Abstract
Cerebral dopamine neurotrophic factor (CDNF) is a recently discovered protein, which belongs to the evolutionarily conserved CDNF/MANF family of neurotrophic factors. The degeneration of dopamine neurons following 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) treatment is well characterized, and efficacy in this model is considered a standard criterion for development of parkinsonian therapies. MPTP is a neurotoxin, which produces parkinsonian symptoms in humans and in C57/Bl6 mice. To date, there are no reports about the effects of CDNF on dopamine neuron survival or function in the MPTP rodent model, a critical gap. Therefore, we studied whether CDNF has neuroprotective and neurorestorative properties for the nigrostriatal dopamine system after MPTP injections in C57/Bl6 mice. We found that bilateral striatal CDNF injections, given 20 h before MPTP, improved horizontal and vertical motor behavior. CDNF pretreatment increased tyrosine hydroxylase (TH) immunoreactivity in the striatum and in the substantia nigra pars reticulata (SNpr), as well as the number of TH-positive cells in substantia nigra pars compacta (SNpc). Posttreatment with CDNF, given 1 week after MPTP injections, increased horizontal and vertical motor behavior of mice, as well as dopamine fiber densities in the striatum and the number of TH-positive cells in SNpc. CDNF did not alter any of the analyzed dopaminergic biomarkers or locomotor behavior in MPTP-untreated animals. We conclude that striatal CDNF administration is both neuroprotective and neurorestorative for the TH-positive cells in the nigrostriatal dopamine system in the MPTP model, which supports the development of CDNF-based treatment for Parkinson's disease.
Keywords
Introduction
Parkinson's disease (PD) is a progressive neurodegenerative disease. Although many different neurons degenerate at various levels of the neuraxis, the major symptoms that lead patients to seek treatment are caused by a loss of nigrostriatal dopamine neurons. At the moment, there is no clinical treatment either to slow down and prevent the progression of the disease or to promote functional recovery by regenerating neurons and neuronal connections.
Rodent models of PD have been well characterized. Loss of dopaminergic circuitry can be induced in C57/Bl6 mice by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) through intraperitoneal (IP) injections (17,40). During the past decades, several neurotrophic factors have been studied in models of PD based on their postulated neuroprotective and neurorestorative properties (5,29). The most studied and promising neurotrophic factor in animal models of PD thus far is glial cell line-derived neurotrophic factor (GDNF), which has been shown to be both neuroprotective and neuroregenerative against 6-hydroxydopamine (6-OHDA) in rats (19) and MPTP in mice (42). However, intraputamenal GDNF treatment in human PD patients was not effective in a double-blind clinical trial (26), although two open label clinical studies, with small numbers of patients, showed clinical improvement (15,38). Thus, other proteins with trophic activity on dopamine neurons need to be studied. One of these has been neurturin, a homologue of GDNF, and it has been shown to have both neuroprotective and neurorestorative properties in several animal models of PD (9,16). Also, novel neurotrophic factors have been identified, like cerebral dopamine neurotrophic factor (CDNF) (31) and its homolog mesencephalic astrocyte-derived neurotrophic factor (MANF) (36); CDNF and MANF are secreted proteins that form a new evolutionary conserved protein family. MANF is protective on embryonic midbrain dopamine neurons in vitro (36). Both CDNF and MANF are neuroprotective against intrastriatal 6-OHDA toxicity in rats and also promote functional recovery (31,43,44). However, the effects of CDNF or MANF in the MPTP mouse model of PD, one of the preclinical standards, have not been examined.
The aim of this study was to investigate whether bilateral CDNF injections into striatum are protective against MPTP and whether CDNF can promote functional recovery in adult mice after MPTP treatment. In mice, MPTP treatment causes greater toxicity on dopaminergic nerve terminals in the striatum compared to the cell bodies in the substantia nigra pars compacta (SNpc) (40). Therefore, we administered CDNF bilaterally into the striata. We found that bilateral striatal CDNF injections of 10 μg, given before MPTP injections, improved motor behavior and that when CDNF (10 μg) was injected 1 week after MPTP there was also improved locomotor behavior. The effects of CDNF to produce behavioral improvement before or after MPTP were associated with increased striatal tyrosine hydroxylase (TH) immunoreactivity and substantia nigra dopamine (DA) cell numbers.
