Abstract
To overcome the weak points of the present cartilage regenerative medicine, we applied a porous scaffold for the production of tissue-engineered cartilage with a greater firmness and a 3D structure. We combined the porous scaffolds with atelocollagen to retain the cells within the porous body. We conducted canine autologous chondrocyte transplants using biodegradable poly-l-lactic acid (PLLA) or poly-dl-lactic-co-glycolic acid (PLGA) polymer scaffolds, and morphologically and biochemically evaluated the time course changes of the transplants. The histological findings showed that the tissue-engineered constructs using PLLA contained abundant cartilage 1, 2, and 6 months after transplantation. However, the PLGA constructs did not possess cartilage and could not maintain their shapes. Biochemical measurement of the proteoglycan and type II collagen also supported the superiority of PLLA. The biodegradation of PLGA progressed much faster than that of PLLA, and the PLGA had almost disappeared by 2 months. The degraded products of PLGA may evoke a more severe tissue reaction at this early stage of transplantation than PLLA. The PLLA scaffolds were suitable for cartilage tissue engineering under immunocompetent conditions, because of the retarded degradation properties and the decrease in the severe tissue reactions during the early stage of transplantation.
Introduction
Recently, regenerative medicine using tissue engineering has attracted social attention as a novel treatment to overcome various congenital or acquired diseases. This therapy is expected to realize a low invasiveness, high biocompatibility, and long-term utility, and so the research and development of tissue engineering has increasingly become pivotal tasks, facing an unprecedented situation with fewer children and an aging society. While many researchers have focused on various tissues and organs in the research fields of tissue engineering, those of bone, cartilage, skin, or cornea have progressed well. This is especially true for cartilage since autologous chondrocyte transplantation (ACT) has propagated around the world. In this method, the healthy chondrocytes were obtained from an isolated area of the injured knee, and were cultured in the laboratory for some weeks. The cultured chondrocytes were injected into the defective area, and covered with a sutured periosteal flap taken from the proximal medial tibia (2). This kind of surgery has been performed on more than 12,000 patients with an athletic injury or osteochondritis disecans (14). However, recent reports have indicated that some complications were related to the periosteal graft detachment, the delamination of the transplants, and the periosteal hypertrophy. Moreover, a systematic review of the classic ACT pointed out a limitation that this treatment has not indicated a clear clinical superiority when compared to the drilling for a cartilage deficit (microfracture) and a cylinder-type or mosaic-type autologous cartilage/bone graft (19).
Also, in the craniofacial areas, Yanaga and collaborators reported that the autologous auricular chondrocytes cultured with 10% autologous serum and fibroblast growth factor 2 (FGF-2) were subcutaneously injected to achieve a cosmetic nasal augmentation (32). Moreover, they applied this injectable cartilage for the treatment of microtia. The chondrocytes cultured in the same manner were injection implanted into the lower abdomen of a patient, and regenerated cartilage was grown for 6 months. The solid regenerated tissue was surgically harvested, formed into an ear framework, and then implanted for auricle reconstruction (31).
Thus, the present form of cartilage tissue-engineered products has been principally a cell suspension or gel. In order to prevent the weak points of the present products, such as cell leakage and periosteal hypertrophy, and to diminish the invasiveness of a two-stage implantation, some groups have attempted the application of the porous scaffold for the production of tissue-engineered cartilage with a greater firmness and a 3D structure. Cao and collaborators produced tissue-engineered cartilage of the human auricular type with porcine auricular chondrocytes and the composites of polyglycolic acid (PGA) and calcium alginate. They transplanted the constructs into the subcutaneous areas of immunodeficient nude mice, revealing the generation of tissue-engineered elastic cartilage (3). Chen's laboratory or other groups also reported that they fabricated a composite scaffold consisting of a poly-dl-lactic-co-glycolic acid (PLGA) sponge with some modified materials, such as collagen or fibrin (5,28). In the former study, bovine articular chondrocytes were seeded into this composite scaffold and then subcutaneously implanted into the dor-sum of the immunodeficient nude mice. As a result, this scaffold realized the formation of the desired shapes with a high mechanical strength and hydrophilicity (5). Thus, many other groups have developed novel porous scaffolds and made tissue-engineered cartilage constructs, showing cartilage regeneration in vivo. However, most experiments in these studies have been carried out in immunodeficient nude mice. The cartilage regeneration in an immunodeficient animal is known to be totally different from that in an immunocompetent human. We should evaluate the porous scaffolds of the tissue-engineered constructs in immunocompetent adult animals, as this will more closely match the clinical treatment situation.
