Abstract
In adults, hematopoiesis takes places in the bone marrow, where specialized niches containing mesenchymal nonhematopoietic cells (stroma) harbor the hematopoietic stem cell (HSC). These niches are responsible and essential for the maintenance of HSCs. Attempts to expand HSCs fail to keep the general properties of stem cells, which depend on several niche components difficult to reproduce in in vitro culture systems. Here, we describe a methodology for in vivo study of hematopoietic stroma. We use stroma-loaded macroporous microcarriers implanted in the subcutaneous tissue of experimental animals and show that the ectopic stroma implant (ESI) is able to support hematopoiesis. Moreover, lethally irradiated mice can be rescued by ESI preloaded with HSCs, showing that they function as an ectopic bone marrow. ESI is also shown as a good system to study the role of different niche components. As an example, we used stromas lacking connexin 43 (Cx43) and confirm the importance of this molecule in the maintenance of the HSC niche in vivo. We believe ESI can work as an ectopic bone marrow allowing in vivo testing of different niches components and opening new avenues for the treatment of a variety of hematologic conditions particularly when stromal cell defects are the main cause of disease.
Introduction
Hematopoietic stem cells (HSCs) are characterized by their self-renewal capacity and ability to differentiate into various hematopoietic progenitor cells (HPCs), giving rise to all blood lineages (57). In adulthood, hematopoiesis takes places inside the bones of the axial skeleton where specialized niches containing mesenchymal and other hematopoietic and nonhematopoietic cells (generally named as stroma) harbor the HSCs. These niches are responsible and essential for maintenance and development of the HSCs (1, 55). Alterations in bone marrow originate a number of pathologies, as malignancies and aplasias. This can be caused by defects in hematopoietic cells or stroma components. Defects in hematopoietic cells are frequently associated with malignancies. Stromal defects are believed to confer pathological alterations in hematopoiesis as well, as has been shown in myelodisplastic syndromes (7) and aplastic anemia (11).
Hematopoietic stem cell reconstitution is widely used to treat a great number of hematologic diseases. However this procedure is hampered by the occurrence of graft versus host disease (GVHD)—a severe disease with high morbidity and mortality rates (6, 9). Transfer of highly purified CD34+ cells can overcome GVHD in cases where immune responses against malignant cells are not mandatory (3, 14). Enrichment in immature progenitors also enables the use of umbilical cord blood (UCB) as transplant product to be used in adults. Transplants using UCB are advantageous over bone marrow or peripheral blood as they are readily available and allow major histocompatibility complex (MHC) disparity with comparable lower incidence of GVHD (52).
However, generation or expansion of high numbers of immature hematopoietic progenitors in vitro has been limited by the complex array interactions present in hematopoietic niches. When cultured in vitro, HSCs quickly lose their characteristics (25, 59). Growth factors, hormones, extracellular matrix proteins, and intercellular interactions has been shown critical for maintenance of long-term hematopoietic stem cells, enlightening the difficulties to reproduce the niche milieu in vitro (24, 31, 32, 46, 49–51, 54). Three-dimensional cultures has also been explored as a way to expand and study HSCs (4, 5, 23, 44, 45) but even though achievements are far from the actual in vivo expansion capacity of HSCs (46).
Hematopoietic stem cell reconstitution gains an even higher level of complexity when the defect is in the stroma, because stroma transplants usually do not seed the bone marrow (29), although in aplastic anemia (21) allogeneic mesenchymal stem cell engraftment had been reported to correct stromal defects (16). Interestingly, in some systems it has been shown to improve HSC engraftment (35) but the mechanism apparently lie in the immunosuppressive effect. Moreover, since mesenchymal cells are highly undifferentiated and can give rise to a number of other tissues, caution must be taken on its therapeutic use (29, 51).
One alternative to correct hematopoietic failures rely on the generation of bone marrow organ systems (BMOS) able to ectopically reproduce a functional bone marrow. In fact, grafts using hydroxyapatite tricalcium phosphate (HA/TCP) support has first been reported as osteogenic support to allow bone formation in vivo (26). More recently, HA-loaded mesenchymal stem cell was shown to give rise to BMOS, characterized by trabecular bone and hematopoietic tissue development, which could rescue lethally irradiated experimental animals (36). This same model system was used in aging studies to show that transplanted mesenchymal cells act in distant sites delaying age related phenomena (58).