Materials and Methods
Subjects and MPTP Injections
Adult male C57/B16 mice (Charles River Laboratories, 10–12 weeks, 23–25 g) were used in the MPTP experiments. Experimental procedures followed the NIH guidelines and were approved by the NIDA Animal Care and Use Committee. MPTP (Sigma) was injected four times at 2-h intervals using 15 and 20 mg/kg IP for pretreatment and posttreatment, respectively. The mouse strain, age, and weight were chosen to produce significant dopamine depletion and a clear behavioral phenotype (7,22,40). The lower dose of MPTP in the pretreatment experiment was used because of a significant loss of mice with higher dose, probably due to weaker physical condition after the survival surgery on the previous day.
Surgical Procedures
Mice were anesthetized with chloral hydrate (400 mg/kg, IP) and placed in a stereotaxic apparatus. Mice were injected bilaterally into striatum (AP 0.5, ML 1.7, DV −3.5) with 10 μg CDNF or phosphate-buffered saline (PBS) in a volume of 2 μl/side. A Hamilton syringe was lowered into striatum and the injection speed was 0.5 μl/min. The needle was left in place for 2 min after each injection. The dose, as well as production and purification of recombinant human CDNF protein, was chosen as previously described (31). In both neuroprotection and neurorestoration experiments mice were divided into the two treatment groups based on balancing their 24-h locomotor behavior before CDNF and PBS injections.
Behavioral Measurements
The locomotor behavior of the mice was measured using an infrared activity monitor (Accuscan, Columbus, OH). Each mouse was individually placed in a 42 × 42 × 31-cm plexiglass chamber for a 24-h period, as previously described (11). The apparatus contains 16 horizontal and 8 vertical infrared sensors placed 2.5 cm apart. Food and water were freely available throughout the experiment. Motor activity was calculated using the number of beams broken by the mice.
TH Immunostaining
After the behavioral experiments, mice were anesthetized and perfused with saline followed by 4% paraformaldehyde (PFA) in phosphate buffer (PB). TH immunostaining was done on free-floating brain sections from striatum and substantia nigra using the ABC method as previously described (10,11). Briefly, sections were rinsed in 0.1 M PB, pH 7.4, blocked with 4% bovine serum albumin (BSA) and 0.3% Triton X-100 in 0.1 M PB. Sections were then incubated in rabbit TH antibody (Millipore, #AB152, 1:500) overnight at 4°C. Sections were rinsed in 0.1 M PB and incubated in biotinylated horse anti-rabbit IgG (1:200; Vector Laboratories, Burlingame, CA) for 1 h, followed by incubation for 1 h with avidin-biotin-horseradish peroxidase complex. Staining was developed with 2,3′-diaminobenzidine tetrahydrochloride (0.5 mg/ml). Sections were then mounted on gelatin/chrome alum-coated slides and coverslipped.
To determine the optical density the sections were scanned with Nikon Super Coolscan 9000. TH fiber density from the dorsal striatum and SNpr was measured from three brain sections and quantitated relative to the control group. For cell counts histological images were acquired using an Infinity 3 camera, NIKON 80i microscope, and QCapture Pro 5.0 software. TH-positive cells were averaged from three sections (AP: −3.16 mm, −3.40 mm, −3.64 mm to bregma) as described previously (11). Image analysis was carried out with NIS elements software (Nikon). The quantitation of TH-positive cells was carried out using stereology and all measurements were done by personnel blinded as to treatment. TH pixel density was obtained by subtracting the background density.
NeuN Immunofluorescent Staining
Sections were rinsed three times with PB and incubated for 1 h with blocking solution (4% BSA in 0.3% Triton X-100 in PB). Sections were then incubated overnight with mouse anti-NeuN antibody (neuronal nuclei, 1:200, Chemicon) and rabbit anti-TH antibody and rinsed three times in PB. The primary antibody was visualized using the AlexaFluor 488 goat anti-mouse or AlexaFluor 568 goat anti-rabbit secondary antibody (Invitrogen). Brain sections were examined with a Nikon eclipse 80i microscope with a Q-imaging camera. Fluorescence intensity of NeuN-positive nuclei was quantified in the SNpc from three sections of each brain area and averaged using NIS Elements software. The images were adjusted equivalently for brightness and contrast for visualization.
