Abstract
Improved posttransplant monitoring and on-time detection of rejection could improve islet transplantation outcome. The present study explored the possibility of detecting harmful events after mouse islet transplantation measuring the immune responsiveness against islet extracts. Mouse islet transplantations were performed using various donor/recipient combinations, exploring autoimmune (NOD/SCID to NOD, n = 6) and alloimmune events (C57BL/6 to BALB/c, n = 20), a combination of both (C57BL/6 to NOD, n = 8), the absence of both (BALB/c to BALB/c, n = 21), or naive, nontransplanted control mice (n = 14). The immune reactivity was measured by ELISPOT, looking at the ex vivo release of IFN-γ from splenocytes stimulated by islet donor extracts (sonicated islets). The immune reactivity was not altered in the syngeneic and autoimmune models, demonstrating similar levels as nontransplanted controls (p = 0.46 and p = 0.6). Conversely, the occurrence of an allogeneic rejection alone or in combination to autoimmunity was associated to an increase in the level of immune reactivity (p = 0.023 and p = 0.003 vs. respective controls). The observed increase was transient and lost in the postrejection period or after treatment with CTLA4-Ig. Overall, allogeneic rejection was associated to a transient increase in the reactivity of splenocytes against islet proteins. Such a strategy has the potential to improve islet graft monitoring in human and should be further explored.
Introduction
The field of islet graft monitoring has grown tremendously over the recent years, with the aims to detect early harmful events, allow tailored treatment, and improve long-term outcomes (6). Various tests have been assessed, including the use of positron emission tomography (PET) (14,38), magnetic resonance imaging (MRI) (26,37), liver biopsy (34), and immunological markers (PCR for granzyme B and/or insulin mRNA, soluble CD30, flow cytometry) (5,16,19,32). At the present time, none of these modalities has gained broad clinical acceptance, and the more complex, time-consuming, and expensive procedures like PET, MRI, and biopsy will likely never be used as first line tests. Conversely, blood-based monitoring is noninvasive, less expensive, and has the potential to be used repeatedly every time the patient is seen in clinic.
Noninvasive immune monitoring has gained similar attention in the area of solid organ transplantation (10,28). Recent studies have established that the level of donor-specific peripheral blood mononuclear cell (PBMC) reactivity does correlate with postkidney transplant outcome, allowing for tailored immunosuppression in high-risk individuals (2,3,20,27,29).
Besides allogeneic events, islet grafts can also be lost due to recurrence of autoimmune diabetes. Here again, ex vivo blood-based monitoring strategies have been established, and patients with type 1 diabetes have higher levels of lymphocyte reactivity against islet-specific peptides and proteins than control individuals (25,31).
This study was designed to assess the feasibility and usefulness of islet donor antigen-stimulated enzyme-linked immunosorbent spot assay (ELISPOT) monitoring after islet transplantation using mouse models. Specific aims were: 1) to define whether harmful immunological events could be detected after islet transplantation; 2) to assess whether the detected events are transient, thus potentially allowing for the detection of signs of cure and/or recurrence; 3) to determine the specificity of the test for allo- and/or autoimmune events; and 4) to assess the impact of immunosuppression on results.
Materials and Methods
Animals
BALB/c, C57BL/6, NOD/ShiLt (NOD), NOD/ShiLtSz-Prkdcscid (NOD/SCID) mice were obtained from the Jackson Labs (Bar Harbor, ME). All animals were cared for according to the international guidelines on animal care, and ethical approval was obtained from the institutional animal welfare committee.
Islet Isolation and Transplantation
Mouse islets were isolated according to established methods (36). Briefly, the pancreas was distended with collagenase, retrieved, and digested at 37°C. Islets were purified in Ficoll density gradient. Aliquots of islets were counted after dithizone staining. Five hundred islets were transplanted under the left kidney capsule using previously described techniques (35).
Transplantations were performed using four donor/recipient combinations as described in Table 1. These combinations were designed to assess the impact of allogeneic rejection episodes and/or of a recurrence of autoimmunity, allowing for a differential read-out of these immunological events.
