Abstract
Articular cartilage, when damaged by degenerative disease or trauma, has limited ability for self-repair. Recently, many trials have demonstrated that gene therapy combined with tissue engineering techniques would be a promising approach for cartilage regeneration. Bone morphogenetic protein 2 (BMP-2) is an important signal for upregulation of osteogenesis and chondrogenesis of stem cells. Sex-determining region Y box gene 9 (SOX-9) has also been reported as one of the key transcription factors for chondrogenesis. We hypothesized that codelivery of BMP-2 and SOX-9 genes would result in improved efficiency of recovery of normal chondrogenic properties in dedifferentiated chondrocytes. To this aim, we constructed a bicistronic vector encoding the BMP-2 and SOX-9 genes linked to the “self-cleaving” 2A peptide sequence. After gene delivery to dedifferentiated chondrocytes using a microporator transfection system, we confirmed over 65% delivery efficiency of the BMP-2 and SOX-9 genes. According to RT-PCR analysis and Alcian blue staining, simultaneous delivery of BMP-2/SOX-9 resulted in significantly increased expression of chondrogenesis-related markers (type II collagen and aggrecan) and GAG matrix formation compared with individual delivery of the BMP-2 or SOX-9 gene. Six weeks after in vivo transplantation, BMP-2/SOX-9 genes also showed a significant increase in cartilage formation compared with the BMP-2 or SOX-9 gene. These results demonstrate that codelivery of two chondrogenic lineage-determining genes can enhance normal chondrogenic properties of dedifferentiated chondrocytes followed by improved cartilage formation.
Keywords
Introduction
Articular cartilage disease is the most common disorder occurring as a result of either traumatic mechanical destruction, such as automobile accidents and sports injuries, or progressive mechanical degeneration (10,33). Unfortunately, articular cartilage tissue rarely regenerates after degeneration and destruction from those events because it is avascular tissue, with restricted supply of blood, oxygen, and nutrients (19,24,34).
In the last decade, many researchers have devoted every effort to generation of cartilage tissues and several techniques have been reported. For cell-based therapy (3,8,11), autologous chondrocytes or various mesenchymal stem cells derived from bone marrow (9) and adipose tissue (38,39) have been used for generation of cartilage tissue. However, harvest of a sufficient amount of normal chondrocytes from autologous tissue for regeneration of an entire defective area is very difficult. Therefore, expansion of harvested chondrocytes into a large amount of chondrocytes using in vitro long-term culture is important (12,14). Despite use of culture techniques, long-term culture would result in serious impairment of inherent characteristics of chondrocytes, followed by dedifferentiation due to two-dimensional (2D) culture conditions (14). Thus, we sought to recover chondrogenic characteristics of dedifferentiated chondrocytes that undergo expansion by in vitro long-term culture.
Bone morphogenetic protein 2 (BMP-2) belongs to the transforming growth factor (TGF)-β superfamily, a potent inducer of cartilage formation, and is an upstream mediator of chondrogenesis in promotion of chondrocyte differentiation (6,7,36). On the other hand, the sex-determining region Y box gene 9 (SOX-9), a downstream mediator of BMP-2 and hedgehog signaling, is the master transcription factor for chondrogenesis in regulation of collagen type II (Col II) and aggrecan (Agg) transcription in stem cells (16,37). For instance, Bi et al. reported that teratomas derived from SOX-9-/- mouse embryonic stem cells did not form cartilage tissue (1).
In this study, we hypothesized that codelivery of the BMP-2 and SOX-9 genes would improve normal chondrogenic properties in dedifferentiated chondrocytes, compared with delivery of one gene, such as one BMP-2 and one SOX-9 gene. To control codelivery of the BMP-2 and SOX-9 genes, we constructed a single 2A bicistronic vector designed for simultaneous expression of two genes from a single promoter using 2A self-cleavage peptides (25,26,31). We investigated whether dedifferentiated chondrocytes delivered with the BMP-2 and SOX-9 genes not only undergo chondrogenic redifferentiation in vitro but also promote cartilage tissue formation in vivo.
