Abstract
Total pancreatectomy with autologous islet cell transplantation (TP with AIT) is an effective treatment for chronic pancreatitis patients with severe abdominal pain. Body mass index (BMI) of the pancreatic donor is proven to be a useful predictor for islet isolation and transplantation outcomes in allogenic islet transplantation. However, the association between BMI and islet isolation outcome and/or metabolism after AIT was previously unclear. Twelve patients who received TP with AIT at our hospital were included in this study. All pancreata were preserved with both pancreatic ductal injection and oxygen-charged static two-layer method using ET-Kyoto solution. The cohort was divided into two groups: low BMI group (BMI < 23 kg/m2, n = 5) and high BMI group (BMI ≥ 23, n = 7). The high BMI group had a significantly higher islet yield per gram than the low BMI group both in pancreas postdigestion and in final product (postdigestion: 7330 ± 539 vs. 3509 ± 563 IE/g; p < 0.001; final product: 6555 ± 585 vs. 3476 ± 546 IE/g; p = 0.004). For islet yield in final product per patient body weight, the high BMI group also had significantly higher islet yield than the low BMI group (7997 ± 779 vs. 4175 ± 750 IE/kg, p = 0.007). Insulin independence rate in the high BMI group (71%) was also higher than that low BMI group (40%), but it did not reach statistical significance. Pancreata from patients with higher BMI could obtain higher islet yield in the setting of autologous islet cell transplantation for chronic pancreatitis.
Introduction
Predicting islet isolation results is important to identifying suitable donors for islet transplantation. Most previous reports on factors that affect human islet isolation were based on data using deceased donors, which aimed for successful islet allotransplantation for patients with type 1 diabetes (4,7,8,13,15,18,20,22,23,32,33,36,40,42, 47). These studies showed a high donor body mass index (BMI) improved islet yields.
Total pancreatectomy and autologous islet cell transplantation is a treatment option for chronic pancreatitis with severe abdominal pain (6,11,38). Predicting islet isolation results is also important in identifying suitable patients. However, there is limited information on the effects of BMI on the outcome of islet isolation for autologous islet transplantation. The University of Minnesota reported that fibrosis and acinar atrophy inversely correlated with islet yield in their pediatric cohort (19). The same group also showed that body weight and fasting blood glucose could predict islet yield in pediatric patients with chronic pancreatitis (3).
In this study we examined whether BMI is an effective predictor for islet isolation from a chronic pancreatitis pancreas.
Materials and Methods
Patients
We started performing autologous islet transplantation for patients with chronic pancreatitis in November 2006. Beginning in December 2007, we adapted novel pancreas preservation and isolation methods resulting in excellent islet isolation outcomes from brain-dead donors (29,35). Because our islet isolation methods substantially changed in December 2007, we only analyzed data after this time.
Twelve patients who fulfilled the following criteria were included in this study: (i) clinically diagnosed chronic pancreatitis with intractable pain, (ii) received total pancreatectomy with islet autotransplantation from December 2007 to October 2009 at Baylor University Medical Center (Dallas, TX), (iii) infused islets that were isolated as described below. This study was approved by institutional review board at Baylor Research Institute (Dallas, TX) and written informed consent was obtained from all patients included in this study.
Islet Preparation
Immediately after total pancreatectomy, chilled ET-Kyoto solution (Otsuka Pharmaceutical Factory Inc., Naruto, Japan) was injected through a cannula inserted into the main pancreatic duct as previously described for ductal injection (16,26,27,35). The pancreas was preserved using the oxygen-charged static two-layer method (ET-Kyoto solution/oxygenated PFC) (30). Liberase MTF with thermolysin (Roche, Indianapolis, IN) or collagenase NB with neutral proteases (Serva Electrophoresis GMbH, Heidelberg, Germany) was infused into the main pancreatic duct (Table 1) (28). Pancreas digestion was performed using the modified Ricordi method (37). If pellet volume was larger than approximately 15 ml, islets were purified with the COBE 2991 cell processor (CaridianBCT, Inc; Lakewood, CO) with continuous density gradient centrifugation (26). The final preparation of islets was assessed for its yield and purity using dithizone staining (2 mg/ml; Sigma Chemical Co., St. Louis, MO). The islet yield was converted into a standard number of islet equivalents (IEQ, diameter standardizing to 150 μm) (21). Islet viability in final product was evaluated with fluorescein diacetate/propidium iodide staining (2). The viability was calculated by averaging the viability of 50 islets.
