Abstract
Although hepatocyte transplantation (HCTx) is expected to become a useful therapy for human liver diseases, allogenic hepatocytes still tend to be rejected within a short period due to host immunosurveillance. In the present study, we investigated the effect of prior bone marrow transplantation (BMTx) for the engraftment of allogenic hepatocytes using the analbuminemic rat transplantation model. The hepatocytes of Lewis (LEW) rats were not accepted in the liver of retrorsine (RS)/partial hepatectomy (PH)-treated analbuminemic F344 (F344-alb) rats, which express the disparate major histocompatibility complex (MHC) against that of LEW rats. Prior BMTx with the LEW bone marrow cells (BMCs) after sublethal irradiation achieved acceptance and repopulation of LEW hepatocytes in the liver of the RS/PH-treated F344-alb rats, associated with elevation of serum albumin. The replacement of hepatic parenchyma with albumin positive (Alb+) donor hepatocytes and elevation of serum albumin levels were dependent on the bone marrow reconstitution by donor BMCs, which was more efficiently achieved by intrabone marrow (IBM)-BMTx than by intravenous (IV)-BMTx. Our results demonstrate that efficient bone marrow reconstitution by IBM-BMTx enables the replacement of the hepatic parenchyma with allogenic hepatocytes in RS/PH-treated analbuminemic rats without immunosuppressants.
Keywords
Introduction
Hepatocyte transplantation (HCTx) is an alternative to orthotropic liver transplantation for the treatment of human liver diseases (25,32,36). The liver is thought to be the most desirable site for HCTx, because transplanted cells could physiologically function by obtaining nutrients, growth factors, and hormones present in the portal blood and interacting with other liver cells and the extracellular matrix (17,27,33). HCTx could be especially effective if the transplanted cells repopulate and replace the host hepatic tissues. This has been observed following HCTx when host hepatocytes possess hereditary defects, such as in urokinase plasminogen activator transgenic mice (31), fumaryl acetoacetate hydrolase-deficient mice (tyrosinemia type I model) (30), multiple drug resistance gene 2 (MDR2) gene knockout deficient mice (9), and Long-Evans Cinnamon rats (Wilson's disease model) (42). Furthermore, transplanted cells can preferentially proliferate over host hepatocytes by regenerating stimuli generated by partial hepatectomy (PH), when host hepatocytes cannot proliferate due to chemical- or irradiation-induced DNA damage (22,40).
One of the main factors that limit the clinical application of HCTx, however, is the large cellular loss following HCTx (18). In the early phase following HCTx, the majority of transplanted hepatocytes infused into the portal circulation are trapped in the hepatic portal venule and the sinusoids around the portal tract, and thereafter are cleared by the cells of the innate immune system including granulocytes, macrophages, and Kupffer cells (18,29). Only a small number of cells thus become integrated into the host hepatic plates by disrupting the sinusoidal endothelium and then survive without being attacked by innate immune cells. In the case of allogenic HCTx, however, transplanted cells stimulate the adaptive immune system and then are rejected mainly by the immune responses mediated by CD4+ and CD8+ T lymphocytes (8,18,37).
HCTx would therefore be much more attractive if the cellular loss following HCTx could be avoided. Up to now, a number of experiments have been performed to attempt to prevent immunological rejection. These have included the administration of immunosuppressants such as cyclosporine and rapamycin (5), depletion of CD4+ or CD8+ T lymphocytes using anti-T-lymphocyte monoclonal antibodies (7), interference with costimulation pathways between antigen-presenting cells and cytotoxic T lymphocytes by the administration of a CTLA4 Ig and/or anti-CD40 antibody (38), and transduction of immunomodulatory genes such as the adenovirus E3 into the donor cells in vitro prior to HCTx (24). However, considering the future application of allogenic HCTx for human liver diseases, prolonged immunosuppressive treatment may therefore be a large long-term obstacle to the successful performance of HCTx.