Western Blotting
Samples were sonicated for 10 s in 100 μl of RIPA lysis buffer with 1× protease inhibitor cocktail (Sigma-Aldrich, P8340) and centrifuged at 10,000 × g for 5 min. The supernatants were collected for TH Western blots and protein concentrations were measured using the DC assay (Bio-Rad). The pellet was eluted with 50 μl 1% sodium dodecyl sulfate (SDS) and used for dopamine transporter (DAT) Western blot and protein concentration measurements. Lysates were diluted with NuPAGE® lithium dodecyl sulfate (LDS) sample buffer and 2-mercaptoethanol and heated at 70°C for 10 min. For TH analysis 20 μg and for DAT analysis 10 μg of protein was loaded into 4–12% Bris-Tris gel (NuPAGE;, Invitrogen) and the electrophoresis was carried out in 1× MOPS [3-(N-morpholino)propanesulfonic acid] buffer (Invitrogen). The proteins in the gel were transferred onto nitrocellulose membranes using NuPAGE transfer buffer (Invitrogen). The Western blot procedures were carried out according to the Odyssey® protocol. Actin mouse antibody (1:1000 MAB1501, Millipore) was used as an endogenous control. TH was detected using an anti-TH rabbit antibody (1:1000, AB152, Millipore) and DAT was detected using rat anti-DAT antibody (1:1000, MAB369, Millipore). Infrared-labeled secondary antibodies of goat anti-mouse, anti-rabbit, and anti-rat were used for actin, TH, and DAT, respectively. After immunostaining, the membranes were scanned by a Li-Cor Odyssey Scanner and the bands were analyzed using ImageJ software.
High Performance Liquid Chromatography (HPLC) Analysis
The tissues obtained from each animal were homogenized in 0.1 M perchloric acid and centrifuged at 13,000 × g for 15 min. Dopamine (DA), 3,4-dihydroxyphenylacetic acid (DOPAC), and homovanillic acid (HVA) were measured by HPLC with electrochemical detection (25). The analytical column was a Symmetry C18 3.5 μm, 4.6 × 150.0 mm (Waters, Milford, MA), and the mobile phase consisted of 0.01 M sodium dihydrogen phosphate, 0.01 M citric acid, 2 mM sodium ethylenediaminetetraacetic acid (EDTA), 1 mM sodium octyl sulfate, 10% methanol, and the pH was 3.5. A flow rate of 0.9 ml/min was used and temperature was set to 25°C. The system consisted of an ESA automated injection system, an ESA 582 pump, and a Coulochem III detector (ESA, Chelmsford, MA). An EZChrom Elite™ chromatography data analysis system (ESA Biosciences, Inc.) was used for data collection and analysis. Levels of DA, DOPAC, and HVA were calculated as nmol/g of tissue weight.
Effects of CDNF in MPTP-Naive Animals
Two additional experiments were carried out to analyze whether CDNF affects the dopaminergic system in MPTP-untreated naive animals. In the first experiment mice were balanced into two groups based on their basal locomotor behavior and injected bilaterally into striatum either with PBS or CDNF 10 μg, and an additional eight normal mice were used as controls. After 2 weeks 24-h locomotor behavior was monitored and mice were decapitated, their brains removed, rapidly frozen in −50°C isopentane, and stored at −80°C. Bilateral tissue punches of the striatum and SN were taken from 1-mm coronal sections cut in a cryostat at −20°C. The other side was used for Western blot experiments and the other side for HPLC analysis.
In another set of experiments, 31 Wistar rats (250–280 g) were injected with vehicle, GDNF 10 μg, or CDNF 10 μg unilaterally into the striatum. These experiments were approved by the Committee for Animal Experiments of the University of Helsinki and the Chief veterinarian of the County Administrative Board. Two weeks later animals were given d-amphetamine (2.5 mg, SC) and, subsequently, motor behavior was measured for 2 h. Afterwards animals were perfused, and TH immunostaining and cell counts in pars compacta and TH optical density in pars reticulata were carried out as described above.
