Abstract
Exenatide is an analog of the incretin hormone glucagon-like peptide (GLP-1) that is used for the treatment of T2D for their metabolic effects. In addition to its insulinotropic effects, exenatide increases functional islet mass and improves their survival. Improved outcomes have been reported in recent clinical islet transplantation trials for the treatment of type 1 diabetes. The purpose of this study was to investigate whether exenatide has anti-inflammatory properties in human islets. Exenatide treatment improved islet function, significantly reduced content of inflammation-related molecules (tissue factor, IFN-γ, IL-17, IL-1β, and IL-2) and caspase 3 activation, whereas increased phosphorylation of ERK1/2, STAT3, and Akt in vitro. Immunostaining showed expression of GLP-1R in β-cells but not in α-cells. IL-1β colocalized with GLP-1R in β-cells. Induction of serine proteinase inhibitor 9 (PI-9) was detected after exposure of human islets to exenatide in vitro and after transplantation into immunodeficient mice. GLP-1 induced PI-9 expression in vitro but to a lower extent than exenatide. This effect was partially blocked by the antagonist exendin-9 in vitro. As assessed by immunostaining PI-9 is mostly expressed in β-cells but not in α-cells. In conclusion, we describe anti-inflammatory and cytoprotective properties of exenatide in human islets. Exenatide-mediated PI-9 expression, the only known granzyme B inhibitor, unveils potential immunoregulatory properties.
Keywords
Introduction
One of the major incretin hormones is the glucagon-like peptide-1 (GLP-1). GLP-1 is secreted in response to oral nutrient ingestion and acts enhancing glucose-dependent stimulation of insulin secretion. GLP-1 also controls blood sugar via inhibition of glucagon secretion and reduction in gastric emptying (23). The biological half-life of GLP-1 is very short (2 min) (20,61), as it is degraded by the ubiquitous enzyme dipeptidyl peptidase IV (DPP-IV) (20). GLP-1 mimetics and incretin enhancers (DPP-IV inhibitors) are a new class of drugs used for the treatment of type 2 diabetes (T2D). Exendin-4, a peptide isolated from the Gila Monster's (Heloderma suspectum) saliva, shares about 50% sequence homology to GLP-1. It is a potent agonist for the GLP-1 receptor and stable against DPP-IV (26). A synthetic exendin-4 (exenatide; Amylin Pharmaceuticals, CA) has been used for the treatment of T2D since 2005 (10,14). The cytoprotective properties of GLP-1 mimetics (29,39) suggest that they may also be beneficial for islet transplantation in patients with brittle T1D (22,28,36,52). GLP-1R stimulation produces direct effects on β-cells, resulting in proliferation, increased cell regeneration, and reduced cell apoptosis in animal models, isolated human islets, and β-cell lines (29,35,67). Whether the improvements observed in clinical studies are the direct effects of GLP-1 agonists on β-cell function or are a result of improved glycemic control remains to be elucidated.
Recent studies have correlated long-term treatment with exenatide with acute pancreatitis (4,19). However, additional clinical trial data and in-depth case report analysis are needed to further evaluate and verify this finding (3).
Previous studies described anti-inflammatory effects of exenatide in different contexts. In adipocytes, exenatide induces the expression of adiponectin and simultaneously inhibits proinflammatory adipokines (46). In an animal model of Parkinson's disease, exenatide protects dopaminergic neurons suppressing expression of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) (47). Likewise, exenatide confers cytoprotection to β-cell lines and pancreatic islets against proinflammatory cytokines such as IL-1β in vitro (30,43,63,64).
The purpose of our study was to characterize the anti-inflammatory properties of exenatide in human islets. Herein we report that exenatide treatment is associated with the reduction of cytokines and tissue factor (TF) production by islets in vitro. Furthermore, we report that exenatide treatment increases the expression of the serine proteinase inhibitor-9 (PI-9; Serpin B9) (8), a critical player in immune responses and the only known human intracellular inhibitor of granzyme B (GB).