Here we present an alternative method to generate BMOS using macroporous microcarriers commonly used in tissue culture. Unlike the use of HA/TCP carriers, which were developed towards bone reconstitution (26), cellulose microcarriers were originally designed to increase tissue culture surface area. In vivo, we used them as a 3D model of stroma implant (ectopic stroma implant, ESI), which supports hematopoiesis. Cellulose microcarriers present two advantages over the HA/TCPA as vehicles for stromal cells: they are rapidly absorbed when empty and allow monitoring cell viability before in vivo implant, warranting the quality of ESI in situations where longer culture periods are necessary prior to implantation. After loaded in vitro with stromal cells, microcarriers can be implanted in the subcutaneous tissue of experimental animals as an ectopic stroma implant (ESI) functionally able to rescue lethally irradiated mice. This alternative 3D model allows the functional study of stromal cells. To test stromal cell alterations in our ESI model, we used Cx43-deficient stromal cells. Cx43 is the most abundant Cx in mammalian tissues and one element of gap junction intercellular communication (GJIC) (40). In humans, Cx43 deficiency causes oculo-dental-dysplasia (39, 43), a disease characterized by bone malformation. In hematopoietic tissue, Cx43 had been shown to be hyperexpressed in the hematopoietic niche after demand (27). Moreover, Cx43-deficient mice have reduced the number of progenitors in their fetal livers (8). Results with bone marrow chimeras using Cx43+/– host mice suggested that Cx43 expression in the radioresistant element was important for thymocyte development (37). Also, with the use of interferon (IFN)-induced gene deletion, it was shown that, after challenge with fluorouracil (5-FU), hematopoietic recovery was impaired and the number of hematopoietic progenitors diminished in experimental mice (41). However, deletion obtained with this model is not stroma specific and affects all BM cells.
One possibility to unequivocally confirm the role of Cx43 in the stroma component of the hematopoietic niche would be the use of Cx43-deficient cells in our ESI model. Cx43-deficient stroma was generated using short hairpin-based specific RNA interference (shRNA) and tested for their hematopoietic support in our ESI model. We show that Cx43 expression in the stroma population is indeed critical to maintain hematopoiesis in vivo, confirming the role of this molecule in the hematopoietic stroma and ascertaining ESI as a suitable model to evaluate individual stromal components.
Materials and Methods
Cell Lines
Cell lines were obtained from the Rio de Janeiro Cell Bank (BCRJ, Federal University of Rio de Janeiro, Brazil). Murine S17 BM stroma cells were used according to authorization by Dr. K. Dorshkind (10). All the experiments were carried out using S17 cells below the 29th passage of the original stock. Viability, assessed by trypan blue exclusion, was always superior to 95% at the beginning of each experiment.
Human Umbilical Cord Blood and Bone Marrow Samples
Human umbilical cord blood units and bone marrow samples were obtained after approval of the UCBB of the National Cancer Institute (INCA-Brazil, project number CEP INCA 31/03) and under donor informed consent. UCB was collected according to institutional standard operating procedures. Normal primary BM were obtained and processed as previously described (38).
Flow Cytometry Analysis
CD45+/CD34+ cell enumeration was done with a dual platform protocol and gating strategy similar to the International Society for Cellular Therapy (formerly ISHAGE-ISCT) guidelines following the method described by Keeney et al. (22). Kit+Sca-1+Lin- (CD117/stem cell antigen/lineage; KSL) cell enumeration was done as described elsewhere (20). Briefly, 2×106 cells/ml were incubated with after staining with anti-c-KIT-allophycocyanin (APC; 2B8 clone, Pharmingen, San Diego, CA, USA), anti-SCA-1-phycoerythrin (PE; D7 clone), anti-CD3-biotin (145-2C1 clone, Pharmingen), anti-CD45/B220-biotin (RA36B2 clone, Pharmingen), TER119-biotin (Ly76 clone, Pharmingen), anti-pan-natural killer (NK)-biotin (CD56 + CD16, DX clone, Pharmingen), anti-CD11b-biotin (M1/70 clone, Pharmingen), and anti-GR1-PECy5 (LY6C clone, Pharmingen), followed by streptavidin PECy5 (Pharmingen).
GJ-mediated intercellular communications between the stroma and hematopoietic cells were monitored by flow cytometry, following the protocol described by Czyz et al. (11) with slight modifications as previously described (38). Briefly, stroma cells were grown to semiconfluence (5×104 cells/cm2) in 24-well plates. They were washed with 0.9% NaCl solution followed by RPMI-1640 culture medium. Stroma monolayers were loaded with 1 μM calcein AM (Molecular Probes/Invitrogen, Eugene, OR, USA) for 2 h and extensively washed with a serum-free followed by serum-supplemented medium. Leukemic cells were then added in 1:1 ratio in relation to stromal cells. After 72 h, cells were trypsinized washed with PBS supplemented with 5% fetal bovine serum (FBS), fixed in 4% paraformaldehyde in PBS, and analyzed by flow cytometry. Where indicated, CBX (Sigma, St. Louis, MO, USA) was used in the coculture system to inhibit GJ-mediated cell communication.