Statistical Analysis
Student's t-test and one- and two-way ANOVA were used for statistical analysis. In the two-way ANOVA the within-subjects factor used was time and the between-subjects factor was CDNF dose. Bonferroni test was used for post hoc analysis. Data are presented as mean ± SEM and a value of p < 0.05 was considered statistically significant.
Results
CDNF Protects TH-Positive Neurites and Cell Bodies Against MPTP-Induced Toxicity
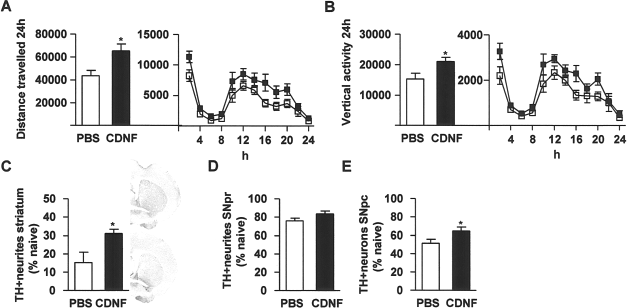
Mice were balanced into treatment groups according to their 24-h behavior (Fig. 1A, B, blue columns). CDNF or PBS injections were given into striatum 20 h prior to MPTP (15 mg/kg, IP ×4, every 2 h). Locomotor behavior over 24 h was measured 2 weeks later. CDNF-treated mice manifested a significantly greater distance traveled (Fig. 1A). Statistical analysis showed significant effects of Time [F(1, 13) = 10.5, p < 0.01] and CDNF dose [F(1, 13) = 5.1, p < 0.05], but not Time × CDNF dose interaction [F(1, 13) = 3.3, two-way ANOVA]. The Bonferroni post hoc test showed a significant difference between PBS and CDNF injected mice on week 2 (p < 0.05). To further analyze the temporal difference between these two groups, a two-way ANOVA was carried out at 2-h intervals. The major difference between the groups was in the dark cycle, hours 12–24, when the mice were more active. Pretreatment with CDNF significantly increased total distance traveled [F(1, 13) = 7.3, p < 0.05] (Fig. 1A, right panel). CDNF-treated mice also showed a significantly greater vertical activity (Fig. 1B). Statistical analysis showed a significant effect of CDNF dose [F(1, 13) = 5.6, p < 0.05], but not Time [F(1, 13) = 2.8, p > 0.05] or Time× CDNF dose interaction [F(1, 13) = 2.9, p > 0.05, two-way ANOVA]. Also, the Bonferroni post hoc test revealed a significant difference between PBS- and CDNF-treated mice (p < 0.05). A two-way ANOVA was carried out at 2-h intervals showing that the major difference between the groups was during the dark cycle [F(1, 13) = 11.7, p < 0.01] (Fig. 1B, right panel).

Striatal cerebral dopamine neurotrophic factor (CDNF) injections protect midbrain dopamine circuitry against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Mice were balanced into two groups according to their 24-h locomotor behavior (blue columns in A and B left panel). Bilateral CDNF 10 μg or PBS injections were given into the striatum 20 h prior to four MPTP injections. White columns and square symbols are PBS + MPTP; black columns and square symbols are CDNF + MPTP. Locomotor behavior and tyrosine hydroxylase (TH) immunoreactivity were measured 2 weeks later. (A) Distance traveled (cm). (B) Vertical activity after MPTP treatment. (C–E) MPTP and PBS white columns and MPTP and CDNF black columns. (C) TH optical density in the striatum. (D) TH optical density in the substantia nigra pars reticulata (SNpr). (E) TH-positive cells in the substantia nigra pars compacta (SNpc). Representative photomicrographs from striatum from normal control (naive), PBS-, and CDNF-treated mice. (A–C) n = 6–9; (D, E) n = 5–8. *Significant difference (p < 0.05) between PBS- and CDNF-treated mice 2 weeks after MPTP treatment, in (A) and (B) with Bonferroni post hoc test and in (C–E) with Student's t-test.