Materials and Methods
Pancreatic Islets
Human pancreata were obtained from deceased multiorgan donors, and islets isolated using the automated method (60) at the Diabetes Research Institute Cell Processing Facility or other centers participating in the National Institute of Health Islet Cell Resources (ICR). Human Islets were used under protocols that received exemption by the University of Miami Institutional Review Board (IRB). Mouse islets were isolated at the Diabetes Research Institute's Preclinical Cell Processing and Translational Models Core from C57BL/6 mouse pancreata by collagenase digestion and purification on density gradients and then cultured in CMRL 1066-based medium (Gibco-Invitrogen). All animal procedures were performed under protocols reviewed and approved by the University of Miami IACUC (Laboratory Animal Welfare Assurance #A-3224-01).
Exenatide Treatment
Human islets were cultured at 37°C, 5% CO2 humidified ambient in CMRL-based Miami modified medium 1 (MM1; Mediatech), containing 5 mM glucose and supplemented with 2% human serum albumin. Mouse islets were cultured in supplemented CMRL containing 10% heat-inactivated fetal bovine serum (56). Human islets were treated with exenatide (0.0, 0.1, 1, 10, and 100 nM) within 2 h from isolation. Akt phosphorylation was measured by Bioplex (Bio-Rad) and/or Western blotting (WB). PI-9 expression was determined by WB or RT-PCR. After dose–response studies, we performed a time course with the most effective dose identified. These results were used to determine the duration of the treatments.
Perifusion
Islets were submitted to a dynamic stimulation assay by perifusing them in a customized apparatus (Biorep Technologies, Miami, FL) with a buffer containing 125 mM NaCl, 5.9 mM KCl, 1.28 mM CaCl2, 1.2 mM MgCl2, 25 mM HEPES, 0.1% bovine serum albumin, and 3 mM glucose at 37°C. Islets were perifused for 1 h (preincubation period) and then sequentially stimulated with 3 (5 min), 11 (10 min), and 3 mM glucose (15 min), respectively, and then with 25 mM KCl (5 min) followed by 3 mM glucose (5 min). Fractions of the perifusates were collected every second minute during the preincubation phase, and every minute during stimulation (13). Human insulin concentrations were determined in the collected perifusates by ELISA (Mercodia, Winston-Salem, NH).
Determination of Cytokine Content
Human islets (500 IEQ) were cultured for 48 h in the presence or absence of 10 nM exenatide. Supernatants and islet pellets were collected at 0, 24, and 48 h. As described previously (40), cytokine content was quantified in the islet supernatants utilizing the Human Cytokine Bioplex kit (Bio-Rad) by Luminex® technology, a dual-laser, flow-based microplate reader system (Bio-Plex® from Bio-Rad). Data were normalized to protein content of islet pellet.
Tissue Factor Content
Mouse islets were cultured for 48 h with or without 10 nM exenatide. Supernatants and islet pellets were collected and TF concentration was quantitatively determined in the supernatant using the IMUBIND® Tissue Factor ELISA Kit (American Diagnostica). Data were corrected for protein content.
Determination of Protein Phosphorylation
Cultured human and mouse islets treated or not with exenatide were harvested at selected time points in lysis buffer and stored until assayed. Phosphorylated and total content of AKT (Ser473), extracellular signal-regulated kinase 1 (ERK1/2) (Thr185/Tyr187), p38 mitogen-activated protein kinase (p38 MAPK) (Thr180/Tyr182), cJun (Ser63), cJun N-terminal kinases (JNK) (Thr183/Tyr185), and Iκβ (Ser32/Ser36) were quantitatively determined using the BioPlex® platform (Bio-Rad), as described (32). This technology is based on bead multiplexable assays that detect the phosphorylated proteins in lysates derived from cell culture or tissue samples.
Western Blotting
Control and exenatide-treated cells were collected, washed with PBS and lysed with Laemmli buffer. Equal protein aliquots (20–25 μg) were submitted to electrophoresis, transferred to nitrocellulose membranes, and incubated overnight with primary antibodies [1:1000 dilution for anti-MAPKs, anti-pSTAT3/STAT3 (Signal transducer and activator of transcription 3) and anticaspase 3 antibodies (from Cell Signaling Technology, Inc. MA) and 1:1000 for anti-PI-9 antibody from MBL International] and subsequently with peroxidase-conjugated secondary antibodies (1:5000 dilution; Amersham Biosciences/GE Healthcare) for 1 h at room temperature in blocking buffer. Chemiluminescence using ECL plus detection reagents (Pierce) was assessed and quantified using ImageJ software (rsbweb.nih.gov/ij/download.html).