Cytopore Microcarriers
Cytopore macroporous microcarriers (GE Healthcare, São Paulo, Brazil) were hydrated with PBS solution (100 ml/g of dry weight) in previously silliconated flasks and autoclaved at 121°C for 20 min. These microcarriers were washed with PBS and resuspended in complete medium (either RPMI or DMEM). Cells were added to the microcarriers at a concentration of 3 × 104 cells/cm2 of microcarrier area, in 2.0 ml cryotubes. These suspensions rested for 2 h at 37°C with 5% CO2. After this time, the tubes were put under mild agitation (20–30 rpm) for 45 min followed by a 15-min period of resting. This 1-h cycle was repeated over the next 12 h.
Animals
BALB/c mice were bred at the Instituto Nacional de Câncer animal facility (Rio de Janeiro, Brazil). Animals used as hosts in the transplantation protocols were females, aged 12–14 weeks old. All other animals were 8–10 weeks. Mice were housed in sterilized microisolator cages and were handled according to our institutional guidelines approval. Each transplant group had at least five mice.
Generation of Connexin 43 Silenced S17 Cell Line
Oligonucleotides corresponding to sense and antisense sequence of Cx43 or scrambled shRNA (see Table S1; available at: http://issuu.com/mbona/docs/supplement-paraguassu-braga-et-al1?mode=a_p) of the shRNA + loop were synthesized (Invitrogen, São Paulo, Brazil), annealed, phosphorilated, and cloned into XhoI and HPAI and alkaline phosphatase-treated pLentiLox pLL3.7 vector (kindly provided by Dr. Luk Van Parijs). Clones were inserted in E. coli XL1, and after confirmation by DNA sequencing, they were transfected into 293T cells using a third gene ration packing system and VSV tropism. Supernatant containing viral vectors were concentrated and S17 cells were transduced by 48-h exposure to viral stock. By this strategy, three cell lines additional to the original S17 were generated: S17.pLL, transfected with empty pLL vector; S17.SCR, transfected with pLL vector containing Cx43 scrambled sequence, and S17.Cx43, transfected with pLL vector containing the specific sequence for Cx43 inhibition.
Cobblestone Area Forming Cell (CAFC) Assay
Stromas were seeded in 96-well plates at 75% confluence, with standard culture medium. Hematopoietic stem cells (mouse bone marrow or human umbilical cord blood) were plated over the stromas in limiting dilution conditions, with 12 repetitions for each different concentration. Cocultures were left for 5 weeks, with a weekly 50% change of the medium. After this period, each well was analyzed under the microscope for the formation of cobblestone areas. The wells were then checked as positive or negative for the growth of CAFC colonies. Frequency was calculated with the L-calc software (Stem Cell, Vancouver, Canada). In experiments where carbenoxolone (CBX) was used, the medium replacement was supplemented with CBX in experimental plates.
Ectopic Stroma Implant (ESI) Model
BALB/c recipients received six subcutaneous injections of 50 μl of a 50% suspension of cytopore microcarrier previously loaded or not with the different stromas as indicated. For hematopoietic recovery studies, 1 day before transplantation, BALB/c recipients received total body irradiation for myeloablation (850 rad, TH780C irradiator with a cobalt Co 60 [60Co] source). On the next day, recipients received six injections of 50 μl of a 50% suspension of cytopore microcarriers previously loaded with stroma and 5 × 106 BALB/C bone marrow cells. Groups consisted of five to eight mice.
ESI Histological Preparation
Whenever histological analysis were necessary, ESI and eventually peri-implant tissue were carefully excised from the subcutaneous tissue of euthanized mice with the help of sterile scissors. ESIs were placed inside tissue processing cassettes and fixed in formalin (formaldehyde 4%). They were routinely embedded in paraffin and processed for hematoxilin and eosin (H&E) staining.
