Abstract
Proper revascularization after transplantation is assumed to be crucial for appropriate islet graft function. We developed a novel noninvasive imaging method, based on adenoviral transduction of islets with a hypoxia responsive reporter gene, for continuous in vivo monitoring of hypoxia in islet grafts in a mouse model. In addition, morphological data were obtained from a deceased patient previously subject to intraportal transplantation. We detected only transient hypoxia in a minority of the animals transplanted. Importantly, a clear response to hypoxia was observed in vitro after removal of the islet grafts on day 28 after transplantation. Also, the morphological data from the deceased patient demonstrated an extensive revascularization of the transplanted islets. In fact, no differences could be seen between native islets, in pancreas biopsies taken prior to islet isolation, and transplanted islets regarding the number, distribution, and shape of the blood vessels. However, fewer small islets (diameter <39 μm) were found in the liver compared to those found in native pancreases. Notably, an absolute majority of the transplanted islets were found remaining within the venous lumen, in direct contact with the vessel wall. In conclusion, the results presented show less pronounced islet graft hypoxia after subcapsular transplantation than previously reported using more invasive methods. Also, formation of an extensive intraislet capillary network, similar to that seen in native islets in the pancreas, was seen after clinical islet transplantation.
Introduction
During the last 10 years, the results of islet transplantation have improved gradually (41). However, in most cases, patients have to receive islets from multiple donors, and long-term results are still not as good as with pancreas transplantation (40). The reason for this is not fully understood, but it has been repeatedly shown that the islets fail to establish proper revascularization after experimental transplantation, resulting in a marked oxygen deficiency remaining even long term after implantation, possibly rendering the transplanted islet even more prone to other insults (6–10, 21). It has also been emphasized that the islet size influences the outcome, suggesting enhanced engraftment for smaller islets (28, 31).
With the aim to substantiate these findings, a novel method using a reporter gene activated by hypoxia-inducible factor-1α (HIF-1α) for continuous monitoring of hypoxia was developed and applied in an experimental islet transplantation model. In contrast to other currently available techniques for studying islet graft blood perfusion and oxygen tension, the presented technique is noninvasive, thus neither changing the inbound milieu of the graft nor requiring the graft to be removed for analysis (6, 9, 10, 35).
Also, morphological data obtained from a deceased patient who had received three islet transplants are presented. With the intention to evaluate islet engraftment, we determined the islet size, the degree of revascularization, and the relation of the islets to the surrounding liver tissue.
Materials and Methods
Patient History
The patient was diagnosed with type 1 diabetes at the age of 16. At the age of 46, he was transplanted with a kidney that had satisfactory function until the patient died at 54 years of age. He received three islet transplantations (January 2002, May 2002, and May 2006) from a total of four donors (islets from two donors were transplanted January 2002). A total of 950,000 islet equivalents (IEQs) were transplanted. At the time for the first islet transplantation, the patient's immunosuppression was changed to a steroid-free regimen with daclizumab, tacrolimus, and sirolimus. Six months after the first islet transplantation, sirolimus was replaced with mycophenolate mofetil because of toxic alveolitis. After the second islet transplantation, he became insulin independent and remained so for almost a year; thereafter, he was reinstated on a low-dose insulin therapy due to postprandial hyperglycemia. The maximum meal-stimulated plasma c-peptide concentration measured was 3.99 ng/ml. Four years later and due to a slowly deteriorating islet graft function, he received a third islet infusion, resulting in a meal-stimulated c-peptide concentration of 2.46 ng/ml and a glycated hemoglobin index (HbA1c) of 6.7%.
Unexpectedly, the patient died 6 months after the last islet transplant. An autopsy was performed approximately 72 h postmortem, establishing acute myocardial infarction as the immediate cause of death.
