Abstract
For pancreatic islet transplantation, the most common method of islet purification is density gradient centrifugation because of the differences in density between islets and acinar tissue. The density of islets/acinar tissue depends on several conditions, such as osmolality of purification solution. In this study, we evaluated the osmolality of iodixanol-controlled density gradients (400, 450, and 500 mOsm/kg) on the islet purification step. The density of the purification solutions was controlled by changing the volumetric ratio of iodixanol and the purification solutions (iodixanol-Kyoto solutions; IK solutions). The osmolality of density gradients was controlled by addition of 10x Hanks balanced salt solution (HBSS) solution. Density of both islets and acinar tissue increased relative to increase of the osmolality of purification solutions. There were no significant differences among the three groups on islet yield after density-adjusted purification and the rate of postpurification recovery. In vitro and in vivo assays suggest that the quality of islets was similar among the three groups. Our data suggest that efficacy of purification and quality of isolated islets is similar when the osmolality of purification solutions is between 400 and 500 mOsm/kg and density adjustment is applied. Since the density of islet and acinar tissue is changed according to osmolality, the density adjustment is important when using several osmolality solutions.
Introduction
Pancreatic islet transplantation is a successful and effective procedure resulting in tight glucose control for type 1 diabetic patients who experience hypoglycemic unawareness despite maximal care (3,4,7,10,22,23). One of the most important steps for islet isolation is islet purification. Purification minimizes the risks associated with islet infusion through the portal vein such as increased portal pressure and thrombosis. By purifying the islets, which represent only 2–5% of pancreas, acinar tissue is removed and the tissue volume transplanted is greatly reduced. The most common method of human islet purification is centrifugation by continuous density gradients in Ficoll solutions with a COBE 2991 cell processor (22,23). Approximately 1.077 g/cm3 of Ficoll is commonly used for a low-density solution and approximately 1.100 g/cm3 of Ficoll for a high-density solution. However, the density of islets/acinar tissue depends on several conditions such as warm ischemic time, cold ischemic time, preincubation time before purification, and osmolality of both preincubation solution and purification solution (2). We recently showed the effectiveness of controlled density gradients using iodixanol (9). According to the outcome of the density determination step, the density of the purification solutions was controlled by changing the volumetric ratio of iodixanol and the purification solutions. Islet yield after purification and postpurification recovery rate were significantly higher with the controlled density gradient purification than with standard continuous gradient purification by Ficoll solutions.
In this study, we evaluated the osmolalities of iodixanol-controlled density gradients (400, 450, and 500 mOsm/kg) on the islet purification step. Osmolality of purification solutions affects the density of islets and acinar tissue as well as purification itself. We previously reported that the purification efficacy of Ficoll (1.097 g/cm3 for a high-density solution) was similar to the 1.090 g/cm3 of iodixanol-based solution for a high-density solution rather than 1.095 g/cm3 (9). This is due to the difference in osmolality between iodixanol-based solution (approximately 400 mOsm) and Ficoll (approximately 500 mOsm). The data suggest that tissue density increases in Ficoll solution more than in iodixanol-based solution because the higher osmolality Ficoll solution leads to greater shrinkage of digested tissue than in iodixanol-based solution. We investigated the influence on tissue density by three different osmolalities of iodixanol-based purification solution.
Materials and Methods
Human Islet Isolation
The islet isolation protocol was approved by the Institutional Review Board of Baylor Health Care System. All pancreata were procured using a standardized technique to minimize warm ischemia. University of Wisconsin (UW) solution (ViaSpan, DuPont Pharmaceuticals, Wilmington, DE) was used for in situ perfusion of each donor. Each pancreas was excised immediately after the liver and before the kidneys. After the removal of the spleen and duodenum, we immediately inserted a cannula into the main pancreatic duct. The pancreas was weighed and 1 ml/g pancreas weight of Kyoto solution (Kyoto solution, Otsuka Pharmaceutical Factory, Inc., Naruto, Japan) was infused through the intraductal cannula (6,14). Pancreata were placed into a modified Kyoto solution/perfluorochemical two-layer preservation container at 4°C (13,16) for less than 8 h until the islet isolation procedure.
