Abstract
Understanding the basic mechanisms that control CNS remyelination is of direct clinical relevance. Suitable model systems include the analysis of naturally occurring and genetically generated mouse mutants and the transplantation of oligodendrocyte precursor cells (OPCs) following experimental demyelination. However, aforementioned studies were exclusively carried out in rats and little is known about the in vivo behavior of transplanted murine OPCs. Therefore in the present study, we (i) established a model of ethidium bromide-induced demyelination of the caudal cerebellar peduncle (CCP) in the adult mouse and (ii) studied the distribution and marker expression of the murine OPC line BO-1 expressing the enhanced green fluorescent protein (eGFP) 10 and 17 days after stereotaxic implantation. Injection of ethidium bromide (0.025%) in the CCP resulted in a severe loss of myelin, marked astrogliosis, and mild to moderate axonal alterations. Transplanted cells formed an invasive and liquorogenic metastasizing tumor, classified as murine giant cell glioblastoma. Transplanted BO-1 cells displayed substantially reduced CNPase expression as compared to their in vitro phenotype, low levels of MBP and GFAP, prominent upregulation of NG2, PDGFRα, nuclear p53, and an unaltered expression of signal transducer and activator of transcription (STAT)-3. Summarized environmental signaling in the brain stem was not sufficient to trigger oligodendrocytic differentiation of BO-1 cells and seemed to block CNPase expression. Moreover, the lack of the remyelinating capacity was associated with tumor formation indicating that BO-1 cells may serve as a versatile experimental model to study tumorigenesis of glial tumors.
Keywords
Introduction
Transplantation of myelin-producing cells into the normal and demyelinated brain and spinal cord has been frequently used to study both the viability of the grafted tissue (16) as well as the potency of these cells to produce myelin (4,10,19,25,31,58,71,73). The analysis of the basic mechanisms that control central nervous system (CNS) remyelination is of particular clinical relevance since it may prompt the development of novel therapeutic strategies to cure demyelinating disorders, such as multiple sclerosis (22,23). Oligodendrocytes arising from oligodendrocyte precursor cells (OPCs) represent the major cell population capable of myelin formation in the CNS (1,9,45,60). Lacking or limited production of myelin under pathophysiological conditions has been ascribed to either a loss of OPCs or a reduced differentiation of OPCs into mature oligodendrocytes (22,23,37,68,69).
Experimental approaches to study demyelination and remyelination include transplantation of OPCs into the demyelinated CNS as well as the analysis of naturally occurring and genetically induced mouse mutants (3,15,17,24,35,41,49). These studies were almost exclusively performed in rats. To fully exploit the potential of this technology and to analyze the relevance of specific molecules, it would be important to use mutant animals for cell isolation or as a host receiving transplants. Though the majority of available transgenic animals represent mice, most OPC transplantation studies and investigations using experimental demyelination of the CNS were carried out in rats (20,74). Historically, this is most likely due to the fact that isolation and culturing of OPCs became first feasible in the rat model, and that isolation and purification as well as marker expression of murine OPCs is different and more elaborate in mice than in rats (12). Thus, little is known about chemically induced demyelination in the CNS of mice and the behavior of murine OPCs following transplantation.
In the present study, we therefore aimed to establish ethidium bromide-induced demyelination in mice as a new model. The target structure we selected was the caudal cerebellar peduncle (CCP). The CCP is an anatomically clearly defined structure that contains a large number or parallel axons of similar caliber allowing morphometric analysis (74). For implantation, the mouse OPC BO-1 cell line was used (56). This cell line was previously generated by spontaneous immortalization of OPCs during long-term in vitro culturing. Although cell lines bear the potential risk of tumor formation, several studies implanted immortalized OPCs and demonstrated remyelination (20,21). Murine OPC lines were invariably created by genetic approaches (6,30,43). In contrast the murine BO-1 cell line was not generated by transfection instead immortalization occurred spontaneously during in vitro culturing. In vitro, BO-1 cells expressed markers typical for immature OPCs and displayed a strong proliferative capacity. However, coculturing with astrocytes as well as treatment with retinoic acid (RA) induced a significant upregulation of oligodendrocyte markers, such as 2′, 3′-cyclic nucleotide 3′-phosphodiesterase (CNPase), and myelin basic protein (MBP), while addition of serum, known to trigger astrocytic differentiation, induced a slight, significant increase in the expression of glial fibrillary acidic protein (GFAP). Transplantation of BO-1 cells into the demyelinated CCP was carried out to demonstrate whether this environment may trigger an even more pronounced oligodendrocytic differentiation.
Materials and Methods
Experimental Animals
All experiments were performed with 8-week-old, female SJL/JOlaHsd mice (Harlan Winkelmann, Borchen, Germany). Animals were housed in isolated ventilated cages (Tecniplast, Hohenpeißenberg, Germany) in a controlled environment (22.5 ± 1°C, 55 ± 5% relative humidity, 12-h light/dark cycle, 74 changes of air per hour), fed a standard rodent diet (R/M-H; Ssniff Spezialdiäten GmbH, Soest, Germany) ad libitum, and had free access to tap water. The study was conducted in accordance with the German Animal Welfare Law and all experiments were approved by the local government (permission number: 33.9-42502-04-09/1627).