Clinical Islet Transplantation
Morphological Examination
The liver was immersed in 5% buffered formaldehyde. Altogether, 40 blocs, each with a mean area of 3 cm2, were obtained from both the right and left lobes and processed for routine histology. Also, islets from biopsies taken from the donor pancreases prior to islet isolation were included in the study. Immunohistochemical staining for chromogranin A (NeoMarkers, Thermo Fisher Scientific, Inc., Fremont, CA) was used to identify transplanted islets (defined as >5 endocrine cells). The size of the islets was determined at 10x magnification using an ocular containing a scale. The longest (x) and shortest (y) diameters of each islet were measured, and the area was calculated using the formula: 0.5(x) ∗ 0.5(y) ∗ pi. The diameter of an assumed spheroid islet was calculated using the formula: √(0.5(x) ∗ 0.5(y) ∗ pi / pi) ∗ 2. Sections were stained with CD31 (Dako Denmark A/S, Glostrup, Denmark), and the level of vascularization was determined by comparing the density of CD31-positive vessels in the islets with the density in the adjacent liver tissue. Islets were also categorized according to their position in the liver tissue (Fig. 1).
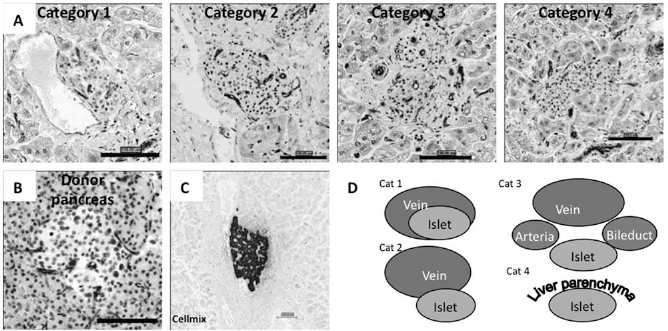
Islet categories and vascularization. (A) Histological sections of typical islets found in the liver of a deceased patient representing the four different categories based on their relation to surrounding structures, stained with CD31 antibodies. Scale bars: 100 μm. (B) Histological section of a typical islet from one of the donor pancreases, stained with CD31 antibodies. Scale bar: 100 μm. (C) Histological section of an islet found in the liver where hepatocyte-like remodulation seems to have occurred, stained with chromogranin A. Scale bar: 100 μm. (D) Schematic illustration of the four different islet categories.
Experimental Islet Transplantation
Islet Preparation
Mouse islets from a total of 48 mice were isolated and handpicked as previously described (3). Female C57BL/6/Bkl mice aged between 12 and 24 weeks (Scanbur, Sollentuna, Sweden) were used as donors. If more than one murine donor was used in an experiment, the islets were pooled.
Human islets from three different human donors were isolated and purified on a continuous density gradient as previously described (16, 39). Islets were not pooled. Donors were of age 47–58, had a body mass index (BMI) of 22–29, and were of blood group A, B, or O. The human islets were used for in vitro experiments to confirm similar behavior to murine islets regarding transduction and response to hypoxia.
All studies were approved by the human and animal ethics committees at Uppsala University Hospital/ Uppsala University.
Viral Vectors
A recombinant hypoxia-responsive promoter-mouse cytomegalovirus (HRE-mCMV) was PCR-amplified from pLA-HRE-mCMV-lacZ (38) (Dr. Chumakov, Lerner Research Institute, Cleveland, OH) and incorporated into pShuttle (Stratagene) together with luciferase (Luc or Luc2CP; Promega) to construct pShuttle (HRE-Luc/ Luc2CP). Ad(HRE-Luc2CP) and Ad(HRE-Luc) were produced through homologous recombination between shuttle plasmids and pAdEasy1. Ad(CMV-GFP) and Ad(CMV-Luc) have been described earlier, and all vectors were produced, purified, titrated, and stored as before (11, 12).
Gene Transduction
Cell Lines
Four different cell lines were used: the mouse intestinal secretinoma cell line STC-1 (kind gift from Dr. D. Hanahan, UCSF, San Francisco, CA), the human cervical cancer cell line HeLa (ATCC, Manassas, VA), the normal human fibroblast cell line 1064SK (ATCC), and the human pancreatic carcinoid cell line BON (a kind gift from Dr. J. C. Thompson and Dr. C. M. Townsend, University of Texas Medical Branch, Galveston, TX). To reach a transduction efficiency of >90%, the cells were transduced with adenovirus (Ad) vectors at a multiplicity of infection (MOI) of 10 (BON, STC-1) or 50 (HeLa, 1064SK) fluorescent forming units (FFU) per cell in a volume of 100–500 μl of complete culture medium in a plastic conical tube for 2 h at 37°C. Thereafter, complete culture medium was added, and the cells were plated in six-well plates and incubated for another 24 h at 37°C before they were used in any experiment.