Immunohistochemical analysis showed that bilateral striatal CDNF injections protected TH-positive nerve fibers against MPTP treatment in the striatum (p < 0.01, Student's t-test) (Fig. 1C). In PBS-treated mice 27% of TH-positive neurites were spared and in CDNF-treated mice 62%, compared to MPTP-naive normal mice. CDNF injections also protected neurites in the SNpr (p < 0.05, t-test) (Fig. 1D). In SNpr TH-positive neurites were 61% in PBS-treated mice and 85% in CDNF-treated mice compared to MPTP-naive normal mice. Intrastriatal CDNF treatment also significantly rescued TH-positive cells in the SNpc in the MPTP-treated mice (p < 0.05, t-test) (Fig. 1E). The numbers of TH-positive cells were 44% in PBS-treated mice and 85% in CDNF-treated mice compared to nonlesioned naive mice.
Selectivity of CDNF-Mediated Neuroprotection in SNpc
A question that arises from the results is whether the CDNF-induced neuroprotective effect is selective to dopamine neurons or is it secondary to the prevention of cell loses in SNpc. To study this, a set of brain sections from experiment one was double stained with NeuN (a marker for neurons, green) and TH (red, Fig. 2). Since NeuN-positive cells do not correlate well with TH-positive cells in SNpc (6,14), NeuN can be used as a marker for other neuronal populations in SNpc. No difference in NeuN fluorescence intensity in SNpc was found between PBS + MPTP-treated mice and CDNF + MPTP-treated mice (p = 0.21, Student's t-test) while a significant increase in TH immunoreactivity was found in mice pretreated with CDNF. These data suggest that CDNF-mediated protection is selective for dopamine neurons in substantia nigra.

NeuN fluorescence intensity after MPTP treatment in CDNF and PBS injected mice. Bilateral CDNF 10 μg or PBS injections were given into the striatum 20 h prior to four MPTP injections. Mice were perfused and the brains were fixed with 4% paraformaldehyde (PFA) 2 weeks later and NeuN (neuronal nuclei) and tyrosine hydroxylase (TH) immunoreactivity were carried. Analysis of NeuN fluorescence intensity in SNpc showed no differences between PBS + MPTP-treated mice and CDNF + MPTP-treated mice. The data are shown in relation to nonlesioned naive mice. Scale bar: 0.5 mm. In the bar graph n = 4 per group.
CDNF Does Not Alter Behavior or TH Immunoreactivity in MPTP Naive Animals
Another question that arises from the previous results is does CDNF increase TH immunoreactivity in MPTP-naive mice that might cause a neuroprotective effect simply by raising TH protein levels? Two experiments were carried out to analyze whether CDNF affects the dopaminergic system in MPTP-untreated naive animals. Sixteen mice were balanced into two groups based on their basal locomotor behavior and injected bilaterally into striatum either with PBS or 10 μg of CDNF, and an additional eight normal mice were used as controls. Two weeks later locomotor behavior, striatal TH and DAT levels, and DA, DOPAC, and HVA concentrations were analyzed (Fig. 3). No difference in horizontal or vertical 24-h locomotor activity was found between PBS and CDNF groups (data not shown). CDNF did not alter TH levels in the striatum (p = 0.25, one-way ANOVA) (Fig. 3A, D), in SN (p = 0.40, one-way ANOVA) (Fig. 3B), or DAT levels in the striatum (p = 0.64, one-way ANOVA) (Fig. 3C, E). There were no differences in DA, DOPAC, and HVA concentrations in the striatum or SN between CDNF- and PBS-treated mice (p > 0.05, Student's t-test) (Table 1). However, DOPAC/DA ratio, an indicator of dopamine metabolism, and DOPAC + HVA/DA ratio, an indicator of dopamine turnover, were significantly higher in the striatum of CDNF-treated mice (p < 0.01 and p < 0.05, respectively, Student's t-test).

