Abstract
Multiple modalities, including injectable bulking agents and surgery, have been used to treat stress urinary incontinence. However, none of these methods is able to fully restore normal striated sphincter muscle function. In this study, we explored the possibility of achieving functional recovery of the urinary sphincter muscle using autologous muscle precursor cells (MPCs) as an injectable, cell-based therapy. A canine model of striated urinary sphincter insufficiency was created by microsurgically removing part of the sphincter muscle in 24 dogs. Autologous MPCs were obtained, expanded in culture, and injected into the damaged sphincter muscles of 12 animals. The animals were followed for up to 6 months after injection, and urodynamic studies, functional organ bath studies, ultrastructural and histological examinations were performed. Animals receiving MPC injections demonstrated sphincter pressures of approximately 80% of normal values, while the pressures in the control animals without cells dropped and remained at 20% of normal values. Histological analysis indicated that the implanted cells survived and formed tissue, including new innervated muscle fibers, within the injected region of the sphincter. These results indicate that autologous muscle precursor cells may be able to restore otherwise irreversibly damaged urinary sphincter function clinically.
Introduction
Urinary incontinence, defined as the involuntary loss of urine, affects more than 200 million patients worldwide (25) and is associated with significant impairment of quality of life, social isolation, and depressive symptoms (20). A conservative estimate is that urinary incontinence affects approximately 20–50% of women during their lifetime (5,6). The most common form of incontinence is stress urinary incontinence (SUI), in which patients lose urine during activities such as laughing, sneezing, or coughing. This insufficiency of the continence mechanism is mainly due to muscle and nerve damage resulting from vaginal delivery and aging (25).
Current treatment plans (27) may include pelvic floor muscle training, pharmacological treatments, or surgical intervention (20). Surgical treatment options for stress urinary incontinence include the endoscopic injection of bulking agents, colposuspension, sling operations, traditional transvaginal needle suspension, and tension-free vaginal tape (TVT). Most of these strategies apply local compression to the urethra in order to improve continence but do not correct the underlying muscle and nerve pathology. In addition, these treatments are performed using nonabsorbable foreign materials, and implantation of foreign materials is associated with a number of possible side effects, including infection and erosion (1,13).
The shortcomings of current therapies have led physicians to consider the application of cell therapy in the treatment of stress urinary incontinence.
The injection of stem cells into the damaged sphincter area may have the potential to regenerate the damaged muscle as well as support re-innervation, which would lead to functional recovery of the sphincter muscle. Cells currently proposed for use in regeneration of the sphincter muscle are committed muscle precursor cells (MPCs) (4,15,34,35) and adult stem cells including skeletal muscle-derived stem cells (MDSCs) (7), bone marrow mesenchymal (11), and adipose-derived stem cells (ADSCs) (17,18,24). Transplantation of muscle precursor cells (MPCs) has been investigated as a treatment for a variety of genetic and acquired muscle disorders (19,23,32,35). MPCs are quiescent adult stem cells and are located under the membrane surrounding the muscle fibers. After trauma or damage, MPCs participate in tissue regeneration by proliferating and differentiating into myoblasts, which later fuse to form new myofibers. The majority of MPCs are committed to the myogenic lineage (3) and are therefore most suitable for muscle tissue engineering. The use of injectable cultured MPCs for the treatment of stress urinary incontinence has been investigated in rodent models (8,35,37). These studies have shown that injected MPCs survive and differentiate into regenerative myofibers and nerves (35) and that they significantly improve leak point pressure (22). Although these results are promising, the small animal models used do not fully reflect the clinical situation in humans. Therefore, we have recently developed a preclinical canine model (14) that would allow researchers to study incontinence in a model that more closely resembles the human bladder.
The main objective of this study was to investigate the feasibility of using a precursor cell-based system for the restoration of sphincter tissue function in a large animal model. A technique for obtaining and transplanting large numbers of MPCs was developed, and we have demonstrated that the injection of autologous MPCs leads to the formation of sphincter muscle tissue and significantly improves sphincter function for up to 6 months after treatment.