Hematopoietic reconstitution by bone marrow transplantation (BMTx) has been shown to provide host tolerance in various models of transplantation (14). It has been demonstrated that when host bone marrow is reconstituted by donor BMTx after lethal irradiation, allogenic HCTx is feasible between the mouse strains expressing the disparate major histocmpatibility complex (MHC) (26,41). In this setting, hepatocytes of the third-party mouse strains were rejected, thus indicating that donor-specific immunotolerance was important for the successful engraftment of allogenic hepatocytes (26). Furthermore, human HCTx to immunodeficient rodent recipients has been achieved by prior human BMTx (4,20). On the other hand, it has been reported that intra-bone marrow-bone marrow transplantation (IBM-BMTx) has been reported to result in more efficient bone marrow reconstitution by donor cells compared to the intravenous infusion of BMCs (IV-BMTx), even after low doses of irradiation in rodents (11,16,21,28).
We have previously reported that the transplantation model using analbuminemic F344 (F344-alb) rats as recipients and F344 rats as donors is an excellent way to monitor the efficiency of intrahepatic HCTx (27,43). F344-alb rats have the genetic background of F344 rats and are otherwise normal except for a seven base pair deletion downstream of the exon H splice site within the ninth intron of the albumin gene, which leads to an inability of hepatocytes to produce albumin (12). In this model, the efficiency of HCTx can be monitored by elevation of serum albumin levels and immunohistochemical albumin staining of hepatic tissues, and hematopoietic reconstitution by donor BMCs can be examined by PCR for albumin genes using DNA isolated from the recipient bone marrow.
In this study, we investigated allogenic intrahepatic HCTx using F344-alb rats as recipients and Lewis (LEW) rats as donors, which express disparate MHC. We used the retrorsine (RS)/partial hepatectomy (PH) treatment for the expansion of the transplanted allogenic hepatocytes, because transplanted normal hepatocytes can expand by responding to the growth stimuli generated by PH, while the host hepatocytes are unable to proliferate due to RS toxicity. We herein demonstrate that efficient bone marrow reconstitution by allogenic BMTx provides tolerance to allogenic HCTx and that IBM-BMTx is superior to IV-BMTx for this purpose.
Materials and Methods
Experimental Animals
F344 and LEW rats were purchased from Charles River Japan (Yokohama, Japan), and specific pathogen-free F344-alb rats were maintained in the Asahikawa Medical University Animal Laboratory. All rats were maintained on daily cycles of an alternating 12-h light/dark cycle with food and water available ad libitum. The procedures performed on the animals were approved by the animal care and use committee of the Asahikawa Medical University according to the guidelines for humane care of laboratory animals.
Animal Treatment and Cell Transplantation
Male F344-alb rats (6 to 8 months old) were assigned to different groups as shown in Figure 1. Group 1 received syngeneic F344 HCTx, while group 2 underwent allogenic LEW HCTx. Group 2 was then further divided into three groups: group 2a without the bone marrow reconstitution, group 2b with bone marrow reconstitution by IV-BMTx, and group 2c with bone marrow reconstitution by IBM-BMTx. Groups 2b and 2c were administered a single dose of irradiation by placing the rats in a container equipped with two opposing 137Cs γ-ray sources (Gamma Cell 40, Atomic Energy of Canada, Ottawa, Canada). BMCs were isolated from the femurs and tibia of 6- to 7-week-old male donor LEW rats, centrifuged with Histopaque 1077 (Sigma-Aldrich, St. Louis, MO) at 1,800 rpm for 30 min, and washed in modified Eagle medium solution (Sigma-Aldrich). A suspension of 5 × 107 BMCs suspended in physiological phosphate-buffered saline was injected into the penile vein for IV-BMTx (group 2b). The same number of cells were suspended in a collagen gel matrix (Nitta Gelatin, Osaka, Japan) and infused into the bone marrow cavity of the femur as described previously for IBM-BMTx (group 2c) (34). The recipient F344-alb rats were administered one dose of RS (Sigma-Aldrich, 30 μg/g body weight), intraperitoneally after 2 weeks, and were subjected to PH and immediately transplanted with 2 × 106 hepatocytes (cellular viability over 98%) via the portal vein after 4 weeks. Hepatocytes were isolated from 8-week-old male donor F344 and LEW rats using the two-step collagenase perfusion technique (3) and were suspended in Hanks' balanced salt solution (Sigma-Aldrich). At the eighth week all the recipient rats that survived were sacrificed under ether anesthesia.
