Adobe® Photoshop® (7.0, Adobe Systems, Inc., San Jose, CA, USA) was used to prepare the figures, with uniform adjustment of contrast, brightness, and sharpness to each part of the figure if necessary.
Results
Single-Cell Dissociation of Adult Canine Olfactory Mucosa and Olfactory Bulb, and Preservation of Antigenicity in the Presence of Applied Enzymes
The accurate determination of immunopositive cells in primary cell suspensions requires the use of enzymes that guarantee single-cell dissociation without interfering with antigenicity of the used marker molecules. To exclude that applied enzymes degraded the cell surface molecules HNK-1 and p75NTR, different enzymes were added to adult canine olfactory mucosa (OM) and bulb (OB) and both the mechanical dissociation and immunoreactivity were analyzed (Table 1). Unfortunately, trypsin efficiently digested the tissue but reduced p75NTR immunoreactivity at both concentrations of 0.5% and 0.25% (Table 1A, B), while cocktails of hyaluronidase IV and collagenase type XI preserved antigenicity but did not allow aggregate-free dissociation of the OM (Table 1C). The mixture of hyaluronidase I V, collagenase IV, and collagenase XI efficiently degraded the extracellular matrix and preserved p75NTR and HNK-1 antigenicity (Table 1D). Accutase®, a ready-to-use solution for gentle dissociation of cultured cells (85), only promoted partial single-cell dissociation and reduced anti-HNK-1 and anti-p75NTR immunoreactivity (Table 1E). On the basis of these experiments, enzyme solutions C and D were used for digestion of OB and OM, respectively.
Identification of Myelinating and Nonmyelinating Schwann Cells in Primary Cell Suspensions of the Adult Canine Olfactory Mucosa and Bulb
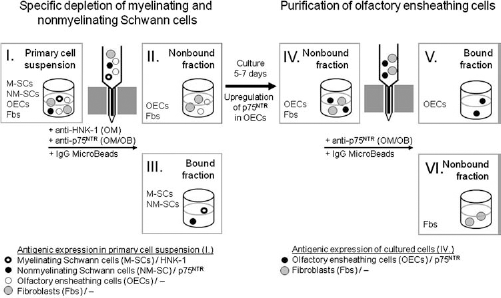
The major aim of the present study was to resolve the long-lasting debate of whether OEC preparations contain substantial numbers of Schwann cells. Since no specific markers were available for the selective identification of cultured Schwann cells, primary cell suspensions immediately after single-cell dissociation were immunostained for HNK-1 and p75NTR, known to be expressed in situ by adult canine myelinating and nonmyelinating Schwann cells, respectively, but not with OECs (10). In OM cell suspensions, 11.3% and 9.4% of the total cells were HNK-1-and p75NTR-immunopositive, respectively, while CNPase, which is synthetized by both OECs and Schwann cells (57), was found in about 37% of all dissociated cells. The percentage of HNK-1- and p75NTR-positive Schwann cells in relation to the number of CNPase-positive cells was 30.6% and 25.7%, respectively, indicating that both Schwann cell phenotypes and OECs were present at a quantitative ratio of about 1:1 (Table 2). HNK-1 and p75NTR were consistently found on distinct cell populations; colocalization of both antigens at the cellular level was only rarely found (Fig. 2A). In OB cell suspensions, 8.1% of the total cells were p75NTR-positive. Since 29.8% of the total cells were CNPase-immunoreactive, this suggested that Schwann cells and OECs displayed a quantitative ratio of about 1:3 (Table 2).
Immunofluorescence staining of olfactory mucosa (OM) (A, C) and olfactory bulb (OB) (B, D) primary cell suspensions with antibodies to HNK-1 (A, green), p75NTR (A, B, red), and 2¢,3¢-cyclic nucleotide 3¢-phosphodiesterase (CNPase) (C, D, red). Labeling of variably sized viable (A, B) and fixed (C, D) cells was punctuate and homogenous, respectively. Note that OM cell suspensions only contained a few aggregates (A, C, arrowheads). Myelin fragments were identified by size and HNK-1 reactivity (A, arrow). No aggregates were observed in OB suspensions (B, D).