Cell Culture and eGFP Transfection
Establishment and maintenance of the spontaneously immortalized OPC line BO-1 derived from SJL/JOlaHsd mice were described previously (56). BO-1 cells, containing plasmid enhanced green fluorescent protein-C1 (pEGFP-C1; Clontech, CA, USA), were constructed by lipofectamine-mediated transfection (Lipofectamine™ 2000; Invitrogen, Karlsruhe, Germany) according to the manufacturer's instructions. Briefly, BO-1 cells were seeded onto six-well microtiter plates (Nunc, Wiesbaden, Germany) at a density of 2 × 105 cells/well 24 h prior to transfection. Cells were incubated with pEGFP-C1 and lipofectamine-containing Dulbecco's modified Eagle medium (DMEM; Invitrogen, Karlsruhe, Germany) for 3 h at 37°C, 5% CO2. Selection of pEGFP-C1-positive cells was done using G 418 (100 μg/ml, Sigma-Aldrich, Munich, Germany) and single cell dilutions in 96 microtiter plates (Nunc, Wiesbaden, Germany). Cells were grown in B104-conditioned DMEM (45,56). Prior to transplantation, cells were washed twice with phosphate-buffered saline (PBS, pH 7.4), mechanically detached, and resuspended in DMEM at a density of 0.5 × 105 cells/μl.
Characterization of BO-1 Cells In Vitro
Immunostaining of BO-1 cells was performed in 96 microtiter plates as previously described (56). Briefly, paraformaldehyde-fixed cells were treated with PBS-Triton X-100 (0.25%) for 15 min and incubated with primary antibodies diluted in PBS containing bovine serum albumin (BSA, 1%) and Triton X-100 (0.25%) for 2 h at room temperature. A monoclonal mouse anti-human CNPase antibody (clone 11-5B, MAB326, diluted 1:800, Millipore, Schwalbach, Germany) and a polyclonal rabbit anti-rat nerve/glial antigen 2 (NG2) antibody (AB5320, diluted 1:200, Millipore) were used. Antigen-antibody reactions were visualized using Cy™3-conjugated goat anti-mouse and Cy™3-conjugated goat anti-rabbit antibodies (Jackson ImmunoResearch, Suffolk, UK) at a dilution of 1:200. Nuclei were counterstained using bisbenzimide (0.01% in ethanol; Sigma-Aldrich, Munich, Germany) (72). Cells were studied under an inverted fluorescence microscope (Olympus IX-70, Olympus, Hamburg, Germany) and microphotographs were taken employing the PM-30 system (Olympus). The total number of positive cells per well was determined by counting five randomly selected areas as described (56).
Ethidium Bromide-Induced Demyelination of the Adult Mouse Caudal Cerebellar Peduncle
Anesthesia of adult mice was induced by intraperitoneal injection of medetomidine (0.5 mg/kg, Domitor; Pfizer, Berlin, Germany) and ketamine (100 mg/kg, ketamin 10%; WDT, Garbsen, Germany). Analgesia was achieved by an intraperitoneal application of tramadol (15 mg/kg; Ratiopharm, Ulm, Germany) and carprofen (4 mg/kg, Rimadyl; Pfizer, Berlin, Germany). Mice were positioned in a flat-skull position (bregma and lambda at the same horizontal level) on a small animal stereotaxic instrument (TSE Systems, Bad Homburg, Germany) and injections were performed employing a 10-μl syringe with 30-gauge needle (Hamilton, Bonaduz, Switzerland) into the left CCP (coordinate values: rostrocaudal −4.9 mm, dorsoventral 4.4 mm, lateral −2.0 mm). For adaption of the previously described model of ethidium bromide-induced demyelination of the CCP in rats (74) to SJL/JOlaHsd mice, animals received injections (2 μl) with varying ethidium bromide concentrations (0.1%, 0.05%, 0.025%, 0.01%, and 0%; Sigma-Aldrich). Animals were inspected daily for clinical signs. These pilot experiments identified 0.025% ethidium bromide as the appropriate concentration to induce demyelination of the CCP. Thus, this concentration was used for all subsequent cell transplantation experiments.
Stereotaxic Implantation of BO-1 Cells Into the Demyelinated and Nondemyelinated CCP
Four days after injection of ethidium bromide in the adult mouse CCP, animals of the BO-1 cell transplantation group (n = 9), received an injection of 105 B0-1 cells in 2 μl of DMEM into the same location, while the mock transplanted control group (n = 4) received the same volume of the vehicle alone. After injection, the needle was left in place for 5 min to prevent back flow of the fluid along the injection canal. Mice were killed 10 days (n = 2, BO-1 cells) and 17 days post-cell transplantation (n = 7, BO-1 cells; n = 4, mock transplanted) under deep anesthesia by perfusion with PBS followed by 4% paraformaldehyde in PBS. To investigate the in vivo behavior of BO1-cells in the nondemyelinated environment, animals (n = 4) received 2 μl DMEM containing 105 BO-1 cells. Animals were killed 10 days after cell transplantation under deep anesthesia, as described above.
