Abstract
The present study was designed to evaluate the clinical outcome of cell-based therapy with cultured adipose-derived stromal cells (ASCs) for the treatment of cutaneous manifestations in patients affected by systemic sclerosis (SSc). ASCs have an extraordinary developmental plasticity, including the ability to undergo multilineage differentiation and self-renewal. Moreover, ASCs can be easily harvested from small volumes of liposuction aspirate, showing great in vitro viability and proliferation rate. Here we isolated, characterized, and expanded ASCs, assessing both their mesenchymal origin and their capability to differentiate towards the adipogenic, osteogenic, and chondrogenic lineage. We developed an effective method for ASCs transplantation into sclerodermic patients by means of a hyaluronic acid (HA) solution, which allowed us to achieve precise structural modifications. ASCs were isolated from subcutaneous adipose tissue of six sclerodermic patients and cultured in a chemical-defined medium before autologous transplantation to restore skin sequelae. The results indicated that transplantation of a combination of ASCs in HA solution determined a significant improvement in tightening of the skin without complications such as anechoic areas, fat necrosis, or infections, thus suggesting that ASCs are a potentially valuable source of cells for skin therapy in rare diseases such as SSc and generally in skin disorders.
Introduction
Systemic sclerosis (SSc) or scleroderma is a rare disease with a chronic trend of probable autoimmune origin, which affects mostly women. It is characterized by progressive skin and internal organ fibrosis due to the accumulation in the connective tissue of extracellular matrix (ECM), mainly type I collagen (23). The etiopathogenesis of SSc is still unclear, involving genetic and environmental factors, alteration in the microvasculature, and abnormalities in the immune system (17). Some studies suggest that SSc might be a multigenic complex disorder (24,52).
SSc predominantly affects the connective tissue of the skin and vessels wall and, to a lesser extent, that of the gastrointestinal tract, heart, lungs, and kidneys.
Cutaneous manifestations occur in most patients and are usually early signs in the course of SSc. The loss of elasticity and tightness of skin (sclerosis) start at acral areas, particularly at the fingers and hands, where it develops in sclerodactyly. Frequently, vascular complications occur, generating digital ulcers that cause severe pain and functional impairment and that could lead to gangrene, atrophy, and eventual amputation. Skin sclerosis may also affect the face including lips. Typical facial features associated with SSc are teleangiectasias, shrunken nose, microcheily, reduced mouth aperture (microstomy), and microglossy. In addition, there may be radial furrowing around the lips. Other skin manifestations include changes in skin pigmentation, hair loss, dryness due to the loss of sebaceous glands, and joint contracture (29,35).
The most widely recognized classification system for SSc has its origins in criteria proposed by the American College of Rheumatology in 1980 (49) and divides patients into two major clinical subsets, limited (lSSc) and diffuse (dSSc), primarily on the basis of the nature and extent of skin involvement, together with the presence of autoantibodies and internal organs' involvement (32). Patients with lSSc have fibrotic skin involvement that is typically limited to the skin distal to the elbows and knees (with or without face involvement). In these patients, skin manifestations can remain confined to acral areas for years. Patients with dSSc are characterized by more rapid onset and progressive course with severe skin involvement that gradually extends from the extremities to the trunk, finally affecting the whole integument. These patients often suffer from severe cardiac, pulmonary, gastrointestinal, and renal involvement that cause significant morbidity and mortality (29,37).
Although not life threatening, cutaneous manifestations of this disease are very plain and hard to conceal. Sclerosis of the extremities is highly disabling and results in significant dysfunction; the facial symptoms bear cosmetic disfigurement and limit expression, leading to a mask-like stiffness of the face. For that reason, many patients suffer from emotional and/or physical sequelae and avoid interpersonal relationship living secluded existences, full of psychological distress (2).
Little data are available about the morphological and histochemical peculiarities of the skin in sclerodermic patients. The most prominent pathological manifestation is an abnormal accumulation of extracellular matrix constituents, principally collagen types I and III. Early skin lesions are characterized by the presence of thickened collagen bundles and inflammatory cell infiltrates within the dermis and around blood vessels, while the epidermis is thin and atrophic. Late skin lesions show evidence of sclerosis, with closely packed collagen, atrophic or absent sweat glands, and few inflammatory cells (29). The treatment of cutaneous defects or lesions remains an unsolved problem in plastic and reconstructive surgery. Possible approaches are local and free flaps (50), dermal fat grafts (28), collagen injections, or synthetic materials (6,19). Unfortunately, all these methods include serious disadvantages. Over the years autologous fat transplantation became the first choice technique to hide cutaneous lesions; this approach, using the patient's own body fat as a natural filler to achieve structural modifications, take advantage of its abundance and accessibility, and avoid complications associated with foreign materials (5). Nevertheless, many studies have shown that free adipose tissue grafts are largely absorbed through time or replaced by fibrous tissue, and they display a low rate of graft survival due to partial necrosis (8). Moreover, elective liposuction for fat transplantation is nowadays considered a safe and well-tolerated procedure, but it is still surgery with all the related risks. Its major drawback is that the fat is harvested and subsequently injected without any expansion or cell concentration. Therefore, the treatment of large areas requires a consistent volume of fat (300–600 cc), and it cannot be applied to thin patients that are left out from this procedure.
Recently, it has been identified a stem cell population within the adipose stromal compartment, termed adipose-derived stromal cells (ASCs). This stem cell reservoir can be easily obtained from a very small amount of liposuction aspirate (1–5 cc), since it is present in any type of white adipose tissue, including subcutaneous and omental fat. Moreover, ASCs possess the ability to differentiate into various cell types, including adipocytes, chondrocytes, osteoblasts, and myoblasts. Therefore, they may represent a promising approach to cell-based therapies, such as tissue engineering and regenerative medicine (14,62,63).
Furthermore, it has been recently demonstrated that ASCs show angiogenic properties and could also exert some immunomodulatory properties, including a suppressive response on collagen-reactive T-cells and the capacity to restore immune tolerance by inhibiting the inflammatory response in vivo (14).
Some clinical data on the use of mesenchymal stromal cells (MSCs) in regenerative medicine within phase I/II trials are now available (3). In particular, the treatment with MSCs of some autoimmune and inflammatory disorders, such as type I diabetes mellitus, SSc, and systemic lupus erythematosus, is now under discussion. Promising results in animal models of diabetes and myasthenia gravis (11,25,59) opened the way to human clinical trials. Recently, a Chinese group demonstrated that transplantation of allogeneic MSCs in patients with lupus erythematosus refractory to conventional treatment options ameliorated disease activity, improving serological markers and stabilizing renal function (33).