Percentage of HNK-1-Positive M-SCs and p75NTR-Positive NM-SCs in Primary Cell Suspensions

Primary cell suspensions of adult canine olfactory mucosa (OM) and olfactory bulb (OB) are either related to the total number of cells identified by bisbenzimide or to the total number of 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase)-immunoreactive cells encompassing both OECs and Schwann cells. Since the OB does not harbor any myelinating Schwann cells, only anti-p75NTR antibodies were used for the analysis. Data represent the mean ± standard deviation. n.d., not done.
Immunofluorescence analysis of OM cell suspensions revealed punctuate staining of relatively small cells with varying size after incubation of viable cells with anti-HNK-1 and anti-p75NTR antibodies (Fig. 2A) and homogenous cytoplasmic labeling after immunostaining of fixed cells with anti-CNPase antibodies (Fig. 2C). Myelin fragments were easily identified by size and HNK-1 immunoreactivity (Fig. 2A, arrow). Despite enzyme treatment and mechanical dissociation, a few aggregates of less than 10 cells could be observed (Fig. 2A, C, arrowheads). In primary OB cell suspensions, anti-p75NTR (Fig. 2B) and anti-CNPase antibodies (Fig. 2D) also visualized small-sized cells after incubation of vital and fixed cells, respectively.
Antibody-Mediated Depletion of Myelinating and Nonmyelinating Schwann Cells From Primary Suspension of the Adult Canine Olfactory Mucosa and Bulb
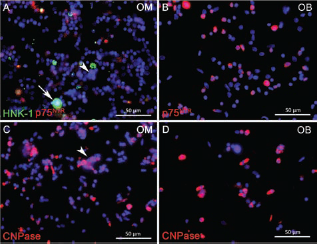
Previously, we used MACS for purification of OECs and their separation from fibroblasts (10, 85). In the present study, this technique was used for the specific depletion of Schwann cells, taking advantage of the fact that adult canine Schwann cells, but not OECs, in situ express HNK-1 and p75NTR (10). To control the efficiency of the separation procedure, antigen-bearing cells in the nonbound and bound fractions were visualized by incubation with immunofluorescent secondary antibodies after their passage over the MiniMACS column (Fig. 3 and Table 3). The observation that the vast majority of immunofluorescent cells were confined to the bound fractions verified the specificity of the separation procedure (Fig. 3). Quantitative evaluation of the number of antigen-bearing cells in the OM nonbound fraction showed that only 1.4% and 1.6% of the total cells were HNK-1- and p75NTR-positive, respectively (Table 3). Compared to the frequencies of immunopositive cells in the primary cell suspension (Table 2), this indicated that MACS allowed an eightfold and sixfold reduction in the number of myelinating (HNK-1-positive) and nonmyelinating (p75NTR-positive) Schwann cells (Table 3). Similarly, the percentage of nonmyelinating Schwann cells (p75NTR-positive) being left in the OB nonbound fraction was only 1.2% (Table 3), which corresponds to a 6.8-fold reduction of Schwann cells compared to the primary cell suspension (Table 2).
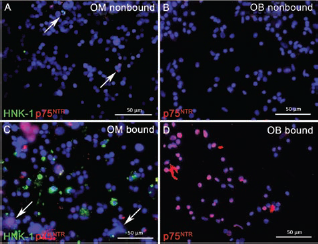
Incubation of the nonbound (A, B) and bound fractions (C, D) of olfactory mucosa (OM) (A, C) and olfactory bulb (OB; B, D) with immunofluorescent secondary antibodies visualizing HNK-1- (A, C, green) and p75NTR-positive cells (A–D, red). Despite formation of some aggregates in the bound fraction of OM (C, arrows), immunoreactive cells were almost quantitatively eliminated from the nonbound fractions (A, B) that only displayed a few positive cells (A, arrows).
Quantitative Determination of M-SCs and NM-SCs in the Nonbound and Bound Fraction of OM and OB a

After magnet-activated cell sorting (MACS) labeled by incubation with the respective secondary fluorescence-coupled antibodies displacing IgG microbeads from their binding of anti-HNK-1 (M-SCs) and anti-p75NTR (NM-SCs) antibodies. Numbers are expressed as percentage of labelled cells in relation to the total number of cells, while the reduction (nonbound) and enrichment (bound) factor was calculated in reference to the frequencies of antigen-positive cells in the primary cell suspensions (see Table 2). Data represent the mean ± standard deviation. n.d., not done.