Enzyme Profiles

BMI, body mass index.
Transplantation
Isolated islets were infused into the portal vein via the mesenteric vein with heparin (70 U/kg body weight) over 30–60 min while the patient was under general anesthesia. During islet infusion, portal vein pressure was monitored intermittently. If portal vein pressure was higher than 22 mmHg the infusion of islets was stopped and then restarted when portal vein pressure was reduced.
Image Studies Before Transplantation
Imaging studies including endoscopic retrograde cholangiopancreatography (ERCP) and endoscopic ultrasonography (EUS) at pretransplantation period were reviewed. ERCP images were classified according to the Cambridge classification: “Normal” (scored as 0) to “Marked” (scored as 4) (1,43). Previous reports showed the EUS is useful to evaluate the diagnosis and severity of chronic pancreatitis (5,12,17,46). EUS criteria included hyperechoic foci, hyperechoic strands, parenchymal lobularity, irregular main pancreatic duct margins, hyperechoic main pancreatic duct margins, visible side branches budding, main pancreatic duct dilatation, shadowing calcifications and cysts, and the presence of four or more criteria was used for diagnosis of chronic pancreatitis (9,41).
Assessment of Transplanted Islet Function
Insulin independence was defined as 2 weeks or longer of no exogenous insulin with good glycemic control by capillary glucose measurements, both available from daily diaries according to the report from Collaborative Islet Transplant Registry (CITR) (10). Blood glucose was controlled with continuous intravenous insulin infusion in intensive care unit immediate after surgery, followed by subcutaneous insulin injection therapy to maintain the following goal: fasting blood glucose <126 mg/dl or HbA1c ≤ 6.5%. HbA1c was measured at 1 month after transplantation.
Transplanted islet function was assessed by C-peptide and secretory unit of islet transplant objects (SUITO) index. The highest value of fasting C-peptide in multiple samples from postoperative days 3 to 10 for each case was evaluated. Because serum C-peptides might be released from injured islets within 48 h after islet transplantation, we excluded the C-peptide value on postoperative days 1 and 2 for this assessment. SUITO index, which has been shown to be a good clinical parameter for engrafted islet function in previous publications, was calculated using the following formula: 1500 × fasting C-peptide level (ng/ml)/[fasting blood glucose (mg/dl) – 63] (25,31).
When the value of C-peptide was below the detective level of the assay (0.1 ng/ml), the islet function was defined as “no function.” When a patient achieved insulin independence, the islet function was defined as “full function.” When neither of the above two conditions were met, the islet function was defined as “partial function.”
Pain Assessment
Pain was scored according to visual analogue scales (VAS) from 0 (no pain) to 10 (severe pain). Opioid doses for pain control were also collected from the different prescriptions and converted to morphine equivalent doses according to published data (14).
Histology and Immunohistochemistry
Biopsy specimens from a pancreatic head were taken in one case with severe chronic pancreatitis (49-year-old female). For control, biopsy specimens from a normal pancreas were also taken (48-year-old female). The samples were embedded in OCT compound (Tissue Tek, Torrance, CA) and stored at −80°C until sectioning. Sections (9 μm) of embedded specimens were taken and stained for insulin (1:75 dilution, Abcam, Cambridge, MA) using standard immunohistochemical (IHC) techniques. After incubation with horseradish peroxidase (HRP)-onjugated secondary antibody, staining was visualized using diaminobenzidine (DAB) and the sections were counterstained with hematoxylin. Additional sections were stained with hematoxylin and eosin (H&E).
Statistical Analysis
Data analyses were performed using SPSS 15.0 for Windows (SPSS, Chicago, IL). Patients were divided into the following two groups according to BMI (kg/m2): low BMI group (BMI < 23) and high BMI group (BMI ≥ 23). The difference of means was tested by Student' t-test and comparisons of rates between groups were done using a two-sided Fisher's exact test. Mann-Whitney U-test was used for nonparametric parameters: pain score, score of Cambridge classification for ERCP, number of positive EUS criteria, and distention score. Correlations were evaluated using Spearman's coefficients. A receiver operating characteristic (ROC) curve was plotted to determine the cut off value of BMI for islet yields. A two-sided value of p < 0.05 was considered statistically significant. Values are expressed as mean ± SE, except for nonparametric parameters, which are expressed as median and range.