Materials and Methods
Human Chondrocyte Isolation and Culture
Human cartilage tissue was obtained from nasal septum by surgical operations from informed and consenting patients (mean age, 34 years; sex, male and female; number of donors, three patients) with the approval of the CHA University Hospital Ethics Committee. Chondrocytes were isolated from cartilage tissue using enzymatic digestion with phosphate-buffered saline (PBS; Hyclone, Thermo Scientific, Logan, UT, USA) containing 0.2% (w/v) bovine serum albumin (Sigma, St. Louis, MO, USA) and 2 mg/ml collagenase type II (Sigma). Undigested tissue was separated from cells using a 40-mm filter (Phenomenex, Torrance, CA, USA); cells were centrifuged, washed at least three times, and suspended in culture media (described below). Freshly isolated chondrocytes were either cultured in cell culture plates (TPP, Trasadingen, Switzerland) for expansion or cryopreserved in liquid nitrogen. Cultures were incubated in Dulbecco's modified Eagle's medium (DMEM, Gibco BRL, Gaithersburg, MD, USA) supplemented with 10% (v/v) fetal bovine serum (FBS, Gibco BRL) and 100 units/ml penicillin (Gibco BRL) in humidified air with 5% (v/v) CO2 at 37°C. At 80% confluency, cells were harvested by 0.5% trypsin-EDTA (Life Technologies, Grand Islands, NY, USA) and seeded on culture plates (2 × 104 cells/cm2), which was considered a two-dimensional (2D) culture condition. For three-dimensional (3D) culture, cell suspension was centrifuged at 1,200 rpm (rotor diameter: 10 cm) for 5 min, and the cell pellet (2 × 105 cells/pellet) was cultured in humidified air with 5% (v/v) CO2 at 37°C.
Construction of Expression Vectors
Full-length human BMP-2 was tagged with the hemagglutinin (HA) epitope site at the C-terminal region and ligated into pcDNA3.0 (Invitrogen, Carlsbad, CA, USA) to generate the pcDNA3.0-BMP-2/HA expression vector. To produce the bicistronic pcDNA3.1 (+)hyg-BMP-2/HA-2A-FLAG/SOX-9 expression vector, pcDNA3.0-BMP-2/HA was used as a template and amplified using the following primers: CTA AAG CTT GCC ACC ATG GTG GCC GGG ACC CGC TG and CTA GGA TCC TGG ACC TGG ATT GCT TTC TAC ATC CCC AGC CAG TTT GAG TAA ATC AAA GTT AAG AGT TTG TTT GAC AGG AGC GAC AAT TTT AGC GTA GTC TGG GAC GTC GTA TGG GTA (italic letters show the 2A sequences, and underlined sequences indicate the HA sequence). These PCR products were cloned into the pc DNA3.1 (+) hygro vector and digested with BamHI and XhoI. The FLAG/SOX-9 region was amplified by PCR using the pCMV6-ENTRY-FLAG/SOX-9 vector (ORIGENE, Rockville, MD, USA) as a template and cloned downstream of BMP-2/HA-2A. Amplified regions of FLAG/SOX-9 were cloned using BMP-2/HA cloning methods, as described above. The enhanced green fluorescence protein (EGFP) open reading frame (ORF) region, amplified from pEGFP-N3 (Clontech, Mountain View, CA USA), was cloned upstream of SOX-9/FLAG-2A for production of the pcDNA4-EGFP-2A-FLAG/SOX-9 expression vector. To produce the pcDNA4-BMP-2/HA-2A-EGFP expression vector, the BMP-2/HA-2A region, derived from the pcDNA3.1 (+) hyg-BMP-2/HA-2A-FLAG/SOX-9 expression vector, was cloned into pcDNA4 (Invitrogen), and the EGFP ORF region, amplified from pEGFP-N3 (Clontech), was cloned downstream of BMP-2/HA-2A. All of the newly generated expression vectors in this work were confirmed by nucleotide sequencing.