Islets
The islets were manually counted and sorted to avoid contamination of exocrine material. Human and mouse islets were transduced 1–9 and 0–2 days after isolation, respectively, by mixing the viral vector and the islets in a droplet of culture medium at 37°C for 90 min at MOIs ranging from 150 to 600 FFU/cell. Thereafter, fresh culture medium was added, and the islets were incubated for another 24 h in a 3-cm Petri dish before any experiments were started. The percentage of cells transduced was assessed by flow cytometry, while integrity and morphology were evaluated by light microscopy.
Induction of Hypoxia
Cells or islets were put in a hypoxia chamber (In Vivo2 400 Hypoxia workstation, Biotrace, Bridgend, UK, with 5% CO2 in humidified air at 37°C) and kept at different oxygen concentrations (0.2–15%) for 1–24 h. Cells or islets were lysed in the hypoxic environment and then immediately taken out and snap-frozen in liquid nitrogen to minimize the protein loss. Luciferase expression in cell lines was analyzed as previously described (11).
Experimental Transplantation Procedure
C57BL/6/Bkl mice aged 12–24 weeks (Scanbur, Sollentuna, Sweden) were made diabetic by IV injection of alloxan (75 mg/kg BW). Animals were anesthetized with isoflurane, and 200–400 islets were transplanted under the left kidney capsule. The animals were left to recover for approximately 1 h before imaging studies were performed. Fifteen animals received Ad(HRE-Luc2CP)-transduced mouse islets, and four received Ad(CMV-Luc)-transduced mouse islets.
In Vivo/In Vitro Imaging of Transduced Islets
For in vivo imaging, an IVIS®-100 system with LivingImage® software (Caliper Life Sciences, Hopkinton, MA) was used. Immediately before imaging, 250 μl of d-luciferin (12 mg/ml) (Caliper Life Sciences) was injected IP in each animal. Pictures were then taken at intervals of 5 min. Exposure time was 1 min, medium binning was used, and f/stop was set to 1. For in vitro analysis, the removed islet grafts were placed in complete culture medium in 3-cm Petri dishes. Just before imaging, 30 μl of d-luciferin (12 mg/ml) was added. Subsequently, retrieved islet grafts were put in the hypoxia chamber for 3 h at 1% oxygen concentration before renewed measurements were performed.
Statistics
Data are presented as median (range). The Mann–Whitney U test was applied for statistical analysis when comparing the size of the transplanted islets found in the liver with the islets from the donor pancreases. For comparison between the four categories of transplanted islets, the Kruskal–Wallis one-way analysis of variance was used with a level of significance of p < 0.05. No post hoc test was used as no significant differences could be observed.
Results
Clinical Islet Transplantation
Islet Localization
A total of 393 islets were identified for morphological studies and divided into four categories (Table 1 and Fig. 1): (1) islets found within a venous lumen, (2) islets found in direct contact with the venous endothelial wall but not inside the lumen, (3) islets in the immediate vicinity of a vein, typically in a portal zone, and (4) islets embedded in the liver parenchyma.
Localization and Vascularization of Transplanted Islets Found in the Liver of a Deceased Patient, Divided Into Four Categories Depending on Their Relation to Surrounding Tissue

Islet Vascularization
All islets stained for CD31 showed positive cells, and in most cases, extensive capillary networks were found penetrating the islets. An absolute majority of the islets had a vascular density higher or comparable to the surrounding liver parenchyma. There were no significant differences between the four different categories regarding vascularization (Table 1 and Fig. 1). Also, compared to the islets from the donor pancreas biopsies, no differences could be seen between native and transplanted islets regarding the number, distribution, and shape of the vessels (Fig. 1).
General Morphological Findings
In the vast majority of islets, no infiltration of immunological cells was seen. Liver cells in the immediate vicinity of the islets often appeared steatotic. Some of the islets were surrounded by fibrosis. Occasionally, signs of liver remodulation were observed, that is, the lumen of a vein containing an islet was completely filled with clusters of hepatocyte-like cells not staining for chromogranin A (Fig. 1).