Culturing of bound and nonbound fractions for 7 days and phase-contrast inspection revealed the presence of equal numbers of spindle-shaped cells in both the nonbound and bound fractions (Fig. 4) presumably consisting of OECs and Schwann cells, respectively. This correlated well with the quantitative data. Since cultured OECs and Schwann cells share expression of marker molecules, immunocytochemical identification of the one or the other cell type was not feasible.

Phase-contrast micrographs of nonbound (A, B, E, F) and bound fraction cells (C, D, G, H) from the olfactory mucosa (OM) (A–D) and olfactory bulb (OB) (E–H) after in vitro culturing for 7 days. Whereas OM nonbound cells (A, B) contained epithelial cells (A, asterisk) in addition to spindle-shaped presumably OECs (A, B, arrows), the bound fraction contained cells that most likely corresponded to Schwann cells (C, D, arrows). OB cultures of the nonbound (E, F) and bound fractions (G, H) contained spindle-shaped cells corresponding to OECs (E, F, arrows) and Schwann cells (G, H, arrows) in addition to fibroblasts present in both fractions (E–H, arrowheads).
Expansion, Antigenic Expression, and Neurite-Promoting Effects of Olfactory Ensheathing Cells From Olfactory Mucosa and Bulb
Highly purified cultures of OB- and OM-OECs were expanded under the same culture conditions and compared with FN-Schwann cells regarding p75NTR expression (Fig. 5) and neurite-promoting effects (Fig. 6). FN was chosen for Schwann cell isolation, since it does not contain OECs and can therefore be used as control for comparison. Immunocytochemical analysis of cells from passage 5 revealed strong expression of p75NTR in about 95% of the cells of each cell type (Fig. 5A, C, E, G, I, K) in the presence (Fig. 5A, E, I) and absence (Fig. 5C, G, K) of FGF-2. The cells displayed a typical spindle-shaped morphology in FGF-2-treated cultures (Fig. 5B, F, J), while there was a shift to a more flattened phenotype upon removal of FGF-2 (Fig. 5D, H, L). Interestingly, these alterations were more striking in OM-OECs (Fig. 5C, D, arrows) and FN-Schwann cells (Fig. 5K, L, arrows) than in OB-OECs (Fig. 5G, H) that only displayed minor changes and retained their spindle-shaped phenotype in the absence of FGF-2.

Immunostaining of MACS-purified OECs from the olfactory mucosa (OM) (A–D), olfactory bulb (OB) (E–H) and of Schwann cells (SCs) from fibular nerve (FN) (I–L) with anti-p75NTR antibodies in the presence (A, B, E, F, I, J) and absence of fibroblast growth factor-2 (FGF-2) (C, D, G, H, K, L). Note that the vast majority of cells expressed p75NTR under both conditions (A, C, E, G, I, K), confirming the high purity of all preparations. FGF-2 deprivation induced a morphological shift to a more flattened phenotype in OM-OECs (C, D, arrows) and FN-SCs (K, L, arrows) but not in OB-OECs (G, H). Moreover, OM-OECs (C, D), but not OB-OECs (G, H) and FN-SCs (K, L), displayed a heterogeneous morphology of p75NTR-immunoreactive cells (C, D, arrowheads).
Neonatal rat DRG neurons survived in the presence of β-NGF and generated neurites in the presence of all glial cell types (Fig. 6). However, neurite growth, defined as the total area covered by class III β-tubulin-immunopositive neurites and the length of the longest neurites, was significantly different between OECs and Schwann cells but not between OM- and OB-OECs (Fig. 6B, C), indicating that OECs supported neurite growth to a higher extent than Schwann cells.