Light Microscopy, Immunocytochemistry, and Statistical Analysis
Cerebrum, cerebellum, and transversal segments of the cervical, thoracic, and lumbar spinal cord were postfixed in formalin (10%) and embedded in paraffin wax. Sections (2–3 μm) were stained with hematoxylin and eosin and Luxol fast blue-cresyl violet (LFB-CV). Immunohistochemistry was performed using different primary rabbit polyclonal and mouse monoclonal antibodies (Table 1) and the avidin-biotin-peroxidase complex method (Vector Laboratories, Burlingame, USA). The Dako Ark™ animal research kit (Dako, Hamburg, Germany) was used for the CNPase antibody only (69). Antibody binding was visualized using 3′3-diaminobenzidine-tetrahydrochloride followed by counterstaining with Mayer's hematoxylin. As negative controls, serial sections were incubated with a mouse IgG1 isotype control antibody (CBL600; Millipore, Schwalbach, Germany), rabbit serum (R4505; Sigma-Aldrich), or rat serum (R9759; Sigma-Aldrich) instead of the primary monoclonal or polyclonal antibody, respectively.
Primary Antibodies: Source, Antigen Retrieval, and Working Dilution

β-APP, β-amyloid precursor protein; eGFP, enhanced green fluorescent protein; CNPase, 2′,3′-cyclic nucleotide 3′-phosphodiesterase; GFAP, glial fibrillary acidic protein; MBP, myelin basic protein; NG2, nerve/glial antigen 2; np-NF, nonphosphorylated neurofilament; p-NF, phosphorylated neurofilament; PDGFRα, platelet-derived growth factor receptor-α; rGFP, recombinant green fluorescent protein; STAT-3, signal transducer and activator of transcription-3.
The percentage of the demyelinated area within the CCP was analyzed by using transverse sections of cerebellum and brain stem after MBP immunohistochemistry. Sections were digitalized with a color video camera (DP72, 12.8 megapixel CCD; Olympus) mounted on a BX51 microscope (Olympus) with 10× objective. The MBP-positive area was determined after manual outlining the CCP using analySIS 3.1 software package (SOFT Imaging System, Münster, Germany) and the same set of threshold values was used for all animals (n = 4). Similarly, the percentage of astrogliosis and axonal integrity/damage was determined using immunostaining for GFAP and phosphorylated (p-NF) or nonphosphorylated (np-NF) neurofilaments, respectively. The total number of CD107b-positive microglia/macrophages and β-amyloid precursor protein (P-APP)-positive axons of the injected and noninjected CCP of the same animal within the same section was manually counted with a 40-fold objective.
Obtained data were subjected to statistical analysis using SPSS for Windows (version 16, SPSS Inc., Chicago, IL, USA) employing the Mann-Whitney U-test. Statistical significance was designated as p < 0.05.
Electron Microscopy
Paraformaldehyde perfusion-fixed tissue was postfixed in glutaraldehyde (5%) and osmium tetroxide (1%) diluted in sodium cacodylate buffer (0.1M, pH 7.3). Osmium-fixed samples were dehydrated and embedded in epon resin as described previously (38,69). Ultrathin sections were stained with uranyl acetate and lead citrate and samples were examined with a transmission electron microscope (EM10 C; Carl Zeiss, Oberkochen, Germany).
Results
Experimental Demyelination of the Adult Mouse Caudal Cerebellar Peduncle
Pilot experiments titrating the concentration of ethidium bromide demonstrated that approximately 100%, 70%, 30%, 0%, and 0% of the animals displayed clinical signs after application of 0.1%, 0.05%, 0.025%, 0.01%, and 0% ethidium bromide, respectively. Mice exhibited a head tilt and temporary circling of varying severity implying an impairment of the ipsilateral vestibular system. Similarly, the size of the lesioned area correlated with the applied ethidium bromide concentration, reaching approximately 17.3-, 3.7-, 0.9-, 0.2-, and 0-fold of the total CCP area after injection of 0.1%, 0.05%, 0.025%, 0.01%, and 0% ethidium bromide, respectively. High concentrations of ethidium bromide (0.1%, 0.05%) induced severe axonal damage and necrosis of the neighboring vestibular nuclei. Based on these results, 0.025% ethidium bromide was used for subsequent experiments. Stereotaxic injection of 0.025% ethidium bromide into the CCP (Fig. 1A) resulted in a significant loss of MBP with a median of 44.35% (range: 21.95–54.83%) compared to the contralateral noninjected site (median 93.32, range: 87.02–95.03%) at 21 days postinjection (Fig. 1B–D).