Materials and Methods
Study Design
A total of 27 adult female beagle dogs (10–12 kg) were used. Three animals served as normal controls (Fig. 1). The sphincter was microsurgically removed in all study animals (n = 24) according to our established dog model (14). One month after surgery, the sphincter damage was confirmed in all animals. This time point served as the starting point for the study. At this time, the treatment group (n = 12) received an injection of autologous MPCs. The damage-only group (n = 12) was followed without cell injections. Routine urodynamic evaluation was performed before sphincter muscle removal and again at 1, 2, 3, and 6 months in all animals. Three animals of each group were sacrificed after urodynamic evaluation at each time point. At the time of tissue retrieval, the bladder neck was assessed grossly and radiographically. Harvested tissues were used for organ bath studies and histological analysis.
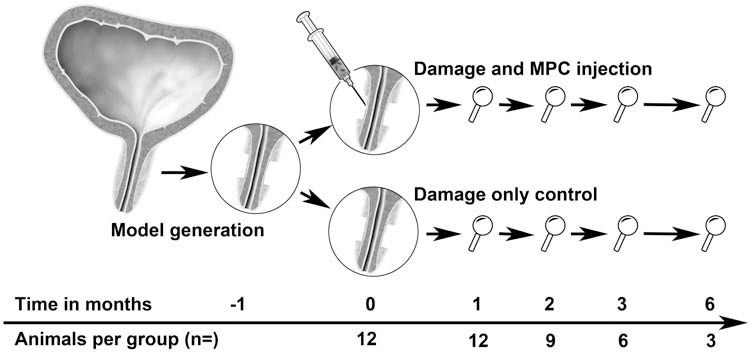
Treatment strategy. The sphincter muscle was microsurgically removed in 24 animals. The loss of sphincter function was confirmed 1 month later. Twelve animals received an injection of muscle precursor cells. Animals were evaluated functionally at 1, 2, 3, and 6 months and compared to 12 animals with damage only. Three additional animals served as normal controls. MPC, muscle precursor cell.
Cell Harvest and Culture
To obtain autologous MPCs, a 1-cm3 muscle biopsy was taken from the lower leg of each animal, and the tissue was placed in sterile Dulbecco's modified Eagle's medium (DMEM, Gibco, Grand Island, NY). The MPCs were isolated and characterized according to well-established protocols in our laboratory (12,28,33–35). Briefly, the muscle was minced into small pieces (≤ 1 mm3). The isolated muscle fibers were incubated for 2 h at 37°C in filter-sterilized 0.2% (wt/vol) type I collagenase (Worthington Biochemical, Lakewood, NJ). After repetitive washes with PBS, the muscle fibers were then transferred into 35-mm dishes containing 3 ml of myogenic medium consisting of 20% fetal bovine serum, 10% horse serum (Gibco), 0.5% chick embryo extract, and 1% penicillin/ streptomycin in DMEM. Single muscle fibers were liberated by repeated pipetting and confirmed by microscopy. The separated muscle fibers were plated on 35-mm dishes precoated with Matrigel (1 mg/ml, Becton Dickinson) and incubated at 37°C and 5% CO2. MPCs migrating from the plated myofibers were trypsinized and plated in 10-cm culture dishes with complete medium containing 10% fetal bovine serum and 1% penicillin/streptomycin in DMEM.
Collagen Preparation and Labeling
After 4 weeks in culture, 10 × 107 cells were trypsinized and transferred into a conical tube for labeling and subsequent mixing with collagen. Cells were labeled using a red fluorescent cell linker kit (PKH26, Sigma, St. Louis, MO) according to the manufacturer's instructions. Collagen type I was used as a carrier for cell injection (final concentration of 0.1 mg/ml). After confirming a neutral pH (phenol red), the cells were mixed with the carrier and placed on ice until injection.