Characteristics of the Studied Transplant Groups

Assessed by log-rank test.
Recipients were treated on days 0, 2, 4, and 6 with IP 0.25 mg CTLA4-Ig.
Spontaneously diabetic NOD females were identified by monitoring our colony two to three times per week, with animals considered diabetic following two consecutive blood glucose readings >18 mmol/L or one reading >25 mmol/L with a One Touch Ultra Glucometer (Lifescan, Mississauga, ON). They were subsequently maintained on insulin (LinBit, LinShin, Toronto, ON) for 2–3 weeks to collect a sufficient cohort for transplant. Linbit was removed at the time of transplantation. The BALB/c recipients included in the ELISPOT studies were nondiabetic. This was elected in order to avoid the suppression of adaptive immunity and the subsequent homeostatic T-cell proliferation induced by chemical induction of diabetes (24). The impact of immunosuppression was monitored in the pure allogeneic combination (C57BL/6 to BALB/c). In this group, recipients were treated with CTLA4-Ig administered on days 0, 2, 4, and 6 posttransplant (0.25 mg, IP), as previously described (12).
In order to assess the kinetics of islet loss (results presented in Table 1), transplants were independently performed in diabetic recipients. For this purpose, diabetes was induced in BALB/c recipients with single IP injections of 200 mg/kg streptozotocin (Sigma-Aldrich, Oakville, ON). Only animals with blood glucose levels ≥20 mmol/L were used.
Splenocyte and Islet Protein Isolation
Posttransplant islet survival (defined as the time to blood glucose ≥ 20 mmol/L) was substantially different between the various donor/recipient combinations, according to the presence of autoimmune recurrence and/or alloimmune rejection (at least p ≤ 0.05, log-rank, Table 1). In order for all recipients to be close to the expected immunological event at the time of study, spleens were retrieved at 4 weeks posttransplant in the pure allogeneic rejection group, at 2 weeks in the pure autoimmune group, and at 1 week in the combined auto- and alloimmune group. Recipients were euthanized at 2 weeks posttransplant in the syngeneic group.
In a subsequent experiment, the kinetics of immune reactivity were assessed prior to and after rejection. Animals were therefore euthanized at 2 weeks (prerejection), 4 weeks (rejection), and 8 weeks (postrejection) in the C57BL/6 to BALB/c model, and at 1 week (at islet loss) and 2 weeks (postislet loss) in the C57/Bl6 to NOD combination. At least four (in one group) and up to nine mice were used per group (median n = 6).
Splenocytes were purified using Lympholyte-M (Cedarlane, Burlington, ON) and cultured at 37°C, 5% CO2 in Roswell Park Memorial Institute (RPMI)-1640 medium containing HEPES (25 mmol/L, final concentration), penicillin (112 kU/L), streptomycin (112 mg/L), L-glutamine (100 mg/L), and fetal calf serum (10%). Overall, 21.4 ± 4 × 106 splenocytes were isolated per animal.
Islet proteins were obtained by sonicating freshly isolated islets on ice for 10 s twice, thus achieving a pure particle-free fluid. The protein content was assessed utilizing the Bradford assay (Sigma-Aldrich, Oakville, ON) and bovine serum albumin as reference. The quality of various surface and intracellular proteins was assessed in a Western immunoblotting experiment, in order to exclude a potential degradation during processing. Twenty micrograms of sonicated islet proteins was resolved in 12%-polyacrylamide-SDS gel, transferred to a nitrocellulose membrane, and incubated overnight at 4°C with antibody to α-ERK and α-AKT (Cell Signaling Technology, Danvers, MA), H2Db (clone KH95, BD Pharmingen, San Jose, CA) and α-actin (Sigma-Aldrich). The average protein content was 0.2 ± 0.12 μg per sonicated islet. The final product was stored at −20°C.