Light microscopic demonstration of ethidium bromide-induced demyelination of the caudal cerebellar peduncle (CCP) of adult mice. Graphic scheme (A, left) indicating the localization of the CCP (dotted line) and the injection site (arrow) and Luxol fast blue-cresyl violet-stained coronal section (A, right). Immunostaining of the injected (B, E, H) and noninjected contralateral CCP (C, F, I) 21 days after application of 0.025% ethidium bromide with antibodies against myelin basic protein (MBP, B, C), CD107b (E, F), and glial fibrillary acidic protein (GFAP, H, I) and quantitative determination of the MBP-positive area (D), CD107b-positive cells (G), and the GFAP-positive area (J). Ethidium bromide injection resulted in a focally extensive demyelination (B–D, p = 0.029) and a significant increase in the number of infiltrating macrophages reactive with anti-CD107b antibodies (E–G, p = 0.029). The GFAP-positive area within the CCP was significantly increased (H–J). Note also the prominent GFAP expression within the adjacent tissue (values represent the median, minimum, and maximum; *p < 0.05, Mann-Whitney U-test). Scale bars: 1 mm (A), 100 μm (B, C, E, F, H, I).
Ethidium bromide-induced demyelination promoted the activation and infiltration of microglia/macrophages, as visualized by anti-CD107b immunostaining (Fig. 1E–G) and resulted in a marked astrogliosis defined as the increased size of the GFAP-positive area within the CCP (Fig. 1H–J).
To determine the degree of axonal pathology, CCP sections were immunostained for β-APP, p-NF, and np-NF (Fig. 2). Ethidium bromide injection induced a significant increase in the number of β-APP-positive axons (Fig. 2A–C) and a decrease in the size of p-NF-positive area (Fig. 2D–F). The increased size of np-NF-positive area in the injected versus the noninjected CCP showed a tendency (p = 0.059) but was statistically not significant (Fig. 2G–I).

Axonal pathology as determined by immunostaining of the injected (A, D, G) and the noninjected contralateral CCP (B, E, H) with antibodies against β-amyloid precursor protein (APP, A, B), phosphorylated neurofilament (p-NF, D, E), and nonphosphorylated neurofilament (np-NF, G, H) and quantitative determination of the immunoreactive axons/area (C, F, I). Injection of ethidium bromide significantly increased the number of β-APP-positive axons (A–C, p = 0.021) while decreasing the p-NF-positive area (D–F, p = 0.029) compared to the nonlesioned control. The increase in the area reactive with anti-np-NF antibodies between the lesioned and nonlesioned side was not statistically significant but revealed a strong tendency (G–I, p = 0.059) (values represent the median, *p < 0.05, Mann-Whitney U-test). Scale bars: 100 μm (A, B, D, E, G, H).
In Vivo-Behavior of BO-1 Cells Following Transplantation Into the Demyelinated and Nondemyelinated Caudal Cerebellar Peduncle
Ethidium bromide-injected mice with BO-1 cell transplants exhibited a reduced general appearance and substantial loss of body weight from day 15 post cell transplantation on. Due to progressive clinical worsening, transplanted animals were humanely killed at 17 days post-cell/mock transplantation. Gross examination of the brains demonstrated the presence of a moderate to severe internal hydrocephalus with mild herniation of the cerebellar vermis into the foramen magnum in all BO-1 cell transplanted animals, while no progressive clinical signs and macroscopic alterations were detected in mock transplanted animals.
Light microscopic analysis of the ethidium bromide-injected animals with BO-1 cell transplants at 10 days posttransplantation (Fig. 3A) revealed tumorigenic cells infiltrating the fourth ventricle. At 17 days after transplantation (Fig. 3B), a round and highly cellular, poorly demarcated, and nonencapsulated neoplasia with a diameter of approximately 0.8 cm, centered in the CCP, infiltrating into the ventral brain stem and laterobasal cerebellum, and compressing the fourth ventricle (Fig. 3B) was observed. Based on this unexpected finding the question arose whether the malignant behavior of the BO-1 cells might be triggered by the environment. To rule out such a tumorigenic process, an investigation using animals receiving BO-1 cell transplants without ethidium bromide application was performed (Fig. 3C). Ten days after cell transplantation, these animals also displayed an infiltrative growing, nonencapsulated neoplasia (Fig. 3C). BO-1 cells displayed identical histological features as detected in the demyelinated environment.