Human islet isolation was conducted as previously described (6,10,13) according to the standard Ricordi technique (20) with modifications later introduced in the Edmonton protocol (7,8,17,18,22,23). In brief, after decontamination of the pancreas, the ducts were perfused in a controlled fashion with a cold enzyme blend of Serva collagenase NB1 (20 PZ U/ml: 1 PZ unit according to Wünsch catalyzes the hydrolysis of 1 μmol 4-phenylazobenzyloxycarbonyl-l-prolyl-l-leucylglycyl-l-prolyl-d-arginine/min at 25°C, pH 7.1) with neutral protease (1 DMC U/ml: 1 DMC unit catalyzes the cleavage of 1 μmol peptide bond from dimethylcasein/min at 25°C, pH 7.0) (SERVA Electrophoresis GmbH, Heidelberg, Germany). The distended pancreas was then cut into 7–9 pieces, placed in a Ricordi chamber, and shaken gently. While the pancreas was being digested by recirculating the enzyme solution through the Ricordi chamber at 37°C, we monitored the extent of digestion with dithizone staining by taking small samples from the system. Once digestion was confirmed to be complete, dilution solution (Mediatech, Inc., Manassas, VA) was introduced into the system, and then the system was cooled to stop further digestive activity. The digested tissue was collected in flasks containing 25% HSA and washed with fresh medium to remove the enzyme. The phase I period was defined as the time between placement of the pancreas in the Ricordi chamber and the start of collecting the digested pancreas. The phase II period was defined as the time between the start and end of collection. The digested tissue was incubated in UW solution prior to purification.
Islet Purification Procedures
Islets were purified with a continuous density gradient of iodixanol (Optiprep®, Sigma-Aldrich, St. Louis, MO)-Kyoto solution as previously reported (6,9,13) in an apheresis system (COBE 2991 cell processor, Gambro Laboratories, Denver, CO). We combined iodixanol with Kyoto solution (IK solution) to generate a new purification solution. Low-density (1.075 g/cm3) and high-density (1.085 g/cm3) solutions are produced by changing the volumetric ratio of iodixanol and Kyoto solution as previously reported (9). The osmolality of density gradients (400, 450, and 500 mOsm/kg) was controlled by addition of 10x Hanks balanced salt solution (HBSS) as shown in Figure 1A, B, and Table 1. Before purification by IK solution, we calculated the density of digested tissue. Digested tissue (0.2 ml) (after incubation in UW solution and prior to purification) was added to six 5-ml test tubes of different densities (1.085, 1.090, 1.095, 1.100, 1.105, 1.110 g/cm3) with each osmolality (total 18 tubes), and these tubes were centrifuge at 1000 rpm for 5 min. The density at which most of the digested tissue floated was defined as the density of the digested tissue. According to the outcome of the density determination step, we determined the necessary density of the high-density IK solution and added an appropriate amount of iodixanol into high-density IK solution. Islet purification was performed by top-loading digested tissue (≤20 ml of tissue/run) in UW solution (9) on the continuous gradients by low-density and density-adjusted high-density solutions in doughnut-shaped COBE purification bags. After a 5-min centrifugation at 1000 rpm, approximately 10 fractions (50 ml each) were collected and examined for purity.
Characteristics of Each Purification Solution and Volumetric Ratio of 10× HBSS and IK Solution

IK, iodixanol-Kyoto solution; HBSS, Hanks balanced salt solution.