Quantitative Real-Time PCR (qRT-PCR)
RNA was isolated using miRNA Isolation kit (Ambion-Applied Biosystems, Foster City, CA). The genes of interest were analyzed following the Applied Biosystem protocol for TaqMan gene expression assays, utilizing TaqMan Universal reagents and 7500 Fast Real-Time PCR system. RQs (relative quantification) were calculated via SDS software as previously described (49). RQ represents the ratio of expression of PI-9 transcripts over the expression of the housekeeping gene β-actin. RT and PCR were done in triplicate using RT universal or specific primers and PCR-specific probes for each gene of interest (Applied Biosystems).
Role of the Protein Kinase A (PKA) Pathway on PI-9 Upregulation Induced by Exenatide
Human islets were preincubated for 1 h with the selective inhibitor of PKA, H89 (100 nM, Tocris Bioscience, MO), or vehicle. After the preincubation period, islets were stimulated either with 10 nM exenatide or forskolin (FRSK 10 mM, Sigma) for 4 h. Samples were collected and stored at −80°C for further analysis by RT-PCR.
In Vivo Effects of Exenatide on Human Islet Grafts
Athymic nu/nu (nude) mice were purchased from Harlan (Indianapolis, IN) and housed in microisolated cages with free access to autoclaved food and water in virus antibody-free rooms in the Department of Veterinary Resources at the University of Miami. Under general anesthesia (isoflurane 2% mixed with oxygen), human islet aliquots of ~1,000 IEQ were implanted under the kidney subcapsular space. Treatment consisted of intraperitoneal injection via an osmotic pump (Alzet, model #2001, infusion rate 1 μl/h) of either phosphate-buffered saline (PBS) or exenatide (3 μg/day). Animals were humanely sacrificed 24 h after transplantation. Grafts were retrieved for molecular studies and stored frozen at −80°C until assayed.
Immunofluorescence
Pancreatic specimens were collected and fixed in 4% (w/v) paraformaldehyde in 0.1 M phosphate buffer, pH 7.4, for 30 min at 4°C, dehydrated in increasing concentrations of alcohol, and embedded in paraffin. Antibodies were used at the following dilutions: rabbit polyclonal anti-human GLP-1R (1:50, Santa Cruz Biotechnologies); rabbit anti-human glucagon (1:250, Dako); guinea pig anti-swine insulin (1:250, Dako); mouse anti-human IL-1β (1:50, Santa Cruz Biotechnologies); mouse anti-human glucagon (1:500, Sigma); and mouse anti-human PI-9 (1:50, Santa Cruz Biotechnologies). Sections were deparaffinized, permeabilized with PBS containing 0.1% (v/v) Triton X-100 (PBS-T) for 10 min and the endogenous binding sites were blocked with universal blocking reagent (Biogenex) for 30 min. After blocking, triple immunofluorescence staining for GLP-1R, insulin, and glucagon, or IL-1β, insulin, and glucagon, or PI-9, insulin, and glucagon was performed. Sections were incubated with primary antibodies overnight. After extensive washes, sections were incubated with secondary antibodies fluorescein isothiocyanate (FITC)-conjugated: goat anti-mouse 568; goat anti-rabbit 488; goat anti-guinea pig 647 (Invitrogen, dilution 1:400). Nucleus was detected by DAPI staining (Invitrogen). After final washes, slides were mounted using vectashield. Images were acquired by confocal microscopy and compiled using Photoshop 7.0. Only contrast and brightness were altered for visualization purposes.
Statistical Analysis
Unless otherwise stated, all experiments were performed at least three times. Differences between experimental conditions yielding p < 0.05 were considered statistically significant. According to the number of factors/variables, a paired t-test or ANOVA were used for the calculations, with the SigmaStat software selecting the appropriate post hoc test where necessary.