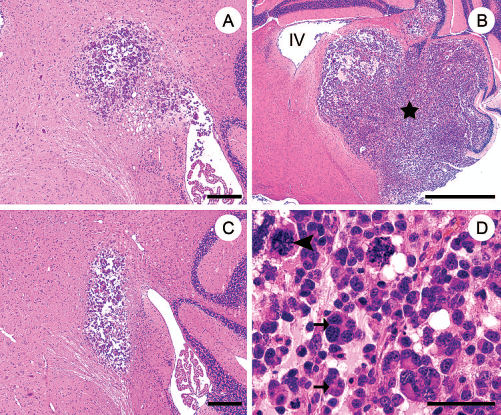
Coronal sections of adult mouse brain stem and cerebellum 10 (A, C) and 17 (B, D) days after transplantation of BO-1 cells into the demyelinated (A, B, D) and normal (C) caudal cerebellar peduncle (CCP). Hematoxylin and eosin staining revealed the presence of a nonencapsulated giant cell glioblastoma (A, B, C, asterisk) infiltrating into the ventral brain stem and laterobasal cerebellum (IV = fourth ventricle). Higher magnification demonstrated oval to caudate neoplastic cells that were organized in solid aggregates, nests, and packets surrounded by a scant fibrovascular stroma (D), bi- and multinucleated giant cells (arrows) as well as bizarre mitoses (arrowhead) were frequently observed (D). Scale bars: 200 μm (A,C), 1 mm (B), 50 μm (D).
At 17 days posttransplantation tumors were composed of solid aggregates, nests, and packets of oval to caudate cells, surrounded by a scant fibrovascular stroma (Fig. 3D). The neoplastic cells exhibited a mean diameter of about 10 μm with distinct cellular borders, few short blunt processes, moderate amounts of eosinophilic cytoplasm, irregularly round to oval, paracentrally arranged, euchromatic nuclei, with coarsely granulated chromatin and one to three variably sized, indistinct, basophilic nucleoli. Neoplastic cells displayed a marked anisocytosis and anisokaryosis. Frequently, binucleated cells and multinucleated giant cells with a diameter of up to 30 μm were present. There were up to five mitoses per high-power field, including frequent bizarre mitotic figures. Neoplastic cells were also found within the lateral ventricles, third ventricle, aqueductus mesencephali, and the meninges of brain and spinal cord. Furthermore there was a mild multifocal infiltration of microglia/ macrophages and lymphocytes within the neoplasm.
Transmission electron microscopy of BO-1 cells revealed oval to caudate neoplastic cells containing one, two, or multiple nuclei (Fig. 4). Cells had distinct cellular borders with either short, blunt processes forming a loose interconnecting meshwork, or they were tightly joined by multiple desmosomes. There was a moderate amount of electron-dense granular cytoplasm, containing a low number of variably sized mitochondria (Fig. 4, arrows), rough endoplasmic reticulum, a Golgi apparatus, and lipid droplets. The euchromatic nuclei were paracentrally located, and of irregular oval, frequently indented shape. The nuclear chromatin exhibited a thin incontinous layer of heterochromatin beneath the nuclear envelope and multiple irregular coarse electron-dense clumps, and one to three nucleoli. Occasionally, nuclei exhibiting a pronounced peripheral aggregation and margination of dark, electron-dense chromatin, interpreted as apoptosis were present. Furthermore, some cells showed a fibrillar spindle apparatus and multiple condensed filaments of chromatin lacking a nuclear envelope interpreted as mitoses.

Transmission electron microscopy of the transplanted BO-1 cells 17 days after implantation into the demyelinated CCP demonstrated variably sized, oval to caudate neoplastic cells with a moderate amount of granular electron-dense cytoplasm. BO-1 cells contained a limited amount of variably sized mitochondria (arrows), rough endoplasmic reticulum, and one to multiple oval, indented nuclei with a thin incontinous layer of heterochromatin beneath the nuclear envelope, multiple coarse clumps of chromatin (arrowheads), and one to three nucleoli. Scale bar: 5 μm.
Identification and Antigenic Expression of Transplanted BO-1 Cells
Immunostaining for eGFP and glial markers (Fig. 5) was performed to identify the transplanted BO-1 cells and to determine whether the microenvironment had any effects on their antigenic expression profile. Approximately 65% of the tumor cells were characterized by a prominent eGFP expression (Fig. 5A) confirming that the tumor arose in fact from the transplanted cells. Furthermore, the majority of cells, including the multinucleated cells expressed NG-2 (Fig. 5B) and platelet derived growth factor receptor α (PDGFRα; Fig. 5C), while only few expressed CNPase (Fig. 5D), GFAP (Fig. 5E), and MBP (Fig. 5F). Almost all of the neoplastic cells exhibited an intranuclear p53 expression (Fig. 6A) and 100% of them displayed a cytoplasmic signal transducer and activator of transcription-3 (STAT-3) expression (19% nuclear immunoreactivity; Fig. 6B). Comparative quantitative evaluations revealed that STAT-3 expression could be detected in the cytoplasm of 100% of cerebellar Purkinje cells (25% nuclear immunoreactivity), 91% of astrocytes (14% nuclear immunoreactivity), 74% of microglia (46% nuclear immunoreactivity), and 50% of oligodendrocytes (0% nuclear immunoreactivity).