Rotter and coworkers formed tissue-engineered cartilage using a polyglycolic acid–polylactic acid copolymer (Ethisorb) and porcine auricular chondrocytes, and autogenously replanted them after the incubation. The transplants showed cartilage regeneration (18). However, as the scaffold of Ethisorb was a membrane-type with a low porosity, the cartilage was formed not in the internal areas of transplants, but only on their surface. We would use the porous scaffold with a sufficient thickness and the capacity to contain the chondrocytes within it. Jung and collaborators also performed the autologous mesenchymal stem cell transplantation with the articular cartilage defects of a minipig. In their experiments, the cartilage defect was sealed with a collagen membrane and a mixture of cells and fibrin glue was injected into the defect cavity. However they used the scaffold of collagen membrane, which was just the sealant, and not the support for the mechanical strength of the tissue-engineered cartilage. This scaffold found it difficult to maintain the arbitrary 3D shape of the regenerated cartilage (11).
Our previous data disclosed that tissue-engineered cartilage constructs consisting of C57BL/6 mice auricular chondrocytes and poly-l-lactic acid (PLLA) scaffolds (MW: 200,000) were syngeneically transplanted into immunocompetent mice of the same genetic background, and showed cartilage maturation in the transplants (7). However, the degree of cartilage regeneration seemed to depend on the species of the experimental animals. Actually, it has been reported that articular chondrocytes have been harvested from sheep, dogs, and humans, and subcutaneously implanted in immunodeficient mice. This result showed that the sheep chondrocyte phenotype tended to be maintained better than that of human or canine chondrocytes (9). The mouse may be an animal in which tissue regeneration or wound healing tends to accelerate. We should use middle- or large-sized animals for the preclinical evaluation.
Thus, the purpose of the present study was to evaluate the feasibility of the scaffold-based tissue-engineered cartilage in experiments using immunocompetent beagles and to elucidate the suitable properties of the scaffolds for the fair cartilage regeneration. Thus, we established an autologous transplantation model of tissue-engineered cartilage using the scaffolds of biodegradable polymers in beagles. To accomplish the experiment, we combined the polymer scaffolds with an atelocollagen hydrogel to retain the cells in the porous body without leaking from the scaffold (30). In the present study, canine auricular cartilage was aseptically harvested by surgery. After the isolation and culturing of the chondrocytes, we made the tissue-engineered cartilage using the cultured chondrocytes with porous scaffolds of PLLA or PLGA, both of which were typical biodegradable polymers for various medical devices. We then autologously implanted it in the subcutaneous areas for 1, 2, and 6 months. We thus morphologically and biochemically examined the time course changes in the cartilage regeneration, and the in vivo behavior of the biodegradable polymers using PLLA and PLGA.
Materials and Methods
Surgical Procedure and Isolation of Chondrocytes
The present study was approved by the animal experiment committee of the University of Tokyo, Graduate School of Medicine (the approval number P09-026). Six male beagles (8 months old, approximately 10 kg, Saitama Experimental Animals Supply Co., Ltd) were used for the experiment. As the first step, we obtained a piece of auricular cartilage and blood sample for the preparation of the tissue-engineered cartilage. Anesthesia was intravenously induced by 1 mg/kg propofol and maintained with 2% halothane in a nonbreathing circuit. The unilateral auricular cartilages (approximately 2–3 g) of the beagles (Fig. 1A, B) were harvested by an aseptic surgical treatment. In addition, approximately 150 ml of autologous blood was collected from the cervical vein, and was allowed to clot over 5 h at room temperature (25). The serum was separated by centrifugation of the clotted blood at 3,000 rpm for 30 min (Fig. 1C). The perichondrium was exfoliated from the native cartilage using a surgical knife, and then the cartilage was minced to a size of 1 mm3. The fragments of cartilage were next incubated for collagenase digestion in a 0.15% collagenase solution at 37°C in 24 h. The collected cells were counted after trypan blue staining to confirm cell viability. The isolated 2.0 × 105 chondrocytes were seeded in a 100-mm collagen type I-coated dish (Iwaki brand Scitech Div. Agc Techno Co., Ltd., Chiba, Japan) and cultured in Dulbecco's modified Eagle medium: Nutrient Mixture F-12 (DMEM/F12) (Sigma-Aldrich Co., St. Louis, MO, USA) containing 5% autologous serum, 100 ng/ml FGF-2, and 5 μlg/ml insulin in a 37°C/5% CO2 incubator (23). The medium was changed two times per week. Passages were performed by treatment with a trypsin-EDTA solution when the cells were approaching confluence.