Enzyme-Linked Immunosorbent Spot (ELISPOT) and Cytokine Profile Assessment
Splenocytes were seeded in triplicate at 106 cells/well in 200 ml completed RPMI-1640 medium. They were stimulated with sonicated islet proteins from the relevant donors (5 μg/ml) or phorbol 12-myristate 13-acetate (PMA, Sigma-Aldrich, 5 μg/ml). No stimulator was added in the negative controls.
After 24 h of culture (37°C, 5% CO2), the release of IFN-γ was assessed by ELISPOT (e-Bioscience, San Diego, CA). Briefly, wells were coated overnight prior to culture with capture anti-IFN antibody. After culture, wells were washed and biotinylated detection antibody added. Following incubation with avidin-horseradish peroxidase, the substrate solution was added and spots were read using an automated Immunospot analyzer (Cellular Technology, Cleveland, OH). Results were expressed as absolute spot counts/106 splenocytes, after removing the background release of IFN-γ found in the negative control.
The cytokine release profile was assessed in the supernatant at the end of the ELISPOT 24-h culture. Quantification was performed using a mouse Multiplex Bead Immunoassay according to the manufacturer protocol (Biosource, Nivelle, Belgium). Of note, IFN-γ could not be assessed in the multiplex assay as it had previously bound to the detection antibody during the ELISPOT assessment.
Statistical Analysis
Results are displayed as median [interquartile range]. Survivals were assessed by the Kaplan-Meier method and groups were compared using the log-rank test. ELISPOT results were assessed by nonparametric tests, including Mann-Whitney test and one-way ANOVA on ranks with Bonferroni's post hoc test. The accuracy of the immune monitoring to predict rejection was assessed utilizing a Receiver Operating Characteristic (ROC) curve and determining its Area Under the Curve (AUC). A standard alpha level of 0.05 was selected. Analyses were performed with the SPSS software package (SPSS 15.0, SPSS, Chicago, IL).
Results
We first assessed the alteration of the immune reactivity of BALB/c splenocytes against islet proteins at the time of islet loss (Fig. 1). As anticipated, a syngeneic transplantation (BALB/c to BALB/c) did not alter the immune reactivity compared to control, nontransplanted BALB/c mice (37 [110] vs. 2 [16] spot/106 cells, p = 0.50). Conversely, the occurrence of an allogeneic rejection (C57BL/6 to BALB/c) was associated with a significant increase in the level of immune reactivity (54 [631] spots/106 cells, p = 0.043 vs. naive BALB/c controls).

Immune reactivity of splenocytes against islet proteins assessed by IFN-γ ELISPOT. Results are displayed as median (lower–upper quartiles). Of note, the spread of the allogeneic rejection data was quite large. *p = 0.043 versus Naive BALB/c; **p = 0.007 versus Naive diabetic NOD.
The impact of a recurrence of autoimmunity was assessed in NOD mice (Fig. 1). The recurrence of autoimmunity alone did not alter the immune reactivity compared to naive, nontransplanted, diabetic NOD mice (11.5 [56] vs. 6 [63] spots/106 cells, p = 1). Conversely, the presence of an allogeneic rejection on top of the recurrence of autoimmunity did induce a significant increase in the level of reactivity (253 [68] spots/106 cells, p = 0.007 vs. naive diabetic NOD).
In an effort to understand whether the observed increase in reactivity was permanent or only transient, we monitored mice prior to and after the occurrence of harmful events (Fig. 2). The reactivity observed at the time of rejection (4 weeks) in the C57BL/6 to BALB/c model was only transient, as testified by the low prerejection (1 week) and postrejection (8 weeks) levels, respectively, at 17 [14] and 18 [14] spots/106 cells (both p ≤ 0.05 vs. rejection) (Fig. 2A). Similarly, the increase in reactivity observed at the time of rejection in the C57BL/6 to NOD combination was lost in the postrejection period (43 [109] spots/106 cells at 2 weeks, p = 0.029 vs. 1 week) (Fig. 2B).