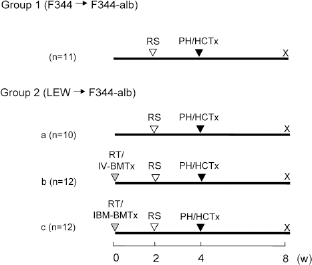
Experimental groups. Group 1: syngeneic hepatocyte transplantation (HCTx) using F344 (Fischer rats) as a donor and F344-alb (analbuminemic) as a recipient. The recipient F344-alb received the retrorsine (RS) treatment at the second week and partial hepatectomy (PH), followed by F344 HCTx at the fourth week. Group 2: allogenic HCTx using LEW (Lewis rats) as a donor and F344-alb as a recipient. Group 2a received RS treatment and PH/HCTx without prior whole body irradiation (RT)/bone marrow transplantation (BMTx), while groups 2b and 2c received RS treatment and PH/HCTx with the prior 8.0 Gy RT plus IV-BMTx and IBM-BMTx, respectively.
Histology and Immunohistochemistry
The livers were perfusion fixed with periodate-lysine-paraformaldehyde (PLP) solution via the portal vein and processed for histological and immunohistochemical analysis (27). Three slices were cut out from each hepatic lobe, fixed in a PLP solution at 4°C overnight, dehydrated through a graded ethanol series, and embedded in paraffin. The tissue specimens were cut into 3-μm-thick sections and stained with hematoxylin and eosin (H&E). For the immunohistochemistry examinations, the slides were deparaffinized, incubated with 3% H2O2, reacted with 1:500 diluted rabbit anti-rat albumin antibody (Cappel, Malver, PA), and incubated with a horseradish peroxidase-conjugated goat anti-rabbit IgG polymer (Dako, Carpinteria, CA). The antibody binding was visualized using diaminobenzidine substrate-chromogen system (Dako), followed by counterstaining with hematoxylin. Digital images of the albumin-stained sections (13.2 ± 1.6 cm2/rat) were obtained on a computer using an image scanner, and the albumin positive (Alb+) hepatocyte areas in the hepatic tissues were measured using the ImageJ software package (rsb.info.nih.gov/ij/), and the rate (%) was determined for each recipient rat.
PCR for the Albumin Genes
The extent of hematopoietic substitution in the recipient rat bone marrow was investigated by PCR using the albumin gene sequence as a marker. PCR primers were designed to amplify a region including the seven base pairs lacking in the analbuminemic albumin gene (2, 19,43), and the forward primer was tagged with 6-fluoreceine amidide (6-FAM). DNA was isolated from bone marrow of the recipient F344-alb rats using DNAzol (Molecular Research Center, Cincinnati, OH), and 40 cycles of PCR were performed using the DNA at 95°C for 5 s, 58°C for 5 s, and 72°C for 5 s with a final extension at 72°C for 10 min by a thermal cycler (iCycler, Bio Rad, Richmond, CA). The PCR products were analyzed on a genetic analyzer (ABI PRISM 310, PE-Applied Biosystems, Foster, CA) using the Genescan software package (PE-Applied Biosystems).
Measurement of Serum Albumin Levels
The serum albumin levels were determined at the fourth week after HCTx by the bromocresol green method (23) using an automatic analyzer (Hitachi 7180, Tokyo, Japan) and by an enzyme-linked immunosorbent assay (ELISA) (Bethyl Lab, Montgomery, TX).
Statistical Analysis
The relationship between Alb+ hepatocyte areas in the liver tissues and the elevation of serum albumin levels in the recipient rats at the fourth week after HCTx was statistically analyzed by a simple linear regression test using the JMP7.0.1 software (SAS Institute, Cary, NC).