Results
Exenatide Has Anti-Inflammatory Properties
Human islets are generally kept in culture for about 48–72 h prior to transplantation. During this time, an estimate of about 30% islet loss is generally observed that is paralleled by a release of proinflammatory cytokines (41,48). Furthermore, these molecules may contribute to increased islet immunogenicity. We observed that treatment with exenatide reduced the levels of IL-1β, IL-2, IL-17, and interferon-γ (IFN-γ) in human islet supernatants after 48 h of culture (Table 1). Although there was a trend already at 12 and 24 h, we didn't observe a statistically significant effect of exenatide in islets before 48 h, perhaps reflecting the time that is necessary to secrete sufficient amount of cytokines into the media for accurate quantification. To further investigate which cell is involved in the production of cytokines we performed immunostaining for IL-1β, insulin and glucagon. The result showed that human β-, but not α-cells colocalize with IL-1β (Fig. 1A). We also observed that GLP-1R is expressed in human β-cells but not in α-cells (Fig. 1B). We did not observe differences in the GLP1-R and IL-1β expression by immunostaining when islets were treated with exenatide 10 nM for 24 h (data not shown). Thus the differences in IL-1β between control and exenatide-treated islets were only detectable by direct cytokine assessment.

Insulin-positive cells coexpress interleukin-1β (IL-1β) and glucagon-like peptide-1 receptor (GLP-1R). (A) Human islets were stained for insulin (INS), glucagon (GCG), and IL-1β or (B) insulin (INS), glucagon (GCG), and GLP-1R. Nuclei were stained with DAPI and are not shown. The images are representative of three independent experiments and show that insulin-positive cells coexpress IL-1β as well as GLP-1R.
Cytokine Profile for Human Islets Treated With Exenatide 10 nM at 12, 24, and 48 Hours

Control islets were treated with vehicle. The values represent the cytokine concentration in islet supernatants (pg ml−1) and are corrected by protein concentrations in islet lysates. Data are representative of four independent human islet preparations assessed. IL, interleukin; IFN, interferon; TNF, tumor necrosis factor.
p < 0.05 (paired t-test) is considered statistically significant.
A phenomenon defined as “instant blood-mediated inflammatory response” (IBMIR) has been implicated in the early loss of functional islets that occurs after intrahepatic islet transplantation (5). This phenomenon has been associated with TF production, which is expressed in isolated islets (6). Our results showed that treatment with exenatide reduced TF content in human islets following 48 h of culture (Fig. 2).

Exenatide reduces tissue factor (TF) content in human islets. Human islets were treated with the indicated increasing concentrations of exenatide for 48 h. Samples were collected and TF content was determined by ELISA. The results indicate that exenatide reduces the content of tissue factor about 30% in all studied concentrations. Results are corrected by protein content (n = 3). *p < 0.05. One-way ANOVA was performed to determine statistically significant differences.
Phosphorylation Levels of Inflammation/Death-Related Kinases Remain Constant Upon Stimulation with Exenatide
Previous studies show that exenatide downregulates inflammation/death-related kinases in islets under cytotoxic conditions (30,45). We studied its ability to modulate the phosphorylation of a panel of stress-activated protein kinases (namely, cJUN, JNK, p38, and Iκβ) in cultured human islets. The phosphorylation status of these kinases was not significantly reduced in our study after 24 h of treatment with 10 nM exenatide (Fig. 3A). It has been reported that exenatide increases Akt and ERK1/2 phosphorylation in islet cells, which parallel improved cell viability in vitro (9,55). To evaluate both time course and dose–response to exenatide, human islets were treated with increasing doses of exenatide and the primary endpoint assessed was Akt phosphorylation changes. The lowest dose studied (0.1 nM) was sufficient to induce a statistically significant increase in Akt phosphorylation, when compared to control islets (Fig. 3B). When a time course study was performed, treatment with exenatide 10 nM increased Akt phosphorylation within 1 h and was sustained for up to 24 h before decreasing (about 20%) by 48 h (data not shown). ERK1/2 phosphorylation was also increased upon treatment with exenatide. Although the lowest dose seemed less effective, there was no statistical difference between the doses studied (Fig. 3C). The time course for ERK1/2 activation exhibited a distinct profile with maximum activation occurring at 24 h (data not shown).