Gene Delivery Using Microporation Transfection
Chondrocytes were transfected with BMP-2 (BMP-2-P5-chondrocyte) and SOX-9 (SOX-9-P5-chondrocyte) and cotransfected with BMP-2 and SOX-9 (BMP-2/SOX-9-P5-chondrocyte) using a Microporator (DigitalBio Technology, Seoul, Korea). First, subconfluent human chondrocytes were harvested and washed with PBS. Cells were resuspended in resuspension buffer R (Sigma) at a density of 1 × 107 cells/ml, followed by incubation of 100 μl of cell suspension solution with 5 μg of plasmid. Subsequently, microporation was performed at a voltage of 1,400 for 20 ms with two pulses. Following electroporation, cells were replated into cell culture dishes and incubated in DMEM supplemented with 10% (v/v) FBS at 37°C in a 5% CO2 humidified atmosphere for 24 h, after which 100 units/ml penicillin were added. Delivery efficiency of the microporator transfection was evaluated by EGFP expression and FACS analysis. Twenty-four hours after EGFP gene transfection, fluorescence was visualized using a Nikon microscope Eclipse 55i (Nikon, Kanagawa, Japan) and quantified by a BD FACS Calibur system (BD Bioscience, San Jose, CA, USA). The cut-off level for definition of EGFP-positive cells was set so that less than 1% of irrelevant antibody stained cells were EGFP positive.
RNA Extraction and Reverse Transcription-Polymerase Chain Reaction
Total RNA was extracted from transfected cells using TRIzol (Invitrogen) and 2 μg of total RNA was used for cDNA synthesis with RT-PreMix (Bioneer, Daejeon, Korea). PCR (MyGenie96 Thermal Block, Bioneer) was performed with PCR-PreMix (Bioneer) under standard PCR conditions: BMP-2, SOX-9, collagen type I (Col I), collagen type II (Col II), aggrecan (Agg), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) primers are shown in Table 1. PCR cycles consisted of an initial denaturation step at 94°C for 5 min, followed by 32 amplification cycles consisting of 30 s of denaturation at 94°C, 30 s of annealing at 62°C, and 1 min of extension at 72°C. Last, a final extension was performed at 72°C for 10 min. PCR products were analyzed by UV irradiation on a 1.2% agarose gel (Invitrogen) stained with ethidium bromide (Sigma).
Nucleotide Sequences of Primer Sets Used for RT-PCR Analysis

bp, base pair.
Western Blots
Cells were incubated with lysis solution from a commercially available Reporter Lysis Buffer (Promega, Madison, WI, USA). Equal amounts of cell extracts were separated in an 8% sodium dodecyl sulfate bisacrylamide gel (Bio-Rad, Hercules, CA, USA) and transferred onto polyvinylidene difluoride (PVDF; Bio-Rad) membranes using the Trans-Blot semi-dry transfer kit (BioRad). Membranes were blocked with 5% nonfat milk (BD Biosciences, Oxford, UK) for 1 h and probed with anti-BMP-2 (1:500, Abcam, Cambridge, UK), anti-SOX-9 (1:1,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), anti-HA (1:1,000, Sigma), anti-mouse FLAG (1:1,000, Sigma), and anti-α-tubulin (1:2,000, Santa Cruz Biotechnology) primary antibodies. Blots were then incubated with horseradish peroxidase-conjugated secondary antibody, and reacting bands were visualized using the WEST-one Western blotting detection system (iNtRON Biotechnology, Seoul, Korea).
Histological Staining
Following harvest of the pellet cultured in vitro for 21 days or in vivo in tissue for 6 weeks, specimens were formalin fixed (Sigma). Blocks of the specimens were embedded with paraffin and cut into 4-μm-thick slices. Sections were stained with 0.5% Alcian blue (Sigma) in 0.1 M HCl (pH 1.0; Sigma) overnight and rinsed with distilled water for microscopy analyses of glycosaminoglycan (GAG) matrix in the regenerated cartilage.