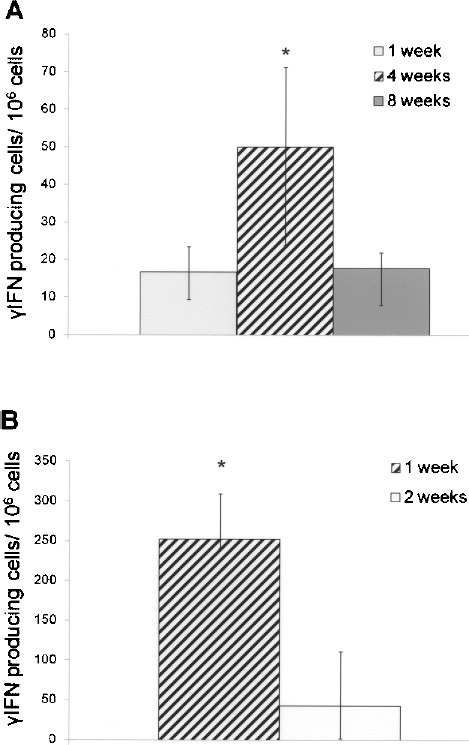
Immune reactivity of splenocytes against C57BL/6 islet proteins assessed by ELISPOT. The increase of reactivity observed at the time of rejection disappeared after rejection, both in the C57BL/6 to BALB/c (A) and C57BL/6 to NOD (B) models. p = 0.007 and p = 0.029 vs. 4-week and 2-week time points, respectively. Results are displayed as median (lower–upper quartiles).
The impact of immunosuppression was assessed after 4 weeks of treatment with CTLA4-Ig. This regimen is associated with significantly improved islet survival (Table 1, p ≤ 0.001). Compared to control, nontreated recipients, CTLA4-Ig treatment was associated with a significantly lower level of reactivity (p = 0.034) (Fig. 3).

Prevention of rejection with CTLA4-Ig is associated with a decreased level of reactivity at 4 weeks after transplantation (p = 0.034). Results are displayed as median (lower–upper quartiles).
The accuracy of the IFN-γ ELISPOT in predicting rejection (4-week values) was assessed using an ROC curve in the C57BL/6 to BALB/c combination (1-, 4-, 8-week and CTLA4-Ig values were entered in the model, n = 30) (Fig. 4). The accuracy of the IFN-γ ELISPOT was good, as the AUC was 0.85 ± 0.087. With a cut-off at 25 spots/106 cells, sensitivity was 70% and specificity 94%.

ROC curve assessing the accuracy of IFN-γ ELISPOT in predicting rejection. AUC = 0.85 ± 0.087. Diagonal segments are produced by ties.
In order to better characterize the profile of the induced immune response at the time of islet loss, and potentially refine the test, various released cytokines were measured in the supernatant after 24-h culture. Both TNF-α and IL-17 demonstrated increased levels and IL-2 decreased levels at the time of islet loss in the C57BL/6 to NOD model (while this was not the case in the pure allogeneic C57BL/6 to BALB/c combination) (Fig. 5).

Cytokine profile after 24 h of culture of recipient splenocytes with donor islet proteins. Naive, nontransplanted mice were used as reference and results were expressed as fold increase/decrease. *IL-2, p = 0.028, TNF-α, p = 0.022, IL-17, p = 0.028. Results are displayed as median (lower–upper quartiles).
Of note, the Western blotting studies for ERK, AKT, H2Db, and α-actin showed appropriate bands (Fig. 6), demonstrating the absence of degradation of these surface and intracellular proteins due to sonicating.
Western immunoblotting analysis of the sonicated islet extracts for α-actin, H2Db, ERK, and AKT proteins.
Discussion
The present study demonstrates that islet protein-stimulated IFN-γ ELISPOT is efficient in monitoring mouse islet transplantation and identifying the occurrence of a rejection.