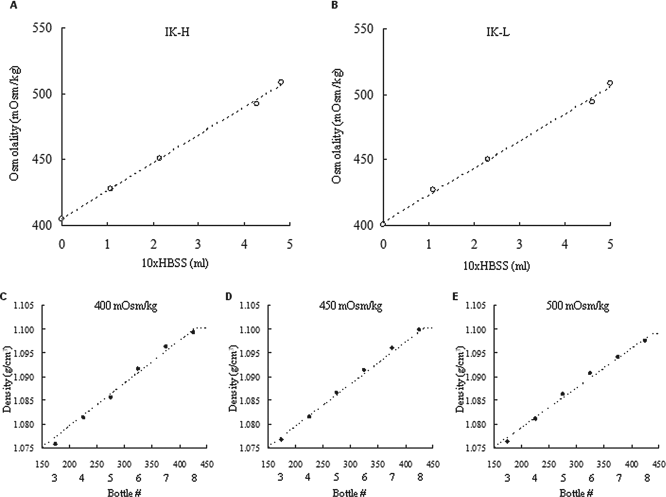
Osmolality changes by 10x HBSS and continuous gradients by each osmolality solution. (A) Osmolality changes of high-density solution (IK-H) by 10x HBSS. Ten times HBSS was added in 130 ml of high-density solution. (B) Osmolality changes of low-density solution (IK-L) by 10x HBSS. We added 10x HBSS to 140 ml of low-density solution. (C–E) Continuous gradient of each solution. Purified tissues were spun in 50-ml tubes and the density of each tube was evaluated by densitometer. Since the densities of the cap and fractions 1, 2, 9, and 10 were constant, data for these fractions are not shown. (C) 400 mOsm/kg solution, (D) 450 mOsm/kg solution, and (E) 500 mOsm/kg solution. HBSS, Hanks balanced salt solution; IK, iodixanol-Kyoto solution.
Densities of islets and acinar tissue were defined by tissue volume, islet purity, and average of density in each tube. For example, when 1.090 g/cm3 of high-density solution was used, the density averages of fractions two through nine were 1.0750, 1.0763, 1.0790, 1.0817, 1.0844, 1.0871, 1.0897, and 1.090, respectively. When the tissue volume of fraction #3 was 0.5 ml and islet purity was 80%, fraction #3 included 0.4 ml of islets and 0.1 ml of acinar tissue. Densities of islets were defined by summary of [the average density of each fraction (e.g., 1.0763 of fraction #3) × tissue volume of islets in each fraction (e.g., 0.4 ml in fraction #3)]/total tissue volume of islets. Densities of acinar tissue were defined by summary of [the average density of each fraction (e.g., 1.0763 of fraction #3) × volume of acinar tissue in each fraction (e.g., 0.1 ml in fraction #3)]/total volume of acinar tissue.
Islet Evaluation
The crude number of islets in each diameter class was determined by counting islets after dithizone staining (2 mg/ml final concentration; Sigma Chemical Co., St. Louis, MO) using an optical graticule. The crude number of islets was then converted to the standard number of IEQs (diameter standardizing to 150 μm) (19). The islet recovery was defined as the percentage of IEQ recovered after purification divided by the IEQ before purification. Islet viability after purification was assessed using a double fluorescein diacetate/propidium iodide (FDA/PI) staining to visualize living and dead islet cells simultaneously (8,19,22,23). Fifty islets were inspected and their individual viability was determined visually, followed by calculation of their average viability (8).
Islet function was assessed by monitoring the insulin secretory response of the purified islets during glucose stimulation using a procedure described by Shapiro and colleagues (22,23). Briefly, 1200 IEQ were incubated with either 2.8 or 25 mM glucose in RPMI-1640 for 2 h at 37°C and 5% CO2. The supernatants were collected and insulin levels were determined using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (ALPCO Insulin ELISA kit; ALPCO Diagnostics, Windham, NH). The stimulation index was calculated by determining the ratio of insulin released from islets in high glucose to the insulin released in low glucose. The data were normalized to total DNA. The data were expressed as mean ± SE.
Determination of ATP Production
To measure adenosine triphosphate (ATP) production, isolated islets in each group were cultured overnight with CMRL plus 5% FBS, washed twice with ice-cold PBS, and solubilized. The amount of ATP was then measured using an ATP assay system (ATP-lite, Perkin Elmer, Groningen, Netherlands), according to the manufacturer's instructions. The data were normalized to islet equivalents. The data were expressed as mean ± SE.