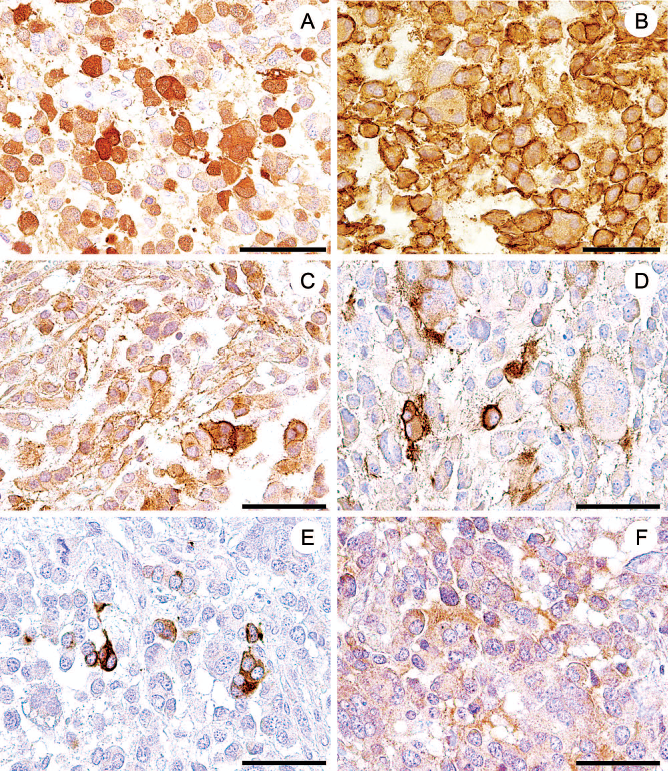
Immunostaining of the murine giant cell glioblastoma 17 days after BO-1 cell transplantation into the demyelinated CCP with antibodies against enhanced green fluorescent protein (eGFP, A), nerve/glial antigen 2 (NG2, B), platelet-derived growth factor receptor a (PDGFRα, C), 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase, D), glial fibrillary acidic protein (GFAP, E), and myelin basic protein (MBP, F). Immunostaining for eGFP demonstrated that the formed tumor arose from the transplanted BO-1 cells (A) that displayed high expression for NG2 (B) and PDGFRα but only low expression for oligodendrocytic (D, F) or astrocytic markers (E). Scale bars (A–F): 50 μm.

Immunostaining of BO-1 cells 17 days after implantation into the demyelinated CCP with antibodies against p53 (A) and signal transducer and activator of transcription (STAT-3, B). Most of the tumor cells exhibited prominent intranuclear p53 immunoreactivity suggestive of an altered conformation, function, and/or degradation of this tumor suppressor gene product (A). All neoplastic cells exhibited a marked cytoplasmic STAT-3 expression (B), whereas only few cells displayed an additional nuclear immunolabeling (B, arrowheads), indicative of an unaltered activation state similar to normal neurons and astrocytes. Nomarski differential interference contrast (B). Scale bars (A, B): 50 μm.
In vitro Characterization of BO-1 Cells
Approximately 65% of the transfected BO-1 cells expressed eGFP in vitro as detected by immunofluorescence (Fig. 7A–D). The cells possessed a bipolar to stellate morphology typical of preoligodendrocytes (Fig. 7B). Expression of NG2 was noted in the vast majority of the transfected BO-1 cells (Fig. 7B), while CNPase immunostaining was detected in approximately 35% of the transfected BO-1 cells, often exhibiting a highly-branched phenotype (Fig. 7D).

Immunostaining of cultured eGFP-transfected BO-1 cells (A–C, green) prior to transplantation with antibodies against nerve/glial antigen 2 (NG2, red, B) and 2′,3′-cyclic nucleotide 3′-phosphodieserase (CNPase, red, D) and nuclear counterstaining using bisbenzimide (A–C, blue). There was strong expression of eGFP in the majority of BO-1 cells (A–C). Whereas the majority of bipolar to stellate BO-1 cells resembled immature OPCs and expressed high levels of NG2 (B), 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) expression was restricted to a minority of a highly-branched cells typical of mature OPCs (D). Scale bars (A–D): 60 μm.
Discussion
In the present study, a chemically induced demyelination model of the murine CCP was established and used for the investigation of the in vivo behavior of the transplanted murine OPC line BO-1. Ethidium bromide-induced demyelination of the CCP and transplantation of OPCs has been described so far only in rats (5,23, 45,74). The present study revealed that the murine CCP also represents a suitable model to investigate the pathogenesis of chemically induced demyelination. The BO-1 cell line, which was previously obtained by spontaneous immortalization (56), showed only limited oligodendrocytic differentiation in response to RA or coculturing with astrocytes in vitro (57). Therefore one aim of the present study was to analyze the response of transplanted BO-1 cells within the demyelinated CCP clarifying the question whether BO-1 cells may be induced to enforce oligodendrocytic differentiation in vivo when exposed to a stimulating environment.
