Abstract
Inflammatory insults following islet transplantation (ITx) hinders engraftment and long-term function of the transplanted (Tx) islets. Using a murine model of ITx, we determined the role of LMP-420, a novel TNF-α inhibitor, both individually and in combination with the immunosuppressant cyclosporine A (CSA) in islet engraftment and survival. Diabetic C57BL/6 mice were Tx with 500 BALB/c islets under the kidney capsule. Four cohorts were used: LMP-420 only, CSA only, combination of LMP-420 and CSA (LMP+CSA), and control (n = 12 per cohort). Serial monitoring of blood glucose levels revealed that LMP+CSA (35 ± 5 days) prolonged stable blood insulin levels compared to control (6 ± 4 days). Immunohistology demonstrated that coadministration (LMP+CSA) results in a significant decrease in CD8+ T-cell infiltration (LMP+CSA: 31 ± 18 vs. control: 224 ± 51 cells, p < 0.001). Serum cytokine analysis revealed that LMP-420 administration resulted in an increase in the anti-inflammatory cytokine IL-10 (2.5-fold), and a decrease in TNF-α (threefold) with no change in IL-2. However, coadministration resulted in a marked decrease in both IL-2 and TNF-α (threefold) along with increase in IL-10 (threefold). Coadministration also demonstrated increase of antiapoptotic SOCS-1 and Mn-SOD expression and significant reduction of donor-specific antibodies (p < 0.005). In conclusion, LMP-420 administration with CSA results in the upregulation of anti-inflammatory and antiapoptotic mechanisms which facilitate islet allograft engraftment and survival.
Keywords
Introduction
Type 1 diabetes mellitus (T1DM) is an autoimmune disease characterized by the destruction of insulin-producing β-islets of the pancreas (41). Parenteral insulin administration still remains the major therapeutic strategy for treatment of patients with T1DM (31). However, this approach leads to long-term complications such as diabetic retinopathy, nephropathy, neuropathy, and macrovascular disease (30). Islet transplantation (ITx) has been proposed to be an effective treatment to achieve sustained blood glucose levels in T1DM patients. However, even with the use of immunosuppressive agents, the long-term success of islet allografts still remains poor with <10% 5-year survival (21,42). One of the major reasons for loss of islet function immediately posttransplant is inflammation caused by ischemia and anoxia (42,43,45). This initial insult, resulting from a proinflammatory milieu, predisposes to alloimmune-and autoimmune-mediated damage of the transplanted islets (14). Therefore, one of the immediate goals in ITx is to identify novel strategies to prevent early damage of these islets.
Among the factors that limit islet graft survival are inflammatory cytokines, mainly interleukin-1β (IL-1β), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α), which are released by the infiltrating mononuclear cells (7). TNF-α inhibitors have proven efficacious towards the treatment and modulation of inflammatory and autoimmune responses in diseases such as rheumatoid arthritis and psoriasis (44). LMP-420 is a novel anti-inflammatory purine nucleobase analogue that transcriptionally inhibits TNF-α production (18). LMP-420 is noncytotoxic to TNF-α-producing cells and does not bind to or interfere with the activity of preformed TNF-α (28). In addition, studies have shown that LMP-420 inhibits several of the IFN-γ-induced chemokines such as CXC chemokine ligand-9 (CXCL-9) and CXCL-10 (17). Based on these unique properties, we postulated that administration of LMP-420 following ITx would be beneficial in preventing damage to the islets and that such treatment might synergize with a currently utilized immunosuppressive regimen in prolonging islet allograft survival. Our results demonstrate that administration of LMP-420 significantly reduces early islet cell loss post-Tx leading to restoration of normoglycemia in chemically induced diabetic animals. We also demonstrate that the beneficial effect of LMP-420 is mediated by its potent anti-inflammatory and antiapoptotic pathways. Furthermore, LMP-420 acts synergistically with the commonly used immunosuppressive, calcineurin inhibitor cyclosporin A (CSA), to further enhance the survival of allogeneic transplanted islets.
Materials and Methods
Animals and Induction of Diabetes
Six- to 8-week-old C57BL/6 (H2b) and BALB/c (H2d) mice were purchased from Jackson Laboratories. Animal studies were performed in accordance with the Animal Studies Committee, Washington University, St. Louis, MO guidelines. Streptozotocin (STZ, Sigma, St. Louis, MO, 200 mg/kg) was administered intraperitoneally (IP) to induce diabetes in C57BL/6 mice. Nonfasting blood glucose (NFBG) was measured using tail snip (One Touch II-glucometer; Lifescan Inc., Milpitas, CA). Mice with two consecutive NFBG ≥ 400 mg/dl were considered diabetic.