In Vivo Assessment
Nude mice were rendered diabetic by a single IP injection of streptozotocin (STZ) at a dose of 220 mg/kg. Hyperglycemia was defined as a glucose level of >350 mg/dl detected twice consecutively after STZ injection. The 2000 IEQ human islets obtained from each group were transplanted into the renal subcapsular space of the left kidney of a diabetic nude mouse, as previously described (11–15). During the 30-day posttransplantation period, the nonfasting blood glucose levels were monitored three times per week. Normoglycemia was defined when two consecutive blood glucose level measurements showed less than 200 mg/dl. No statistical differences in either pretransplantation blood glucose levels or pretransplantation body weight were observed among the four groups of mice. Mouse studies were approved by the Baylor Institutional Animal Care and Use Committee (IACUC).
Statistics
Three groups were compared by means of analysis of variance (ANOVA) followed by Bonferroni test or the Kaplan-Meier log-rank test. The differences between each group were considered significant if the value
Results
Density Determination for Purification
In this study, we used 400, 450, and 500 mOsm/kg of iodixanol-controlled density gradients on the islet purification step. The osmolality of density gradients was controlled by addition of 10x HBSS solution as shown in Figure 1A, B and Table 1. The continuous gradient of each osmolality solution was constructed as shown in Figure 1C-E.
Human islet isolations from three pancreata were performed in this study. In first islet isolation, we used six test tubes of different densities (1.085, 1.090, 1.095, 1.100, 1.105, 1.110 g/cm3) with 400 mOsm/kg for density determination. After the determination at 1.100 g/cm3, we purified the islets using 1.100 g/cm3 of high-density solution with all three osomolalities. The high pure fraction was shifted as two fractions with rising osmolality (tube number of the highest pure fraction: 400 mOsm/kg—#3, 450 mOsm/kg—#5, 500 mOsm/kg—#7). Therefore, we calculated the tissue density using the six test tubes of different densities with each osmolality (total 18 tubes) in second and third islet isolations. In the second isolation, we determined 1.095 g/cm3 for 400 mOsm/kg solution, 1.100 g/cm3 for 450 mOsm/kg solution, and 1.105 g/cm3 for 500 mOsm/kg solution. In the third isolation, we determined 1.090 g/cm3 for 400 mOsm/kg solution, 1.095 g/cm3 for 450 mOsm/kg solution, and 1.100 g/cm3 for 500 mOsm/kg solution. These data suggest that 50 mOsm/kg increases of purification solutions induces 0.005 g/cm3 increase of digested tissue (≥ 95% acinar tissue).
Tissue Density Differences in Each Solution
The characteristics of human pancreata and islet isolation are shown in Table 2. In each isolation, the digested tissue was split in thirds and each third was purified by either 400, 450, or 500 mOsm/kg of density gradients. As expected, the density of both islets and acinar tissue increased with rising osmolality (Fig. 2A). Acinar density was increased as about 0.005 g/cm3 relative to 50 mOsm/kg increase of purification solutions, similar to the data of density determination step. The differences in the density of both tissues were not significant among three groups (Fig. 2B).

Density of islets and acinar tissue in each solution. (A) Density of islets and acinar tissue at different osmolalities of the iodixanol-based continuous gradient. (B) Differences in density of islets and acinar tissue at different osmolalities of the iodixanol-based continuous gradient. Data are expressed as the mean ± SE.
Human Islet Isolation Characteristics (Before Purification)
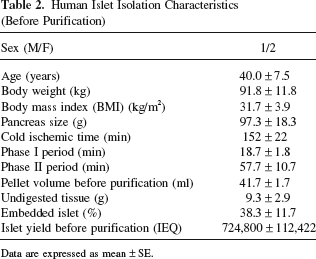
Data are expressed as mean ± SE.
Human Islet Isolation Characteristics
The characteristics of islet purifications are shown in Table 3. There were no significant differences in islet yield after purification, recovery rate, and purity of high pure fraction. These data suggest that efficiency of islet purification is similar among the three groups.
Human Islet Isolation Characteristics (After Purification)
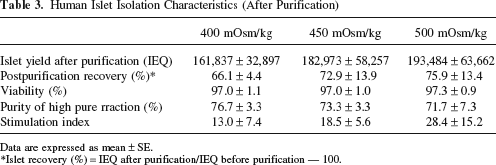
Data are expressed as mean ± SE.
Islet recovery (%) = IEQ after purification/IEQ before purification — 100.