Animal Anesthesia and Surgery
All experimental procedures were reviewed and approved by the Wake Forest University Institutional Animal Care and Use Committee and were performed in compliance with the Animal Welfare Act and the Guide for the Care and Use of laboratory Animals. Anesthesia was induced by injection of telazol (12 mg/kg IM) (Fort Dodge, Fort Dodge, IA). All animals were placed in supine position, intubated, and ventilated. The anesthesia was maintained using 1–2% isoflurane. The surgical area was shaved and scrubbed with povidone–iodine solution. According to our recently established model of irreversible sphincter damage, approximately 25% of the circumferential muscle fibers in the sphincter were removed (14). At the same time, a muscle biopsy was taken via a 2-cm long incision over the soleus muscle of the hind leg. One month after model generation, urodynamic analysis was used to confirm sphincter damage, and the treatment group received an injection of autologous MPCs. A low midline abdominal incision was made to expose the damaged sphincter region. The exact location of the damage was located using the marking sutures left during model generation. To increase tissue porosity at the recipient location, 2 ml of saline was injected into the remaining periurethral tissue (hydrodissection). Cells mixed with a low concentration collagen carrier (5 × 107 cells/ml, total of 2 ml) were carefully injected at multiple locations into the sphincter using a 22-gauge needle. Care was taken not to release cells into the urethral lumen or the peritoneal cavity. After all surgeries, observations were made every 15 min until the animals were fully awake and active. Animals were carefully monitored for signs of bladder infection, and urine samples were taken if there was any concern.
Urodynamic Evaluation
All examinations were done under general anesthesia in the supine position. The anesthesia depth was carefully monitored by evaluation of eye reflex, heart rate, and systolic blood pressure. We used the life-Tech Urolab Opus System V (life-Tech, Inc., Stafford, TX) in combination with a Millar microtip transducer catheter (Millar Instruments, Inc., Houston, TX) and a rectal balloon catheter. For the assessment of the detrusor leak point (DLP = lowest bladder pressure where leakage occurs), the bladder was emptied and filled at a speed of 12 ml/min and the pressure at the first leakage was noted. Static urethra pressure profilometry (UPP, pressure curve along the length of the urethra) and urethra stress profile (USP, pressure curve along the length of the urethra with repetitive abdominal compression, simulating coughing) were used to measure the sphincter pressure at the site of sphincter damage. These were performed at a speed of 0.5 mm/s using a pulling device at one-third of maximal bladder volume. The average of three closure pressures within the sphincter region (2 cm) was used for statistical analysis. Furthermore, sphincter contraction during electrical stimulation of the pudendal nerve was assessed before model generation and at the time of sacrifice. The pudendal nerve was located alongside the rectum, as described by Creed and Tulloch (9). The best stimulation location was defined as the place with minimal leg twitching and maximal sphincteric response. We used a Grass S48 Stimulator (Grass, West Warwick, RI) with 1 ms delay and 1 ms pulse duration at 8 Hz, 24 V for 2 s. Every urodynamic assessment was performed at least twice per animal.
Cystourethrogram
In order to visualize the sphincter, we performed cystourethrograms at the time of sacrifice in all animals. Contrast media were injected into the bladder with a syringe until a pressure of 40 cm H2O was reached. Anterior–posterior images of the lower urinary tract were taken (Siremobil Compact, Malvern, PA), and the configuration of the sphincter and bladder neck was assessed.
Tissue Harvest and Gross Anatomy
Animals were sacrificed by injection of Fatal-Plus (Vortech Pharmaceuticals, Dearborn, MI, 86 mg/kg IV). A long midline incision was made, and the symphysis pubis was removed using a bone saw. The urethra was dissected from the vagina, and the bladder and urethra were removed en bloc. The entire specimen was placed in cold DMEM to preserve the tissue for physiological organ bath studies.
Histology
Tissue samples were taken from the center of the damaged area in the sphincter, placed in Tissue-Tek® O.C.T. (Sakura®), and frozen in liquid nitrogen. The frozen blocks were sectioned into 6-μm slices using a cryostat (Model CM 1850, Leica, Bannockburn, IL). The sections were fixed and stained with hematoxylin and eosin (H&E) and Masson's trichrome stain. Sphincter muscle cells were identified via immunocytochemistry using an anti-desmin antibody (BD Biosciences, San Jose, CA) at a dilution of 1:50. Nerve fibers were identified through staining with polyclonal anti-S100 (DAKO, Carpinteria, CA) at a dilution of 1:400 and anti-protein gene product 9.5 (DAKO) at a dilution of 1:2,000. Immunolabeling was performed using an avidin–biotin detection kit (Vectastain Elite ABC, Vector, Bulingame, CA) according to the manufacturer's instructions. All sections were counterstained with Gill's hematoxylin. Native sphincter tissue sections served as positive controls. Tissue sections without primary antibody were used as negative controls. PKH26-labeled cells were identified by fluorescence microscopy using an excitation wavelength of 550 nm. Nuclei were stained using the VectaShield kit for DAPI staining (Vector). Digital images were taken (Zeiss Axio, Thornwood, NY) at various magnifications.