Effects of exenatide on phosphorylation levels of kinases related to inflammation/death and survival in human islets. Human islets were treated for 24 h with the indicated concentrations of exenatide. Phosphorylation levels of cJun N-terminal kinase (JNK), cJun, p38 [p38 mitogen activated protein kinase (MAPK)], and IKB (A), Akt (B), extracellular signal-regulated kinases 1/2 (ERK1/2) (C), and were determined by Bioplex (n = 3; *p < 0.05). One-way ANOVA was performed to determine statistically significant differences.
STAT3 Phosphorylation Is Increased in the Presence of Exenatide
Recent data suggest a cytoprotective role for the STAT3-SOCS3 (suppressor of cytokine signaling 3) pathway in rat pancreatic islets (59). Neonatal rat islets treated with ciliary neurotrophic factor (CNTF) signal through STAT3 pathway inhibiting caspase-3 activation induced in the presence of cytokines. We investigated the effects of different concentrations of exenatide in STAT3 phosphorylation in cultured human islets. Our results showed that exenatide increased STAT3 phosphorylation in human islets (Fig. 4A). Likewise, we observed that caspase-3, which is activated upon islet culture (51), was reduced upon treatment with exenatide (Fig. 4B). In addition, we tested whether exenatide affects human islets function by measuring the glucose-mediated insulin secretion in dynamic perifusion assays (Fig. 5). After 48 h of treatment with exenatide, islets were perifused and the insulin content assessed in the perifusate. Four out of six experiments showed that exenatide could enhance islet function in a dose-dependent manner.
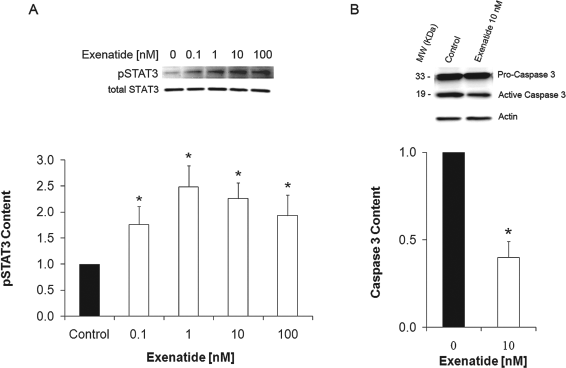
Exenatide activates signal transducer and activator of transcription 3 (STAT3) and inhibits caspase 3 activation. (A) Human islets were treated for 24 h with different concentrations of exenatide. Control islets were treated with vehicle and are shown as exenatide 0 nM. Representative Western blotting for phospho-STAT3 immunocontent (upper panel). Optical density quantification of three independent experiments (lower panel). Data are presented as folds versus control, *p < 0.05 (one-way ANOVA). (B) Human islets were treated with exenatide 10 nM or vehicle (Control) for 24 h. Representative western blot analysis of caspase-3 immunocontent (upper panel). Optical density quantification of three independent experiments (lower panel). Data are presented as folds versus control, *p < 0.05 (t-test).
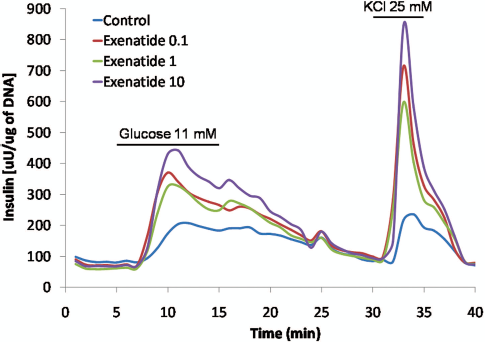
Insulin release after glucose and KCl challenge. Exenatide increases glucose induced insulin release in a concentration dependent manner. Whereas otherwise noted, glucose concentration is 3 mM. Islets are stimulated with 11 mM glucose and 25 mM KCl. Human islets were cultured for 48 h with the indicated increasing concentrations of exenatide. After washout incubation in the perifusion buffer not containing exenatide, islets were subject to a dynamic glucose-stimulated insulin release assay (perifusion). Note that the same islet number was utilized for the assay and insulin concentration was corrected for DNA content. The figure shows a representative experiment out of six independent assays performed.