ELISA for Cx43
Immunoenzimatic 96-well plates (Corning, Glendale, CA, USA) were prepared with 100 μl of 100 μg/ml rabbit polyclonal antibody against Cx43, diluted in carbonate/bicarbonate buffer (Na2CO3 0.06 M mixed with NaHCO3 0.06 M in 1:4 proportion), incubated at 37°C for 2 h then at 4°C overnight. Wells were washed with PBS/ Tween 20 0.05% nonfat dry milk 1% for three times. Cell extracts made by treatment of S17 stromas with Tris.Cl 100 mmol/L and Triton X-100 0.01% then adjusted to a protein concentration of 100 mg/ml. Cells extracts were incubated for 2 h at room temperature and washed with PBS/Tween/milk. Next, wells were incubated with 50 μg/ml monoclonal antibody anti-Cx43 (Invitrogen) for 2 h at room temperature then washed. Detection was made with a TMB chromogenic system (BD Biosciences, Franklin Lakes, NJ, USA). Reading was at 620–650 nm.
Immunocytochemistry of Cx43
Cells for immunocytochemistry analysis were cultured in multiplate slides (NUNC, Rochester, NY, USA) until 75% confluence, when cells were fixed in methanol 100% v/v. After 10 min methanol was drained, slides were dried at room temperature and washed for 2 h with PBS/Tween/Milk. Next, slides were incubated with 50 μg/ml monoclonal antibody anti-Cx43 (Invitrogen) for 2 h at room temperature and washed with PBS/ Tween/Milk, followed by antimouse IgG-peroxidase. Detection was made with DAB chromogenic system (BD Biosciences, San Jose, CA, USA).
Statistical Analysis
For cobblestone area forming cell (CAFC) assay statistics were calculated using L-calc software (Pearson's statistic). For ectopic stroma implant (ESI) model and ELISA for Cx43 ANOVA test was used under Prisma Software (Graphpad Prism V5.0, Graphpad Software, La Jolla, CA, USA), with Dunn posttest.
Results
Macroporous Microcarriers Are Substrates for S17 Stromal Seeding
Macroporous microcarriers (Cytopore) used in this report are cotton cellulose cross-linked matrix covered with hydrophilic DEAE exchanger for positive charge (18). They are designed as substrates for high-density culture, which allow expansion of available growth surface area for plastic adherent cell lines. Here, we used S17 stromal cell line seeded on macroporous microcarriers under agitation for 24 h. After that, the cells can easily be seen under an optical microscope (Fig. 1A). The use of vital dyes (Fig. 1B) or crystal violet (Fig. 1C) allows morphological identification and evaluation of cell viability. In vitro the microcarriers resembles an ex vivo explant confirming the viability of the seeded cells. When taken out of the suspension culture and set in static culture conditions, cells will migrate, colonize the surrounding area, and further give rise to a new monolayer culture (Fig. 1D). This finding supports the idea that microcarrier suspension culture keeps cell adhesion properties and viability. Under histological examination (Fig. 1E), cells can be seen all over the microcarrier, with mitotic (Fig. 1F) and apoptotic (Fig. 1G) events suggesting that the dynamic maintenance of a three-dimensional culture is reproducible in this model of cell culture.

S17 stromal cells can be visualized inside the microcarriers under inverted microscopy and by histological examination. S17 stroma was seeded in microspheres incubated in cryotube vials in a density of 5 × 103 to 1 × 104 cells/cm2 for 7 days. Medium was changed every other day. (A) Aspect of cytopore microcarrier loaded with S17 cells under standard culture conditions by contrast phase microscopy. (B) Same optical field as in (A) under fluorescent microscopy, where viable cells were stained with calcein AM vital dye. (C) Cytopore were sampled and fixed with 10% paraformaldehyde and stained with violet crystal. (D) Cytopore under continuous agitation cultures were sampled and put in static cultures for 7 days. Viable cells migrate and grow as explants cultures. Leica DMIL inverted microscope, 10×40x/0.50 with Sony Cybershot DSC-W310. (E) S17 stromal cells were seeded as previously mentioned and cultured on our standard homogenization system for 5 days. After that they were fixed and processed for standard H&E histological routine and submitted to microscopic examination (10×40x/0.65). (F) Zoom-in of a section of cytopore microcarrier showing mitosis (arrow) (G) and (F) apoptotic cell (arrow). Nikon Eclipse E20 and 10×100×/1.25 with Sony Cybershot DSC-W310.