Effects of striatal CDNF injections in MPTP-naive animals. Mice were balanced according to their behavior and placed into two groups (A–E). MPTP-naive mice were injected bilaterally into striatum with either PBS or CDNF 10 μg. Two weeks later striata were collected and TH and dopamine transporter (DAT) levels were analyzed by Western blotting. (A, D) TH levels in the striatum in normal naive, PBS-, and CDNF-treated mice. (D) Lane 1: Cerebral cortex negative control, lane 2: naive, lane 3: PBS, lane 4: CDNF. TH (58.5 kDa) shown in red and actin (42 kDa) in green. (B) TH levels in substantia nigra (SN). (C, E) DAT levels in striatum. (E) Lane 1: Cerebral cortex negative control, lane 2: naive, lane 3: PBS, lane 4: CDNF. DAT (68.5 kDa) shown in red and actin in green. Rats were given unilateral intrastriatal injections of vehicle, CDNF (10 μg), or glial cell line-derived neurotrophic factor (GDNF; 10 μg). (F) Two weeks later animals were given d-amphetamine (2.5 mg/kg, SC) and locomotor activity was measured for 2 h. Activity is shown as related to the control group. (G) Number of TH-positive cells in substantia nigra pars compacta shown in relation to the contralateral untreated side. (H) TH optical density in substantia nigra pars reticulata shown as normalized to the control group.
The Effect of Cerebral Dopamine Neurotrophic Factor (CDNF) on Dopamine (DA) Concentrations.?

DA, 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA) tissue concentrations (nmol/g) in dorsal striatum and substantia nigra were measured in unlesioned mice 2 weeks after PBS or CDNF injections.
p < 0.01 versus PBS (t-test).
p < 0.05 versus PBS (t-test).
Thirty-one rats were injected with vehicle, GDNF 10 μg, or CDNF 10 μg unilaterally into the striatum. Two weeks later animals were given d-amphetamine (2.5 mg/kg, SC) and, subsequently, motor behavior was measured for 2 h. Afterwards animals were perfused, and TH immunostaining and cell counts in pars compacta and TH optical density in pars reticulata were carried out as described above. Locomotor activity, induced by d-amphetamine, did not differ between vehicle, CDNF, and GDNF treatment groups [vehicle: 100 ± 14, CDNF: 113 ± 24, GDNF: 123 ± 26; values are related to vehicle, F(2, 28) = 0.26, p = 0.74, one-way ANOVA] (Fig. 3F). The number of TH-positive cells was similar in all treatment groups [vehicle: 89 ± 5, CDNF 95 ± 8, GDNF: 98 ± 8; values are related to the number of TH-positive cells in the nontreated hemisphere, F(2, 26) = 0.43, p = 0.65, one-way ANOVA] (Fig. 3G). In addition, TH optical density in the SNpr was similar in all treatment groups [vehicle: 100 ± 10, CDNF: 91 ± 7, GDNF: 109 ± 8; values are normalized to the TH optical density in the control group, F(2, 25) = 0.93, p = 0.41, one-way ANOVA] (Fig. 3H).
CDNF Promotes Recovery After MPTP Injections
Twenty-eight mice were injected with four doses of MPTP at 2-h intervals. Animals were equally separated into two groups according to their locomotor activity 1 week after MPTP injections and were treated with either CDNF or PBS into the striatum. Posttreatment with CDNF, compared to PBS, significantly improved total distance traveled (p < 0.05, t-test) (Fig. 4A) and vertical activity (p < 0.05, t-test) (Fig. 4B) over 24 h at 1 week after injection. The analysis at 2-h intervals with two-way ANOVA further indicated that there was a significant difference in distance traveled [F(1, 26) = 5.7, p < 0.05] (Fig. 4A) and vertical activity [F(1, 26) = 7.1, p < 0.05] (Fig. 4B) after CDNF treatment during the dark period. Bilateral striatal CDNF 10 μg injections incremented TH-positive nerve fiber immunoreactivity after MPTP-induced toxicity in the striatum (p < 0.05, t-test) (Fig. 4C). In striatum TH-positive neurites were 15% in PBS-treated mice and 31% in CDNF-treated mice compared to MPTP-naive mice. CDNF did not alter TH optical density in the SNpr (Fig. 4D). However, CDNF treatment significantly increased the number of TH-positive cells in the SNpc compared to PBS treatment (p < 0.05, t-test) (Fig. 4E). In SNpc TH-positive cells were 51% in PBS-treated mice and 65% in CDNF-treated mice as relation to MPTP-naive normal mice.