Islet Isolation, Transplantation, and Treatment
Islets from murine pancreata were isolated by collagenase digestion and transplanted under the kidney capsule as described previously (29). In brief, donor pancreata were digested using collagenase-XI (Sigma, 4 mg/ml). Islets washed and purified on a Ficoll-gradient were handpicked and cultured for 24 h before Tx. The reversal of diabetes was defined as a reduction in consecutive NFBG <200 mg/dl post-ITx, and rejection was defined as an increase in NFBG > 250 mg/dl. LMP-420 (96–98%) was synthesized by Scynexis Inc. (Research Triangle Park, NC). The LMP-420 and CSA stock solutions were made using 5% sorbitol (Sigma). The animals were administered IP with either 5% sorbitol (n = 12) or LMP-420 alone (25 mg/kg, n = 12) or CSA alone (25 mg/kg, n = 12) or a combination of LMP-420 and CSA (both 25 mg/kg, n = 12) every day from day −1 of ITx.
Determination of Serum Insulin, Chemokines, and Cytokines
The concentrations of insulin were measured using an insulin ELISA (enzyme-linked immunosorbent assay) kit (Mercodia Inc., Winston Salem, NC). Serum obtained (stored at −20°C) from 60–80 μl blood collected by retro-orbital puncture was used. Standard insulin solution and serum (25 μl) were added into a monoclonal anti-insulin antibody (Ab)-coated ELISA plate along with enzyme conjugate and incubated for 2 h at room temperature. After washing six times, TMB (3,3′,5,5″-tetramethylbenzidine) substrate (200 μl) was added and incubated for another 5 min. The reaction was stopped by adding 50 μl of stopping solution and read at 450 nm on a microplate reader.
Serum levels of cytokines were analyzed using a multiplex bead immunoassay (Biosource International Inc., Camarillo, CA) as per the manufacturer's protocol (10). Briefly, primary Ab-coated beads with standards and samples were incubated for 2 h at room temperature. The wells were washed and incubated with biotinylated Abs for 30 min. The streptavidin-R-phycoerythrin solution was added and read using the Luminex-xMAP-system.
Analysis of T-Cell Frequency
Transplanted mice splenocytes were stimulated with irradiated donor splenocytes; the number of T cells secreting IFN-γ, IL-10, IL-4, and TNF-α was enumerated by ELISPOT (Enzyme-linked immunosorbent spot) (5). Briefly, Millipore MultiScreen® filter plates (Millipore Corporation, Billerica, MA) were coated overnight with 5 μl capture mouse cytokine-specific mAb (BD Biosciences Pharmingen™, San Diego, CA). Subsequently, 3 × 105 recipient splenocytes were cultured in triplicate in the presence of irradiated donor splenocytes (1:1 ratio). After 48–72 h, 2 μ/ml biotinylated mAb was added to the wells and incubated overnight. The plates were washed and 100 μl streptavidin-horseradish peroxidase (HRP) was added. After 2 h, the assay was developed by 3-amino-9-ethylcarbazole substrate reagent and the spots were analyzed.
Immunohistochemistry for Graft-Infiltrating CD4+ and CD8+ T Cells
Explanted kidneys containing transplanted islets were embedded in frozen tissue matrix and sections cut at a 6-μm thickness. They were fixed in cold alcohol for 2 min (-20°C), air dried, treated with 3% H2O2, and blocked with biotin/avidin (Avidin/Biotin Blocking Kit; Vector Laboratories, Burlingame, CA). The sections were incubated overnight with rat anti-mouse mAbs against cluster of differentiation 4 (CD4) and CD8 T cells (5.0 μg/ml, BD Biosciences Pharmingen™) or isotype Ab (Chemicon International, Billerica, MA). Washed sections were treated with biotin-conjugated goat anti-rat IgG followed by streptravidin-HRP. The presence of positive cells was detected with the DAB (3,3′-diaminodbenzidine) substrate kit (BD Biosciences Pharmingen™). Photomicrographs were taken of 10 random field stained sections using a Nikon Eclipse 50i microscope (10x) from each group and were analyzed using morphometric software (NIS-Elements BR 3.2 program) in a blinded fashion. Sections were also prepared from control islet grafts from nontreated mice with and without incubation with mouse CD4+ and CD8+ Abs.