Results
Patient Characteristics
Patient characteristics before transplantation are shown in Table 2. All patients were administered a regular narcotic medication. There were no diabetic patients before transplantation in this cohort. The low BMI group had significantly higher scores of severity of pancreatitis assessed by both ERCP (p = 0.007) and EUS (p = 0.02) than the high BMI group. Mean duration of symptoms in the low BMI group were longer than the high BMI group, which did not reach statistical significance. There was a significant correlation between BMI and EUS score (Spearman's coefficient r = −0.928, p = 0.008), although there was no significant correlation with duration of symptoms (r = −0.169, p = 0.64) and ERCP (r = −0.544, p = 0.16).
Patient Characteristics

AIT, autologous islet cell transplantation; ERCP, endoscopic retrograde cholangiopancreatography; EUS, endoscopic ultrasonography.
BMI < 23 group included chronic pancreatitis due to cystic fibrosis, ERCP, and alcohol. BMI ≥ 23 group included secondary due to pancreatic divisum.
BMI < 23 group included two patients with a history of Whipple resection.
Values are expressed as median and range with p-value by Mann-Whitney U-test.
Outcome of Islet Isolation
There were no significant differences in pancreas weight and cold ischemia time (Table 3). The low BMI group had significantly poorer scores of distension when compared with the high BMI group (p = 0.04). Islet yield (IE) and islet yield per pancreas weight (IE/g) at both postdigestion and final product had significant differences between the two groups. Total islet yield per patient body weight (IE/kg) was also significantly different among the two groups (p = 0.007). In order to eliminate the effect of previous operations we eliminated two cases with previous operations and re-analyzed the data. Even excluding the two cases with past history of pancreatic operation from the low BMI group, there were still significant differences in islet yield (IE), islet yield per pancreas weight (IE/g), and islet yield per patient body weight (IE/kg) at both postdigestion and final product between the low- and high BMI groups (p < 0.05) (Table 4). There were no statistical differences in purity and viability. There was a significant correlation between BMI and islet yield in final product (r = 0.74, p = 0.006) and at postdigestion (r = 0.83, p = 0.001) (Fig. 1).
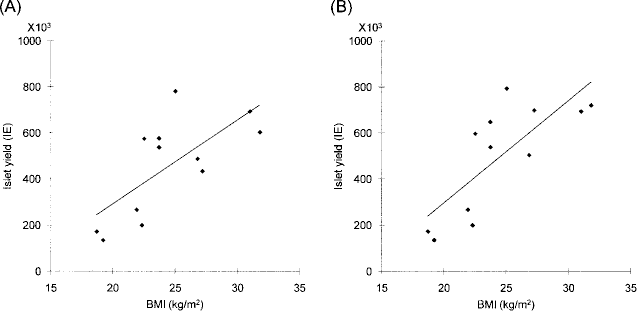
Correlation between islet yield and BMI. There was a significant correlation between BMI and islet yield both in final product (A, Spearman's coefficient r = 0.74, p = 0.006) and postdigest islet yield (B, r = 0.83, p = 0.001).
Outcomes of Islet Isolation
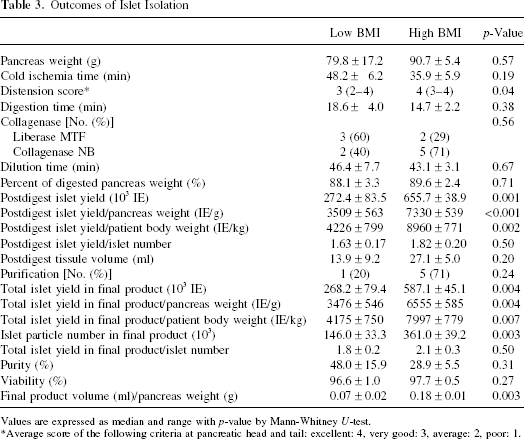
Values are expressed as median and range with p-value by Mann-Whitney U-test.
Average score of the following criteria at pancreatic head and tail: excellent: 4, very good: 3, average: 2, poor: 1.
Islet Yields Except for Two Cases With Previous Pancreatic Surgery
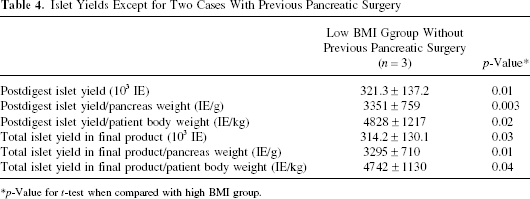
p-Value for t-test when compared with high BMI group.
The rate of isolated islet size of 100–150 μm at postdigestion in the high BMI group was significantly higher than the low BMI group (p = 0.04), although that of 300–350 μm in the high BMI group was significantly lower than the low BMI group (p = 0.05) (Table 5).