As concerning the question if autologous or allogeneic mesenchymal cells should be preferred in regenerative medicine applications, it seems reasonable that for chronic pathologies, in which there is sufficient time for cell harvest and in vitro expansion, autologous cells may be preferentially used. Autologous cells should be preferred especially for autoimmune and inflammatory disorders, provided that they are functionally active, because of the potential immunogenicity of allogeneic cells, which might be rejected after infusion. Contradictory results have been published on the properties of mesenchymal cells isolated from patients affected by autoimmune and chronic inflammatory diseases. Nevertheless, some reports documented that mesenchymal cells of SSc patients, as well as those from patients with various autoimmune diseases, exhibit the same phenotype, proliferative and differentiation potential, and immunosuppressive properties as their healthy counterparts (31).
For that reason, here we assessed viability, proliferation rate, and capability to differentiate toward the adipogenic, osteogenic, and chondrogenic lineage of ASCs harvested from liposuction aspirate of sclerodermic patients and suggested the use of cell-based therapy with those functional expanded ASCs into a scaffold of hyaluronic acid (HA) as an ideal and safe source of autologous stem cells for the treatment of sclerodermic patients, with the goal to contribute in ameliorating their cosmetic disfigurement and physical sequelae, such as facial atrophy, muscle wasting, and joint contractures.
A key element for tissue engineering is an ideal scaffold that can provide structural support and a favorable environment for growing cells. HA is a natural component of the extracellular matrix of the dermis and other soft tissues and has proven to be well tolerated. Moreover, it has been previously demonstrated the in vitro compatibility of a commonly used injectable soft-tissue filler, nonanimal stabilized HA, with tissue-resident adipose tissue-derived stem cells harvested from adipose tissue (1).
Materials and Methods
Patients
The clinical trial protocol conformed to the guidelines of the 1975 Declaration of Helsinki, as revised in 1983, and was approved by the ethical committee of our institution (Ref. 1834/25.03.10). We classified the patients according to the Mayo Clinic classification criteria (42), as follows: plaque morphea, generalized morphea, bullous morphea, linear scleroderma [including the head–face subtypes En Coup De Sabre (ECDS) and Parry-Romberg Syndrome] and deep morphea.
Six patients, four females and two males, were enrolled in this study. One patient had been diagnosed with generalized morphea, two with linear scleroderma, one with ECDS, and the last two with both linear and plaque scleroderma. All patients were recruited from dermatologic outpatient clinics. All the selected patients were White (skin type II or III). In all patients, the diagnosis was established according to accepted clinical and histopathological features.
In addition, inclusion criteria called for signs of no active disease expressed by increasing size of lesions, appearance of new lesions, and/or clinical signs of inflammation within the last 6 months.
Exclusion criteria were as follows: pregnancy or lactation, any immunomodulating or immunosuppressive therapy within the last 4 weeks and any topical therapy within the last 2 weeks, except the use of emollients.
No skin biopsies were carried out across the study. Four of six patients had previously performed biopsies in other clinical centers to confirm the diagnosis.
The mean onset age of the disease was 7 years. All patients exhibited a variety of deformities: skin atrophy was found in all of them, muscle wasting in four, and restricted joint movement was noticed in three patients. One patient had also extracutaneous manifestation. One reported a family history of autoimmune disorder. Four patients had been previously treated with corticosteroids (methylprednisolone and methotrexate), but such therapies had not resulted in a sufficient improvement and have been stopped more than 4 weeks before entering the study. Therefore, the effects of these drugs were not considered to be relevant in our protocol. Relevant laboratory test results during the disease course were recorded. At diagnosis, two patients showed mildly increased erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), none had high white blood cell (WBC), eosinophil count, and creatinine kinase, but one patient had a slightly raised aldolase level. One of six patients was antinuclear antibody (ANA) positive, none were extract-able nuclear antigens (ENA) positive. Written informed consent was obtained from all patients before entering the study. Patients' data are summarized in Table 1.
Summary of Patients' Data

ASCs, adipose-derived stromal cells; HA, hyaluronic acid; M, male; F, female; ECDS, en coup de sabre; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; ANA, antinuclear antibody.
Surgical Techniques
Preoperative sedation was administered to all patients. The lower abdomen served as donor site for all patients. After the administration of local modified Klein solution [NaCl 0.9% (Fresenius Kabi, Verona, Italy), Naropin 7.5 mg/ml: 5F polyamp 10 (ropivacaine hydrochloride monohydrate), Xylocaine for injection 5f 10 ml 20 mg/M, (both from Astrazeneca Spa, Basiglio, Italy)] containing 1:500,000 of adrenaline, adipose tissue was harvested using hand-generated suction by means of a one-hole blunt 4-mm cannula (BD Italia, Milan, Italy) attached to a 10-cc Luer-lock syringe (BD Italia). Such nontraumatic low-negative pressure drain method preserves adipocytes intact and viable for transfer. In each patient, 10 ml of adipose tissue sufficed the entire procedure. The liposuction aspirates were then spun at 600 rpm for 2 min. Strict sterile technique was followed throughout the entire process. Prolonged exposure to air was minimized. After centrifugation, fat was sent to the laboratory for cell cultivation within 1 h and processed for the isolation of ASCs. Antibiotics were given to all patients as a precautionary measure.
Usually after 3 weeks, the patient went back to the operating room for the injection of the expanded ASCs. This procedure did not require anesthesia, only two patients asked for blunt sedation to calm down. The injection technique relied on preoperative topographic markings. Small aliquots of cell-enriched HA (1.6%, Wipeline, Fasel s.r.l., Bologna, Italy) were infiltrated in the chosen areas. As concerning the choice of the treated area, the principles that supported our choice were the easier access, the lower sensibility at the pain, and the request of the patient. In most of the cases, we tried to comply with patient's requests treating the areas that were most unsatisfying; in a few cases, we suggested, with the patient agreement, the most appropriate site according with the previously indicated principles.
The infiltration was done using 2.5-ml syringes provided with a 22-gauge needle, by means of multiple passes along several planes between the skin and the muscle tissue to avoid the creation of lumps. We employed different amounts of HA for each patient, as described in Table 1, keeping a constant rate of 8 × 105 expanded ASCs for each ml of HA. This technique does not induce fat necrosis and subsequent calcification, as lipofilling does, and there is no need to overcorrect the treated area.
In patients where there were several areas affected, we chose, with the patient consent, to treat only one site, to test the patient reaction and to compare the modification of the treated area with the nontreated zones.