Determination of SOCS-1 and Mn-SOD by RT-PCR and Western Blots
A significant portion of pancreatic islet grafts can be destroyed by apoptosis, failing to engraft in the early period immediately post-ITx. Overexpression of suppressor of cytokine signaling-1 (SOCS-1) and manganese-dependent superoxide dismutase (Mn-SOD) in islet grafts achieve an antiapoptotic effect to prolong graft survival. Expression profiles of SOCS-1, Mn-SOD, and actin were analyzed by FAM-labeled RT-PCR primers (Applied Biosystems, Foster City, CA). Briefly, total RNA was extracted from islet grafts using TRIzol reagent. The RNA was reverse transcribed and real-time RT-PCR was performed using iCycler 480 Probes Master (Bio-Rad Laboratories, Inc., Hercules, CA). Cycling conditions consisted of an initial denaturation of 95°C for 15 min, followed by 40 cycles of 95°C for 30 s, followed by 61°C for 1 min.
Protein levels of SOCS-1 and Mn-SOD were analyzed by Western blot. The graft extracts were obtained using 4% SDS cell lysis buffer containing protease inhibitor cocktail. Extracts were boiled for 20 min in sample buffer [200 mmol/L Tris (pH 6.8), 20% glycerol, 2% SDS, 0.1% bromophenol blue, and 10% β-mercapto-ethanol], centrifuged for 30 min, and run on 4–12% gradient Bis-Tris denaturing gels (NuPage®; Invitrogen). The gels were transferred onto nitrocellulose membranes, and blocked overnight with 5% nonfat milk in PBS-T (0.1% Tween20). Thereafter, the membranes were incubated for 1 h with the appropriate Abs. After labeling with HRP-labeled secondary Ab (1:10,000 dilution; Jackson ImmunoResearch Laboratory Inc., West Grove, PA), the membranes were developed using chemiluminescence detection kit (Millipore) and analyzed using Bio-Rad Universal Hood-II (Bio-Rad Laboratories, Inc.). Morphometric analysis was performed using the software provided by the company.
Detection of Donor-Specific Abs (DSA) in the Recipient's Sera by Immunofluorescence
Sera from sensitized C57BL/6 mice were assayed for allo-Abs by their ability to bind BALB/c splenocytes using flow cytometry. Donor splenocytes (0.5 × 106) were incubated in 50 μl fluorescence-activated cell sorter (FACS) buffer (5% FBS, 1% HEPES, 0.2% sodiumazide in HBSS), containing 1:10 recipient serum for 30 min at 4°C. Anti-mouse phycoerythrin (PE)-labeled secondary Ab was added and further incubated for 30 min. The cells were then washed and read on a fluorescence-activated cell sorter (FACS) caliber machine with instrumental settings for mouse splenocytes.
Statistical Analysis
Data are expressed as mean ± SD. Statistical differences between means were analyzed using a paired or unpaired Student's t-test, or subjected to Scheffe's post hoc test in analysis of variance (ANOVA). Using the Scheffe procedure all possible comparisons between the means of controls with individual treatment groups (LMP-420 alone, CSA alone, and LMP+CSA group) were conducted. A value of p < 0.05 was considered significant.
Results
LMP-420 and CSA Prolongs Islet Allograft Survival
To determine the effect of LMP-420, diabetic C57BL/6 mice were treated with LMP-420 1 day prior to ITx (500 BALB/c islets) and daily thereafter. As shown in Figure 1, LMP-420 significantly prolonged islet allograft survival (22 ± 3 days), whereas the vehicle-treated cohort rejected the allograft on 7 ± 2 days post-Tx (p = 0.005). To confirm that normoglycemia was achieved by the transplanted islets, a group of LMP-420-treated mice was nephrectomized 14 days post-ITx. Nephrectomy resulted in prompt elevation of NFBG levels, demonstrating that normoglycemia was being achieved by the transplanted islets (Fig. 1A). Daily administration of CSA also significantly prolonged graft survival (24 ± 3 days) compared with control (p = 0.001) (Fig. 1B). These results demonstrate that LMP-420 prolongs islet allograft survival similar to that observed with CSA.

Effect of administration of LMP-420 (A), cyclosporin A (CSA), and LMP+CSA (B) on islet allograft survival. Diabetic C57BL/6 mice were transplanted (Tx) with 500 BALB/c islets. LMP-420 (25 mg/kg), CSA (25 mg/kg), and LMP+CSA (25 mg/ kg each) were administered by IP injection daily from day −1. Nonfasting blood glucose (NFBG) levels were monitored twice a week by glucometer utilizing a tail snip method. Data represents mean ± SD for n = 12.