Ectopic Stroma Implant Model (ESI): Macroporous Microcarriers Loaded with S17 Are Kept In Vivo and Support Local Hematopoiesis
After being sure that the microcarriers chosen could provide an appropriate environmental support for stromal cell growth, we injected microcarriers containing the S17 cell line into the subcutaneous tissue of a BALB/c mouse syngeneic to the stroma. Figure 2 illustrates the external appearance of such transplants and compares transplants of microcarriers where S17 cells are present or not (Fig. 2A, B). When S17 stromal cells are present, a transplant nodule can be seen and measured (Fig. 2B). However, in the absence of S17 cells, the microcarriers can hardly be seen and in 2 weeks they become barely detectable (Fig. 2A, F). After 4–6 weeks, empty microcarriers are absorbed not being detectable at all. Macroanatomically, S17 full microcarriers seem to be surrounded by a membrane resembling a vascularized granuloma (Fig. 2B). At histological level, it is surrounded by mononuclear infiltrate and blood vessels can be observed on the edge of the microcarrier area (Fig. 2C). The S17 positive microcarriers are fully populated and the presence of hematopoietic cells, especially granulocytes, in different maturation stages can be observed (Fig. 2D, E).
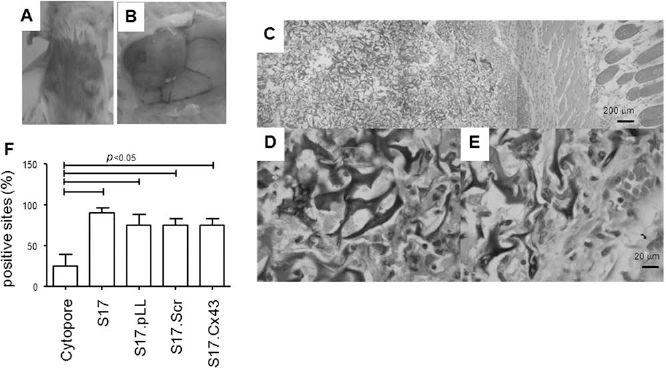
Establishment of stroma implant in a microcarrier support—ectopic stroma implant model. (A) development of ectopic stroma implant (ESI) depends on the presence of stroma: a cytopore microcarrier suspension loaded (right side) or not (left side) with stromal cells was injected in the subcutaneous tissue of BALB/c mouse for 6 weeks. Microcarriers implanted free of stroma cells do not develop. (B) Macroscopic aspect of subcutaneous tissue injected with stroma containing cytopore, showing vascular development towards the ESI. (C) Panoramic histological view of skin and subcutaneous compartment (H&E). Leica DMIL inverted microscope, 20x/0.30 with Sony Cybershot DSC-W310. (D) Section of subcutaneous area showing trabeculae-like cytopore microcarrier where fibroblastic cells can be seen. Leica DMIL inverted microscope, 40x/0.50 with Sony Cybershot DSC-W310. (E) Another section of subcutaneous area demonstrating not only fibroblastic cells, but also blood vessels and granulocytes. Mature (arrow) and immature granulocytes (arrow head) are indicated. Leica DMIL inverted microscope, 10×40x/0.50 with Sony Cybershot DSC-W310. (F) Microcarriers were injected ate six different sites in each mouse. The number of sites where a nodule was formed was counted after 4 weeks. Notice that stroma-free microcarriers hardly develop into nodes.
These results suggest that transplanted microcarriers alone are not enough to support hematopoiesis in vivo and that in the absence of stromal cells they are absorbed without any evident damage to the host.
The stromal cells transplanted inside the microcarriers might be essential to attract and support hematopoietic progenitor development. These progenitors can differentiate inside the stroma-loaded microcarriers, allowing the in vivo study of the hematopoietic supporting activity of the stroma, mimicking an ectopic bone marrow.
Short Hairpin Inhibition of Cx43 Expression in S17 Stromal Cell Lines Inhibit Hematopoietic Support In Vitro
We chose Connexin 43 (Cx43) as target molecule to test the viability of our ESI model in studies of single stromal elements important for hematopoietic support. Cx43 is the most abundant Cx in mammalian tissues and is highly expressed in the hematopoietic stem cell niche (27). Moreover, Cx43 has been suggested to be critically important to maintain hematopoietic homeostasis in different reports (8, 37). Finally, it has been recently shown that Cx43 is an important regulator of CXCL12 secretion indicating an important role for this molecule in hematopoietic stem cell niche maintenance (47).