Immunohistochemical Staining
Sections, prepared as described above, were deparaffinized and hydrated by sequential incubations in xylene and ethanol. After washing with PBS for 2 min, the sections were preblocked with 3% H2O2 (Ambion, Austion, TX, USA) for 10 min. Following application of the primary antibody against type II collagen (1/100 polyclonal, Millipore, Billerica, MA, USA), the sections were incubated for 1 h at room temperature, followed subsequently by application of fluorescein isothiocyanate (FITC)-conjugated secondary antibodies (Abcam, Cambridge, MA, USA). Samples were examined with the Zeiss LSM 510 Meta laser scanning confocal microscope (Carl Zeiss Microimaging Inc., Göttingen, Germany).
Chondrocyte Implantation and In Vivo Cartilage Formation
Seven-week-old female athymic mice (BAL b/c-nude, Orientbio, Seoul, Korea) were anesthetized with ketamine (8 mg/kg; Bedford Laboratories, Bedford, OH, USA) and xylazine (1.15 mg/kg; Cervizine, Wildlife Pharmaceuticals, Fort Collins, CO, USA). Each group of nontransfected P5-chondrocytes, BMP-2-P5-chondrocytes, SOX-9-P5-chondrocytes, and BMP-2/SOX-9-P5-chondrocytes was injected at 1 × 106 cells per site into the dorsal subcutaneous spaces of the mice (n = 4 per group) with fibrin gel (Greenplast™ kit, Green Cross, Chungbuk, Korea). Each animal was injected at four sites (right and left at the top and bottom). Six weeks after implantation, the mice were sacrificed and all implants were retrieved. All procedures carried out on animals were subject to the guidelines of an approved protocol from CHA University Institutional Animal Care and Use Committee. Histological specimens were fixed in formalin, embedded in paraffin, and sectioned transversely at a thickness of 4 μm. Specimens were treated with Alcian blue staining, where blue color indicates GAG matrix in cartilage tissues. The area of cartilage formation was measured using an image analysis system (KS400; Zeiss, Munich, Germany) coupled to a light microscope and then quantified as the percentage of cartilage formation area (blue color staining) in the available pore space and other tissue areas [(cartilage area/pore and other tissue areas) × 100%]. Half of each specimen was utilized for evaluation of chondrogenic gene expression by RT-PCR analysis.
Statistical Analysis
At least three independent sets of experiments for each condition were performed in triplicate. Data were pooled and statistically expressed as mean ± SD. One-way analysis of variance (ANOVA) was used for analysis of quantitative values, and the Tukey's post hoc test was used for all pair-wise comparisons among groups. Values of p < 0.05 were considered statistically significant.
Results
Characterization of Dedifferentiated Chondrocytes
Normal and dedifferentiated chondrocytes were defined depending on passage number. Morphology and chondrogenic gene levels were compared in order to determine whether normal chondrocytes [passage 1 (P1)] and dedifferentiated chondrocytes (P5) show different chondrogenic characteristics (Fig. 1). Normal (P1) chondrocytes exhibited a polygonal morphology representing a typical chondrogenic phenotype, whereas dedifferentiated (P5) chondrocytes changed into a fibroblast-like morphology, with hypertropic cell bodies (Fig. 1A). In comparison with P5-chondrocytes, higher expression of Col II and Agg (chondrogenic markers) and lower expression of Col I (osteogenic marker) were observed in P1-chondrocytes (Fig. 1B). These results demonstrate chondrocytes cultured on 2D conditions readily lose their chondrogenic characteristics resulting in dedifferentiation.

Characterization of normal chondrocytes [passage 1 (P1)] and dedifferentiated chondrocytes (P5). (A) Repre sentative images of phenotypes of P1-chondrocytes and P5-chondrocytes. (B) RT-PCR analysis of chondrogenic genes, including collagen type I (Col I), type II (Col II), and the aggrecan (Agg) gene. Scale bar: 100 μm. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is shown as the housekeeping gene.