As seen in Table 1, the first patient showed signs of disease on the face, trunk, and both arms, and we proceeded to treat the face. The second patient was affected on the trunk and left arm, and the latter was the chosen treated site. In the third patient, the afflicted areas were the trunk and face, and we treated the face. The fourth patient showed signs of disease on the abdomen and on the right arm, and we proceeded by treating the arm. The fifth patient was affected on the face and feet, which we decided to focus on. Finally, in the last patient, both lower and upper limbs were involved, and we treated only the upper limbs.
Before surgery, a template of the chosen region was performed and possibly modeled to obtain a symmetric and pleasant result. All patients were dismissed the following day.
Cell Isolation and Culture
ASCs were isolated and subsequently expanded following protocols in accordance with Good Manufacturing Practice (GMP) guidelines. The liposuction aspirates were transferred into a sterile tube and washed extensively with sterile phosphate-buffered saline (PBS) containing 2% penicillin–streptomycin–glutamine (PSG; Gibco, Paisley, Renfrewshire, UK) to remove contaminating debris and red blood cells. Then, the floating adipose tissue (about 10 ml for each patient) was transferred into a Petri dish and digested with 0.075% collagenase (type I; Gibco) in PBS at 37°C for 30–60 min, with gentle agitation, to break down the extracellular matrix. The collagenase was inactivated with an equal volume of Dulbecco's modified Eagle's medium (DMEM; Gibco) supplemented with 10% fetal bovine serum (FBS; Gibco), and the suspension was gently pipetted until disintegration and filtered through a 100-μm mesh filter to remove debris. The stromal vascular fraction (SVF) that contains ASCs was then pelleted by centrifugation for 5 min at 2,000 rpm. The pelleted cells were resuspended in DMEM-Ham's F-12 (v/v, 1:1) (DMEM/F12; Gibco) supplemented with 10% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin, and 2 mM l-glutamine and plated in a 75-cm2 tissue culture flask coated with collagen (type IV; Sigma-Aldrich, Milan, Italy). The SVF fraction contains an unpurified population of stromal cells, which includes ASCs, but also circulating cell types, hematopoietic stem cells, and endothelial progenitor cells. ASCs were self-selected out of the SVF during subsequent tissue culture passages, since they are adherent to the plastic tissue cultureware.
The same procedure was also performed on liposuction aspirates from four different nonsclerodermic donors, and these isolated ASCs were indicated as CTR and used as control in all the experiments.
ASCs were maintained in a 5% CO2 incubator at 37°C in a humidified atmosphere, with medium change twice a week. When reaching 80–90% confluency, cells were detached with 0.5 mM EDTA/0.05% trypsin (Gibco) for 5 min at 37°C and replated. ASCs were expanded, and cell viability was assessed by using the trypan blue exclusion assay. A homogeneous population of ASCs was subsequently checked by determining growth kinetics and by analyzing the surface marker expression profile. For both patients' treatment and cell analyses, ASCs were used between passages 2 and 3.
Flow Cytometry
Flow cytometric analyses were performed by using a FACSCalibur cytometer (BD Biosciences, San Jose, CA, USA), as previously described (63). Briefly, cultured ASCs were harvested, centrifuged, and fixed for 30 min in ice-cold 2% paraformaldehyde (Electron Microscope Sciences, Hatfield, PA, USA). The single-cell suspensions were washed in flow cytometry buffer containing PBS, 2% FBS, and 0.2% Tween 20 (Bio-Rad Laboratories S. r. l., Segrate (MI), Italy) and then incubated for 30 min with monoclonal antibodies to CD14, CD34, CD45, CD44, CD90, CD166, CD105, CD29, and CD73, conjugated to fluorescein isothiocyanate, phycoerythrin, or phycoerythrin-Cy5 (BD Biosciences). All monoclonal antibodies were of the IgG1 isotype. Nonspecific fluorescence was determined by incubating the cells with conjugated mAb anti-human IgG1 (DakoCytomation, Glostrup, Denmark).
Growth Kinetics
ASCs obtained from sclerodermic patients (SCL) and nonsclerodermic donors (CTR), at passages 2–3, were seeded on a 24-well plate (BD Biosciences) at a density of 1 × 1 0 4 cells/well. At several time points (days 3, 6, 9, and 12) after seeding, cells from three duplicate wells were harvested and counted. Nonviable cells were excluded by using the trypan blue (Sigma-Aldrich) assay. The number of cells was plotted against the time points, and the exponential growing phase of the cells was determined. The population doubling time of both sclerodermic- and nonsclerodermic-derived cells was calculated by using the formula:

Adipogenic, Chondrogenic, and Osteogenic Differentiation
The adipogenic, chondrogenic, and osteogenic differentiation capacities of the cultured ASCs from the sclerodermic patients were assessed using the Human Mesenchymal Stem Cell Functional Identification Kit (R&D Systems, Inc., Minneapolis, MN, USA), that contains specially formulated media supplements and a panel of antibodies to define the mature phenotypes of adipocytes, chondrocytes, and osteocytes. ASC differentiation was achieved following manufacturer's instructions.
Briefly, for adipogenic differentiation cells were resuspended in αMEM (Gibco) supplemented with 10% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin, and 2 mM l-glutamine and seeded on coverslips (Electron Microscopy Sciences) onto 24-well plates, at a density of approximately 3.7 × 104 cells/well. Medium was replaced every 2–3 days until 100% confluency was reached, then cells were incubated with adipogenic differentiation medium (ADM), that is, αMEM supplemented with a solution containing hydrocortisone, isobutylmethylxanthine, and indomethacin (all R&D Systems), to induce adipogenesis. The ADM was replaced every 3–4 days. The appearance of lipid vacuoles was monitored by microscopic examination.
Chondrogenic differentiation of pelleted ASCs was performed in 15-ml centrifuge tubes. About 2.5 × 105 cells were transferred to each tube and centrifuged at 1,000 rpm for 5 min at room temperature. The medium was removed, and cell pellets were resuspended with DMEM/F-12 supplemented with 1% insulin–transferrin–selenium (ITS; R&D Systems), 100 U/ml penicillin, 100 μg/ml streptomycin, and 2 mM l-glutamine. Cells were pelleted, resuspended with the chondrogenic differentiation medium (CDM), that is, DMEM/F-12 supplemented with a solution containing dexamethasone, ascorbate-phosphate, proline, pyruvate, and TGF-β3 (all R&D Systems), and pelleted again without removing the medium. The tubes were then incubated at 37°C and 5% CO2. The CDM was replaced every 2–3 days.