Striatal CDNF injections restore midbrain dopamine circuitry in the MPTP toxicity model. In the neurorestoration experiment, mice were balanced into two groups (PBS and CDNF 10 μg) according to their 24-h behavior 1 week after MPTP injections. Bilateral PBS and CDNF injections were given into striatum and locomotor behavior and TH immunoreactivity were measured 1 week later. (A) Distance traveled. (B) Vertical activity. (C) TH optical density in the striatum. (D) TH optical density in the SNpr. (E) TH-positive cells in the SNpc. (A, B, E) n = 13–16, (C, D) n = 4. White bars and symbols represent PBS and black bars and symbols CDNF. The upper photomicrograph in (C) is from the striatum of a CDNF-treated mouse and lower photomicrograph from a PBS-treated mouse.
Discussion
The present results show that CDNF can protect against MPTP toxicity and restore motor function in mice when given before or after MPTP. Here we provide both histochemical and behavioral evidence indicating that CDNF exerts positive neurotrophic effects on adult dopamine neurons in MPTP-lesioned mice. We found that CDNF (10 μg) injections, given bilaterally into the striatum before MPTP injections, improved locomotor behavior and increased TH immunoreactivity in the striatum and SNpr, and also protected TH-positive cells in the SNpc significantly. Furthermore, when administered 1 week after MPTP, CDNF improved locomotor behavior and increased TH immunoreactivity in the striatum and the number of TH-positive cells in SNpc. These results suggest that CDNF treatment not only protects the cell bodies of midbrain dopamine neurons from degeneration but also restores dopaminergic circuitry.
We used MPTP in C57/Bl6 mice to elicit disruption of midbrain dopamine circuits. MPTP is a neurotoxin that affects nigrostriatal dopamine neurons in humans, in nonhuman primates, and in mice, and produces motor symptoms resembling PD (22,39). In humans and nonhuman primates MPTP irreversibly destroys the midbrain dopamine neurons. C57/Bl6 mice are susceptible to MPTP and dopamine terminals are destroyed transiently; there is some spontaneous regeneration from the surviving cells at many weeks to months after lesioning. Hence, it is important to examine toxicity within a 2–4-week window (12). There is still ongoing debate whether MPTP treatment in mice does cause death of dopamine neurons or loss of dopamine phenotype. In two studies it has been indicated that there is loss of dopamine cells in SNpc followed up to 2 or 4 weeks after MPTP administration, respectively (3,21). However, in other studies where mice have been followed up to 5 and 6 months it has been reported that there is partial spontaneous recovery (35,37). Our finding that CDNF can promote recovery is in line with the latter studies and suggests that some of the MPTP-induced toxic effects can be reversed. If dopamine cells would be totally degenerated, CDNF and any other neurotrophic factor may not show restorative properties. It should be kept in mind that in advanced human PD many neuronal elements are still present, albeit with a loss of the dopamine phenotype (28). Indeed, the concept of neurorestoration therapy in PD is to restore the function of neurons that are still viable. The present experiments do not distinguish between whether CDNF facilitates restoration of the dopamine phenotype or regeneration of dopamine neurons, which could occur through neural progenitor cells (20). To answer this question would require an extensive time course follow-up and prelabeling of dopamine neurons.
The finding that CDNF-induced neuroprotection was stronger than the effects found in the neurorestorative experiments is also in line with previous findings with CDNF (31), MANF (44), and GDNF (42). It is likely that early treatment with these trophic factors may more effectively reduce DA fiber degeneration.
Although GDNF has been shown to be a potent survival factor for embryonic dopaminergic neurons in vitro, there is some controversy about whether GDNF provides neuroprotection in addition to its well-established neurorestoration properties in mouse MPTP models. Tomac and coworkers, using a similar experimental design as in this study, found that exogenous GDNF is protective against MPTP and promotes recovery (42). In a study by Cheng and coworkers MPTP (35 mg/kg, SC) was injected once daily for 7 days and GDNF was injected unilaterally into striatum either before, during, or after MPTP treatment (8). Although this study failed to show a correlation between behavior and the extent of the lesion, GDNF pretreatment and posttreatment increased dopamine concentrations in the striatum. A study by Kojima and coworkers found that adenovirus mediated GDNF overexpression increased dopamine levels in striatum when the virus was given 1 week after MPTP (2 doses of 40 mg/kg, SC, on consecutive days) exposure (23). In contrast, a study by Eberhardt and coworkers, where adenovirus GDNF was injected into striatum and 1 week later MPTP (30 mg/kg, IP) was injected daily for 5 consecutive days, it was found that GDNF overexpression alone did not produce neuroprotection of dopamine neurons (13). Also, another study by the same group, where the same MPTP treatment protocol and a conditional rearranged during transfection (RET)-deficient mouse model was used, indicated that endogenous GDNF-RET signaling does not provide protection against MPTP-induced cell loss (24). Taken together these studies suggest that GDNF's most prominent effect in the MPTP-lesioned mouse midbrain is to facilitate regrowth of the dopaminergic axon terminals. Consistent with clinical findings (26) it is likely that GDNF's beneficial effect may be limited to selective aspects of PD. In this study, we report that CDNF protects the cell bodies of dopamine neurons when given before or after lesioning, revealing beneficial properties of this novel neurotrophic factor that may provide additional beneficial effects in PD patients.