Synergism of LMP-420 with Low-Dose CSA in Prolonging Islet Allograft Survival
The above results demonstrated that either LMP-420 or CSA alone significantly prolonged islet allograft survival. However, islets were rejected between days 23 and 27 post-Tx, respectively. Therefore, we sought to determine whether LMP-420 could act synergistically with CSA to prolong allograft survival. Combined treatment (LMP+CSA) significantly prolonged allograft survival (35 ± 5 days; p = 0.0001) compared to control (Fig. 1B). These results demonstrate that combination of the anti-inflammatory LMP-420 with immunosuppressive agents such as calcineurin inhibitors can result in enhanced islet graft survival. Furthermore, the sera from the control cohort had significantly lower (p = 0.0002) (Fig. 2A) insulin on day 8 compared with all three treatment cohorts (LMP, CSA, or LMP+CSA). All three treatment regimens demonstrated insulin levels up to 0.5 ± 0.1 pg/ml from day 8 through day 20. Additionally, serial histology sections of transplanted islets from treatment groups showed positive staining for insulin on day 8, signifying the presence of functionally healthy islets (Fig. 2B). Insulin levels in LMP-420 and CSA treatment cohorts reached pre-Tx levels by day 22, indicating the rejection. In contrast, the combined treatment cohort retained high serum insulin levels at 32 days post-Tx, corresponding to the graft survival curve (Fig. 1B).

Determination of insulin content in islet allograft recipient's serum by insulin ELISA. Sera (25 μl) were added to monoclonal anti-insulin Ab-coated ELISA plates (Mercodia Inc., Winston Salem, NC) with enzyme conjugate and incubated for 2 h. After washing (6x), they was incubated with TMB substrate (200 μI) for 5 min. After adding 50 μl of stopping solution the plate was read at 450 nm. (B) Average numbers of insulin-positive [anti-mouse fluorescein isothiocyanate (FITC) Abs] cells in serial histology sections of explanted grafts from control, LMP-420, CSA, and LMP+CSA treatment cohorts on day 8 post-Tx, respectively (**p < 0.01).
LMP-420, CSA, and LMP+CSA Administration Delay the Infiltration of Donor-Specific CD4+ and CD8+ Cells Into the Islet Grafts
To identify the kinetics and specificity of cells infiltrating the islet allografts, we performed immunohistology of the grafts on day 8 post-Tx, a time point when controls start rejecting. Grafts from normoglycemic mice were removed from all three treatment cohorts and sections were stained for CD4+ and CD8+ cells. As shown in Figure 3, the control grafts showed marked infiltrating CD4+ and CD8+ cells (152 ± 42 and 224 ± 51 cells, respectively, p = 0.0001) enumerated by morphometric analysis using NIS-Elements BR 3.2 software. In contrast, the LMP-420 and CSA groups had 42 ± 20, 58 ± 30 and 76 ± 24, 112 ± 42 CD4+ and CD8+ cells, respectively, which though reduced was not very significantly different from controls. In contrast, infiltrations of CD4+ and CD8+ cells (22 ± 10 and 31 ± 18 cells, respectively) following combined treatment with LMP+CSA were significantly decreased compared to control (p = 0.0007) as well as LMP-420- or CSA-treated cohorts (p = 0.004). These results demonstrate that the infiltration of T cells into the allograft was significantly reduced when LMP-420 is combined with CSA, which agrees with the prolonged islet allograft survival noted with this regimen.
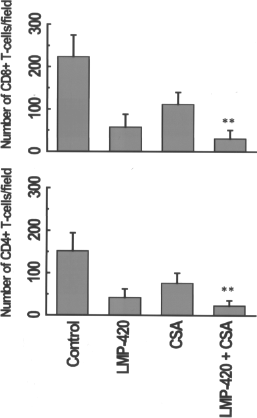
Immunohistochemical staining for graft infiltrating T cells. Cryosections of islet grafts from mice on day 8 post-Tx were stained with rat mAbs specific to mouse CD4+ and CD8+ T cells. The secondary biotinylated goat anti-rat Ab was added followed by streptavidin-HRP and developed using DAB substrate. Pictures taken (10X; Nikon Eclipse 50i microscope) were used for morphometric analysis using NIS-Elements BR 3.2 program. The data represents mean ± SD (**p < 0.01).