Exenatide Induces Sustained and Dose-Dependent PI-9 Upregulation in Human Islets
The serine proteinase inhibitor PI-9 can protect target cells from the attack of cytotoxic T lymphocytes and natural killer cells (7,18,42). Our preliminary screening of inflammation-related molecules led to the novel finding that exenatide induced an increase in PI-9 expression in human islets in culture. Treatment of human islets with 10 nM exenatide for 24 h induced more than twofold increase in PI-9 expression. Dose–response for exenatide (0, 0.1, 1, 10, and 100 nM) and time course studies were performed to determine the optimal culture conditions to attain a maximal anti-inflammatory effect on human islets. Twenty-four-hour exenatide treatment resulted in increased PI-9 content in a dose-dependent manner (Fig. 6A). The minimal dose at which PI-9 was induced was 0.05 nM (data not shown). Exenatide 10 nM increased PI-9 expression within 1 h and the effect was sustained for 48 h (Fig. 6B). A similar sustained upregulation of PI-9 mRNA steady-state level was detected also in murine islets exposed to exenatide for 48 h in vitro (data not shown). PI-9 overexpression was also induced in human islets by GLP-1, although to a lesser degree than with exenatide. Exenatide-mediated PI-9 expression was partially blocked by exendin-9, an antagonist of the canonical GLP-1R (37) (Fig. 6C). Notably, exendin-9 by itself also slightly increases the level of PI-9 expression.

Exenatide induces serine proteinase inhibitor 9 (PI-9) expression via GLP-1R activation. (A) Human islets were treated for 24 h with different concentrations of exenatide. Control islets were treated with vehicle and are shown as exenatide 0 nM. A representative Western blotting for PI-9 immunocontent is shown (upper panel). Optical density quantification of three independent experiments (lower panel). Data are presented as folds versus control, *p < 0.05. (B) Human islets were treated with exenatide 10 nM for the indicated time points. A representative Western blot analysis of PI-9 immunocontent is shown (upper panel). Optical density quantification of three independent experiments (lower panel). Data are presented as folds versus control, *p < 0.05. (C) Human islets were treated with the selective antagonist of the GLP-1R, exendin-9 (Ex-9), exenatide, GLP-1, GLP-1 + Ex-9, and exenatide + Ex-9. The results show the GLP-1R involvement in PI-9 expression induced by exenatide. Statistical analyses were conducted by one-way ANOVA. *,**,***Difference between groups at p < 0.05.
PI-9 Is Expressed in Human β-Cells
To assess which cell type expresses PI-9, we conducted immunostaining in freshly isolated human islets. The results show that PI-9 is mainly expressed in β-cells (Fig. 7).

Insulin-positive cells coexpress PI-9. Human islets were stained for insulin (INS), glucagon (GCG), and PI-9. Nuclei were stained with DAPI and are not shown. The images are representative of three independent experiments and show that insulin-positive cells coexpress PI-9.
Exenatide-Induced PI-9 Upregulation Is Mediated by cAMP
We investigated the effect of cAMP, a critical second messenger mediating GLP-1 analogues effects, in the induction of PI-9 expression. We observed an increase of PI-9 expression in islets treated with the cAMP rising agent forskolin (Fig. 8), while an opposite effect was found when the islets were treated with exenatide in the presence of the PKA inhibitor H89.

Protein kinase A (PKA) mediates exenatide induced PI-9 upregulation. Human islets were preincubated with H89, a selective inhibitor for PKA, or vehicle for 1 h. Islets were subsequently stimulated either with exenatide or forskolin (FRSK), a PKA pathway activator. Samples were collected after 4 h and then assayed by RT-PCR (n = 3). Data are representative of three independent experiments. One-way ANOVA was performed to determine statistically significant differences. *,**Differences between groups at p < 0.05.
Exenatide Upregulates PI-9 Expression in Human Islet Grafts In Vivo
We sought to further investigate whether PI-9 upregulation could be induced by exenatide in vivo in human islets transplanted into immunodeficient mice. Untreated human islets were transplanted under the kidney capsule of nude mice receiving either saline (control) or exenatide via an intraperitoneal osmotic pump (3 μg/day). The grafts were harvested 24 h after transplantation (n = 4 islet grafts/experimental condition) for RT-PCR analysis of PI-9. Control, nontransplanted islet aliquots from the same human islet preparations were used to determine basal levels of expression. Our results indicate that in vivo treatment with exenatide is associated with an increase in PI-9 expression in human islet grafts when comparing to control grafts (Fig. 9).