Organ Bath Studies
Physiological organ bath studies were performed at 3 and 6 months (n = 3). Ring-shaped sections of the sphincter were cut into three equal pieces and placed into Kreb's solution (NaCl 119 mM, KCl 4.4 mM, NaHCO3 20 mM, NaH2PO4 1.2 mM, MgCl2 1.2 mM, CaCl2 2.5 mM, and glucose 11 mM in distilled water, pH 7.2). The sphincter muscle strips (2 mm × 3 mm × 1 mm) were cut and suspended in a Radnoti Isolated Tissue Bath Chamber (Radnoti, Monrovia, CA) containing Kreb's solution aerated with 95% O2 and 5% CO2 at 37°C. The strips were allowed to equilibrate for 10 min under tension (1.5 g) before experimentation. Sphincter muscle strips were stimulated by electrical field stimulation (EFS) using recessed platinum electrodes connected to a Grass S48 Stimulator (Grass, West Warwick, RI), which provided stimulation with a 2-ms delay and 2-ms pulse duration at 100 Hz, 100 V for 1.5 s. The tetanic contractility was recorded and normalized to the sample weight.
Statistical Analysis
All experimental groups contained three animals at each time point. Data values are expressed as averages ± the corresponding standard error of the mean (SEM). For statistical analysis, SPSS v11 software (SPSS, Inc., Chicago, IL) was used. To increase the statistical quality of this large animal study, we performed multiple observations for every value measured. Averages of all observations of each animal were then calculated and used as one sample. Data comparing the MPC-treated to the damage-only group were analyzed by independent sample t tests. Multiple time points were assessed by one-way ANOVA and Bonferroni post hoc analysis. An a of p < 0.05 was considered significant.
Results
Animal Surgery and Urodynamic Evaluation
The surgeries and the postoperative phase were uneventful. No wound infections or bladder infections were recorded. Profilometry was used to precisely measure the pressure in the sphincter area and showed a resting sphincter pressure of 14.4 ± 0.7 cm H2O (n = 24) in normal animals. None of the study animals showed signs of detrusor overactivity. Urodynamic analysis indicated that the sphincter function in the animals treated with MPCs was improved compared to the damage-only controls (Table 1, Fig. 2A, B). This difference was statistically significant after 1, 2, 3, and 6 months (p < 0.001, p < 0.001, p = 0.025, and p = 0.011, respectively). The sphincter pressure values obtained by stress urethra profile analysis showed similar results, with values of p = 0.511 at 0 months, p < 0.001 at 1 month, p < 0.001 at 2 months, p = 0.014 at 3 months, and p = 0.032 at 6 months.

Functional assessment of sphincter function. (A) Representative urethra profiles showing the increase of sphincter pressure in the sphincter area after cell treatment. N, normal control; D6, damage-only control at 6 months; M6, MPC-treated animal at 6 months. (B) Graph showing sphincter pressures over time. Animals treated with MPC injection showed a significant functional recovery with pressures of approximately 80% of normal, while the damage-only group remained low at 20% (p < 0.025). (C) Pressure increase during electrical stimulation of the pudendal nerve showed a significant improvement for the cell-treated group, when compared to the damage-only control (p < 0.003).
Urodynamic Findings

Static urethral pressures demonstrate a statistically significant improvement of sphincter function in the animals with cell therapy during the entire study period. Detrusor leak point pressure, a measure of the function of the entire sphincter apparatus, showed a clear trend toward higher pressures in animals after cell injection with a significant improvement 1 month after cell injection.