Results
Irradiation Doses and Survival Rates
We first investigated the optimal irradiation dose for bone marrow reconstitution by BMTx using F344-alb rats. After 7.5 or 8.0 Gy whole body irradiation, 6/6 and 3/6 F344-alb rats survived until 2 weeks without BMTx, respectively (data not shown). When F344-alb rats received BMTx (5 × 107 cells), either by IV-BMTx (group 2b) or IBM-BMTx (group 2c) (Fig. 2) after 8.0 Gy irradiation, all the rats (12/12 in each group) survived until the fourth week and appeared healthy. When irradiated with 8.5 Gy, however, all F344-alb rats (6/6) died within 2 weeks even after F344 IBM-BMTx (data not shown). Bone marrow reconstitution by BMTx was therefore performed after 8.0 Gy irradiation.

Survival rates of the recipient A344-alb. All recipient F344-alb rats survived until the fourth week, while the fair numbers of them died after PH/HCTx at the fourth week.
Although all recipient F344-alb rats survived until the fourth week, several of rats (5/11 in group 1, 4/10 in group 2a, 6/12 in group 2b, and 6/12 in group 2c) died within 2 weeks after PH/HCTx during the fourth week (Fig. 2). This is likely due to persistent hepatic damage by irradiation/RS (group 2b and 2c) or RS (group 1 and 2a) and PH impaired liver functions, and it is possible that the transplanted hepatocytes could not compensate for liver functions, as it takes time for the transplanted hepatocytes to repopulate the liver following transplantation (6).
Bone Marrow Reconstitution After BMTx
We next investigated bone marrow reconstitution using the albumin gene sequence as a marker in the recipient F344-alb rats that survived until the eighth week. When PCR was performed to target the sequences spanning the seven base pair (bp) deletion in the analbuminemic albumin gene, the normal DNA fragment (68 bp) was generated from the F344 genomic DNA, while the analbuminemic DNA fragment (61 bp) was observed for the F344-alb genomic DNA as described previously (2,19,43) (Fig. 3a, b). When DNA from F344 and F344-alb was mixed in various ratios, the one pattern became dominant, when the amount of one DNA was present in a 9:1 excess ratio to the other (Fig. 3b). One of six of the recipients with LEW IV-BMTx (group 2b) showed a normal pattern, while 5/6 rats showed an aberrant pattern (Table 1, Fig. 3c). In the group with LEW IBM-BMTx (group 2c), 6/6 rats showed a normal pattern (Table 1, Fig. 3c). Therefore, bone marrow reconstitution was much more efficiently achieved by IBM-BMTx than IV-BMTx.

Analysis of bone marrow reconstruction by donor bone marrow cells (BMCs) using the albumin gene sequences as a marker. (a) PCR primer design to detect the albumin gene sequences. The forward primer is tagged with 6-FAM (6-carboxyfluorescein). (b) The normal 68 bp peak is observed for F344 DNA, while the aberrant 61 bp peak was generated from the F344-alb DNA. If the two DNAs were mixed in various ratio, one pattern became dominant, when one DNA was present over 9:1 excess against the other. Molecular markers show 50 bp and 60 bp in the red peaks. (c) In Group 2b, one F344-alb (#1 in Table 1) showed the normal pattern, while the others (represented by #2 in Table 1) showed aberrant patterns. All rats in group 2c (represented by #6 in Table 1) showed only the normal pattern.
Serum Albumin Levels, Albumin-Positive Areas in the Livers, and Donor Albumin Genes of F344-Alb Rats 4 Weeks After Hepatocyte Transplantation (HCTx)

Serum albumin levels determined by the bromcresol green method. The values for untreated F344 rats were 3.8 ±0.1 g/dl. —: values comparable to those of untreated F344-alb (1.0 g/dl).
Serum albumin levels determined by ELISA. The values for untreated F344 rats were 3.76 ± 0.60 g/dl.
+: F344 albumin gene dominant; -: F344-alb albumin gene dominant; nd, not done.