Administration of LMP-420 or CSA and Combined Treatment Leads to Decreased Production of Proinflammatory Mediators
Ischemia-reperfusion injury results in increased production of proinflammatory cytokines that accelerate islet cell death (5,13). To test whether LMP-420, CSA, and the combination decreases the levels of inflammatory cytokines and chemokines, sera collected from all four groups on day 8 post-Tx were analyzed using a Mouse Twenty-plex Luminex kit. As shown in Figure 4A, cytokines IL-1β and IL-4 were 1.3- to 2-fold lower in all three treatment cohorts compared to control (p = 0.01). Furthermore, the T-cell proliferative cytokine IL-2 was 2.5- to 3-fold lower in the CSA and LMP+CSA cohorts (p = 0.01) but not in the LMP-420 treatment cohort compared to control (144 ± 18 vs. 178 ± 21 pg/ ml, p = 0.145). In contrast, serum IL-10 levels in the LMP-420 treatment cohort were 2.5-fold (221 ± 9.8 vs. 86 ± 14.3 pg/ml, p = 0.0001) and 1.6-fold (221 ± 9.8 vs. 136 ± 18 pg/ml, p = 0.004) higher than those in the control and CSA administered mice, respectively. Furthermore, the LMP+CSA group had 3-fold higher concentrations of IL-10 in their sera compared to controls (86 ± 14.3 vs. 240.6 ± 35 pg/ml, p = 0.00001), indicating a potentially significant role for LMP-420 in increasing IL-10-mediated anti-inflammatory effects. Additionally, LMP-420 alone and LMP+CSA cohorts had 3- and 2-fold reductions of TNF-α, respectively, in their sera compared with the control cohort (p = 0.001). The CSA alone-treated group also showed a decrease in TNF-α concentration (372.3 ± 47 vs. 284.3 ± 27 pg/ml, p < 0.05) compared to controls.

Analysis of cytokines and chemokines in transplanted mouse sera by Luminex. Sera collected on day 8 post-Tx were used for comparative screening of various cytokine levels using a Luminex kit according to the protocol provided by the company. The data are representative of three separate experiments done in triplicate, and the SD bars are illustrated for the Luminex assay (*p < 0.05, **p < 0.01).
Chemokines produced after Tx play a pivotal role in recruitment of inflammatory cells (25). To determine the effect of LMP-420 on chemokine production, sera were analyzed for the levels of chemokines that play critical roles in directing mononuclear cells to islet allografts (48). As depicted in Figure 4B, LMP-420 and LMP+CSA decreased the levels of IFN-γ-inducible protein-10 (IP-10), monocyte chemotactic protein-1 (MCP-1), and macrophage inflammatory protein (MIP) by 1.5- to 2.9-fold (p < 0.001) compared to control. In contrast, there was no significant difference in the concentrations of the above chemokines between control and CSA cohorts. In addition, the concentrations of chemokines keratinocyte chemoattractant (KC) and monokine induced by IFN-γ (MIG) were found to be significantly less (1.6- to 2.5-fold, p < 0.001) in all LMP-420, CSA, and LMP+CSA treatment groups compared to controls.
Low Frequency of Donor Reactive T-Cells in the Recipient Spleen After Treatment with LMP-420, CSA, and LMP+CSA
To study the effect of treatment with the above agents on the initiation of alloimmune responses after ITx, we determined the frequency of IL-4-, IL-10-, IFN-γ-, and TNF-α-secreting T cells upon stimulation with donor cells by ELISPOT on day 8 post-Tx (Fig. 5). We observed 2.3-, 2-, and 4.1-fold reductions (p < 0.001) in the frequency of IL-4-producing T cells in LMP-420, CSA, and LMP+CSA compared with controls, respectively (Fig. 5A). Similarly, the IFN-γ-producing T-cell frequency was reduced by 1.6-, 2.0-, and 2.9-fold (p < 0.001) compared with control, respectively (Fig. 5C). The frequency of TNF-α-producing T cells in LMP-420 and LMP+CSA administered mice was 3-fold less (p < 0.01), whereas the CSA alone group demonstrated a 1.3fold decrease (p < 0.05) (Fig. 5D). In contrast, LMP-420, CSA, and LMP+CSA groups had 2-, 1.4-, and 2.4-fold increases in the frequency of IL-10-producing T cells, respectively (Fig. 5B), compared to controls. These ELISPOT results correlated well with the decreased levels of proinflammatory and increased levels of anti-inflammatory cytokines in the sera of treatment cohorts.