Gene Delivery on Dedifferentiated Chondrocytes by Microporation Transfecion
We constructed a single 2A bicistronic vector designed for simultaneous expression of two genes of BMP-2 and SOX-9 from a single promoter using 2A peptides (Fig. 2A) (25,26,31). To avoid the potential risks associated with a viral vector, P5-chondrocytes were transfected using a nonviral delivery system by the microporator. Fluorescence images of BMP-2/EGFP vector-delivered chondrocytes confirmed that our 2A bicistronic vectors were introduced successfully and showed continuous expression in P5-chondrocytes (Fig. 2B, a–c). Delivery efficiency of various tested conditions was evaluated by flow cytometric analysis after transfection of a plasmid encoding EGFP, as described in a previous study (15) (data not shown). The optimized transfection conditions were determined to be a voltage of 1,400 V for 20 ms and two pulses. According to FACS analysis, the EGFP-positive population was observed to be above 65% (Fig. 2B, d).

Construction of the 2A bicistronic vector and verification. (A) Schematic of 2A bicistronic vectors. BMP-2, bone morphogenetic protein; HA, hemaglutinin; EGFP, enhanced green fluorescent protein; SOX-9, sex-determining region Y box gene 9. (B) Representative photomicrograph of P5-chondrocytes with pcDNA4-BMP-2/HA-2A-EGFP using a microporator. (a) Phase contrast image. (b) Fluorescence image. (c) Merged image. (d) Flow cytometry histograms of EGFP transfected P5-chondrocytes (shaded), compared with untransfected P5-chondrocytes (unshaded). Scale bar: 100 μm. (C) Representative images of RT-PCR for P5-chondrocytes (control), BMP-2, SOX-9 delivery groups and the BMP-2/SOX-9 codelivery group on 2D culture (4 and 14 days) and 3D culture (8 and 21 days). (D) Representative images of Western blots in the control, one BMP-2, SOX-9 transfection groups, and BMP-2/SOX-9 codelivery group on 2D culture at 7 days. α-Tubulin was used as loading control.
Expression of BMP-2 and SOX-9 Encoding Bicistronic Vector
As shown at Figure 2C, RT-PCR analysis was performed in P5-chondrocytes transfected with BMP-2, SOX-9, and BMP-2/SOX-9, respectively. Under 2D (4 and 14 days) and 3D culture conditions (8 and 21 days), the BMP-2 and SOX-9 groups exhibited BMP-2 and SOX-9 expression, respectively. The BMP-2/SOX-9 codelivery group expressed both BMP-2 and SOX-9, whereas the control group did not. This result demonstrates that BMP-2, SOX-9, and BMP-2/SOX-9 genes were successfully transfected and were stably expressed in P5-chondrocytes under 2D as well as 3D culture conditions over the incubation period. In addition, Western blot analysis confirmed that one BMP-2, SOX-9, and the BMP-2/SOX-9 codelivery group successfully expressed BMP-2, SOX-9, and both BMP-2 and SOX-9 proteins, respectively, including HA and FLAG epitope sites at 7 days after transfection (Fig. 2D). Expression of HA and FLAG indicates that the detected proteins were a product of the transfected vectors, as opposed to the endogenously produced proteins in transfected chondrocytes. This result demonstrates that the BMP-2/SOX-9 vector linked by the 2A peptide was stably delivered into P5-chondrocytes.