Transplantation of BO-1 cells into the demyelinated CCP did not elicit remyelination as expected but an invasive and metastasizing murine giant cell glioblastoma was noticed at the injection site. The expression of NG2 and PDGFRα, by most of the tumor cells confirmed their neuroektodermal origin (50,61) and visualization of eGFP verified that the tumor cells arose from the transplant. Control experiments of BO-1 cells into the normal CCP revealed the same malignant behavior of the transplant excluding the possibility that experimental demyelination was causally related to the rapid tumor growth.
Notably, history, histology, ultrastructure, immunophenotype, and malignant biological behavior of the BO-1 cells are in accordance with the evolving concept of partially lineage-restricted cancer stem cells derived from glial progenitors in glial tumors (9,11). Thus, transplantation of BO-1 cells may serve as a suitable animal model for investigating both the pathogenesis and the therapy of glial tumors.
Ethidium Bromide-Induced Demyelination in Adult Mice
Experimental demyelination of brain stem and spinal cord has so far mainly been performed in rats (5,25, 53,63). Due to the fact that isolation of primary OPCs has first become feasible in neonatal rats (59), this species was mainly used for in vivo remyelination studies (20,25). However, mice as experimental model species offer several advantages over rats due to the existence of several natural mouse mutants with myelination deficits (2,24,33,44,46). In addition, the transgenic technology has generated a large number of different mice strains overexpressing or lacking expression of a variety of molecules involved in the control of myelination that represent highly interesting candidates to investigate glial–axonal interactions in transplantation studies. Unfortunately, murine OPCs in contrast to their rat counterpart are difficult to obtain and maintain (12).
To test the remyelinating potential of transplanted BO-1 cells, the CCP of adult mice was demyelinated by injection of the DNA-intercalating substance ethidium bromide. Injection of this reagent resulted in a focal demyelinated lesion at the injection site. Though a lower dose was required compared to rats, the extent and time course of the demyelination process was comparable to that described in the rat (74). Injection of 0.025% ethidium bromide induced marginal clinical signs and distinct demyelination within the CCP. Higher doses were found to create lesions that extended into the adjacent lateral, spinal, or superior vestibular nucleus.
In Vivo Behavior of Cell Lines Following Transplantation
Transplantation of mitogen-expanded primary cells or immortalized cell lines generally bears the increased risk of tumor formation (18). Studies in the peripheral nervous system have provided evidence for species-specific differences in the malignant potential of transplanted cells (18,39). In vivo, BO-1 cells maintained a high growth rate and formed tumors instead of differentiating into mature oligodendrocytes. Similarily, rat in contrast to human mitogen-expanded primary Schwann cells formed tumors following injection into the sciatic nerve of immunodeficient mice (18). In contrast, immortalized rat OPCs, such as the CG4 cell line, have been frequently used for implantation into the demyelinated brain stem and spinal cord (20,21,52,66,67) causing extensive remyelination mediated by the transplanted cells. None of the studies described a tumorigenic growth. Previous studies have shown that BO-1 cells have a strong inherent proliferative capacity (56,57). Contrary to the rat CG4 cell line, BO-1 cells did neither require the addition of the growth-promoting B104 conditioned medium for maximum in vitro proliferation nor did they respond to thyroid hormones known to increase oligodendrocytic differentiation (56,57). However, proliferation of BO-1 cells was effectively modified in vitro by RA and fetal calf serum (FCS) (56). Moreover, CNPase expression in vitro was dramatically increased upon coculturing with astrocytes (56), which induced CNPase expression in 60–90% of BO-1 cells compared to about 30–40% in the absence of astrocytes while the percentage of MBP-positive BO-1 cells remained unaltered at about 5% (56). Interestingly, the majority of transplanted BO-1 cells retained a highly proliferative NG-2- and PDGFRα-positive OPC phenotype. In contrast, less than 10% of the cells expressed CNPase, MBP, or GFAP characteristic of oligodendroglial or astrocytic differentiation, respectively (48). This unexpected finding indicates that the demyelinated CNS environment did not only fail to promote oligodendrocytic development as expected but instead reduced the expression of CNPase as compared to the in vitro condition (56).
Ultrastructural examination revealed poorly differentiated neoplastic cells lacking morphological characteristics of differentiated astrocytes or oligodendrocytes. The observed ultrastructure of the neoplastic cells is in accordance with features described for a putative adult glial progenitor cell population in the central nervous system of monkeys and rats consisting of pale irregular nuclei with a thin layer of heterochromatin beneath the nuclear envelope, clumps of heterochromatin, irregular cellular shape, abundant mitochondria, rough endoplasmic reticulum, and lack of intermediate filaments (54,57). Interestingly, intracytoplasmic bundles of glial intermediate filaments, which are generally considered to be a diagnostically relevant ultrastructural feature for astroglial tumors in humans (26,42), were also lacking in a previous study detailing the morphology of cell transplantation-induced murine astrocytomas (36). Therefore, the lack of intermediate filaments including GFAP-immunoreactivity as observed in mouse astrocytomas (36,55) may be either due to poor differentiation or species-specific properties. Based on these results, it can be concluded that the local environment of the CCP was insufficient to stop proliferation and induce oligodendroglial or astrocytic differentiation of the transplanted BO-1 cells. Immortalization of cells being genetically engineered or naturally occurring does not necessarily indicate neoplastic transformation. Accordingly, a number of different cell lines created by genetic manipulations and exhibiting high growth rates in vitro did not induce tumor formation after transplantation into the brain, spinal cord, or the peripheral nerve (32,48,51, 64,65). This implies that additional factors are defining the malignant potential of the transplanted cells. Although proliferation of BO-1 cells could be reduced in vitro, the percentage of BO-1 cells undergoing oligodendrocytic and astrocytic differentiation was limited upon treatment with RA or FCS, respectively (56). This is in striking contrast to studies using the rat CG4 cell line, where a strong expression of MBP after treatment with thyroid hormones was noted.