For osteogenic differentiation, ASCs were seeded on coverslips onto 24-well plates at a density of 7.4 × 103 cells/well and incubated with αMEM supplemented with 10% FBS, 100 U/ml penicillin, 100 mg/ml streptomycin, and 2 mM l-glutamine until they reached about 50–70% of confluence (1–2 days). Then, the medium in each well was replaced with the osteogenic differentiation medium (ODM), that is, αMEM supplemented with a solution containing dexamethasone, ascorbate-phosphate, and β-glycerolphosphate (all R&D Systems), to induce osteogenesis. The ODM was replaced every 3–4 days.
Immunofluorescence Analysis
Cultured ASCs were phenotypically characterized by using immunofluorescence analysis to assay for the expression of specific cluster of differentiation (CD) markers. Briefly, cells grown on coverslips onto 24-well plates were fixed in 4% paraformaldehyde for 30 min at room temperature, followed by treatment with 0.1 M glycine (Sigma-Aldrich) in PBS for 20 min and with 0.1% Triton X-100 (Sigma-Aldrich) in PBS for additional 5 min to allow permeabilization. Cells were then incubated with the following primary antibodies: anti-human CD29 monoclonal antibody (1:100 in PBS; Bio Legend, San Diego, CA, USA), anti-human CD166 monoclonal antibody (1:20 in PBS; BioLegend), and anti-human CD34 monoclonal antibody (1:10 in PBS; BD Biosciences). After appropriate washing in PBS, primary antibodies were visualized using FITC-conjugated goat anti-mouse IgG (Cappel Research Products, Durham, NC, USA).
The acquisition of a mature phenotype of adipocytes and osteoblasts was assessed after 21 days from induction. Briefly, coverslips were fixed in 4% paraformaldehyde for 20 min at room temperature, permeabilized with 0.1% Triton X-100, and incubated with goat anti-mouse fatty acid binding protein 4 (FABP-4) antibody (for adipocytes) or with mouse anti-human osteocalcin antibody (for osteoblasts) overnight at 4°C (both 1:10 in PBS; R&D Systems). Then the primary antibodies were visualized using Texas Red-conjugated rabbit anti-goat IgG and Texas Red-conjugated goat anti-mouse IgG (Jackson Immunoresearch Laboratories, West Grove, PA, USA), respectively.
The evaluation of chondrogenesis was performed through immunofluorescence on frozen sections (5 μm) of the induced pellets, obtained using a cryomicrotome (Microm HM 505N, Thermo Fisher Scientific, Inc., Waltham, MA). Briefly, sections were permeabilized with 0.1% Triton X-100 and then incubated overnight at 4°C with goat anti-human aggrecan antibody (1:10 in PBS; R&D Systems), followed by Texas Red-conjugated secondary antibody (Jackson Immunoresearch).
For all immunofluorescence experiments, nonspecific fluorescence was determined by omitting primary antibody. Nuclei were visualized using 4′,6-diamido-2-phenylindole dihydrochloride (DAPI) (Sigma-Aldrich). Fluorescence signals were analyzed by recording stained images using a CCD camera (Zeiss, Oberkochen, Germany) and IAS2000/ H1 software (Delta Sistemi, Rome, Italy).
Histology
All compounds were from Sigma-Aldrich. After 21 days from induction with ADM, coverslips were fixed in 10% formalin for 30–60 min at room temperature, incubated in 60% isopropanol for 5 min and stained with Oil Red O solution, which identifies lipids, for 5 min. Coverslips were then rinsed in tap water, counterstained with hematoxylin for 1 min, and mounted.
At the same time, coverslips induced with ODM were ethanol fixed for staining with Alizarin red S, which identifies calcium deposition. Briefly, coverslips were incubated with the Alizarin red solution for 2 min, extensively washed, and mounted.
Finally, the pellets subjected to CDM induction were fixed in 4% paraformaldehyde for 20 min, washed twice in PBS, frozen in liquid nitrogen, and sectioned using a cryomicrotome. Sections (5 μm) were stained in Alcian blue solution for 30 min or with 1% toluidine blue for detection of proteoglycans. Slides were then rinsed in tap water and mounted.
Osteogenic and chondrogenic differentiation of ASCs cultured in monolayers was additionally assessed through staining with both Alizarin red and toluidine blue of cells undifferentiated or after 14 and 21 days of differentiation.
Preparation of Cell-Enriched HA
Primary cultures of ASCs derived from each sclerodermic patient were expanded following the guidelines of current GMP in order to obtain an adequate number of 75-cm2 flasks at 95–100% confluence. On the day of transplantation, cells were detached with 0.5 mM EDTA/0.05% trypsin for 5 min at 37°C and counted. Then, ASCs were centrifuged at 1,500 rpm for 10 min, washed twice in PBS to remove serum, and finally resuspended in an adequate volume of synthetic stabilized HA solution (1.6%, Wipeline) at a concentration of 8 × 1 0 5 cells/ml. After gentle mixing, the suspension was kept under ambient conditions for 10–15 min to allow cell adherence to the hyaluronan matrix. Homogeneous dispersion of the cells within the gel was ensured by microscopical observation. Then the cell-supplemented HA solution was loaded into an injection syringe and carried to the operating room.
Clinical Outcomes Evaluation
All patients were followed postoperatively at least for 1 year with periodical medical assessments, weekly for the first month and then monthly.
Clinical Evaluation
Clinical evaluation was performed using five parameters to assess skin wellness: arrest of local disease progression, regression of dyschromia, softening (skin elasticity), sensitivity, and erythema reduction.
Independent Evaluation
The degree of improvement was also graded by two independent dermatologists who did not participate in the medical care of the patients and who did not have any knowledge of the employed procedures. We asked to assess each patient by assigning one of five degrees: considerable worsening, moderate worsening, any noticeable modification, moderate improvement, and considerable improvement. They evaluated the patients using pictures taken before entering the study and 1 year after surgery. Photographs were taken with the same parameters [same camera Canon EOS 500D (Canon Europe, Uxbridge, Middlesex, UK), same shooting distance of 50 cm, and same anatomical landmarks].
Subjective Evaluation
Each patient also graded his/her own results after 1 year, reporting their satisfaction degree by assigning one of four degrees: not satisfied, moderately satisfied, satisfied, extremely satisfied, based on estimation of tightness, itching, and appearance of the treated skin.