Distribution of Isolated Islet Size After Digestion

The percentages of categorized size/total islets were shown.
Validation of the Cut Off Value of BMI
Previous large cohort studies showed that islet yields in final product per patient body weight (IE/kg) were significantly correlated with graft function; in patients with more than 5,000 IE/kg, 86% had graft function and 63% remained insulin independent 1 year after transplant (44). Herein, we validated cut off value of BMI for islet yield in final product per patient body weight more than 5,000 IE/kg using ROC analysis. Both sensitivity and specificity to determine islet yield > 5,000 IE/kg were 100% when cut off value was BMI 23 (area under the curve 1.0 ± 0.0, p = 0.007) (Fig. 2).
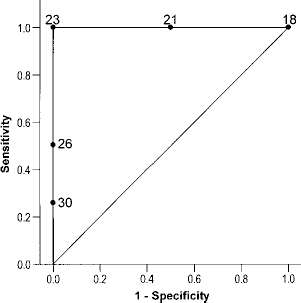
ROC curve of BMI for islet yield >5,000 IE/kg. Each number in the graph shows BMI at the point on ROC curve. BMI 23 is the most appropriate for cut off value to determine islet yield >5,000 IE/kg.
Metabolic Outcome
Follow-up period in the low BMI group was significantly shorter than the high BMI group (p = 0.02) (Table 6). All patients maintained graft function assessed by positive C-peptide. Insulin independence rate and average SUITO index in the high BMI group was higher than in the low BMI group, but it did not reach statistical significance, whereas the highest fasting C-peptide in the high BMI group was significantly higher than the low BMI group (p = 0.01). Average HbA1c in both groups remained less than 6.5% and there was no significant difference between two groups (p = 0.74).
Clinical Outcomes

SUITO index, secretory unit of islet transplant objects index.
Histology in the Low BMI Group
The majority of exocrine tissue was already destroyed in pancreas with severe chronic pancreatitis as seen in H&E staining (Fig. 3A) and insulin staining (Fig. 3B) compared to normal pancreas (Fig. 3C, D). This patient had the lowest islet yield per body weight (2,559 IE/kg) and low BMI (BMI 19.2).
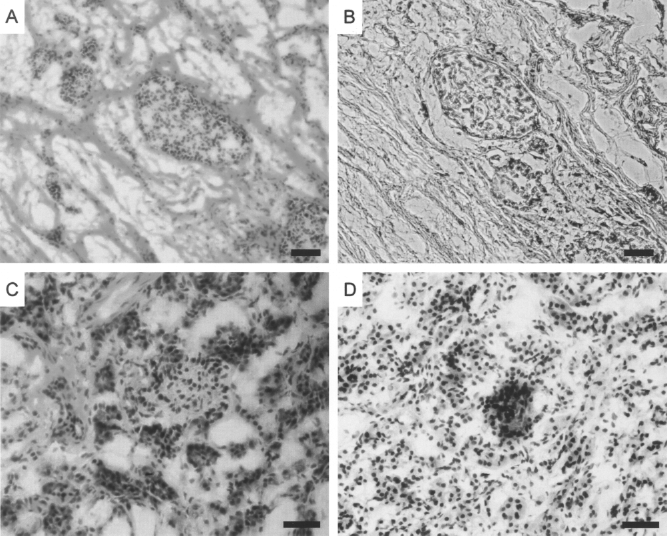
Histology in low BMI group. Pancreas biopsies from a 49-year-old female with chronic pancreatitis (A, B) and a 48-year-old healthy female (C, D). Hematoxylin and eosin (A, C) and insulin staining (B, D) were performed on frozen sections. Scale bar: 50 μm.
Discussion
The retrospective study presented here supports an association among BMI, islet yields, and clinical outcomes in autologous islet cell transplantation for patients with chronic pancreatitis. This is the first report to mention the association between BMI and islet isolation and metabolic outcome for autologous islet cell transplantation. This study showed that patients in the high BMI group had better islet isolation and autologous islet cell transplantation outcomes, similar to allogeneic islet transplantation findings previously reported (23,33,42).
For allogeneic islet isolation, pancreas distension is usually excellent because only pancreata of reasonable quality were selected for islet isolation. However, in the case of auto-islet transplantation, patients were selected because of severity of abdominal pain irrespective of the status of pancreatitis. Therefore, we encountered low-quality pancreata for auto-islet transplantation.