Transplantation of BO-1 Cells as a Model to Study the Tumorigenesis of Malignant Gliomas
Based on the high frequency of multinucleated cells and the abundance of bizarre mitoses, the tumor was classified as a murine giant cell glioblastoma (70). A giant cell glioblastoma has been experimentally induced by intracranial application of 3-methylcholanthrene in the parietal lobe of an albino mouse (70). Glioblastomas in humans are considered the most frequent and malignant neoplasms in the CNS and are predominantly localized at the subcortical white matter of the cerebral hemispheres (34). However, in contrast to human glioblastomas the tumor lacked areas of necrosis and vascular proliferations and therefore might represent more likely a counterpart to a human malignant glioma with little astrocytic and oligodendrocytic differentiation. The giant cell glioblastoma represents one histological subtype of glioblastomas with a predominance of multinucleated giant cells, and a high frequency of tumor suppressor gene 53 (TP53) mutations (34). The main functions of p53 include induction of apoptotic cell death, irreversible cell cycle exit, and enhancement of DNA repair (40). Accumulation of p53 in tumors is due to mutations of the p53 gene and subsequent synthesis of proteins with altered conformation, function, and degradation as compared to the wild-type protein (27). In the current study, all giant cell glioblastoma cells exhibited a strong nuclear p53 expression, despite a high mitotic index indicating an altered p53 function as described for 70–90% of human giant cell glioblastomas (34). A recently published meta-analysis of protein–protein interactions revealed a central role of p53 in glioblastomas (13). Interestingly, normal astrocyte precursors have an autonomous feedback system, including p53 that limits proliferation and suggests that loss of p53 function may be an early event in astrocytoma genesis (9). Recently, STAT-3 (member of the Jak/STAT-pathway downstream of EGFR) was found to represent an important molecular hub controlling astrogliogenesis during developmental and reparative conditions (28,29). STAT-3 seems to operate as a tumor suppressor gene in many tumors outside the CNS but its function in glial tumors is still discussed controversially (7,14). In this context, the marked expression of STAT-3 within the tumor cells with a cytoplasmic to nuclear ratio comparable to that observed in the neighboring normal neurons and astrocytes suggests an unaltered expression and activation state of the STAT-3 signaling pathway in BO-1-derived giant cell glioblastoma. Similarly, very little activated STAT-3 is found in multiple glioblastoma cell lines in vitro under unstimulated conditions (62). However, others report the occurrence of activated, tyrosine-phosphorylated STAT-3 in human glioblastomas in association with the expression of a mutated, autophosphorylated, truncated epidermal growth factor receptor (EGFRvIII) (47).
In summary, demonstration of a robust PDGFRα expression, an increased nuclear p53 expression, and an unaltered STAT-3 activation state suggests that BO-1 cells harbor molecular changes characteristic for secondary glioblastomas in humans (8).
Conclusively, transplantation of the spontaneously immortalized murine glial cell line BO-1 did not only fail to remyelinate ethidium bromide-induced lesions of the murine CCP, they also lacked oligodendrocytic differentiation as identified by a dramatic reduction of CNPase expression compared to in vitro conditions. Moreover, BO-1 cells induced a giant cell glioblastoma in the demyelinated and nondemyelinated CCP. It is suggested that tumor formation follows a failure of molecular control mechanisms in BO-1 cells, responsible for a shift from proliferation to differentiation. Future studies should reveal whether an activation of the STAT-3 signaling pathway can induce astrocytic differentiation and thereby limit the malignant potential of transplanted BO-1 cells.
Footnotes
Acknowledgments
The authors thank D. Waschke, K. Rohn, B. Buck, and P. Grünig for excellent technical support. The outstanding help of Prof. Tanja Kuhlmann, Institute for Neuropathology, Universitätsklinikum Münster, Germany, for further characterization of the neoplasm is gratefully acknowledged. F. Hansmann, K. Pringproa, and Y. Sun were supported by scholarships from the National Academic Research Foundation (Germany), the Deutscher Akademischer Austausch Dienst (DAAD, Germany), and the China Scholarship Council (China), respectively. This study was in part supported by the German Research Foundation (BA 815/10-3; FOR 1103). The authors declare no conflicts of interest.