12-MHz Ultrasound
To measure both structure and thickness of the skin, ultrasound measurements were performed using the high-resolution ultrasound linear array transducer of 12 MHz (Apogee 3800, SIUI Ltd., Shantou, China). For each patient, scans were obtained only from the area we decided to treat. The usable depth of signal penetration was 8 mm. Sonographic gel was used to prevent flattening of the lesions and to keep the transducer and the skin surface apart. Sonographic examination was performed before surgery and 1, 4, and 12 months after the end of the treatment on the same skin site. Image analysis of the ultrasound measurements was performed by two experienced investigators who were blinded to any treatment detail.
Statistical Analysis
Values for sclerodermic- and nonsclerodermic-derived cells were compared using Student's t test for unpaired samples assuming a two-tailed distribution.
Results
Phenotypic Characterization and Growth Kinetics of Cultured ASCs
Cultured ASCs obtained from the processing of adipose tissue were selected on the basis of their adherent property and subsequently characterized in order to assess their purity and to exclude the presence of contaminating elements, such as hematopoietic stem cells. First, we compared morphologically ASCs obtained from sclerodermic patients (Fig. 1A, a, c) and from healthy donors (Fig. 1A, b, d). At early passages, they both displayed a typical fibroblast-like, spindle-shaped morphology in vitro (Fig. 1A, a, b), with only minimal alterations up to passage 30, when we observed the presence of larger cells and a tendency to grow in multilayered clusters (Fig. 1A, c, d). We also performed a growth kinetics assay to assess possible differences in cell proliferation. Cells at passage 2 were counted at several time points (days 3, 6, 9, and 12) after seeding, and their number was plotted against the time points, showing a very similar growth rate between sclerodermic (SCL)- and nonsclerodermic (CTR)-derived cells (Fig. 1B). For both the cell populations the mean population doubling time, calculated on the basis of the exponential growing phase, was about 2.1 days (Fig. 1C), consistent with that indicated by previous works (21,26), with no significant differences among the two (p = 0.67).
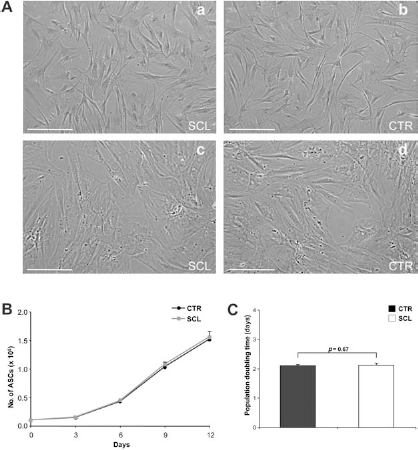
Morphology and growth kinetics of adipose-derived stromal cells (ASCs). (A) Representative phase-contrast photomicrographs of cells derived from sclerodermic patients (a, c) and from healthy donors (b, d) at passage 3 (a, b) and at passage 30 (c, d). The two cell populations displayed a similar, homogeneous, and spindle-shaped morphology, with minor alterations until high passages. Scale bars: 100 μm. (B) Growth kinetics of ASCs harvested from sclerodermic patients (SCL, ■, gray line) and from healthy donors (CTR, ●, black line). The graph shows the mean cell number obtained from three independent experiments for every patient or donor; error bars indicate standard deviation. (C) Population doubling time was calculated from the exponential growing phase of ASCs. Two-sided Student's t test was used to compare ASCs derived from healthy donors (CTR, black bar) and sclerodermic patients (SCL, white bar), showing no statistically significant difference (p = 0.67).
Then, ASCs harvested from sclerodermic patients were characterized by flow cytometry (Fig. 2A, B) and immunofluorescence (Fig. 2C). CD marker profile was examined by means of FACS analysis. Representative histograms are shown in Figure 2A. A fluorescence intensity greater than 99% of that obtained with the isotype-matched control antibody was considered as a positive staining and indicated by M1 window in histogram panels. The mean results obtained using a panel of nine antibodies on ASCs derived from the six patients are summarized in Table 2. ASCs displayed their specific pattern of expression, characterized by a high expression of the mesenchymal lineage cell markers CD105, CD73, CD29, CD90, CD44, and CD166, similar to the pattern of bone marrow-derived mesenchymal stromal cells (BM-MSCs) (62). In contrast, little or no expression of the hematopoietic lineage markers CD45, CD14, and CD34 was observed in the cultures.
Flow Cytometric Analysis for Cluster of Differentiation (CD) Markers Expression of ASCs at Passages 2–3

Results are expressed as mean percentage of positive cells ± standard deviation of flow cytometry of 10,000 cells obtained from the six sclerodermic patients. Percent positive is defined as fluorescence intensity greater than 99% of isotype-matched antibody control.

Phenotypical characterization of ASCs. (A) Flow cytometric analysis of ASCs. Cells were stained with monoclonal antibodies directed against selected mesenchymal (a–e) and hematopoietic (f–g) markers. Histograms are representative of all patients. Gray areas represent patterns obtained with antibodies against the indicated markers, whereas black lines represent the isotype-matched monoclonal antibody that served as a control. The M1 window represents fluorescence intensity exceeding that of 99% of the control antibody. (B) Dual parameter flow cytometric analysis of ASCs. Cells were double-stained with antibodies for two different mesenchymal markers (a–c) or with an antibody for a hematopoietic marker (x-axis) and an antibody for a mesenchymal antigen (y-axis) (d–g). (C) Immunofluorescence analysis of ASCs. Cells stained positively for the mesenchymal antigens CD29 (green, a) and CD166 (green, b) and negatively to CD34 antigen (c). Nuclei (blue) were visualized with 4,6-diamidino-2-phenylindole (DAPI). Scale bars: 100 μm.
Dual parameter flow cytometric analyses were also performed using combinations of the same antibodies to assess that most of the cells were at the same time positive for mesenchymal markers and negative for haemopoietic markers or double-positive for two different mesenchymal markers. Representative histograms are shown in Figure 2B. Flow cytometry results were also confirmed by immunofluorescence analysis, showing the expression of CD29 and CD166 and the absence of CD34 antigen in ASC cultures (Fig. 2C).