Canine auricular cartilage and cell culture. The ear auricle of the beagle (A), the collected auricle (B), canine serum (C), and chondrocytes of canine in vitro (D). (E) Growth curve of canine. Scale bars: 1 cm (A, B), 50 μm (D).
Biodegradable Polymer Scaffold Preparation
We prepared two kinds of porous biodegradable polymer scaffolds, PLLA and PLGA, with the dimensions of 2 × 0.5 × 0.3 cm. The porous scaffolds of PLLA (molecular weight 200 kDa) or PLGA (30:70, molecular weight 200 kDa) with the average porosity of 95% (26) were produced by KRI, Inc. (Kyoto, Japan).
Canine Autologous Chondrocyte Transplantation
The P1 chondrocytes proliferated at a 1000-fold increase in numbers and were suspended in a 1% atelocollagen solution at a density of 108 cells/ml. To make the regenerated construct, the atelocollagen aliquot containing the chondrocytes was added to the PLLA and PLGA scaffolds to form a gel after about 1-h incubation at 37°C (30). For transplantation of the tissue-engineered cartilage, the anesthesia was inducted in a similar manner to that described previously. The regenerative constructs were placed in a fibrous pocket formed by silicone templates. The regenerative constructs were operatively harvested after transplantation for 1, 2, and 6 months.
Histological and Histochemical Analyses
The samples were fixed with paraformaldehyde, and then successively immersed in 10% sucrose in phosphate-buffered saline (PBS), 20% sucrose in PBS, and a 2:1 mixture of 20% sucrose of PBS and OCT compound. The samples were frozen at −30°C, then cryosectioned at a thickness of 5 μm (CM1850-Kryostat, LEICA, Solms, Germany). After staining with hematoxylin and eosin (H&E), toluidine blue O (TB), and safranin O (SO), the sections were observed by optical microscopy (Olympus DP 70, Tokyo, Japan). To examine the biodegradability of the polymer scaffolds, we observed the crystallization of the polymers using a polarizing microscope.
We also examined the enzyme histochemical localizations against acid phosphatase (ACP) for the detection of macrophages. The sections were immersed in distilled water for 10 min to remove the sucrose, then incubated in acid phosphatase solution (Naphtol AS-BI phosphate, N,N-dimethylfolmamide, 0.2 M acetate buffer, distilled water, Fast Red Violet LB salt) for 5 min. Finally, methyl green was used for counterstaining.
In order to support the findings of the foreign body reactions, we also examined the immunohistochemical localizations for calcitonin gene-related peptide (CGRP) and tumor necrosis factor-α (TNF-α), both of which could be detected even in the canine experiments (4). The histological sections were quenched in a mixture of 0.3% H2O2 in methanol for 15 min at room temperature. The sections were treated with 10% normal fetal bovine serum for 1 h at room temperature to reduce the nonspecific binding of the antibodies. Incubation with primary antibodies [goat anti-TNF-α antibody (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), 1:100 dilution; rat anti-calcitonin gene-related peptide antibody (Sigma-Aldrich Co.), 1:1000 dilution] was carried out for 3 h at room temperature, 48 h at 4°C. The sections were then incubated with the biotinylated secondary antibodies to each IgG (1:200 dilution) for 60 min at room temperature. Consecutive steps for the visualization of positive reactions were conducted using a Vectastain ABC-kit and peroxidase substrate kit DAB following the manufacturer's instructions. Subsequently, the sections were counterstained with hematoxylin or methyl green. Nonimmune goat or rat antibodies at the same concentration were used as a negative control instead of the primary antibodies.