Histological Changes of the Recipient Livers
The livers of the recipient rats that received either syngeneic (group 1) or allogenic HCTx (group 2) showed megalocytosis (enlarged hepatocytes with huge nuclei) and frequent clusters of small-sized hepatocytes, as previously reported (22). Although the livers of F344-alb with syngeneic F344 HCTx (group 1) showed either no or only weak inflammation (Fig. 4a-1), those of F344-alb with allogenic LEW HCTx showed a strong degree of inflammatory cell infiltration, consisting mainly of granulocytes, in the portal tract and occasionally in sinusoids regardless to the prior BMTx (Fig. 4b-1). No inflammatory cell infiltration, however, was seen within the Alb+ hepatocyte areas in group 1 or in group 2b and 2c (Fig. 4a-2, b-2).

Histology of the recipient F344-alb livers. (a) Liver of the F344-alb with syngeneic F344 HCTx (group 1). No inflammatory cell infiltration was seen within the hepatic lobule (1), portal tract (inset), or Alb+ hepatocyte area (2). (b) Liver of F344-alb with allogenic HCTx with IBM-BMTx (intra-bone marrow; group 2c). Strong inflammatory cell infiltration was seen in the portal tract (1 and inset 1) and occasionally within the sinusoid (inset 2). However, no inflammatory cell infiltration was seen in the Alb+ hepatocyte area (2). (a-1, b-1) H&E staining; (a-2, b-2) albumin immunostaining. (a-1, b-1) 400×; insets (a-2, b-2) 1000×. Scale bar: 100 μm.
Replacement of Hepatic Parenchyma with Alb+ Hepatocytes
The hepatic tissue areas of recipient F344-alb rats that received syngeneic F344 HCTx (group 1) were replaced by Alb+ hepatocytes, ranging 4.5–28.7% (Table 1, Fig. 5a). In group 2a that received LEW HCTx without BMTx, their liver tissues did not contain any Alb+ hepatocyte areas (Fig. 5b). In 1/6 recipient F344-alb rats that received LEW IV-BMTx followed by LEW HCTx (group 2b), 44.5% of the liver tissue areas were replaced by Alb+ hepatoyctes, while the liver tissues of other five rats did not contain any Alb+ hepatocyte areas (Table 1, Fig. 5c, d). The six recipient F344-alb rats with LEW IBM-BMTx followed by LEW HCTx (group 2c) had liver tissues that were replaced with Alb+ hepatocytes, ranging from 4.8% to 58.4% (Table 1, Fig. 5e, f). The replacement of the liver tissues by the allogenic LEW Alb+ hepatocytes was clearly dependent on the bone marrow reconstitution by the donor LEW BMCs (Table 1).

Replacement of the liver tissue by Alb+ hepatocytes. F344-alb rat livers at the fourth week after PH/HCTx. The liver tissue was extensively replaced by Alb+ hepatocytes by syngeneic F344 HCTx (group 1) (a), but not by allogenic HCTx without BMTx (group 2) (b). Upon allogenic HCTx with prior IV-BMTx (group 2b), the liver tissue was extensively replaced by Alb+ hepatocytes in one F344-alb (#1 in Table 1) (c), but not in the other recipients (represented by #2 in Table 1) (d). In F344-alb rats that received allogenic HCTx with prior IBM-BMTx (group 2c), all the livers (represented by #1 and #4) were replaced by Alb+ hepatocytes to various degrees (e, f). (a–f) Albumin immunostaining, 20×. Scale bar: 1000 μm.
Elevation of Serum Albumin Levels
Although the serum albumin levels of analbuminemic rats have been reported to be less than 0.0005 g/dl (10), those of untreated F344-alb detected by the bromocresol green method was around 1.0 g/dl, most likely because this method is not very specific to albumin (23). We therefore examined the serum albumin levels also by ELISA, which can specifically detect rat albumin. The bromocresol green method demonstrated the albumin levels to increase above the levels in untreated F344-alb in 6/6 recipient rats with syngeneic F344 HCTx (group 1), 1/6 rats with LEW IV-BMTx/HCTx (group 2b), and 6/6 rats with LEW IBM-BMTx/HCTx (group 2c) at the eighth week (Table 1). ELISA showed the albumin levels to also increase in the same recipient rats (6/6 rats in group 1, 1/6 rat in group 2b, and 6/6 rats in group 2c) (Table 1). These values roughly corresponded to 5–80% of the serum albumin levels of untreated F344. The increase in the serum albumin levels in the recipient F344-alb rats was correlated with the degree of replacement of hepatic parenchyma with Alb+ hepatocytes (p = 0.007) (Fig. 6).