To address the specific role of Cx43 in stromal cells, we used Cx43 shRNA silenced stromas. shRNA was transduced to the stromal cell line S17 using the lentiviral vector pLL 3.7 lentilox carrying previously described shRNA sequences targeting Cx43 (48). Silencing with shRNA for Cx43 inhibited protein expression evaluated by Cx43-specific ELISA and immunocytochemistry (see Supplementary Fig. 1A, B; available at: http://issuu.com/mbona/docs/supplement-paraguassu-braga-et-al1?mode=a_p), without affecting doubling time or phosphatase activity (see Supplementary Fig. 1C, D; available at: http://issuu.com/mbona/docs/supplement-paraguassu-braga-et-al1?mode=a_p). This indicates that at least no major modifications on S17 cell line behavior were taking place after lentiviral transduction. To study the effect of Cx43 in GJIC, immature hematopoietic progenitors (cKit+sca1+lin– or KSL) were cocultured with calcein loaded stromal cells and the number of calcein+ KSL after the coculture period was addressed. GJIC was compromised between stroma and hematopoietic progenitor cells as seen by a reduction from 80% to 30% in KSL communicating cell (calcein+) cocultured with Cx43 silenced cultures compared to control (see Supplementary Fig. 1E; available at: http://issuu.com/mbona/docs/supplement-paraguassu-braga-et-al1?mode=a_p). These results suggest that Cx43 is an important mediator of cell-cell communication between bone marrow stroma and hematopoietic progenitors.
The S17 stromal cell line silenced for Cx43 (S17.Cx43) was tested for its functionality regarding hematopoietic support. Stromas were cultured for 5 weeks with bone marrow cells in limiting dilution conditions. CAFC colonies from S17.Cx43 were less than one-fifth that of control stromas, indicating that Cx43 is indeed important to maintain immature hematopoietic progenitors (Fig. 3A). This result confirms previous results from Cancelas' group using fetal liver stroma cell lines from Cx43 knockout (KO) mice (8).

Cx43 silenced stroma cannot sustain the maintenance of immature hematopoietic progenitors. (A) Diminished hematopoietic progenitor support activity in the absence of Cx43. Total BALB/c mouse bone marrow was cocultured with the various S17 stromas in limiting dilution conditions to ascertain cobblestone area forming cells (CAFCs) as described in material and methods. Analysis was performed using L-calc software (p < 0.05). (B) Inhibition of gap junction intercellular communication (GJIC) between S17 stroma or S17 silenced for connexin 43 (Cx43) (S17.cx43) and KSL progenitor cells. Calcein AM-positive stromas were cocultured with total mouse bone marrow for 2 h. Cell suspensions were prepared after collagenase treatment and labeled for KSL. Results are expressed as percentage of total KSL cells. (C, left panel) Capacity to sustain KSL progenitors in vitro depends on Cx43. S17 stromas were cultivated to 75% confluence in 24-well plate. At this time, 106 total BALB/c BM cells were added to each well and cocultivated for 3 days. After this period the percentage of Kit+Sca-1+Lin– (KSL) cells was determined. ANOVA test with Dunn posttest (p < 0.05). (C, right panel) Kinetic of hematopoietic production on long-term bone marrow cultures confirm the importance of Cx43. The different S17 stromas were cocultured with BALB/c BM as in (C). Total (left) and weekly (right) cell production was accessed by counting the cells present in the supernatant in a hematocytometer using trypan blue dye exclusion.
We had previously shown that GJIC was important to keep leukemic cells in a quiescent state. We postulated that this would be important to maintain stem cells and in the absence of Cxs, differentiation would take place (38). This correlates with the results shown in Figure 3B where smaller numbers of immature KSL+ hematopoietic progenitors are recovered from S17.Cx43 supporting cultures. In fact, the total amount of mature cells generated when the supporting stroma is deficient in Cx43 expression is higher when compared to control cultures (Fig. 3C, left). Moreover, with time the number of cells generated on a weekly basis drops more acutely in the absence of Cx43 than in the presence (Fig. 3C, right). By the fifth week basically no nucleated cells come out of Cx43 silenced cultures and the stroma is devoid of CAFCs (see Supplementary Fig. 2; available at: http://issuu.com/mbona/docs/supplement-paraguassu-braga-et-al1?mode=a_p).
Altogether, these results indicate that the presence of Cx43 in the hematopoietic supporting stroma is indeed critical to maintain hematopoietic stem cell progenitors, at least in vitro.
ESI Can Be Used to Reconstitute Hematopoiesis and to Address the Role of Single Stromal Element in Hematopoiesis In Vivo
ESI empty microcarriers or carriers loaded with the stromal cells S17, S17.pLL, S17.Scr, or S17.Cx43 were implanted in the subcutaneous tissue of regular or irradiated mice to evaluate hematopoietic supporting activities.