Exenatide upregulates expression of PI-9 in human islet grafts explanted from nude mice. Human islets were transplanted under the kidney capsule of nude mice. Mice were treated intraperitoneally with either saline or exenatide (3 μg/day). Grafts were explanted after 24 h of treatment. Aliquots of the same human islet preparations (not transplanted) were used to determine baseline levels (Control). Samples were analyzed by RT-PCR (n = 4 grafts per group). *Statistical difference at p < 0.05 (one-way ANOVA).
Discussion
Proinflammatory cytokines, such as IL-1β, IFN-γ, and TNF-α, play a critical role in the pathophysiology of type 1 diabetes (21,24,38). Isolated islets produce proinflammatory cytokines during the early postisolation culture period as a consequence of the activation of stress signal transduction pathways resulting from pancreas ischemia, islet isolation process, and culture conditions (2,12,31,32,53,54,57,62). In order to elucidate the possible mechanisms involved in the increased survival and function of islets treated with exenatide, we investigated its effects on basal secretion of cytokines by freshly isolated human islets in culture and observed that exenatide significantly diminishes human islet production of IL-1β and IFN-γ by 30% and 45%, respectively.
Using immunohistochemistry we also confirmed that proinflammatory cytokines are produced by human β-cells in vitro. We performed triple immunostaining for glucagon, insulin and IL-1β that showed colocalization of IL-1β with insulin-positive but not with glucagon-positive cells. Notably, we did not observe IL-1β in other islet cell types (Fig. 1A). In another set of experiments, and in agreement with previous report (65), we found that insulin, but not glucagon, colocalized with GLP-1R (Fig. 1B). These observations suggest that exenatide may preclude IL-1β secretion by activating GLP-1R in the human β-cells. GLP-1R was also expressed in islet cells not stained for insulin. TF expression in islets is believed to be one of the major triggers of IBMIR (5). We observed that TF content in human islets is reduced by 30% upon 48 h treatment with exenatide in culture (Fig. 2). After 48 h the plateau of inhibition was reached even with the smallest dose studied (0.1 nM), which is close to the levels found in humans treated with exenatide.
Signaling molecules such as JNK, p38, NF-κB, and cJUN mediate death-related events including those triggered by cytokines in islet cells (1,11,16,24,30). In fact, selective inhibition of JNK phosphorylation in human islets is paralleled by improved functional β-cell mass (32). Exendin-4 protects β-cells from IL-1β-induced apoptosis by interfering with the JNK pathway (30). Studies designed to evaluate the effect of exenatide or other GLP-1 analogs in cytokine induced-apoptosis have shown protection from both apoptosis and activation of JNK (9,30). We studied the effect of exenatide on JNK, p38, NF-κB and cJun activation and did not observe any alteration of the basal phosphorylation levels of these kinases. Collectively, these results indicate that although exenatide protect islets against the toxic effect of cytokines in vitro (9,30), it does not affect the expression of the kinases in culture.
In addition, exenatide activated STAT3 in human islets. This protein has been implicated in insulin sensitization (50), α-cell mass expansion (25), and protection from cytokine-induced apoptosis (59). Another important mediator in apoptosis is caspase-3, considered to be a central protein in the execution of apoptosis. Within 24 h exenatide inhibited caspase-3 activation about 50%, when compared to controls. Furthermore, culturing human islets with exenatide improves islet glucose-induced insulin secretion as assessed by dynamic perifusion stimulation. These results are in agreement with previous studies showing that GLP-1 activation protects islet cells against apoptosis via inhibition of caspase-3 activation and improvement of glucose responsiveness (29,39). This effect was observed in four out of six human islet preparations, perhaps reflecting the quality and potency of the islet isolation.
Prior to transplantation, human islets are routinely cultured for up to 72 h (41). Based on our results, we speculate that pretreatment of human islets with exenatide prior to transplantation could reduce the state of inflammation and in turn improve transplantation outcome.