Islet Size and Shape
For islets stained with chromogranin A (n = 133), the median diameter was 87.7 μm (range 32–345 μm). There was no significant difference (p < 0.05) in size of the islets in the four categories, having a median diameter (μm) of 80.3 (32–311), 90.5 (35–345), 98.0 (42–325), and 70.3 (45–232) in categories 1–4, respectively. Most of the islets (84%, n = 112) were less than 160 μm in diameter. A small number (4%, n = 5) of large islets (diameter >280 μm) were also found. There was no significant difference in islet size or shape when the same measurements were made on the islets stained with H&E (n = 177).
The islets found in the donor pancreases tended to be smaller with a median diameter of 64.8 μm (range 24–245 μm) (p = 0.0001) when compared to those found within the liver. Notably, the smallest islets (diameter 20–39 μm) constituted 25% (n = 30) of the islets in the donor pancreas, while only 5% (n = 6) of the transplanted islets were found within the liver (Fig. 2).

Islet size distribution. Transplanted islets found in the liver of a deceased patient versus islets recovered from biopsies of the donor pancreases prior to isolation, stained with chromogranin A. The diameter was calculated from the area.
The vast majority of the islets found in the liver had an elliptical shape. The median (x)/(y) ratio of the islets was 1.8 (range 1.0–5.0). There was no significant difference in the shape between the four categories of islets, with an (x)/(y) ratio of 1.8 (1.0–4.0), 1.7 (1.0–5.0), 1.7 (1.1–3.4), and 1.5 (1.1–2.6) in categories 1–4, respectively. The islets from the donor pancreas appeared more spherical, with a median (x)/(y) ratio of 1.3 (1.0–4.7).
Experimental Islet Transplantation
Adenoviral Transduction of Islets
Islets were efficiently transduced at an MOI of 300 (27% transduced cells), remained intact in culture, and responded accurately to glucose in perifusion assays. As previously described (5), a slight impairment in glucose-stimulated insulin release from transduced islets was observed when compared with nontransduced control islets, with no difference between Ad(HRE-Luc2CP)- and Ad(CMV-luc)-transduced islets. Even so, the stimulation index of the transduced islets was above 20, and the islets cured diabetic nude mice as efficiently as did the nontransduced islets when transplanted under the kidney capsule. An MOI of 300 was chosen for all islet transduction experiments presented below.
Characteristics of the HRE Reporter Gene
Response from the HRE promoter was observed at low oxygen concentrations (Fig. 3). Hypoxic incubation time to reach maximal signal was 24 h, but there was an increase in luciferase activity already after 3 h (Fig. 3). The oxygen concentration in the chamber correlated well with the oxygen tension in the culture medium, and the cutoff value for activation of the HRE promoter at 1% oxygen concentration corresponded to an oxygen tension of approximately 10 mmHg [measured with a blood-gas analyzer (Avl Omnitm9, Avl List GmbH Medizintechnik, Graz, Austria)]. Islets transduced with Ad(HRE-Luc) showed an approximately 50-fold induction in response to hypoxia, while the cell lines (HeLa, BON, STC-1, and 1064SK) responded with a 30- to 150-fold induction.

Luciferase induction in Ad(HRE-Luc2CP)-transduced cells. (A) HeLa cells were transduced with adenovirus containing recombinant hypoxia-responsive promoter and luciferase [Ad(HRE-Luc2CP)] and incubated at different oxygen concentrations (%) for 24 h. Cells were then lysed, and luciferase activity was measured. The activity was adjusted for by the total protein content of the lysates [relative luminescence unit (RLU)/μg protein] and expressed in relation to the activity at 0.2% oxygen (set to 100). (B) HeLa cells were transduced with Ad(HRE-Luc2CP) and incubated for different times in 0.2% oxygen. Cells were then lysed, and luciferase activity was measured. The activity was adjusted for by the total protein content of the lysates (RLU/μg protein) and expressed in relation to the activity obtained after 24 h (set to 100).