In Vitro Redifferentiation of Dedifferentiated Chondroctyes by BMP-2/SOX-9 Codelivery
In order to confirm whether BMP-2 and SOX-9 delivery indeed induced redifferentiation of dedifferentiated chondrocytes in 2D (4 and 14 days) as well as 3D (8 and 21 days) culture conditions, characteristic genes of chondrocytes (Col II and Agg) were investigated and compared by RT-PCR analysis (Fig. 3). In 2D culture conditions, P5-chondrocytes in all groups, including the BMP-2/SOX-9 codelivery group, rarely expressed Col II and Agg at 4 and 14 days. On the other hand, 3D culture conditions induced strong expression of both Col II and Agg in all groups, especially as BMP-2/SOX-9 was delivered (Fig. 3B). Alcian blue and immunohistochemical staining (Fig. 3C, D) showed significantly higher expression of GAG and Col II protein in the BMP-2/SOX-9 codelivery group, compared with the control, BMP-2, and SOX-9 groups alone. Taken together, these results demonstrated that BMP-2/SOX-9 codelivery resulted in significantly enhanced redifferentiation of dedifferentiated chondrocytes, followed by higher expression of chondrocyte marker proteins.

In vitro bicistronic vector-induced chondrogenic redifferentiation of P5-chondrocytes. (A) Representative images of RT-PCR in the control, one BMP-2, SOX-9 delivery groups, and BMP-2/SOX-9 codelivery group on 2D culture at 4 and 14 days. (B) Representative images of RT-PCR in the control, one BMP-2, SOX-9 delivery groups, and BMP-2/SOX-9 codelivery groups on 3D culture at 8 and 21 days. (C) Alcian blue staining of histological sections formed on 3D culture of the control, one BMP-2, SOX-9 delivery groups, and BMP-2/SOX-9 codelivery group for 21 days. (D) Type-II collagen immunostaining of histological sections formed on 3D culture of the control, one BMP-2, SOX-9 delivery groups, and BMP-2/SOX-9 codelivery groups at 21 days. Scale bar: 100 μm. n = 4.
In Vivo Cartilage Formation by BMP-2/SOX-9 Codelivery
For in vivo transplantation, the BMP-2, SOX-9, and BMP-2/SOX-9 codelivery groups were injected with fibrin gels (fibrinogen to thrombin ratio = 1:1) into the dorsal part of BAL b/c nude mice (n = 4 per groups). After 6 weeks of transplantation, mice were sacrificed and the retrieved samples were biopsied. Alcian blue staining (Fig. 4A, B) and RT-PCR analysis (Fig. 4C) revealed that the control and BMP-2 delivery groups generated immature cartilage tissues, while the SOX-9 and BMP-2/SOX-9 codelivery groups exhibited formation of mature cartilage tissue with lacuna structure. In particular, the BMP-2/SOX-9 codelivery group produced native cartilage-like tissue with strong GAG expression and lacuna structure. Figure 4B shows quantitative values of cartilage tissue formation. The BMP-2/SOX-9 codelivery group produced approximately twofold, fourfold, or ninefold higher cartilage tissue formation, compared with the control, BMP-2, and SOX-9 delivery groups, respectively (*p < 0.05) (Fig. 4B). In addition, RT-PCR results confirmed that the BMP-2/SOX-9 codelivery group induced higher expression of chondrogenic markers, Col II and Agg, compared with others (Fig. 4C).
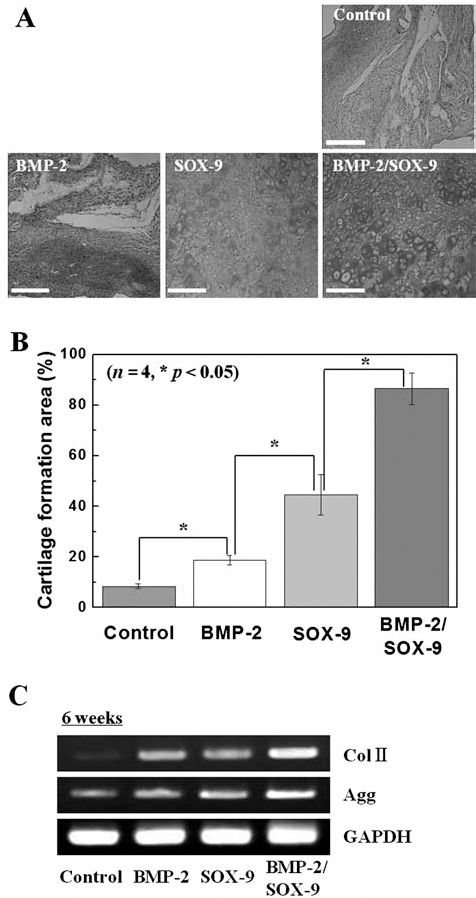
In vivo bicistronic vector-induced redifferentiation of P5-chondrocytes. (A) Alcian blue staining of histological sections formed by implantation of the control, one BMP-2, SOX-9 delivery groups, and BMP-2/SOX-9 codelivery group into the dorsal subcutaneous parts of athymic mice for 6 weeks. Scale bar: 50 μm. (B) Area of cartilage formation in the implants at 6 weeks post implantation. The difference was statistically significant (*p < 0.05; n = 4). (C) RT-PCR analysis of chondrogenic marker genes in the formation tissue at 6 weeks postimplantation.