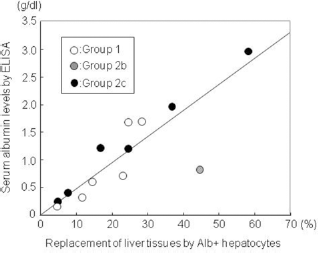
Correlation between the serum albumin levels and the replacement rates of liver tissues by Alb+ hepatocytes in the recipient F344-alb (p = 0.007). Open circles: group 1; gray circle: group 2b; black circles: group 2c.
Discussion
In the present study, we succeeded in performing the transplantation of allogenic LEW hepatocytes into the F344-alb liver and in replacing the RS/PH-treated liver tissues with allogenic HCTx by prior BMTx after sublethal irradiation. This was confirmed by immunohistochemistry, which showed an increase in Alb+ hepatocyte areas in the liver tissues (Table 1, Fig. 5) and an elevation of serum albumin levels in recipient rats (Table 1), and there was a correlation between the results (Fig. 6). Furthermore, the replacement of host liver tissue with the allogenic LEW Alb+ hepatocytes was dependent on the bone marrow reconstitution by the donor LEW BMCs (Table 1). No replacement of liver tissues by the donor hepatocytes occurred without the prior donor BMTx (group 2a), consistent with previous reports in which allogenic hepatocytes survived only few days after HCTx without immunosuppression or BMTx (7, 18). Our results are in line with those obtained in previous studies reporting either allogenic or xenogenic HCTx to be feasible when combined with the prior donor BMTx (4,20,26,28,35).
A histological analysis revealed that, although there were no or minimal inflammatory signs in the liver tissues of the recipient F344-alb with syngeneic HCTx (group 1) (Fig. 4a-1), those of the recipient F344-alb with allogenic HCTx (groups 2a, 2b, and 2c) exhibited strong inflammatory cell infiltration in the portal areas and occasionally in sinusoids regardless of BMTx status (Fig. 4b-1). Most hepatocytes infused into the portal circulation have been known to be trapped within the portal veins and sinusoids around the portal areas and thus are killed by innate immune cells such as granulocytes, macrophages, and Kupffer cells in early phases following allogenic and syngeneic HCTx (18). Strong inflammatory infiltration after allogenic HCTx suggested that stronger early rejection might have more occurred more frequently than with syngeneic HCTx (groups 2a, 2b, and 2c). It is also possible that the graft versus host reaction might have occurred in the liver in groups 2b and 2c, which received the allogenic BMTx, although no such reaction was detected in other tissues (data not shown). However, no inflammatory cell infiltration was observed in the Alb+ hepatocyte areas of the liver that received allogenic HCTx with BMTx (groups 2b and 2c), thus indicating that there might have been no immunological attack against the repopulated allogenic hepatocytes (Fig. 4b-2).
The replacement rate of host liver tissues by syngenic HCTx in RS/PH-treated F344 rats is approximately 40–60% at 4 weeks after HCTx and as much as 95% at 1 year after HCTx, respectively (22,39). However, the replacement rate varied greatly between the syngeneic HCTx group (group 1; 4.6–28.7%) as well as the allogenic HCTx group (group 1; 4.8–58.4%) (Table 1). Such variation might be due to the inconsistent efficiency regarding the infusion of hepatocytes into the portal vein rather than as a result of the immunological mechanism, because almost the same degree of variation was seen in both the syngeneic F344 HCTx without BMTx (group 1) and allogenic LEW HCTx with IBM-BMTx (group 2c). On the other hand, the liver of the recipient rats (group 1 and groups 2b, 2c) contained about 15–150 albumin+ areas/cm2 liver section 4 weeks after HCTx (data not shown), thus implying that about 600–18,000 clusters of Alb+ hepatocytes were present within the liver. If each Alb+ hepatocyte cluster was formed by the expansion of a single transplanted hepatocyte, then the engraftment of about 600–18,000 donor hepatocytes per liver might be sufficient for the extensive replacement of the liver tissue by HCTx.