Detrusor leak point (DLP) pressures were measured to assess the overall function of the sphincter apparatus (Table 1). The normal value for the leak point pressure was 45.9 ± 9.1 cm H2O (n = 24) in control animals. In the damage-only group, the DLP dropped to 23.0 ± 4.0 cm H2O at 1 month, 32.2 ± 5.0 cm H2O at 2 months, 34.5 ± 6.8 cm H2O at 3 months, and 32.5 ± 9.3 cm H2O at 6 months postoperatively. Animals that received autologous MPC injections showed a higher DLP with 42.6 ± 4.6 cm H2O at 1 month (p = 0.005), 42.3 ± 3.7 cm H2O at 2 months (p = 0.128), 41.0 ± 5.7 cm H2O at 3 months (p = 0.491), and 43.3 ± 4.9 cm H2O at 6 months (p = 0.397), indicating that the injected MPCs support the overall continence apparatus.
Sphincter power generation was assessed by measuring the pressure increase during pudendal nerve stimulation (Fig. 2C). Before model generation, the animals were able to generate an increase in sphincter pressure of 19.5 ± 1.7 cm H2O (n = 18) in response to pudendal nerve stimulation. However, the average pressure increase in the damage-only controls was only 5.8 ± 0.9 cm H2O at 1 month, 5.0 ± 1.3 cm H2O at 2 months, 6.3 ± 1.4 cm H2O at 3 months, and 5.5 ± 0.5 cm H2O at 6 months postoperatively. Interestingly, the sphincter pressure increase was nearly equivalent to normal values in the animals that received autologous MPC injections [15.8 ± 0.4 cm H2O at 1 month (p = 0.003), 19.4 ± 1.3 cm H2O (p = 0.001) at 2 months, 20.3 ± 2.7 cm H2O (p = 0.002) at 3 months, and 17.6 ± 0.8 cm H2O (p = 0.002) at 6 months], indicating that the injected MPCs participated in functional regeneration of the urinary sphincter.
Organ Bath Studies
Sphincter strips taken from animals injected with MPCs showed a higher myogenic contraction when compared to strips from damage-only controls.
At 3 months, the contractility of strips from damage-only animals was 2.7 ± 0.7 mg/mg; however, strips from MPC-treated sphincters demonstrated a contractility of 6.8 ± 3.7 mg/mg at the same time point. In addition, the same pattern was observed at the 6-month time point (3.0 ± 2.1 mg/mg for damage-only strips and 5.2 ± 0.7 mg/mg for MPC-injected strips). Although these results indicate a trend, they did not reach statistical significance in the present study.
Cystourethrogram
Radiograms of the sphincter area were performed to visualize the sphincter apparatus and evaluate the contour, gross morphology, and sphincter anatomy (Fig. 3). None of the investigated animals showed signs of stricture formation. All animals in the damage-only group showed a bulging of the sphincter area, demonstrating a loss of anatomic integrity. This widening was more prominent in the later time points. Animals in the damage + MPC group showed less widening of the urethra and maintained a contour comparable to normal sphincter anatomy.

Radiogram of the sphincter area (cystourethrogram) at 6 months. Animals treated with MPC injection were able to maintain a normal anatomical sphincter (arrows in B) and bladder neck region, while animals without treatment showed a widening of the sphincter area (arrows in C), indicating a loss of anatomic integrity. (A) Normal animal. (B) Damaged sphincter. (C) Damaged sphincter and MPC treatment.
Gross Anatomy and Histology
No macroscopic inflammation or fibrosis was seen at the site of sphincter damage. Histologically, at 1 month, the muscle bundles were clustered in groups throughout the mucosal layer. The sphincteric myofibers were centrally nucleated, a phenomenon known to be visible in early myofiber regeneration (Fig. 4). A mild inflammatory response with an increased number of lymphocytes was visible in early time points, but this decreased over time. The presence of PKH26-labeled MPCs confirmed cell survival and tissue formation at 1 month. The injected cells were distributed throughout the sphincter area and appeared to be forming new muscle fibers (Fig. 4). At 3 and 6 months, cell-treated animals showed increased muscle content with new muscle fiber bundles in the periphery (Fig. 5). When compared to normal sphincter tissue, the muscle layer was thicker and less organized. However, a tendency toward normal circular alignment of the muscle tissue over time was visible.