Discussion
In the present study, the goal of codelivery of BMP-2 and SOX-9 was not only to induce more efficient re differentiation of dedifferentiated chondrocytes than that achieved with individual BMP-2 and SOX-9 delivery but also to investigate the question of whether the BMP-2/SOX-9 codelivery group can maintain redifferentiation status under both 2D and 3D culture conditions. First, differentiated chondrocytes were defined as having a high passage number (P5) of chondrocytes and were induced by 2D culture conditions. Rapid loss of the characteristic phenotype (polygonal morphology) and chondrogenic gene expression in P1-chondrocytes was observed during 2D culture conditional passages (Fig. 1A, B). This observation is similar to those of previous reports concluding that use of a monolayer culture system results in rapid loss of the characteristic phenotype of chondrocytes, followed by hypertropic cell morphology and a decrease in synthesis of type II collagen (12,14,17).
The phenomenon of dedifferentiation in chondrocytes is a major difficulty in application of chondrocyte expansion for cartilage tissue regeneration. Several culture conditions, including use of cytokines, pellet culture, 3D suspension culture, and bioreactor systems, have been suggested by many researchers as an alternative to conventional 2D culture conditions. These culture conditions are effective for use in maintenance of the cellular phenotype of chondrocytes; however, they are not satisfactory for achievement of sufficient expansion of cell numbers. Several cytokines or growth factors, including insulin-like growth factor I (IGF-I) (30), platelet-derived growth factor (PDGF) (32), fibroblast growth factor 2 (FGF-2) (21), and bone morphogenetic protein 2 (BMP-2) (28) have the capacity for enhancement of redifferentiation of dedifferentiated chondrocytes, accompanied by a new extracellular matrix. However, due to a short half-life of activity and high cost, use of cytokine treatment would not solve fundamental problems. Meanwhile, use of 3D culture methods in chondrocytes resulted in maintenance of both chondrogenic properties and chondrogenic redifferentiation because it allows for establishment of the microenvironments observed in native tissues, including low oxygen tension and 3D cellular interaction, when compared with 2D conditions (18,20,22,35). DeLise et al. reported that use of a 3D environment in pellet culture can result in much higher numbers of cell–cell interactions, resulting in higher redifferentiation and matrix synthesis (5).
In the present study, we hypothesized that codelivery of the BMP-2 and SOX-9 genes would improve normal chondrogenic properties in dedifferentiated chondrocytes. To this aim, we constructed a single bicistronic vector encoding the BMP-2 and SOX-9 genes linked to the “self-cleaving” 2A peptide sequence (Fig. 2A) (25,26). According to RT-PCR results in each BMP-2, SOX-9, and BMP-2/SOX-9 codelivery group (Fig. 3A, B), the effect of gene delivery on P5-chondrocytes was investigated by comparison under 2D and 3D conditions. Despite BMP-2/SOX-9 codelivery, use of 2D culture conditions resulted in weak expression of Col II and Agg. However, use of 3D culture conditions resulted in strong expression of Col II and Agg, indicating that use of 3D culture conditions is critical to redifferentiation of chondrocytes, even after gene delivery. Of particular interest, expression levels of Col II and Agg showed attenuation at 21 days, presumably due to the long period of culture, which is coincident with results of the previous study (14). Compared with the other groups, the BMP-2/SOX-9 group cultured under 3D conditions showed stronger GAG matrix and type II collagen expression, demonstrating stimulation of more efficient redifferentiation of the BMP-2/SOX-9 codelivery group under 3D culture conditions (Fig. 3C, D).