Recently, we implemented novel methods for pancreas procurement and islet isolation, which were originally developed for allogeneic islet cell transplantation using a non-heart-beating donor pancreas (29), into autologous islet cell transplantation for patients with chronic pancreatitis to maximize islet yield and graft function. In order to avoid the effect of differences in pancreas procurement and islet isolation methods, only cases in which the novel methods were fully performed were included in this study. These new methods including ductal injection with ET-Kyoto solution and two-layer method with oxygenated perfluorochemical (PFC) could obtain significantly higher islet yields than the previous methods used (34). For cadaveric pancreas procurement, the pancreas is immediately chilled by the perfusion of cold preservation solution. Conversely, for autologous islet transplantation, the pancreas is procured without perfusion of cold preservation solution; therefore, the pancreas suffers from warm ischemic injury. Because warm ischemic injury of more than 30 min had a significant impact on islet isolation (22), minimizing warm ischemic time is critical. We postulated that ductal injection of chilled ET-Kyoto solution minimized warm ischemic time for autologous islet isolation and transplantation. It was shown that two-layer method can alleviate warm ischemic injury (24). However, because average cold ischemic time was only 45 min in this study, the effect of the two-layer method is uncertain.
In this study, the low BMI group had significantly higher scores in clinical image studies for severity of pancreatitis suggesting late stage inflammation. Previously it was demonstrated that pancreatic fibrosis was associated with poor islet yield (22) and number of EUS criteria was well associated with histological findings (45). In this study, fibrotic change was observed in patients with lowest islet yield per body weight. Lower BMI might be associated with inflammatory change of pancreas. In addition, in this study pancreas distension was significantly worse in the low BMI group. This difficulty in distension is most likely due to fibrosis and rigidity because of severe pancreatitis. Damage to the pancreas due to inflammation plus distension difficulty due to rigidity in the low BMI group should be the main reason for lower islet yield. Symptom duration in patients with the low BMI was longer than those with high BMI, although it was not statistically significant. Both patients with a past history of pancreas surgery were included in the low BMI group, which also might suggest that the low BMI group had advanced stage of chronic pancreatitis. Because patients with chronic pancreatitis had poor digestive enzymes, those patients might lose their body weight due to malnutrition. This malnutrition might be another reason for poor islet yield in the low BMI group. On the other hand, the high BMI group may have had more islets secondary to compensation for overweight/obesity, as is probably the case with high BMI allo-islet cadaveric donors.
Interestingly, the low BMI group had lower rates of small size of islets and higher rates of large size islets than the high BMI group (Table 5). Final production volume per pancreas was significantly smaller in the low BMI group (Table 3). It was demonstrated that semiob-struction of the pancreatic duct stimulated islet growth and destroyed exocrine tissue (39). We speculated that islets in the low BMI group suffered prolonged chronic pancreatitis with pancreatic ductal obstruction. This stimulated growth of islets and destroyed the pancreatic acinar cells. This phenomenon seems to be a compensatory mechanism to protect important insulin secretion ability. However, even with this compensation, the quality of islets cannot be maintained after prolonged chronic pancreatitis. Clinical results of significantly lower C-peptide levels in the low BMI group supported this scenario.
Final tissue volume was significantly higher in the high BMI group; therefore, the ratio of purification was higher in the high BMI group. Even with high ratio of purification procedure, final products of isolated islets had lower purity in high BMI. These findings would suggest that advanced chronic pancreatitis, which was associated with low BMI, destroyed exocrine tissues but still maintained islets. In other words, advanced pancreatitis associated with low BMI might have the advantage of increasing purity and decreasing tissue volume, although advanced pancreatitis eventually destroyed islets, which is definitely a disadvantage.
There are some limitations in this study: small number of cohort, retrospective design, and relatively short follow-up periods. In addition, there is a possibility that image scores on EUS and ERCP were confounding factors to the association among BMI, islet yields, and clinical outcomes in autologous islet cell transplantation. Further investigations are needed to determine the clinical impact of BMI on long-term graft function. In summary, higher BMI was associated with greater islet yields and better islet function. BMI was also associated with the score of clinical imaging studies such as ERCP and EUS, which suggests that patients with lower BMI might have a more advanced phase of chronic pancreatitis. Therefore, BMI seems to be one candidate to predict autologous islet transplantation success.
Footnotes
Acknowledgments
This work was supported in part by All Saints Health Foundation. The authors thank Ms. Melanie Corcoran for checking English grammar of the manuscript.