Differentiation Potential of ASCs
We then proceeded to assess the capability of ASCs derived from each patient to differentiate into various cell types. The adipogenic, osteogenic, and chondrogenic differentiation was induced by replacing the culture medium with a specific differentiation medium, which was changed twice a week for the full induction period. In particular, for adipogenic differentiation, cells cultured in ADM for 2–3 weeks and then stained with Oil Red O showed evident intracellular lipid accumulation (Fig. 3A, a). Moreover, adipogenic induction resulted in the expression of the lineage-specific FABP4 (Fig. 3A, b), which was not detected in noninduced ASCs (Fig. 3A, c). To assess osteogenic differentiation, cells cultured in ODM for 3 weeks were stained with Alizarin red S, which showed the deposition of de novo bone matrix (Fig. 3B, a). Moreover, immunofluorescence analysis showed the expression of the bone-specific protein osteocalcin in induced ASCs (Fig. 3B, b) and not in uninduced control cells (Fig. 3B, c). Finally, serial sections of cell pellets induced toward chondrogenic differentiation by culturing in CDM for 3 weeks showed a positive stain for Alcian blue, thus demonstrating the secretion of sulfated glycosaminoglycans (Fig. 3C, a). Immunofluorescence analysis of cell pellets revealed the expression of the large proteoglycan aggrecan in induced samples (Fig. 3C, b), while no specific signal was evident in uninduced cells (Fig. 3C, c). Sections of the cell pellets also stained positively for toluidine blue, which is specific for the highly sulfated proteoglycans of cartilage matrices (Fig. 3D, a).

Multilineage differentiation of ASCs. (A) Adipogenic differentiation. ASCs cultured in adipogenic medium were stained with Oil Red O to visualize lipid accumulation (a, red). Immunofluorescence analysis revealed the expression of the adipocytespecific fatty acid binding protein 4 (FABP4) in induced cells (b, red) and not in uninduced ASCs derived from the same patient (c). (B) Osteogenic differentiation. ASC cultures subjected to osteogenic conditions showed positive histological staining for Alizarin red (a, red) and positive immunofluorescence reactivity to antibodies detecting osteocalcin (b, red). No immunofluorescence signal was obtained in uninduced cells (c). (C) Chondrogenic differentiation. Pellets of ASCs induced toward the chondrogenic lineage were characterized by proteoglycans deposition, assessed by positive staining for Alcian blue (a, blue) and by immunofluorescence with anti-aggrecan antibodies (b, red). Pellets of uninduced cells were negative for aggrecan expression (c). (D) Pellets of ASCs induced toward the chondrogenic lineage were further characterized by proteoglycans production, assessed by positive staining for toluidine blue (a, blue violet). Monolayers of ASCs were also analyzed at different times (ND, undifferentiated cells; 14d, 14 days of differentiation; 21d, 21 days of differentiation) of both osteogenic (b) and chondrogenic (c) differentiation. Scale bars: 100 μm.
Moreover, we performed an additional osteogenic and chondrogenic differentiation on ASCs cultured in monolayers, assessing the faint staining with both Alizarin red (Fig. 3D, b) and toluidine blue (Fig. 3D, c) in undifferentiated cells and the progressive increase in cell staining after 14 and 21 days of differentiation.
HA Scaffold
We then proceeded to include ASC suspension into a matrix of injectable HA. Previous studies have proved that local transplantation of stem cells via a scaffold has some advantages over direct transplantation such as local injection or intravenous infusion (34,36). The scaffold we used to deliver ASCs is a 1.6% solution of synthetic HA, without chemical modifications and with a molecular weight of 1 × 103 kDa, very similar to the endogenous HA. That solution showed great biocompatibility (41) and represented a good way to vehicol autologous stem cells into each patient, since observation of an effect in supporting their growth and differentiation in vitro suggests that this process may unfold likewise in vivo. In fact, the specific composition of HA used for our cell-based therapy showed to possess both the mechanical strength necessary to sustain the pressure of surrounding tissues and the biochemical property of slow biodegradability, thus allowing the full maturation of the neomesenchymal tissue before graft degradation, also creating a “biomimetic” environment during tissue regeneration (1,4,9).
Clinical Outcomes
All patients were followed postoperatively at least for 1 year with periodical medical assessments, weekly for the first month and then monthly. The obtained results were satisfactory both for the patients and the physicians. The main complications noticed were small areas of ecchymosis. No viral complications or infections were detected.
Clinical Evaluation
As shown in Table 3, all patients treated with this technique presented a favorable outcome with improvement in wellness of the skin judged by clinical examination.
Summary of Patients' Clinical Outcomes Assessed at 1-year Follow-Up

For each patient are reported the clinical evaluation based on the parameters of skin wellness that presented a favorable response, the degree of improvement according to two independent surgeons, and the patient's subjective evaluation based on their satisfaction.
All patients showed arrest of local disease progression (100%), four of them presented regression of dyschromia (67%), five patients increased skin softening (83%), four patients showed better sensitivity (67%), and one patient reported erythema reduction (17%). Three representative patients are shown in Figures 4–6.
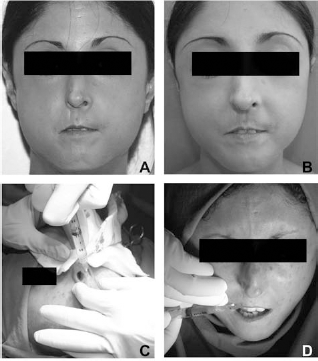
Clinical views of Patient 1 (a 27-year-old woman). (A) Preoperative view. The patient was diagnosed with scleroderma when she was 4 years old. It started with Raynaud's phenomenon and skin patches on the body and then diffuse SSc rapidly progressed, affecting a large area of the skin, until she was 17, when it stabilized. The cutaneous manifestations were localized to the face (in particular nose, mouth, chin, and cheekbones), trunk, and arms, and there was no evidence of internal organs being affected. The patient never underwent any treatment before coming to our observation. (B) Postoperative view at 1 year. The facial defect was significantly improved. The patient was particularly satisfied with the nose result where the irregular cartilages underneath were minimized. The same result could not be obtained using fat transplantation because of the extreme thinness of the skin. (C, D) Intraoperative views. Injection of HA (5 ml) enriched with autologous ASCs into the nose (C), mouth (D), and cheeks.

Clinical views of Patient 4 (a 21-year-old woman). (A) Preoperative view. The patient was diagnosed with linear scleroderma when she was 9 years old. The cutaneous manifestations were localized to the right arm and abdomen, with no internal organs involvement. The patient reported no rheumatic or autoimmune disorders, and she never underwent any treatment before. Injection of HA (8 ml) enriched with autologous ASCs was performed into the right elbow. (B) Postoperative view at 1 year. The treated area shows a distinctive improvement, with the complete remission of the ulcer and greater coverage of the bones.