Qualitative (A, D–F) and quantitative analysis (B, C) of neurite growth of neonatal rat dorsal root ganglion (DRG) neurons cocultured with olfactory mucosa-derived (OM) (A, D) and olfactory bulb-derived (OB) (E) OECs and fibular nerve (FN) (F) Schwann cells (SCs) using anti-class III β-tubulin (A, D–F) and anti-p75NTR (A) antibodies for visualization of neurons and canine glial cells, respectively. Although DRG neurons exhibited substantial neurite growth in the presence of all glial cell populations, both the total area of neurites (B) and the length of the longest neurites (C) in DRG/OM-OEC cultures were significantly increased compared to DRG/FN-SC cultures. DRG/OB-OEC cultures differed significantly from DRG/FN-SC cultures regarding length of the longest neurites (C). (D–F) Display representative DRG explants and schematic division of the area for neurite growth analysis. For the sake of clarity, p75NTR immunostaining identifying canine glia was omitted (D–F). ∗p < 0.05.
Discussion
The olfactory mucosa (OM) is a complex tissue that contains OECs and Schwann cells intermingled with each other and associated with olfactory receptor neurons and trigeminal afferents, respectively (51, 53). Both cell types have not only been shown to display specific properties (25, 44, 46, 71, 77), but they also develop a closely related antigenic phenotype during culturing (21, 38, 83, 85), making it difficult to distinguish between them in vitro. To define the specific regenerative potential of OM- and olfactory bulb (OB)-derived OECs, it is essential to gain control over the cellular composition of the cell cultures that are used for in vitro and in vivo analyses. However, attempts to identify Schwann cells in OEC cultures by the use of specific markers [e.g., calponin (12, 68)] have not proven successful so far (30, 80).
In the present study, we identified and quantified Schwann cells in OEC preparations from the adult canine OM and OB and developed a novel two-step protocol for the generation of highly purified, Schwann cell-free OEC preparations. The novelty of this approach is that freshly dissociated instead of cultured cells that undergo alterations of their antigenic phenotype in vitro were used for quantitative analysis. This isolation procedure was designed in full realization of the fact that transplantation of purified cell preparations into the lesioned CNS may not be optimal for neural repair (6, 45, 63). However, specific effects of OM- and OB-derived OECs cannot be rigorously defined in the presence of contaminating Schwann cells that share at least some of the beneficial effects of OECs and that may be present in different numbers. Adult canine cells were used, since we and others have shown that the dog is an attractive translational model for human research (26, 32, 33, 35–37, 75, 76, 78, 86) and because previous in situ antigen expression mapping of OECs and Schwann cells as direct forerunner to this study was also carried out in the dog (10, 11, 57).
Primary Cell Suspensions of the Adult Canine Olfactory Mucosa and Bulb Contain Substantial and Differing Numbers of Schwann Cells
Several studies have demonstrated that OECs can be isolated from the OM of different species, including, mouse, rat, dog, and humans (2, 10, 34, 55). To address the question of whether OM-OECs and OB-OECs display distinct properties, OM and OB cell cultures were subjected to a comparative in vitro analysis (8, 10, 33, 34, 43) in addition to transplantation into the lesioned nervous system (64, 67). However, although a number of interesting aspects arose from these studies, it remained unclear how far the results had been biased by contaminating trigeminal Schwann cells.
The present analysis revealed that adult canine OM and OB primary cell suspensions substantially differed with respect to the number of Schwann-like cells. Whereas in OM suspensions, presumptive myelinating (HNK-1-positive) and nonmyelinating (p75NTR-positive) Schwann cells correlated to about 21% of the total cells, in OB cell suspensions, only 8% of the cells were immunopositive for the nonmyelinating cell marker p75NTR. Calculation of Schwann cells in relation to CNPase expression, encompassing OECs and Schwann cells (57), demonstrated that Schwann cells and OECs were present at quantitative ratios of 1:1 and 1:3 in the OM and OB cell suspensions, respectively. The higher number of Schwann cells in OM preparations roughly correlates with the presence of myelinated nerve fibers in the OM (10, 53). Although these data clearly show that Schwann cell contamination in OECs is not negligible and should be taken into account when comparing OECs from both origins, direct extrapolation to the rodent and human system is not allowed in the light of species-specific expression patterns (39, 57). Moreover, it is not yet clear how far other cells express Schwann cell-typical antigens, which may result in overestimation of cell numbers. There is evidence, for example, that p75NTR expression is associated with perineural and perivascular cells (32, 57) as well as with pluripotent cells (74). Several studies in humans demonstrated that mucosa stem cells may express p75NTR (54, 56). Thus, it cannot be excluded that the values for nonmyelinating Schwann cells in the OM might be slightly overestimated. However, this limitation does not refer to HNK-1-positive cells and, therefore, does not weaken the principal finding that Schwann cell contamination in OM is substantial and distinct from OB. What also has to be kept in mind is that the present quantitative analysis was done once prior to cell seeding and, therefore, represents a snap shot on the numerical relations of OECs and Schwann cells. Further studies have to determine the proliferation rates of Schwann cell-depleted OEC cultures.