Biochemical Measurement of the Collagen Types II (COL2) and Glycosaminoglycan (GAG)
The samples were minced by scissors and homogenized twice for 30 s each using an homogenizer (IKA®-T10 basic ULTRA-TURRAX®, Staufen, Germany). These samples were dissolved in 10 mg/ml pepsin/0.05 M acetic acid at 4°C for 48 h and then in 1 mg/ml pancreatic elastase/1x TSB (0.1 M Tris, 0.2 M NaCl, 5 mM CaCl2, pH 7.8–8.0; Sigma) at 4°C. Any cell debris and insoluble materials were removed by centrifugation at 6,000 × g for 30 min. We then measured these conditioned samples.
The COL2 proteins were quantified by ELISA according to the protocol of the Type II Collagen Detection Kit (Chondrex, Redmond, WA, USA). For the ELISA measurement, we diluted the supernatant at 1: 100 with dilution buffer. In the sample mixture, the collagen proteins were captured by polyclonal anti-canine COL2 antibodies and detected by the biotinylated counterparts and streptavidin peroxidase. o-Phenylenediamine (OPD) and H2O2 were first added to the mixture, then sulfuric acid was added after 30 min to stop the reaction. The spectrophotometric absorbance of the mixture was measured at a wavelength of 490 nm by a spectral photometer (ARVOTMSX 1420 MULTI LABEL COUNTER, Perkin-Elmer™, Waltham, MA, USA).
We evaluated the GAG content using the Alcian blue binding assay (Wieslab AB, Lund, Sweden), according to the supplier's protocol. GAG in the supernatant was precipitated in Alcian blue solution, and the sediments after centrifugation at 6,000 × g for 15 min were dissolved again in a 4 M GuHCl-33% propanol solution. The spectrophotometric absorbance of the mixture was measured at a wavelength of 600 nm by a spectral photometer.
Results
Observation of the Procedure for Tissue-Engineered Cartilage Consisting of Autologous Canine Auricular Chondrocytes
The chondrocytes isolated from the canine auricles (Fig. 1D) rapidly proliferated in the culture medium containing 5% autologous serum with 100 ng/ml FGF-2 and 5 μg/ml insulin. It took two passages until the cell numbers increased 1,000-fold (Fig. 1E). The cells multiplying approximately 1,000-fold were harvested and mixed with 1% atelocollagen (8 × 107 cells/800 μl), then administered into the porous scaffolds made of PLLA or PLGA (20 × 5 × 3 mm) (Fig. 2A, B). The tissue-engineered constructs using PLLA (Fig. 2A) or PLGA (Fig. 2B) were made with or without chondrocytes. They all possessed a sufficient mechanical strength and maintained their 3D shapes, even before transplantation into the body. Both constructs could be tractably transplanted in the subcutaneous areas of the donor animals (Fig. 2C). After 1–6 months of transplantation, the constructs were excised and were histologically and biochemically examined (Fig. 2D).

Before and after cell transplantation. (A) Poly-l-lactic acid (PLLA) with the tissue-engineered cartilage before transplantation. (C) Transplanted tissue-engineered cartilage just after that (arrow head). (B) Poly-dl-lactic-co-glycolic acid (PLGA) with the tissue-engineered cartilage before transplantation. (D) Arrow head shows the transplanted tissue-engineered cartilage under the skin (D). Scale bar: 1 cm.
Time Course Changes in Tissue-Engineered Constructs
The macroscopic findings of the tissue-engineered constructs after transplantation showed that the constructs of PLLA with cells [Fig. 3A, cell (+)] maintained their height throughout the observation period (6 months), although those without cells [Fig. 3A, cell (-)] as well as the constructs of PLGA (Fig. 3B) gradually decreased in thickness. In particular, the PLGA constructs without cells at 6 months after transplantation had lost their stiffness and could not maintain their shapes. In the histological analysis, a cartilage-like structure was recognized only in the PLLA constructs with cells (Fig. 3A) after 1 month of transplantation. The chondrogenesis in the PLLA constructs was confirmed by toluidine blue staining (Fig. 4). Metachromasia, suggesting the accumulation of GAG, was widely observed over all the areas of the PLLA constructs with cells (Fig. 4A), although those of PLGA did not exhibit any metachromasia (Fig. 4B). The cartilage regeneration in the tissue-engineered constructs using PLLA could be maintained for 6 months based on the histological findings of both the toluidine blue staining and safranin O staining (Fig. 5). We summarized the results of the histological observations in Table 1. The abundant cartilage regeneration was noted in almost all samples of the PLLA constructs after 1 month, although those of PLGA hardly showed any effective accumulation of the cartilage matrices (Table 1).