ESI implants were performed at multiple sites in each individual mouse. At the site of injection, a nodule is formed as shown above (Fig. 2B). Basically, 100% of injected sites develop into nodules after 2 weeks, as long as the microcarriers are loaded with functional S17 stromas (Fig. 2F). However, the nodule size, measured at 2 weeks after injection, is dependent on the presence of functional stroma and empty carriers as well as ESI carrying Cx43-deficient stromas are smaller than ESI carrying Cx43-sufficient cells (Fig. 4A), suggesting impairment on the hematopoietic support activity in the absence of Cx43.

ESI rescue hematopoiesis, but recovery is limited in the absence of Cx43. Hematopoietic recovery is limited in the absence of Cx43. (A) ESI size depends on the stroma. Suspensions of either empty cytopores or cytopores loaded with various S17 stromas were injected subcutaneously in BALB/c mice at four to six different points. After 5 weeks, ESI was measured. (B) Development of colonized ESI depends on Cx43. ESI was performed as in (A). After 5 weeks, the subcutaneous nodules were excised and processed for histological examination. Leica DMIL inverted microscope, 40x/0.50 with Sony Cybershot DSC-W310. (C) Cx43 silenced stroma does not protect mice from radiation-induced death. Irradiated BALB/c mice received ESI with cytopores previously loaded with the various S17 stromas and BALB/c bone marrow cells. Mice receiving stroma-free cytopore/bone marrow and Cytopore/S17.Cx43/bone marrow had a lower median survival rate (20 and 14 days, respectively). (D) Peripheral blood count of irradiated BALB/c mice shown in (C). Total peripheral blood cell counts were accessed at 2, 4, and 6 weeks after transplant (n = 8/mice/group). One-way ANOVA test (p < 0.05). All cell counts were performed in a hematocytometer using trypan blue dye exclusion and, whenever necessary, acetic acid to deplete red cells.
One advantage of ESI is the possibility of recovery, which enables the analysis of the ESI inner content. ESIs were recovered and analyzed by histopathology. In the presence of control stromas, an intense hematopoietic activity is observed (Fig. 4B), which at a macroscopic level is seen as larger subcutaneous nodules (Fig. 4A). However, in the presence of S17.Cx43 the ESI is devoid of hematopoietic cells confirming the inability to either sustain hematopoiesis or attract progenitor cells (Fig. 4B).
To better investigate both, the hematopoietic reconstitution potential of the ESI and Cx43 dependence of the stroma, BALB/c mice were irradiated and reconstituted with microcarriers loaded with stroma and BALB/c total bone marrow cells. By the end of a 6-week follow-up period groups containing S17 control stroma achieved an overall survival of nearly 70% against the 40% observed for the group receiving either empty microcarriers or microcarriers loaded with S17.Cx43 stroma (Fig. 4C).
Hematological reconstitution was assessed through total nucleated blood cell count. After 2 weeks, all groups showed aplasia, even though S17.Cx43 group had a significant higher blood count compared to other groups (3% of control BALB/c leukogram against 1% in the control Cx43 positive). By the fourth week, no significant difference was seen among all groups although Cx43 silenced group tend to be smaller. By the sixth week, lack of Cx43 lead to lower blood counts in reference to all other groups (Fig. 4D). This pattern is similar to that seen in vitro, where an initial proliferation boom followed by the extinction of cell production was observed when the stroma is deficient in Cx43 (Fig. 3C), suggesting a role in stem cell maintenance for Cx43.
Discussion
Given the complexity of the stem cell niche (34, 51, 60), we wish to design a simple strategy to enable the study of stromal elements in vivo.
Our model was based on the in vivo use of cellulose matrix macroporous microcarriers (Cytopore) developed to increase tissue culture area in vitro. Empty microcarriers implanted in the subcutaneous tissue are rapidly reabsorbed suggesting that it can be employed without major risks/toxicity to the host. This could represent an advantage over ceramic HA/TCP supports. The latter has been widely used in orthopedics and experimental hematology where it serves as support to bone (26, 33) and bone marrow formation (36, 58). HA particles are bigger than the Cytopore used here, and its absorption is very slow, usually accompanied by bone formation or replacement (2). Another advantage of using cellulose microcarriers is the possibility to monitor cell viability in vitro prior to the in vivo studies, warranting the quality of the biological material to be implanted. Stromal cells can easily be identified inside the microcarriers where they are able to proliferate, apoptose, and support hematopoiesis (data not shown).