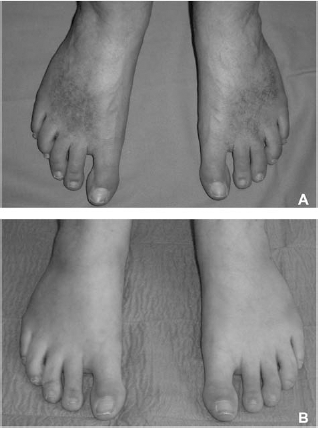
Clinical views of Patient 5 (a 31-year-old woman). (A) Preoperative view. The patient was diagnosed with linear scleroderma when she was 7 years old. The cutaneous manifestations were localized on the face and on the feet, with no involvement of internal organs. The patient never underwent any treatment before. Injection of HA (10 ml) enriched with autologous ASCs was performed on both feet. (B) Postoperative view at 1 year. The treated area shows a distinctive improvement, with no erythema and complete remission of the lesion.
Independent Evaluation
Two independent dermatologists evaluated the patients using pictures taken before entering the study and 1 year after surgery. They graded four patients as considerably improved, one patient as moderately improved, and in one case they did not observe any modification (Table 3).
Subjective Evaluation
Four patients were extremely satisfied with the results, one patient was satisfied and one was moderately satisfied with the outcome (Table 3).
Ultrasound Scanner Evaluation
Sonographic examination showed any anechoic area or fat necrosis for all the patients. We noticed that 1 month after surgery, when the HA reabsorption was almost completed, the subcutaneous thickness was unmodified. By the fourth month, instead, it resulted increased, and we observed a more regular skin structure. Such improvement could be ascribed to the starting growth and differentiation of transplanted adipose stem cells. The registered thickness parameters were confirmed in two patients after 1 year, while in the other four it resulted further increased (data not shown).
Discussion
Scleroderma is a clinically heterogeneous disease with a complex phenotype. It is characterized by vascular dysfunction, tissue fibrosis, internal organ, and immune disorder, often resulting in autoantibody production, whose significance in the pathogenesis of SSc is not known and whose titers do not seem to correlate with disease course or severity (10,61).
The complexity of the disease is made worse by the limited therapeutic options available and the lack of evidence for clinical effectiveness of treatments that are currently in use. However, there are emerging data supporting efficacy for some treatment approaches, such as high-dose immunosuppressive therapy coupled with autologous hematopoietic stem cell transplantation, in order to ablate immune responses driving disease activity and to generate a new non-auto reactive immune system (39).
The skin involvement is the hallmark of SSc; therefore, the treatments of skin symptoms have been the focus of investigation in many clinical trials. Recent studies have assessed different biological agents for the treatment of skin thickness: molecules that interfere with platelet derived growth factor (PDGF) or transforming growth factor beta 1 (TGFβ1) downstream signaling, such as neutralizing antibodies or tyrosine kinase inhibitors; proteins with antifibrotic properties, such as relaxin; proteins that induce immune tolerance, such as oral bovine collagen type I. All these treatments, however generally well tolerated, did not show significant efficacy (27).
Such considerations prompted us to evaluate the use of stem cell-based therapy in sclerodermic patients. In particular, we focused our attention on adipose tissue, which represents an abundant, practical, and appealing source of autologous stem cells, the ASCs. Several groups have demonstrated ASCs to be very similar to BMSCs in morphology, growth rate, and surface markers profile (16,21,62). In fact, ASCs are characterized by the high expression of MSC markers, such as CD29, CD44, and CD90, and the lack of expression of markers restricted to hematopoietic cells, such as CD14 and CD45 (16). Moreover, this cell population displays a developmental plasticity, with the potential to differentiate into various lineages including adipocytes, osteoblasts, chondrocytes, myoblasts, and endothelial cells (12,40,62).
The clinical use of ASCs is advantageous with respect to that of BMSCs because they can be obtained through liposuction, a procedure generally less invasive and painful than a marrow harvest. Moreover, the frequency of stem cells in the adipose tissue is higher than in bone marrow compartment, and ASCs display nonselective media requirements, faster culture expandability, and higher retaining of stem cell phenotypes and mesenchymal multipotency at higher passages (60).
We detected no differences in growth kinetics and population doubling time of ASCs from sclerodermic patients and healthy donors, suggesting that stromal cells in the adipose tissue are not involved in the phenotypic alterations determined by the disease on mesenchymal cells (e.g., fibroblasts) (45). In addition to the determination of growth kinetics, we have phenotypically characterized the cultured ASCs and assessed their multipotency. The surface-marker expression profile is in accordance with that found by others (16,60,62), and our ASCs were able to differentiate into adipocytes, chondrocytes, and osteoblasts.
Current methods for expanding ASCs rely on media containing bovine serum, which represents a potential risk of viral transmission and immunological reactions (34). A completely serum-free medium have been recently presented and its ability to promote ASC proliferation is currently being explored, but a standard expansion method without animal products has not been yet established (48). Nevertheless, to avoid the contamination of our cell-enriched HA scaffold with animal-derived products, we performed an extensive wash on the cell pellet just before the inclusion.
The choice to treat only one site for each patient, instead of all the affected ones, allowed us to better assess the disease progression. All the patients enrolled showed no illness progression over the past 2 years. After 1 year from the procedure, there was no sign of scleroderma resumption in the treated area, while two of six patients showed disease progression in the nontreated area, which would have also affected the zones we focused on and that was avoided thanks to our approach.
In the field of translational research, stromal cells derived from adipose tissue have been previously used in combination with standard approaches (lipoinjection) for soft-tissue reconstruction in humans, such as cosmetic breast augmentation or treatment of facial lipoatrophy and other disfiguring problems (57,58). However, MSCs derived from adipose tissue are not only confined to plastic and reconstructive surgery applications but also represent possible therapeutic solutions for several conditions affecting various tissues (bone, cartilage, muscle, liver, kidney, cardiac, and neural systems). In fact, ASC-based therapies are now under consideration for the treatment of Parkinson's and Alzheimer's disease, osteoarthritis, and multiple sclerosis (13,34,47). Their ability to repair injured tissues, together with their immunomodulatory and anti-inflammatory properties, means that MSCs have been tested in several animal models of tissue injury and degenerative disorders, including systemic lupus erythematosus, diabetes and glomerulonephritis (11,30,33). In all these pathologies, mesenchymal cells were able to ameliorate the signs and symptoms of the disease by inhibiting autoreactive T and B cells or by stimulating the healing process, thus resulting into the repair of the damaged tissue.
Topical implantation of BMSCs has been shown to promote the healing process of experimental colitis in rats, confirming the ability of these cells to induce tissue repair (18). In similar models, mesenchymal cells of adipose tissue origin also displayed immunomodulatory functions and ameliorated inflammation-related tissue destruction (15).
The mechanism through which ASCs exert their therapeutic potential in tissue repair is not yet fully defined, but it might be related to the ability of these cells to secrete soluble factors capable to stimulate survival and functional recovery of injured cells. The release of soluble factors and chemokines (such as hepatocyte growth factor, insulin-like growth factor, prostaglandin E2, nitric oxide) by ASCs has been previously demonstrated (55).