Early histological evaluation of cell survival and myofiber formation (1 month). (A) Maison's trichome stain showing muscle cells in a darker shade of color than the collagen-rich extracellular matrix. The injected MPC formed new muscle fiber bundles with central localization of the nuclei, a sign of early muscle fiber formation. Scale bar: 50 μm. (B) Detection of PHK26-labeled MPCs 1 month after injection shows survival and arranging in tissue bundles. Scale bar: 100 μm. (C) Macroscopic composite image of a cross-section through the sphincter area showing the distribution of injected cells around the urethra. Asterisk indicates the urethral lumen. Scale bar: 2 mm.

Histomorphological assessment at 3 months. (A–C) Maison's trichrome staining showing the morphological advances of MPC-injected animals with reconstruction of the muscle layer in the sphincter area. Stain shows muscle cells and mucus in a darker color than the extracellular matrix. The sphincter area of damage-only control consisted mainly of lamina propria and urothelium. (D–F) Imunohistochemical staining for anti-desmin revealed a significantly higher sphincter muscle mass in cell-injected animals. The newly formed muscle bundles were wider and more randomly aligned when compared with normal sphincter. (G–I) Imunohistochemical staining for anti-protein gene product 9.5, a ubiquitin hydrolase highly expressed in neural tissue, showed the presence of nerve fibers (arrows) between the regenerated sphincter muscle. Scale bar: 200 μm (A–F) and 100 μm (G–I).
In the damage-only group, histology revealed a sustained loss of muscle tissue in the sphincter region. The sphincter area was mainly composed of the urothelial layer and the lamina propria. In general, the wall thickness was significantly thinner and the lumen was wider, indicating a loss of wall strength. There were only minimal signs of muscle regeneration with minimal muscle content at the periphery without centrally nucleated muscle cells. There were no signs of inflammation or fibrosis at any time point.
Staining with nerve-specific antibodies showed that nerve fibers of different sizes were present in between the newly formed muscle tissue in the cell treated group (Fig. 5).
Discussion
Stress urinary incontinence is the most common form of incontinence, and it frequently results from repetitive vaginal deliveries with deterioration of the pelvic floor and sphincter muscles in women (5,6). The complex pathologic changes include muscle denervation, muscle fiber loss, fibrosis, and alteration of connective tissue (29,30). Surgical treatment options include the endoscopic injection of bulking agents, anatomical repositioning of the bladder neck, and local compression of the bladder outlet through nonabsorbable materials (1,13,20). However, none of these therapies addresses the underlying pathologies causing the incontinence, and they cannot be considered cures. On the other hand, tissue engineering using autologous cells might offer new possibilities in the functional treatment of stress urinary incontinence. Therefore, we have investigated the applicability of cell transplantation and tissue engineering techniques in the treatment of urinary incontinence in a preclinical setting.
We chose to use autologous muscle precursor cells for these studies because MPCs have excellent growth potential and are easy to expand in the undifferentiated state in culture (10,35). In addition, it has been demonstrated that MPCs are capable of long-term survival in vivo and that they retain the ability to form new myotubes (31) and new motor units (35). After myotube formation, the cells become postmitotic and further differentiate into mature fibers, thus preventing an unwanted volume effect that could lead to bladder outlet obstruction (36).
In this study, a low concentration collagen was used as a carrier material to offer a suitable microenvironment to the MPCs injected. The concentration used was 350 times lower than the concentration of collagen in commercially available bulking agents (Contigen®, Brad, Inc., Covington, GA) and therefore does not induce any bulking effect by itself. In an effort to minimize the number of dogs used for this research, this additional control group was therefore not included.
For this study, we employed a model of irreversible sphincter damage that was established at our institute (14). We have used microsurgical removal of the sphincter muscle because this technique allows the removal of myofibers and the progenitor cells together. Furthermore, this model resembles the clinical situation resulting from accidental surgical damage and allows for injection of MPCs into an environment with low levels of inflammation and fibrosis, resembling the chronic incontinence situation. There is a limitation to how much volume can be injected into the remaining sphincter tissue after damage. In our hands, the injection of more than 2 ml was not feasible. However, 2 ml of injectable volume might well be sufficient for the majority of patients suffering from incontinence. Finally, the sphincter damage can be examined by routine urethra pressure profilometry.