When implanted in the subcutaneous tissue, cellulose microcarriers provide different outcomes determined by the feeder stroma. If the microcarriers are used empty, without S17 cells, they are rapidly absorbed, and within 4 weeks, no implant is left. These results indicate that microcarriers by themselves are afunctional and atoxic when used in vivo. It also tells us that the stroma, in this case, prevents a fast absorbance. This could be related to the fact that mesenquimal cells are potent immunosuppressors (42) and in fact the S17 cell line has mesenquimal characteristics.
The histological examination of an ESI shows organized tissue surrounded by inflammatory mononuclear cells limiting the implant externally, resembling a granuloma. Some blood vessels are observed in the outer layer. It is important to note that the microstructure of the microcarriers resembles a BM trabeculi with hematopoietic precursors in different developmental stages being observed, especially neutrophils, indicating that progenitor cells migrate to and differentiate inside the implant. Besides, when tested in reconstitution experiments ESI were able to rescue more than 70% of animals from death, at least 30% more than control (empty microcarriers). These observations encouraged us to test if this model was suitable to in vivo study of stromal cells modifications.
The molecule chosen was Cx43, as its importance in hematopoiesis had been pointed by several groups (8, 37, 41), but its role on the stroma had not been unequivocally addressed in vivo.
Cx43-deficient stroma was generated using shRNA and tested for their hematopoietic support. In vitro, Cx43-deficient cells do not couple to KSL and have diminished CAFC support ability. Moreover, in Dexter's type cultures (13) early differentiation ascertained by increased numbers of floating cells is accompanied by decreased generative capacity, or exhaustion.
In vivo, Cx43-deficient stromas were not colonized by hematopoietic cells. This could be due to Cx43 dependent migration of hematopoietic progenitors. However, when ESI containing Cx43 deficient stroma and bone marrow cells were performed, impairment in maintenance or differentiation was evident by histopathology. Moreover, Cx43 deficient ESI were not as efficient as Cx43 sufficient ESI to rescue irradiated mice. When total nucleated cell were examined in the peripheral blood, a more efficient reconstitution is seen early (3% against 1% of nonirradiated controls). This early recovery is hampered by important loss on blood counts. At 4 weeks after implant, there is no significant difference among the various groups. However, by the sixth week mice reconstituted with Cx43-deficient ESI are severely cytopenic indicating the importance of this molecule to maintain the stem cells.
Various mechanisms had been proposed to explain the role of Cx43 in cell fate (8, 12, 17, 19, 30, 56), and this had gained attention with identification of mutations related to human oculo-dental-dysplasia (39). More than 60 Gja1 mutations had been reported with several skeletal malformations (28, 39). In experimental models, hematological defects appear as the animals age, raising the possibility of immunological defects either in senior years of after demand (15, 28). Very recently, Cx43 and Cx45 were shown to be important for CXCL12 secretion (47), a chemokine critical for osteoblastic/hematopoietic niche organization (53). In the absence of CXCL12, an increase in numbers of short-term HSCs and decreased long-term HSCs was observed (53). The Cx43 regulation of chemokine (C-X-C motif) ligand 12 (CXCL12) fits well with our results showing that, in Dexter's type cultures, differentiation and exhaustion are faster in the absence of Cx43. Moreover, faster reconstitutions with abrupt decay of hematopoiesis support is seen in vivo using ESI, showing the importance of Cx43 in the stroma and validating ESI as a model system to study the stromal elements of the hematopoietic niche.
In summary, the proposed ESI used here represents a simple alternative way to reconstitute hematopoiesis in vivo using a 3D stroma implant model, especially in the case of stroma-related diseases. As important as an ectopic hematopoietic device, it is a support for stromal cells that can receive hematopoietic cell and promote hematopoiesis. As so, it provides a mean to study and manipulate individual components of hematopoietic supportive cells present in the bone marrow environment in vivo.
Footnotes
Acknowledgments
We thank Romulo Areal Braga for helping with figures preparation, Antonio Carlos Campos de Carvalho and Regina Goldenberg for providing the anti-Cx43 antibodies, and Ana Carolina Leal and Leonardo Chicaybam for assistance with shRNA techniques. This work was supported by CNPq, FAPERJ, INCT Câncer, Ministry of Health/INCA, and Swiss Bridge Foundations grants. The following author contributions are recognized: designed and performed the experiments and analyzed the data and wrote the paper (Flávio Henrique Paraguassú-Braga), performed experiments (Ana Paula Gregório Alves), performed experiments (Isabela Maria Alvim Andrade Santos), designed shRNA experiments and analyzed data (Martin Bonamino), designed experiments and analyzed data and wrote the paper (Adriana Bonomo). The authors declare no conflicts of interest.