Quality of the Isolated Islets
To assess the islet quality in each group in vitro, the ATP content and stimulation index of isolated islets was measured. There was no significant difference in ATP content (400 mOsm/kg, 0.59 ± 0.26 pmol/IEQ; 450 mOsm/kg, 0.69 ± 0.14 pmol/IEQ; 500 mOsm/kg, 0.77 ± 0.23 pmol/IEQ) (Fig. 3A) as well as stimulation index (Table 3) among the three groups. To assess the islet graft function of each group in vivo, 2000 IEQ from each group were transplanted below the kidney capsule of STZ-induced diabetic nude mice. The blood glucose levels of 8 of the 11 mice (72.7%) receiving islets from the 400 mOsm/kg group, 6 of the 10 mice (60.0%) receiving islets from the 450 mOsm/kg group, and 7 of the 11 mice (63.6%) from the 500 mOsm/kg group, decreased gradually and reached normoglycemia. The blood glucose levels remained stable thereafter and returned to pretransplantation levels after islet-bearing kidneys were removed (30 days after transplantation) (Fig. 3B). The attainability of posttransplantation normoglycemia was similar among the three groups. These data suggest that the quality of islets was similar among the three groups.

Quality of purified islets in each solution. (A) The adenosine triphosphate (ATP) concentration of the cell lysate after islet purification was measured using an ATP assay system. The ATP was normalized to islet equivalents (IEQ) [400 mOsm/kg group (
Discussion
Our study shows that the density of both islets and acinar tissue changes depending on the osmolality of purification solution and that efficacy of purification and quality of isolated islets are similar when the osmolality of purification solutions is between 400 and 500 mOsm/kg. Osmolality is known as one of the most critical variables in human islet purification (24). It has been reported that endocrine and exocrine tissues of pancreata have distinct osmotic sensitivity (1,5). However, in our study, the density differences between islets and acinar tissue are similar among the three groups. We speculated that poorly preserved pancreas resulted in edematous due to hypoxia-induced cell swelling during cold storage. Since exocrine tissues are more susceptible than islets to cold ischemic injury (15), exocrine tissues should be more edematous than islets. In such case, the effect of high osmolality solution is more apparent in exocrine tissue. On the contrary, all our pancreata were preserved with ductal injection followed by oxygen-charged static two-layer method. Therefore, our pancreata were not edematous at all and the ratio of shrinkage was similar between the islet and exocrine tissue.
Eckhard et al. showed that the mean density was similar between the endocrine and exocrine tissues in an iso-osmolar environment (300 mOsm/kg) and in 600 mOsm/kg solutions (2), resulting in insufficient purification. Osmolality of Ficoll solutions, as standard solutions for human islet purification, is about 500 mOsm/kg after addition of 10x HBSS (22) and that of IK solutions in our institute is about 400 mOsm/kg. We previously reported that our purification method by IK solutions led to a higher recovery rate (84.9%) than the standard Ficoll purification (55.6%) (9). Based on the data in this study, the differences of osmolalities themselves between IK solution and Ficoll solution may not affect the efficacy of islet purification.
Since the density of digested tissue is increased by about 0.005 g/cm3 relative to 50 mOsm/kg increase of purification solutions in this study, we should consider the density adjustment when we use different osmolality solutions for purification. We previously reported that the density of digested tissue varies with each isolation and that this has profound implications for the difficulty of islet purification (9). Many variables, such as osmolality of purification solutions as well as donor characteristics, preservation, and cold ischemic time, affect densities of endocrine and exocrine tissues and the islet recovery rate (21). Therefore, the density determination before islet purification by loading the digested tissue into six test tubes of different densities (1.085, 1.090, 1.095, 1.100, 1.105, 1.110 g/cm3) is extremely important for human islet purification.
In conclusion, the efficacy of human islet purification by different osmolality IK solution is similar between 400 and 500 mOsm/kg after density adjustment of high-density solution. On the other hand, the densities of both islets and acinar tissue are changed in each osmolality solution. Therefore, we should take into consideration that the osmolality of purification solutions affects the tissue density and that it may affect the efficacy of purification without density adjustment.
Footnotes
Acknowledgments