Since we had previously established the optimal cell preparation (14,16,29,36) and large animal model, the ultimate goal of this study was to assess the influence of injected MPCs morphologically, functionally, and anatomically. Our histological results demonstrate that the transplanted MPCs survive and form new muscle tissue after injection. At 1 month, PKH-labeled MPCs had grown in clusters and had participated in myofiber formation. In addition, immunohistochemistry confirmed the formation of muscle bundles and nerve fibers in cell-transplanted animals. The innervation is crucial not only for muscle contraction but also for activation of the MPCs (2) and for the maturation of the regenerated myotubes (26).
The mechanisms governing the beneficial effects of MPC transplantation may involve multiple pathways. First, as demonstrated in this study, the differentiation of the injected cells into the cellular constituents of the sphincter muscle can contribute to functional restoration, and second, the injected cells may release factors capable of paracrine signaling and recruiting host satellite cells, thus triggering an additional wound healing response. This process has been shown to occur in the heart, where injected stem cells not only participated in muscle formation but recruited surrounding cells to form new muscle as well (2,26).
The functionality of the engineered sphincter muscle was assessed by a variety of analyses, including urodynamic assessments, power generation upon electrical stimulation, and organ bath studies. To prevent incontinence, the sphincter muscle has to be able to maintain a sufficient resting tone and be able to rapidly increase outlet resistance during coughing. Urodynamic evaluation demonstrated a significant increase in pressures in the sphincter area in animals treated with the cell transplantation protocol. The early increase in sphincter pressure at 1 month is probably due to a combined effect of early muscle tissue formation and postoperative swelling. Over the following months, the pressure stabilized at approximately 80% of normal sphincter pressures. The results of the in vivo contraction of the sphincter muscle suggest that the engineered muscle is innervated with functional nerves at 2 months after cell injection. Detrusor leak point pressure is a measure of the function of the entire sphincter apparatus, and it is possible that we did not see significant changes in this parameter because a compensatory increase of sphincter muscle tone in the more proximal and distal areas occurred after injury. In addition, the evaluation of incontinence using other clinical tests (such as the pad test) is not practical. In our experience, the animals did not leak urine after sphincter damage.
The anatomical assessment and radiographic studies underlined these findings. The injection of MPCs appeared to prevent the remodeling of the sphincter and the bladder neck and allowed for the maintenance of an anatomy comparable to normal.
The proposed injectable cell therapy is especially appealing in the treatment of patients with surgical damage to the sphincter. This group of patients usually has localized damage to the sphincter muscle with intact innervation and blood supply. On the other hand, patients with dense fibrosis after radiation treatment might require combination treatments. Recently, it has been shown that some patients with stress urinary incontinence have a higher expression level of collagen degrading proteins (16,21), which leads to weakened pelvic floor support. It is not known whether autologous cell therapy is able to improve continence in this patient population. We have shown that injected MPCs are able to restore sphincter function for up to 6 months. Although this study, to the best of our knowledge, presents the longest published large animal trial using cellular therapy for the treatment of urinary incontinence, longer time points are desirable to see if cell transplantation can cure the disease.
In conclusion, autologous MPCs injected into irreversibly damaged sphincter muscle are able to form new muscle tissue and functionally improve the sphincter for up to 6 months. This study provides the basis for a clinical trial using MPCs for the treatment of sphincter incontinence.
Footnotes
Acknowledgments
We thank Luiz Freitas Filho, M.D. Sergio Rodriguez, M.D., Ph.D., Cindy Andrews, Paul Sikoski, D.V.M., and Mandy Lockard for their technical assistance. We also thank Karl-Erik Andersson, M.D., Ph.D., for his scientific advice and Jennifer Olson, Ph.D., for her editorial assistance. This research was supported, in part, by Crown and Joshua Frase Foundation. Support for the first author came from the GEBERT RÜF STIFTUNG and the Swiss National Research Foundation. Authors declare no conflicts of interest.