Histological Examination Score

LA, lactic acid; GA, glycolic acid. I: no metachromatia, II: observed only part of the metachromatic matrix, III: observed extensive metachromatic matrix.

Histological and gross findings of tissue-engineered constructs. (A) PLLA with tissue-engineered cartilage [cell (+), cell (-)] of the sectioned surfaces and hematoxylin and eosin (H&E) findings at 1, 2, and 6 months. (B) PLGA with tissue-engineered cartilage [cell (+), cell (-)] of the sectioned surfaces and H&E findings at 1, 2, and 6 months. Scale bar: 1 mm.

The histological findings indicated that PLLA with tissue-engineered cartilage enhanced the metachromasia, implying the accumulation of proteoglycan in tissue-engineered cartilage (A) in comparison with PLGA (B) by toluidine blue staining. Scale bar: 500 μm.
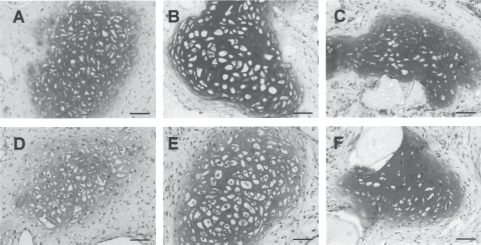
PLLA with tissue engineered cartilage were stained with toluidine blue impling the metachromasia and with safranin O for 1 month (A, D), 2 months (B, E), and 6 months (C, F). Scale bar: 50 μm.
In order to biochemically evaluate the accumulation of cartilage matrices, we examined the GAG and COL2 contents in both the PLLA and PLGA constructs (Fig. 6). As the PLGA possesses poly-anionic properties and may increase the baseline of the measurement value in GAG, we compared the ratios of the values in the constructs with cells to those without cells. As a result, the tissue-engineered constructs of PLLA showed higher contents of both GAG and COL2 when compared to those of PLGA (Fig. 6A, B).

Biochemical findings. Glycosaminoglycan (GAG) accumulation (A) and type II collagen (B) of PLLA, PLGA with tissue-engineered cartilage were compared with control groups for 1, 2, and 6 months. Relative ratio = cell (+)/cell (-).
Evaluation of Polymer Degradation and Tissue Reaction
In order to determine the reason why the cartilage regeneration of the PLGA constructs was inferior to that of PLLA, we evaluated the degradation of polymers using a polarizing microscope. Figure 7A shows that the majority of the crystal structures of PLLA remained in the constructs at 1 month of transplantation. Even 2 or 6 months later, the PLLA constructs did not completely diminish in the regenerative tissue (Fig. 7B, C). In contrast, the crystal structures of PLGA were noted within the constructs at 1 month (Fig. 7D), but after 2 months, the crystals of PLGA had almost totally disappeared from the regenerated tissue areas (Fig. 7E). By 6 months, no crystal structures of PLGA were observed at all within the construct (Fig. 7F).

Evaluation of degradable polymers. Crystals of both PLLA and PLGA were detected at 1 month (A, D), although the latter rapidly degraded thereafter. Crystallites of PLLA and PLGA constructs at 2 months (B, E) and 6 months later (C, F). Scale bar: 50 μm.
We assessed the localization of the acid phosphatase (ACP)-positive macrophages as a parameter of the tissue reactions in order to propose reasons why the PLGA constructs hardly contained any regenerative cartilage. The ACP-positive macrophages were significantly increased in the PLGA constructs in comparison with those of the PLLA constructs at 1 month, when the early stage of cartilage regeneration could be seen in the PLLA constructs with cells (Fig. 8A–D). TNF-α, which is a typical proinflammatory cytokine and also a putative catabolic factor for the cartilage (10), was detected more intensively in the cells of PLGA constructs than in those of PLLA (Fig. 8E, F). The metabolic products of PLGA, which were rapidly released into the tissue at this stage, may enhance migration of the macrophages, which played catabolic roles in the regenerated tissues. In addition, CGRP has previously been reported to decrease the local inflammation and to promote the cartilage regeneration (7) and was immunohistochemically observed in the cells of PLLA constructs, but hardly in those of PLGA, also supporting the tendency of the severe tissue reactions in PLGA (Fig. 8G, H). Negative controls using nonimmune antibodies detected specific localization in neither TNF-α nor CGRP (data not shown).