Structural analysis of CDNF and MANF indicates that they are two-domain proteins (34). The CXXC disulphide bridge of the carboxy-terminal domain resembles the active site of oxidoreductases and isomerases, which may be involved in endoplasmic reticulum (ER) stress function; the amino-terminal domain is homologous to saposins, which are proteins known to bind lipids (34). Recent studies indicate that the C-terminal part of MANF is crucial for protective effects against apoptotic cell death and it resembles SAP domain of Ku70 that can bind to and block the proapoptotic molecule Bax (18). MANF and CDNF are expressed in neurons and in nonneuronal tissue (30,31), and the expression of MANF is increased by tunicamycin, ischemia, and status epilepticus (4,27,30,32,41). MANF protects cells from tunicamycin, an inhibitor of N-linked glycosylation and an agent known to produce ER stress (4). MANF also protects neurons against cerebral ischemia in vivo (1,2). The mechanisms underlying these functions may also be involved in the neuroprotective and restorative effects of CDNF against MPTP.
The role of CDNF in the normal development or maintenance of dopamine neurons is not well characterized. Vertebrates encode for both MANF and CDNF genes, whereas invertebrates contain only a single homolog (30). In Drosophila it has been shown that deletion of MANF results in degeneration of dopamine axons and a significant decrease in dopamine concentration (33). In the adult mouse brain, both CDNF and MANF proteins are detected mostly in the neurons in the striatum, substantia nigra, cortex, hippocampus, and cerebellum, and MANF has also been localized to some dopamine neurons (30,31). These data suggest that CDNF is present endogenously in adult brain. In this study, we found that exogenously applied CDNF did not upregulate TH and DAT or alter dopamine concentrations in MPTP-naive animals. We found that DOPAC/DA and DOPAC + HVA/DA ratios were increased after CDNF treatment, suggesting that dopamine metabolism and turnover is increased. However, the effect of increased dopamine turnover was not associated with increased horizontal or vertical locomotor activity, and therefore the physiological role of CDNF in dopamine neurons in nonlesioned animals is not clear at present. Furthermore, our results in nonlesioned animals suggest that the neuroprotective effect observed is not due to simply increased TH or DAT levels.
In rats it has been reported that NeuN-positive cells do not correlate well with TH-positive cells in SNpc (6) and a study with cynomolgus monkeys showed no correlation between histological markers of cellular death and NeuN in SNpc after MPTP administration (14). As we did not find any differences in the levels of NeuN-positive cells in SNpc, our results suggest that the neuroprotective effect of CDNF is selective for dopamine neurons. However, this conclusion should be considered with caution since there are no published data about the effects of MPTP on the number of NeuN-positive cells in SNpc in mice.
In conclusion, the present study is the first to report the positive neuroprotective and neurorestorative effects of intrastriatal CDNF administration in the nigrostriatal dopamine system against MPTP. CDNF had neuroprotective effects on dopamine cell bodies when MPTP was given either before or after striatal CDNF injections. Furthermore, CDNF neither altered TH and DAT expression levels nor dopamine and its metabolite concentrations in the nigrostriatal dopamine system in MPTP-naive animals. These results further support the development of CDNF-based treatment strategies to stop progression or to promote recovery in PD.
Footnotes
Acknowledgments
This research was supported by the IRP of NIDA, NIH, DHHS. We would like to thank Carl Lupica, Alex Hoffman, and Christopher Richie for their help with the study. The authors declare no conflicts of interest.