Only a few previous studies of allogenic HCTx with BMTx have performed special treatments for expansion of transplanted cells. Streez et al. (35), however, reported that when allogenic hepatocytes of human α1-antitrypsin transgenic FVB mice were transplanted in the liver of BALB/C mice with prior BMTx, CD4+ lymphocytes infiltrating in the host liver augmented apoptosis of host hepatocytes, which may have facilitated liver regeneration and the expansion of donor cells. It is possible that, in addition to the RS/PH treatment, the liver damage caused by the inflammatory cell infiltration as observed in the allogenic LEW HCTx group (groups 2b and 2c) in the present study, might have contributed to the expansion of transplanted allogenic hepatocytes. The results of Streez et al. (35) and our present study suggest that allogenic hepatocytes can therefore expand within the host liver if appropriate growth stimuli and growth advantages are present.
According to the site of allogenic HCTx, some studies have used the spleen or subrenal capsules (26,28,41), while other studies used the liver (13,24,35,39). Because the expansion of transplanted cells in sufficient numbers is necessary to compensate for the impaired host liver functions, the liver might be an ideal site not only as a physiological site for hepatocytes to function, but also contain sufficient space for the transplanted hepatocytes to expand.
PCR examination of the albumin gene for host bone marrow DNA revealed 1/6 rats in group 2b and 6/6 rats in group 2c showed a normal albumin gene pattern, suggesting that more than 90% BMCs were of donor origin, because the normal albumin gene pattern was observed when donor DNA was present at a 9:1 ratio excess against the recipient DNA (Fig. 3b). The replacement of hepatic tissues by Alb+ hepatocytes was only observed in the recipient rats of which bone marrow was reconstituted by the donor BMCs (Table 1, Fig. 5c). Bone marrow reconstitution was much more efficient by IBM-BMTx than IV-BMTx (6/6 vs. 1/6 rats) as consistent with previous reports (11,16,21,28).
F344-alb and LEW rats used in this study express disparate MHC, RT1lv1, and RT1l, respectively. However, this combination demonstrates partial rather than complete allogenicity (15), and reciprocal heart transplantation between F344 and LEW rats induced a weaker rejection than the complete MHC mismatched combination, such as F344 (RT1l) and Black Norway rats (RT1n) (1). When considering the clinical applications of allogenic HCTx, it is necessary to confirm whether or not allogenic HCTx would be feasible between rats with complete MHC mismatched combinations.
There are many hurdles that must still be overcome to achieve the successful clinical application of allogenic HCTx, such as the efficient isolation of viable hepatocytes from small donor hepatic tissues, the protection of transplanted hepatocytes from the innate and acquired immune system, the efficient engraftment of transplanted hepatocytes into chronically injured hepatic tissues, and efficient methods for the massive repopulation of transplanted hepatocytes. However, HCTx might be especially efficient under conditions in which there are large selective advantages for transplanted hepatocytes, such as with acute and chronic hepatic failure or inherited liver disorders (i.e., ornithine transcarbamylase deficiency, tyrosinemia type I, α1-antitrypsin deficiency, Crygler-Najjar syndrome type 1, Wilson's disease, etc.). It is important to achieve the successful replacement of liver parenchyma by the allogenic IBM-BMTx/HCTx without use of RS in animal models. Shifting the paradigm for hepatic transplantation therapies from orthotopic liver transplantation to allogenic intrahepatic HCTx, it is therefore expected to yield great benefits for the overall treatment of human liver diseases.
Footnotes
Acknowledgments
The authors are grateful to Dr. Kazuhiro Nakaya and Mr. Hironobu Chiba for their valuable help in the animal care. This work was supported by a Grant-in-Aid for Scientific Research (C) (20591509) from Japan Society for the Promotion of Science.