Evaluation of tissue reaction. H&E staining (A, B), acid phosphatase staining (C, D), and immunostaining staining for tumor necrosis factor-α (TNF-α) (E, F) and calcitonin gene-related peptide (CGRP) (G, H) were observed 1 month after transplantation in PLLA (A, C, E, G) or PLGA (B, D, F, H). As seen in the inset (square of D), the acid phosphatase was positive in the mononuclear cells or the multinuclear cells attached to the polymer surface, indicating its histochemical localizations in the macrophage or the multinucleated giant cells. The findings of acid phosphatase showed the intensive reaction by the macrophages in the PLGA constructs. The remnants of PLGA polymers were stained in blue with methyl green 1 month after transplantation (D, H), which suggested that the metabolic products due to the biodegradation, regarded as polyanions, were present on the surface of the PLGA scaffold. Although TNF-α was detected more intensively in the cells of PLGA constructs than in those in PLLA, CGRP was hardly observed in the PLGA constructs. Scale bar: 200 μm (A, B, C, D), 50 μm (inset of D), 10 μm (E, F), or 100 μm (G, H).
Discussion
This study investigated the effects of a biodegradable polymer scaffold on cartilage regeneration in the canine autologous transplantation model of implant type tissue-engineered cartilage using the auricular chondrocytes and biodegradable polymer scaffold. Although the scaffolds provided a sufficient stiffness and the 3D shapes for the engineered tissues, the biodegradable polymers may cause an immune response (rejection) as the degradation products are recognized as foreign substances in spite of the fact that the final products are water or carbon dioxide. This reaction may deteriorate the cartilage regeneration.
PLLA is a typical biodegradable polymer that has been clinically applied as the material of bone plates and screws. PLLA is produced by polymerization of the lactic acid (LA) monomers, which is heated under reduced pressure by catalytic agents of stannic octanoate, aluminium, or lanthanoid. The low-molecular compound is hydrolyzed into a LA oligomer by microorganisms, in environmental moisture. This kind of polymer is characterized by a high mechanical strength and biodegradability at a slow rate (more than a year) (17,24). The PLLA has been widely available for various purposes including an agricultural film, sheet, or optical disc, because PLLA is soluble in organic solvents or a thermoplastic and because it can be fabricated into arbitrary shapes.
Salyer and collaborators implanted a biodegradable plate and screws consisting of PLLA with other polymers into the skull of beagles and observed it at 6 and 12 weeks. The histological findings disclosed that the implanted screws were evident by 12 weeks and that there were several multinuclear giant cells around the material, indicating chronic inflammation (20). Another report described the treatment of mandibular fractures in 12 dogs, which were successfully stabilized with resorbable PLLA plates and screws. Histologically, the biomaterials evoked a mild inflammatory response during the first week. The molecular weight showed a rapid decrease to approximately one eighth within the first 4 weeks after implantation, although the flexural strength was maintained within 50% (8). These findings suggested that although PLLA is gradually biodegraded and evokes a mild inflammation during the early stage of transplantation, it maintained its structure and strength for some months or years.
On the other hand, PLGA is a hydroxyacetic acid–lactic acid copolymer of LA and glycolic acid (GA), which is clinically used as the material of absorbable sutures. The degradation speed of PLGA is much faster since this polymer is hydrolyzed within 2–6 months when transplanted into living bodies (15). Both monomers, LA and GA, are common metabolic products in the living body. PLGA shows a greater biocompatibility, and so it can be used as various biomaterials. PLGA is generally used as a barrier membrane for guided tissue regeneration (GTR)/guided bone regeneration (GBR) procedures. Park and coworkers assessed the PLGA films as periodontal barrier membranes for GBR in the bone defects with a calvarial critical size in SD rats, revealing that the PLGA film was completely degraded and absorbed within 8 weeks (16). As another application, PLGA stents were used for common bile duct repair and the construction of canines. These data showed that the molecular weight loss of PLGA was quite rapid during the first 6 days (29).