The ELISPOT assay has been previously used after clinical kidney transplantation as a high sensitivity functional tool assessing primed memory/effector T cells and predicting kidney function (7,20,27–29). In the present study, islet proteins were used as the stimulator, allowing for the indirect activation of lymphocytes (antigens presented by self APCs). The combined presence of donor major histocompatibility complex (MHC) and islet antigens was designed to trigger the activation of both allo- and autoimmune primed memory/effector cells. This strategy combined properties of the mixed lymphocyte reaction (MLR) using donor MHCs on PBMCs, and of tests using purified islet proteins like GAD (21,22). Despite this combined stimulation, only allogeneic rejection could be detected. This low level of IFN-γ release contrasts with the early loss of islets in the pure autoimmunity transplantation model (NOD/SCID to NOD, median islet loss: 14 days [0–19)], and may be related in part to a low threshold of detection of these events with the current design of the test. When appropriate, the detection of autoimmunity may be improved with the use of purified islet-related proteins as stimulator and/or with the study of alternative cytokines or cytokine combinations. The observed cytokine profile suggests a shift from Th1 to Th17 together with an increase of TNF-α in case of autoimmune diabetes (higher IL-17 and TNF-α, lower IL-2). These data are consistent with previous reports suggesting that IL-17 is associated with the development of diabetes and IL-17 inhibition with its prevention (9,11). In addition, TNF-α has been associated with the development of diabetes in NOD mice (23,39).
The studied test was extremely efficient in detecting rejection episodes, both in BALB/c and NOD mice. In the C57BL/6 to BALB/c model, the AUC of the ROC curve was high at 0.85 ± 0.087. In addition, the detected immune response was back to baseline after rejection. This observation is linked to an increased frequency of the studied memory/effector cells at the time of rejection, followed by a rapid compression (likely when all islet antigens have been cleared). This transient pattern can not only allow for the detection of rejection, but also for the monitoring of its resolution and of a possible recurrence. The use of immunosuppression (CTLA4-Ig) was also linked to a lower response, likely linked to a lower frequency of responder cells and/or to their previous exposure to immunossuppression in the recipient. If applied clinically, this would allow for a better tuning of immunossuppression after islet transplantation.
While encouraging, these mouse observations need further validation in human. One of the main obstacles may be the less pure and slower rejection kinetics in human, related to the background immunosuppression.
In addition, in order to have access to large numbers of cells, only splenocytes were used in the present study and the validation with PBMCs is still required. However, based on a previous study, one can speculate that similar results would be achieved (13).
Of note, the study design did not assess whether the observed immune reactivity was specifically directed against islet antigens, as the appropriate controls were missing. This assessment remains to be performed, potentially including stimulation with controls such as islet antigens from a third party, heat-inactivated islet-specific antigens to denature the proteins, and donor-specific proteins from the acinar tissue or splenocytes.
The current trend in the field of islet transplantation is to use more potent (thus often with more side effects) immunosuppression protocols, often utilizing stronger induction therapies, including alemtuzumab (anti-CD52), efalizumab (anti-LFA-1), and anti-thymocyte globulins (4,8,15–18,30,33). The availability of an accurate monitoring test would help better modulate and tune immunosuppression and decrease side effects. In addition, the on-time detection of rejection could lead to an appropriate treatment (still to be defined), leading to better long-term rates of insulin independence. Besides islet transplantation, similar tests would also be of great help in other types of cell and solid organ transplantations; thus allowing for the detection of rejection or of patients at low risk, eligible for immunosuppression minimization or drug withdrawal (1).
Overall, this study demonstrates that islet protein-stimulated IFN-γ ELISPOT can accurately detect allogeneic rejection after murine islet transplantation. As the observed signal is transient, repeated measurements can be performed. Although translation from mouse to human cannot be predicted, similar experiments should be conducted in humans, as they have the potential to be of substantial help in the management of islet graft recipients.
Footnotes
Acknowledgments
C.T. was supported by the Swiss National Science Foundation (SCORE grant 3232230-126233). A.M.J.S. was supported by an AHFMR Senior Clinical Scholarship. The study was funded in part by a grant “Projet Recherche et Développement” from the University of Geneva Hospitals.