Isolation and Characterization of Schwann Cell-Free Cultures of Purified Olfactory Ensheathing Cells From Adult Canine Olfactory Mucosa and Bulb
The specific depletion of Schwann cells from OM and OB cell suspensions was done on the basis of two recent observations: First, antibody-coupled magnetic particles were suitable for selection of cells from acutely prepared cell suspensions (85), and second, p75NTR-negative OECs upregulated p75NTR expression during culturing (10, 21, 85). We show here that the number of Schwann cells in OM and OB suspensions could be reduced using MACS by about sevenfold, suggesting that the developed protocol efficiently and quantitatively eliminated Schwann cells from OEC suspensions. Efficiency of the procedure was verified by incubation of the nonbound and bound fractions with fluorescent secondary antibodies. The relatively low enrichment factor of the bound fraction of about twofold is most likely explained by the fact that purification was done from primary cell suspensions containing some aggregates and not from a homogenous population of cultured cells (10, 42). As already discussed above, targeting of cells with antibodies against p75NTR may also have eliminated some precursor cells (54, 56, 82) in addition to Schwann cells. Since it could not be excluded that the bound fraction contained OECs, Schwann cells isolated from the FN were used as a control in further comparative in vitro analysis.
Several authors argued that OECs may display advantages over Schwann cells for neural repair (22, 44, 63). However, specific depletion of Schwann cells so far has not been carried out, and the majority of in vitro (16, 69, 79) and in vivo studies (41, 59, 72, 73) analyzed the neurite-promoting effects of the glial cell types separately. To establish OM-OEC, OB-OEC, and FN-Schwann cell preparations suitable for direct comparative analyses, we expanded the cells under identical culture conditions, as described previously (78). Preliminary analysis revealed that the cells displayed a typical spindle-shaped morphology only in the presence of FGF-2, while its removal induced a morphological shift to a more flattened phenotype. This is in agreement with a previous study on canine OB-OECs (42). Interestingly, these morphological alterations were more pronounced in OM-OECs than in OB-OECs, indicating subtle differences between the two populations. Moreover, neurite growth of neonatal rat DRG neurons was higher in the presence of OM-OECs and OB-OECs compared to Schwann cells, while no differences were noted between the two OEC preparations. This provides experimental evidence for previous assumptions implying that OECs may be more beneficial to axonal regeneration than Schwann cells (18, 22).
The isolated Schwann cell-free OEC preparations will be essential for defining the regenerative properties of OM-OECs and OB-OECs. Moreover, they may help to solve the long-lasting controversy of whether OEC are able to form myelin (29). Several in vitro and in vivo studies provided conflicting results regarding the inherent myelinogenic capacity of OECs (4, 13, 24, 31, 50, 58, 59, 72). It was argued that the detected peripheral type myelin after transplantation of OECs may be in fact due to Schwann cells contaminating the cell preparations, rather than to OECs (29). The transplantation of genetically labeled OEC preparations proved that the formed myelin was in fact due to transplanted cells but did not allow to verify that it originated directly from OECs (1, 72).
Taken together, we generated Schwann cell-free OEC preparations and identified subtle differences in the morphology but striking differences in neurite-promoting capacities of OECs and Schwann cells. Currently, both OEC preparations are analyzed and compared with FN Schwann cells regarding remyelination and axon growth-promoting capacities after transplantation into the lesioned spinal cord.
Footnotes
Acknowledgments
This study was supported by a grant from the German Research Foundation to W.B. and K.W. (Research unit 1103, BA815/10-1). S.Z. received a grant from the Akademie für Tiergesundheit e.V. (Bonn, Germany). The excellent technical assistance of K. Schöne, D. Waschke, C. Schütz, and C. Herrmann is gratefully acknowledged. The authors declare no conflict of interest.