Both PLLA and PLGA reflect a low pH when they are degraded, and may affect tissue regeneration and cell viability. Crow and coworkers examined the changes in pH of the supernatant using an HPLC pH monitor. Although the solution pH of both the PLLA and PLGA fibers was approximately pH 7.0 at the first pH measurement, it decreased to approximately pH 5.7 for the PLLA fibers by 15 weeks. In contrast, the PLGA fibers reduced the solution pH to 3.6, even by 9 weeks, suggesting that the degradation of the PLGA fibers results in a more acidic solution than the PLLA ones. This was due to the greater percent release or more rapid decrease in the molecular weight of the PLGA fibers (6). Meanwhile, Sittinger and coworkers examined the chondrocytes that were incubated for 12 days with GA and LA at different concentrations of 0.5, 1, and 2 mg/ml. Cell viability significantly decreased in the MTT assay. They suggested that at concentrations above 2 mg/ml, GA was more cytotoxic than LA (21). The difference in the local concentration of the monomers, which is determined by the degradation rate, may affect the pH environment or the cytotoxicity. These reports suggested that the degradation and absorption speed of PLGA is rapidly increased in comparison to that of PLLA. For PLGA, it is speculated that the local density of the degradation products becomes high, even during the early stage after transplantation. In fact, crystallization in the scaffold of PLLA remained even 6 months after transplantation under differential interference contrast (DIC) observation, although that of PLGA was not detected (Fig. 6). These differences among PLLA and PLGA may be responsible for the changes in the degree of tissue response and cartilage regeneration.
The oligomers or monomers of PLLA or PLGA are the immunogens for the tissue reaction, because of the decrease in the local pH or immunoreactivity to themselves. The behavior of the polymer degradation products is known to be crucial for the surrounding cells and tissues (27). Against these degraded substances, the macrophages were mainly accumulated to form multinucleated giant cells. The macrophages or the giant cells may reduce the survival of the chondrocytes, as they produce cytotoxic factors including nitric oxides (NOs), proteases, and other catabolic factors (1). The macrophages are also known to secrete the inflammatory cytokines of TNF-α, transforming growth factor-β (TGF-β), and prostaglandin E2 (PGE2) (12,22), which make the various cells significantly produce catabolic enzymes including matrix metalloproteinases, leading to the degradation of the proteoglycan and type II collagen in the cartilage matrix (10). Actually, TNF-α was detected more intensively in the cells of PLGA constructs than in those in PLLA (Fig. 8C, G).
On the other hand, the chondrocytes seemingly enhance the resistance to attack of the macrophages when they become mature. As the transplanted chondrocytes begin to express some factors related to immune privilege, such as macrophage migration inhibitory factor (MIF), FasL, TGF-β, and CGRP in response to the host-derived macrophages, these factors may induce immune privilege and regulate the macrophages (7). In the present study, we could detect CGRP in the cells of PLLA constructs, but not in those of PLGA, 1 month after transplantation (Fig. 8D, H). The chondrocytes hardly produce immunoprivilege factors immediately after transplantation, while the immature chondrocytes following the monolayer culture are highly sensitive to macrophage attack and tend to have a low survival rate (7). In fact, apoptosis was often noted in the dedifferentiated chondrocytes under the monolayer culture when compared to the mature ones with the induction of differentiation (13). Therefore, the rapid increase in macrophages at the early stage, in which the transplanted chondrocytes still remained dedifferentiated, may reduce the viability of the chondrocytes. For these reasons, the PLGA constructs could not promote cartilage regeneration, although the maturation of the PLLA one was evident even in the immunocompetent beagles.
In conclusion, this study established a canine autologous cartilage transplantation model for the purpose of clarifying the biodegradable polymer scaffold suitable for cartilage reproduction, and evaluated PLLA and PLGA that are used for the biodegradable polymer scaffold. Based on these results, the PLLA scaffolds were suitable for the autologous chondrocyte transplantation for cartilage tissue engineering under the immunocompetent condition, because of the retarded degradation properties and the decrease in the severe tissue reactions during the early stage of transplantation.
Footnotes
Acknowledgments
This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (No. 21390532 and 21659462), and Research and Development Programs for Three-dimensional Complex Organ Structures from the New Energy and Industrial Technology Development Organization. The authors declare no conflicts of interest.