Determination of frequency of donor-specific T cell by ELISPOT. Recipient (C57BL/6) splenocytes (3 × 105) isolated on day 8 post-Tx from treated and control cohorts were stimulated with irradiated donor (BALB/c) splenocytes (3 × 105) and analyzed for production of interleukin (IL)-4 (A), IL-10 (B), interferon-γ (IFN-γ) (C), and tumor necrosis factor-α (TNF-α) (D). The data are representative of three separate experiments performed in triplicate, and SD bars are illustrated for these assays (*p < 0.05, **p < 0.01).
Enhanced SOCS-1 and Mn-SOD Levels with LMP and CSA Combination
The results presented above indicated enhanced production of anti-inflammatory molecules after treatment with LMP-420 and CSA. To further determine the potential mechanisms involved in prolonged allograft survival, we analyzed for the expression of SOCS-1 and Mn-SOD, two important molecules involved in the prevention of islet apoptosis. Mn-SOD is a mitochondrial antioxidant enzyme, increased expression of which protects islet cells from inflammatory damage (19). The transcription factor SOCS-1 has the ability to negatively regulate inflammatory cytokine signaling and can also inhibit islets apoptosis through suppression of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway (23). The effect of combined treatment with LMP+CSA on the intracellular expression of these molecules was determined using both Western blot and RT-PCR analyses of explanted grafts on day 8 post-Tx. As shown in Figure 6A and B, Western blot and densitometry analysis clearly demonstrated that there are 2.8 ± 0.6- and 1.6 ± 0.4-fold increases in the levels of SOCS-1 and Mn-SOD (p < 0.001) in allografts from LMP-420-treated mice. Furthermore, the LMP+CSA demonstrated a very significant 6.2 ± 0.6-and 4.3 ± 0.5-fold increase of SOCS-1 and Mn-SOD levels, respectively (p < 0.0001). RT-PCR analysis of mRNA expression levels of SOCS-1 and Mn-SOD in treatment cohorts also demonstrated enhanced expression of these transcription factors (11.8 ± 3.1 and 6.8 ± 2.1, respectively) with coadministration over LMP-420 alone (4.3 ± 1.1 and 3.2 ± 0.5, respectively) (Fig.6C, D). In addition, blinded morphometric analysis of the explanted islet grafts (day 8) upon terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining demonstrated 3.2-fold (12 ± 7 vs. 47 ± 13 cells) decreased numbers of apoptotic cells in LMP+CSA treatment group (data not shown) compared to the control, respectively. These results further support that LMP-420 induces upregulation of the antiapoptotic and anti-inflammatory transcriptional factors Mn-SOD and SOCS-1, which is beneficial for the continued function of transplanted islets.
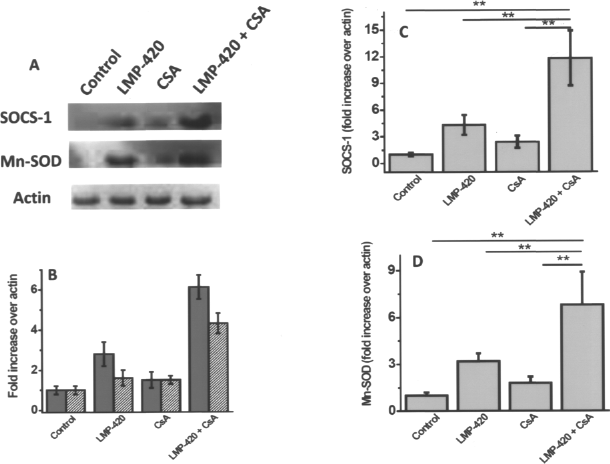
RT-PCR and Western blot analysis of SOCS-1 and Mn-SOD with LMP-420 and CSA treatment. (A) Western blot analysis of suppressor of cytokine signaling-1 (SOCS-1) and manganese-dependent superoxide dismutase (Mn-SOD) on the explanted islets grafts obtained on day 8 post-Tx. Islets grafts were lysed with lysis buffer and the cytoplasmic extracts from all four cohorts were analyzed for SOCS-1 and Mn-SOD proteins using mAbs. (B) Densitometric analysis of the protein expression (gray bars represent SOCS-1 and hatched bars represent Mn-SOD). (C, D) RT-PCR on the mRNA extracts of the explanted islets was performed for SOCS-1 and Mn-SOD, respectively, expression using FAM-labeled primers. Data are from four different experiments and mean ± SD is reported.