Islet Transplantation and In Vivo Response of Reporter Gene
The functional quality and integrity of the islets were clearly demonstrated by the fact that all diabetic mice (mean pre-tx blood glucose 26.4 ± 1.7 mmol/L) became normoglycemic after transplantation (mean post-tx blood glucose 9.0 ± 0.8) and by the reoccurrence of hyperglycemia following islet transplantectomy (mean post-ectomy blood glucose 19.9 ± 1.6).
A hypoxic response with reporter gene expression could be observed in only 33% of the animals transplanted with Ad(HRE-Luc2CP)-transduced mouse islets (n = 15). In these animals, the signal reached its maximum 2–3 days after implantation and became nondetectable 7 days after transplantation (Fig. 4, mouse to the right and graph). To verify reporter gene expression prior to transplantation, some of the mice received Ad(HRE-Luc2CP)-transduced islets that showed a response to hypoxia in vitro. In one of these animals, we detected a markedly higher hypoxic signal from the graft on the day of transplantation. However, this signal also decreased and became nondetectable after transplantation. At the same time, all of the mice receiving Ad(CMV-Luc)-transduced islets (n = 4) had a stable light emission (Fig. 4, mouse to the left).

Hypoxia in mouse islets during the first days after transplantation (all values in photons/s/cm2/sr; scale bars in gray). Freshly isolated mouse islets were transduced with either Ad(HRE-Luc2CP) or adenovirus containing cytomegalovirus-luciferase [Ad(CMV-Luc)] at a multiplicity of infection (MOI) of 300. The following day (24 h later), they were transplanted under the kidney capsule of syngeneic mice. Luciferase activity was measured approximately 1 h after transplantation (day 0) and 1, 2, 3, and 7 days after transplantation in the same mice. (A) The mouse to the left is an example of a mouse transplanted with Ad(CMV-Luc)-transduced islets, while the mouse to the right was transplanted with Ad(HRE-Luc2CP)-transduced islets. The graph shows the light emission (photons/s/cm2/sr) from the area of the graft [region of interest (ROI)] from the five mice transplanted with Ad(HRE-Luc2CP)-transduced islets that responded with reporter gene expression in vivo. (B) The mice were sacrificed, and the grafted islets were recovered. Luciferase activity was measured in the islets directly after removal (two top images) and after 3 h in the hypoxia chamber (1% O2) (two bottom images). The panel to the left shows an example of Ad(CMV-Luc)-transduced islets, while the panel to the right shows Ad(HRE-Luc2CP)-transduced islets.
At day 28 after transplantation, the graft-bearing kidneys of all animals (n = 19) were removed and exposed to hypoxia. No signal was detectable in the Ad(HRE-Luc2CP)-transduced grafts (n = 15) immediately after graftectomy, except from the animal with the highest signal on day 0, which had a weakly positive signal. However, after 3 h of incubation in vitro at 1% oxygen concentration (pO2 < 10 mmHg), clear signals were detected from all of the Ad(HRE-Luc2CP)-transduced grafts (Fig. 4), including the grafts from the 10 animals that did not display any reporter gene expression in vivo. The excised Ad(CMV-luc)-transduced grafts (n = 4), showed a somewhat weaker signal after incubation in hypoxia (Fig. 4).
Discussion
An absolute majority of the transplanted islets in the liver of the deceased recipient were found within the venous lumen, in direct contact with the vessel wall. This finding is in concordance with the results of previous studies both in humans (13, 42) and in nonhuman primates (1, 2, 19, 20, 22–24, 26, 29, 46), demonstrating islets within the vascular compartment after intraportal transplantation.
In contrast, most studies in small-animal models describe islets incorporated in the liver parenchyma (25). It may be speculated that, in small animals, where the transplanted islets completely block the blood flow of small portal branches, a subsequent degeneration of the venous wall may occur allowing direct contact between the islets and the surrounding liver cells. This type of islet engraftment was only rarely observed in the liver of the deceased patient. However, the observed accumulation of hepatocytes within the vessel lumen adjacent to the transplanted islets most likely represents persistent remodulation of the liver parenchyma. In addition, the implanted islets appeared to have affected some of the surrounding hepatocytes, resulting in periportal steatosis, a phenomenon probably due to local insulin secretion (4, 32).