Various reports have demonstrated an association of BMP-2 and SOX-9, either for recovery of chondrogenic properties or chondrogenic differentiation. For example, Sailor et al. reported on BMP-2 induced chondrogenic redifferentiation in chondrocytes, followed by enhancement of Col II expression and proteoglycan synthesis (27). In another study, BMP-2 was reported to induce an increase in expression of Col II and aggrecan and to stimulate proteoglycan synthesis up to threefold in vivo, in damaged knee joints of mice (2). And, SOX-9 overexpression in hMSCs led to upregulation of its known cartilage-specific target genes (Col II and Agg) with synthesis of GAGs (13). Cao et al. reported on bone marrow mesenchymal stem cell (BMSCs)-based SOX-9 gene therapy for enhancement of the healing of full-thickness articular cartilage defects in vivo (4). However, this trial was not conducted for recovery of chondrogenic properties from dedifferentiated chondrocytes using codelivery of the BMP-2 and SOX-9 genes.
In this study, we observed that gene delivery by BMP-2 and SOX-9 rarely affects chondrogenic redifferentiation under in vitro culture conditions (Fig. 3A, B). However, after in vivo transplantation, redifferentiation of P5-chondrocytes was observed more clearly (Fig. 4A, B). BMP-2 delivered chondrocytes generated immature cartilage tissues, while SOX-9 transfected chondrocytes stimulated formation of mature cartilage tissue with a lacuna structure. SOX-9 delivery appears to provide effects that are more conducive to achievement of chondrogenic redifferentiation. This is presumably due to the fact that the SOX-9 signal, as a transcription factor of the downstream of BMP signal, plays a direct pivotal role in regulation of chondrogenic redifferentiation, compared with BMP-2 (23,29). Meanwhile, the BMP-2/SOX-9 codelivery group exclusively showed the highest areas of GAG expression and cartilage formation (p < 0.05) with lacuna structure, verifying chondrogenic redifferentiation (Fig. 4A, B). This result might be associated with that of a previous report in which combination of BMP-2 and SOX-9 showed a significant synergistic effect on chondrogenesis in mouse embryonic fibroblasts (MEFs) (23). BMP-2 and SOX-9 are believed to play the role of initiator and inducer, respectively, during chondrogenic redifferentiation. Therefore, codelivery using BMP-2/SOX-9 would be a useful tool for recovery of chondrogenic characteristics of differentiated chondrocytes.
In summary, BMP-2 and SOX-9 genes were delivered simultaneously into dedifferentiated chondrocytes using the 2A bicistronic vector encoding BMP-2 and SOX-9 from a single promoter by microporation transfection, resulting in over 65% delivery efficiency. Expression of genes and proteins of delivered BMP-2 and SOX-9 occurred during the incubation period under both 2D and 3D culture conditions. For in vitro study, chondrogenic marker expression was found to be significantly higher in the BMP-2/SOX-9 codelivery group, especially under 3D culture conditions. In addition, when compared with BMP-2 and SOX-9 alone, more significant induction of in vivo cartilage formation was observed in the BMP-2/SOX-9 codelivery group. Thus, in the effort to enhance chondrogenic redifferentiation of dedifferentiated chondrocytes, both in vitro and in vivo, codelivery of BMP-2 and SOX-9 is a useful tool.
Footnotes
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ITC, & Future Planning (No. 2012M3A9B4028569). The authors declare no conflicts of interest.