LMP-420, CSA, and LMP+CSA Administration Delay Formation of Abs to Donor Major Histocompatibility Complex (MHC) (DSA) in the Recipient
Even after continued treatment with LMP-420 and LMP+CSA allograft rejection occurred at 23 and 35 days post-Tx, respectively. Studies from our laboratory, and others, have shown that the development of DSA correlates with the rejection of transplanted islets (6,29). Therefore, sera collected at the time when glucose levels were rising were analyzed for development of DSA by FACS. The sera of mice treated with LMP-420 or CSA alone showed DSA on day 22 post-Tx (Fig. 7A, B). This paralleled the increase in NFBG and decreased insulin levels, indicating rejection of transplanted islets. In contrast, LMP+CSA-treated sera showed no Abs binding to the donor splenocytes on day 22 (Fig. 7C), with both normoglycemia and insulin levels significantly higher, indicating functional islets. However, the presence of DSA was detected on day 33, when all the mice became hyperglycemic (Fig. 7D). These results indicate that combined treatment of LMP-420 with CSA can not only downregulate the cellular immune recognition of the graft but also delay the humoral response against the graft.
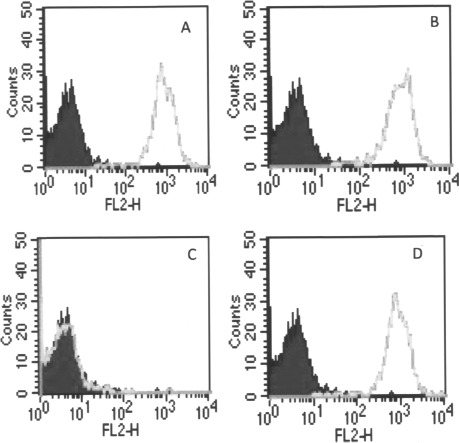
Analysis of donor-specific antibodies (DSA) in recipient sera by fluorescence-activated cell sorter (FACS). Donor splenocytes (5 × 105) were incubated in 50 μl FACS buffer containing 1:10 dilution of recipient serum for 30 min at 4°C. Phycoerythrin (PE)-labeled secondary Ab was added and further incubated for 30 min. The cells were washed three times with FACS buffer and read on FACS caliber machine with instrumental setting for mouse splenocytes. (A–C) LMP-420, CSA, and LMP+CSA on day 22. (D) LMP+CSA on day 33.
Discussion
Islet transplants are subjected to ischemia and reperfusion injuries as a consequence of both their isolation and Tx procedures (4,22,38). Prior to vascularization, transplanted islets are susceptible to inflammation and apoptosis due to lack of nutrition and compromised blood flow. Furthermore, the processes of coagulation and thrombosis contribute to multiple insults suffered by the islets during early post-Tx period (38). These factors induce inflammation, apoptosis, and loss of islets, subsequently inducing adaptive immune responses that compromise the long-term survival. It is estimated that up to 70% of the islets are lost in the early post-Tx period, contributing to the need for islets from multiple pancreata to achieve insulin independence in a single recipient (2). Interest in clinical ITx has contributed to development of improved harvesting, purification procedures, and new steroid-free immunosuppressive drug regimens that minimize β-cell toxicity and insulin resistance (45). This has resulted in excellent rates of engraftment and achievement of normoglycemia. Nonetheless, recent longer term follow-up data of ITx recipients have shown that most recipients who are rendered free from insulin therapy at 1 year need to reinstitute insulin therapy within 5 years post-Tx (42).
Traditionally, CSA is an immunomodulator that is effectively utilized to prevent rejection in solid organ Tx such as heart, liver, and kidney (3,9,12). It has a high specificity for T lymphocytes and inhibits cell-mediated immune responses. We previously identified a novel small molecule, LMP-420, which effectively inhibits TNF-α production both in vitro and in vivo (17). In our current study using a murine model of ITx we demonstrate that a combination of LMP-420 with low-dose CSA results in enhanced islet engraftment resulting in significant prolongation of islet allograft function. This enhanced engraftment is a result of both downregulation of proinflammatory cytokines/chemokines and upregulation of anti-inflammatory cytokines/chemokines. More importantly, this combined treatment results in the upregulation of both the transcriptional and protein levels of SOCS-1 and Mn-SOD, two molecules previously demonstrated to be important in allograft survival (11,24).