The size of the transplanted islets found in the liver as well as the islets in the donor pancreases was in concordance with other previous studies of native islets (17, 18). However, a discrepancy regarding the number of the smallest islet was observed. Islets with a diameter of less than 50 μm are not included in the currently applied algorithm for determining the number of IEQ after islet isolation. These islets represent a small volume of the total islet volume; even so, it has been hypothesized that they contribute significantly to the islet graft function due to superior engraftment and survival when compared to large islets (28, 31). No evidence supporting this hypothesis was found in the present study. In contrast, fewer small islets (diameter <39 μm) were found in the liver compared to that found in native pancreases. It has previously been described that transplanted islets may engraft as groups of islets forming large clusters (45); if so, conglomerates of small islets could have been detected as single islets. Alternatively, a preferential loss of smaller islets could occur during isolation and in vitro culture or after transplantation.
Notably, random sectioning, that is, cutting through the islets at any level, does not necessarily represent the widest diameter, theoretically underestimating the diameter to only around 80% of its true size. The average diameter of the transplanted islets of 107 μm should therefore theoretically be corrected to 136 μm. The shape of the engrafted islets was somewhat more elliptical than the shape of native islets. This finding could possibly be explained as a result of the islets physically being squeezed into the small/end branches of the portal vein during transplantation.
Most importantly and in sharp contrast with previous experimental studies (9, 10, 21, 33, 34), a majority of the transplanted islets found in the liver of the diseased patient were penetrated by an extensive capillary network similar to that seen in islets in the pancreas. Our experimental studies also suggest a less pronounced islet graft hypoxia after subcapsular transplantation than previously reported using more invasive methods (6–10, 21). Importantly, there are also recent studies supporting our findings, showing that transplanted islets have a high level of revascularization and oxygenation when transplanted to the pancreas and into striated muscle (27, 44).
HIF-1α is involved in responses to hypoxia in most mammalian cell types including human islet cells (36), and transient HIF-1α accumulation has been detected by immunohistochemistry in the early posttransplantation period after subcapsular islet implantation (35). HIF-1α activated expression of luciferase for in vivo monitoring hypoxia has previously been used successfully in cancer research (15, 30, 47, 48). The threshold for reporter gene expression in islets was found to be below an oxygen tension (pO2) of 10 mmHg; that is, pO2 would only be reported if below this threshold. However, the present detection level is about twice as high as the oxygen tension previously reported to be present in subcapsular islet grafts (6–10).
Notably, transduction of islets in vitro mainly engages the peripheral cells, leaving inner cells nontransduced (37). Since the degree of hypoxia is more pronounced in the cells in the core of an islet prior to revascularization, the technique potentially underestimates the “true” degree of islet graft hypoxia. However, in the experimental model used, the graft is composed of 200–400 islets densely packed into a small volume (approximately 2 × 2 × 1 mm), leaving the transduced islet cells randomly distributed within the entire graft. Another potential confounding factor is that HIF-1α transcriptional activity can be activated also by other factors, such as insulin, cytokines, and nitric oxide (43, 49).
Importantly, even mild islet hypoxia causes significant functional impairment of glucose-induced insulin release. In comparison with islets cultured in normoxia, insulin release is reduced by 50% already in islets cultured at a pO2 of 27 mmHg and by 98% in islets cultured at a pO2 of 5 mmHg (14). The present findings, with formation of an extensive intraislet capillary network after intraportal clinical islet transplantation and with only transient islet graft hypoxia (pO2 < 10 mmHg) in experimental islet transplantation, are in accordance with the capacity of an islet graft to respond with insulin secretion in response to glucose, repeatedly shown in numerous clinical and experimental islet transplantation studies.
Footnotes
Acknowledgments
The authors wish to thank Berith Nilsson for assistance with viral vector production, Margareta Engkvist for assistance with islet isolation and transplantation, Fredrik Carlsson for assistance with islet transplantation, Mansour Naghibi for assistance with morphological examinations, and Di Yu for assistance with the statistical analysis. The study was financed by the Swedish Medical Research Council and the Juvenile Diabetes Research Foundation. The authors declare no conflict of interest.