In particular, the rationale for the use of ASCs in autoimmune disease is based on the assumption that immune dysregulation could be restored by ex vivo expansion and reinfusion of cells with immune-regulatory function. ASCs can modulate immune responses by inhibiting the proliferation and proinflammatory cytokines [interferon (IFN)-γ and tumor necrosis factor (TNF)-α] production by T cells, B cells, natural killer (NK) cells, and dendritic cells via multiple mechanisms (7). The inhibition of inflammatory and immune responses by ASCs might also be displayed through the generation of antigen-specific regulatory T cells and suppression of humoral immunity (15). Finally, they also inhibited the production of matrix-degrading enzymes, collagenase and gelatinase, which are involved in the inflammatory response.
Indeed, in most of the reported studies, the therapeutic effect of ASCs seemed to be mostly related to their antiproliferative and anti-inflammatory effect and only scarcely associated with their differentiation into the resident cell types. However, mechanisms linked to the multipotentiality of ASCs, such as their differentiation into endothelial cells in ameliorating lupus nephritis, may also be involved (54).
In the light of these considerations, we hypothesize that the disease improvement obtained in our patients by injection of ASCs might be induced by several mechanisms. First, ASCs might exert an immunomodulatory effect by producing soluble factors induced upon stimulation, somehow inhibiting the inflammatory response in vivo. Second, we speculate that ASCs may be associated with expansion of regulatory T cells, which suppress the activity of autoreactive T cells and play a pivotal role in the maintenance of self-tolerance. The third presumed mechanism of effect in SSc is based on the multipotentiality of ASCs, and in particular on their capability of differentiating into various mesenchymal cell types in vitro, as demonstrated by our work. In particular, ASCs can differentiate into adipocytes, giving volume to the treated skin and ameliorating patients' soft tissues defects; they can release angiogenic factors and even differentiate into endothelial cells, thus contributing to the promotion of angiogenesis in the surrounding tissues (51), and finally, they could represent a stable niche of self-renewing stem cells (38,43,44,46).
A great number of scaffolds have been used for restoring soft-tissue loss, and various biomaterials have been proposed for adipose tissue engineering (20). In particular, injectable hydrogels have been extensively used as cell delivery systems, since they provide readily integration and homogenous distribution of cells into the matrix. In addition, they allow minimally invasive approaches for cell delivery (53).
Right now, we have also several studies going on with ASCs in physiologic solution, but we chose to employ a nonanimal stabilized HA solution as scaffold for patients with scleroderma, since it has proven to be biocompatible, well tolerated, with persistent volume fill up to 6 months (4,9), and it has been previously demonstrated its in vitro compatibility with adipose tissue-derived stem cells. These cells were shown to proliferate equally when grown on a nonanimal stabilized HA substrate compared with standard tissue culture conditions on a plastic substrate, and hyaluronan-based ASC engraftments are associated with increased angiogenesis in vivo (1). Therefore, HA is capable of successfully delivering adipose tissue-derived stem cells in a targeted fashion, with adipose tissue-derived stem cells focally established at the graft site (20), allowing us to distribute the cells smoothly on extended areas and to precisely set the needed amount to inject, according to the area we decided to treat. Another advantage is given by the transitory aesthetic result reached with the injection of HA, highly appreciated by the patient, who could also foresee the final aesthetic result that would be obtained once the adipocytes would take on and proliferate.
Recent studies demonstrated that HA regulates cytokines and other inflammatory substances, but its influence on inflammatory cell recruitment, chemotaxis, and ultimately immune response is strictly dependent on its molecular weight. Moreover, HA and HA-binding proteins play an active role in maintaining and differentiation of MSCs. In particular, MSCs (and also ASCs, as demonstrated by our FACS analysis) express high levels of HA receptor (CD44), which has long been used as a marker for these cells and facilitates cell migration through interaction with extracellular HA. Such a migratory mechanism could be important for the recruitment of stem cells into wound sites to improve tissue regeneration. Therefore, several HA-based scaffolds have been developed for tissue regeneration, since CD44–HA interactions help MSCs to move to injury area and also recruit exogenous MSCs to promote tissue regeneration (22).
Nevertheless, in our clinical setting the importance of recruiting and migrating function of HA is less significant, since we performed topical applications of small amounts of product directly in the injured sites. Moreover, previous observations suggested that nonanimal stabilized HA may confer volume-filling effects, attributable to its stimulatory effect on collagen production (56), but such effects are only temporary since it was almost completely reabsorbed after 1 month. Furthermore, Altman et al. (1) reported a significant increase in vascular density after grafts with combined adipose tissue-derived stem cell and nonanimal stabilized HA compared with nonanimal stabilized HA alone, thus suggesting that MSCs are directly responsible for the establishment of a neovascular network at the site of tissue repair. In this work, we have demonstrated that human adipose tissue-derived stem cells successfully integrate within nonanimal stabilized HA for an injectable, cell-enhanced, soft-tissue augmentation strategy. Further studies will allow greater understanding on the mechanisms of adipose tissue-derived stem cell and nonanimal stabilized HA interactions and will clarify their specific contribution to the clinically relevant benefit in soft-tissue augmentation and reconstruction.
Conclusions
The present study assessed the availability and the efficacy of the use of ASCs for therapeutic treatment of sclerodermic patients.
Nowadays, there is still no cure for this disease, but only palliative treatments that aim to lessen the impact of the cutaneous symptoms. Among these, autologous fat transplantation guarantees good results. The technique we describe has the advantage of removing the risks tied to extensive liposuction and of being easily applied, even more than once, to patients with scarce adipose tissue that cannot turn to fat transplantation.
The significant improvement in four patients and moderate improvement in one patient demonstrate that ASCs could be successfully transplanted into an injectable scaffold of synthetic stabilized HA, thus representing a good, feasible, and efficient cell-based soft-tissue augmentation strategy.
We think that our findings would allow further future applications of ASC-based cell therapies to the clinical setting and could open the way for the development of new therapeutical strategies in the treatment of skin diseases such as SSc. Nevertheless, further longer-term studies will be needed to clarify the potential for a clinically relevant benefit in the treatment of other pathologies involving skin disfigurement, but we believe that ASCs will be a good opportunity for regenerative cell-based therapy in the near future.
Footnotes
Acknowledgments
The authors would like to thank Dr. Giuseppe Mucci at Bioscience Institute S.p.A., Republic of San Marino, for his contribution and support. The authors confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