PI-9 is a protein that plays a critical role in the survival of cells exposed to the attack of the immune system. PI-9 is unique for its ability to inhibit both inflammation and apoptosis. It also inhibits caspases-1 and −3 and thereby reduces the production of inflammatory cytokines (44,68). Expression of increasing levels of PI-9 in target cells might progressively inhibit the immune surveillance by blocking natural killer cells (NKs) and cytotoxic T lymphocytes (CTLs) through the inhibition of perforin/granzyme B as well as Fas/FasL-mediated death pathways (18,42). Notably, elevation of PI-9 levels has been described into immune-privileged sites (8,18). Our results show that treatment with exenatide increased PI-9 expression in a dose-dependent and sustained manner. Both in human and mouse islets we found that exenatide induces a rapid and sustained increase in PI-9 content. PI-9 appeared as a very fast and quite potent agent because a 50% increase in protein content was observed within 1 h (Fig. 6). RT-PCR showed that after 48 h of treatment with exenatide the mRNA level of PI-9 was still 40% higher than in control mouse islets (data not shown). Exenatide is a better mediator of PI-9 expression than GLP-1; it induces up to 25% more of PI-9 (Fig. 6C). The antagonist exendin-9 partially inhibits the induction of PI-9 expression. Unexpectedly, exendin-9 by itself also mediates PI-9 expression compared to control islets although not up to the extent induced by GLP-1 or exenatide. Interestingly, in the same study reporting that exendin-9 is a potent antagonist at the GLP-1 receptor in insulin-producing cells, the authors observed in long-term experiments a small agonistic activity of exendin-9 (37). Thus, it might be possible that binding of the exendin-9 to GLP-1R might be in part agonistic for some effects mediated through this receptor under special conditions. Further studies will be necessary to shed light on this topic. Notably, immunostaining with human islets of PI-9, insulin, and glucagon suggest that PI-9 is constitutively expressed in β-cells but not in α-cells (Fig. 7).
Exenatide-mediated PI-9 induction was mediated by cAMP, similarly to what has been previously reported for exenatide-mediated mitogenic effects in β-cells (21,66) and for exenatide-mediated inhibition of chemokine (C-X-C motif) ligand (CXCL)10 (58), which has a role in insulitis in type 1 diabetes (15). Importantly, exenatide also induced PI-9 upregulation in human islets after transplantation into immunodeficient animals, ruling out the possibility that it was an in vitro artifact and confirming the relevance of this phenomenon in an experimental transplantation setting (Fig. 9). Notably, we used a continuous dose of exenatide in the in vivo experiments achieved by osmotic pump implantation. This was based on the consideration that the half-life of exenatide is estimated of 2.4 h and that metabolic rates in mice are higher than in humans and several drugs are required in higher doses to achieve their efficacy range in this model. In fact, it has been estimated that degradation of exenatide by kidney membranes in mice was 15fold slower in human than in mouse (17). For our in vivo experiments, we reasoned that having continuous exposure during the early posttransplant period via osmotic pump delivery would provide more efficient availability of the drug to assess its effect on transplanted human islets.
Collectively, our study contributes to the elucidation of novel anti-inflammatory properties of the GLP-1 agonist exenatide that may have immediate implications for the clinical arena of islet transplantation and autoimmune diabetes. The scheme shown in Figure 10 summarizes our findings on the anti-inflammatory, antiapoptotic, and potential immunoregulatory effects of exenatide in human islets, which may reduce human islet graft immunogenicity. These effects could be related to the improved outcome observed in islet transplantation (27,33,34). Expression of increasing levels of PI-9 in target cells may contribute to mitigate/block NK and CTL-induced cytotoxicity through the perforin/granzyme pathway and the Fas/FasL pathway. Although not formally addressed in our study, it is conceivable that the observed PI-9 upregulation by human islets after exposure to exenatide both in vitro and, importantly, after transplantation into immunodeficient animals, may represent a desirable phenomenon particularly in the context of islet transplantation and autoimmunity.

Working model showing the multiple anti-inflammatory and antiapoptotic pathways modulated by exenatide. Summary of the findings presented in this study supporting the anti-inflammatory and immunomodulatory properties of exenatide in human pancreatic islets. IBMIR, instant blood-mediated inflammatory response.
Footnotes
Acknowledgments
This study was possible thanks to a grant from Amylin Pharmaceutical (to E.F. and R.L.P.) and to the generous support from the Diabetes Research Institute Foundation (www.DiabetesResearch.org).