Initial insult to β-cells following the Tx reduces their functional efficiency. In our murine model, animals continually treated with LMP-420 and CSA showed better islet allograft survival as evidenced by continued normoglycemia and increased serum insulin levels (Figs. 1 and 2). Proinflammatory cytokines have been shown to play a significant role in acute graft rejection. Pascher et al. (33) demonstrated that anti-TNF therapy attenuated the acute rejection episode. Studies have shown that the local expression of the inflammatory cytokine TNF-α can impair tolerance to both auto- and alloreactivity (46). Endogenous overexpression of TNF-α resulted in accelerated rejection of transplanted islets in both autoimmune and chemically induced diabetic mice, compared to wild-type, by accelerating the infiltration of CD4+ and CD8+ T cells (34). Our results demonstrate (Fig. 3) reduced infiltration of both CD4+ and CD8+ T cells by 70% on day 8 following administration of LMP-420 alone. However, coadministration of LMP-420 and CSA reduced the T-cell infiltration by 95%. This decreased cellular infiltration and significantly decreased serum TNF-α noted following administration of LMP-420 indicated that these mechanisms play an important role in the noted prolongation of islet allografts following LMP-420 administration.
The role of proinflammatory responses in graft rejection is well defined (1,26). T cells are important in the pathogenesis of rejection, and constitute a predominant infiltrating cell type. However, other mononuclear cell subpopulations, including B cells, natural killer (NK) cells, and monocytes/macrophages also have been shown to contribute in rejection (37,39). Proinflammatory cytokines/chemokines released from the initial infiltrating cells plays a major role in the continued infiltration of cells to the graft (5,20,25,49). In this study we demonstrated that LMP-420 not only attenuates proinflammatory TNF-α, IL-1β, and MCP-1 responses, but also augments an anti-inflammatory IL-10 response (Fig. 4). We also demonstrated that the combination of LMP-420 and CSA provided a synergistic effect on the cytokine environment that facilitated islet allograft survival. Thus, our results demonstrated that the delayed islet allograft rejection is due to a skewing of the equilibrium towards an enhanced IL-10 with decline in proinflammatory cytokines including TNF-α, IL-1β, IL-2, and IL-4. Based on our data, we conclude that regulatory mechanisms were induced by LMP-420, which inhibits the proinflammatory TNF-α and simultaneously turns on the anti-inflammatory IL-10. A similar reduction in proinflammatory cytokine responses following administration of LMP-420 has been reported by Haraguchi et.al. (18) for the potential use of this agent to treat HIV patients.
Various studies demonstrate that TNF-α causes decreased expression of Mn-SOD and SOCS-1, both of which are known to reduce TNF-α-induced apoptotic effect of caspases (15,23,35). Suo et al. have demonstrated that SOCS-1 inhibits apoptosis in islet grafts through caspase 3-dependent and apoptosis-inducing factor caspase-independent pathways (47). In animal models inactivation of Mn-SOD has been correlated with Tx rejection in cardiac (32) and renal (36) allografts. In our current study, we demonstrate that LMP-420 downregulates chemokines down stream of TNF-α signaling, resulting in enhanced expression of Mn-SOD (1.6 ± 0.4-fold) and SOCS-1 (2.8 ± 0.6-fold). These data clearly demonstrate the antiapoptotic effect of LMP-420, which is crucial for the enhanced survival and functionality of Tx islets.
Previous studies from our laboratory, and studies by others, have demonstrated that Abs to HLA in human ITx has a positive correlation with graft rejection (8,27,40). Studies in animal models of ITx also indicate that Tx across sensitization to major histocompatibility (MHC) antigens is unfavorable towards islet allograft survival and function (6). Earlier studies in our laboratory demonstrated that ITx into MHC-mismatched STZ-treated diabetic mice resulted in the rejection of islets with mean survival time 2 days when compared to unsensitized mice with mean survival time of 10 days (16). In our current report, we demonstrate that the enhanced protection and survival is also evidenced by delayed circulating levels of DSA in the cotreatment cohort. FACS analysis demonstrated appearance of DSA during the rejection (22 days post-Tx) in LMP-420 and CSA alone groups while no DSA was detected in serum from cotreatment mice. The serum DSA development was detected during the final stages of graft rejection (35 days post-Tx), indicating that combined treatment not only downregulates cellular immune response but also delays the humoral immune response against the graft.
Our results conclusively demonstrate that LMP-420 has a potent anti-inflammatory and antiapoptotic effect. LMP-420 has been further demonstrated to enhance islet survival and function by inhibiting the alloimmune humoral and cellular responses. Based on our data, we propose a novel therapeutic application for LMP-420 in combination with current therapeutic regimens using calcineurin inhibitors, such as CSA, in the amelioration of rejection following human islet transplantation.
Footnotes
Acknowledgments
This work was supported by JDRF 1-2007-565 and BJC foundation (T.M.). N.A. is the recipient of JDRF Postdoctoral Fellowship (3-2009-218). The authors thank Ms. Billie Glasscock for her help in preparing this article. The authors declare no conflict of interest.
