Abstract
Cryoprotective agents (CPAs) such as dimethyl sulfoxide (DMSO), glycerol, ethylene glycol, and propylene glycol have been used for the cryopreservation of cells and tissues. DMSO is the most effective CPA but shows high cytotoxicity and can effect differentiation. ∈-Poly-l-lysine (PLL) derivatives show higher cryopreservation efficiency than the conventional CPAs. Culture medium solutions with 7.5 w/w% of PLL whose amino groups of more than 50 mol% were converted to carboxyl groups by succinic anhydride showed higher postthaw survival efficiency of L929 cells than those of current CPAs without the addition of any proteins. In addition, rat mesenchymal stem cells were cryopreserved more effectively than with DMSO and fully retained the potential for proliferation and differentiation. Furthermore, many kinds of cells could be cryopreserved with PLL having the appropriate ratio of COOH groups, regardless of the cell types, including adhesive and floating cells, human- and mouse-derived cells, primary cells, and established cell lines. The properties might be associated with the antifreeze protein properties. These results indicate that these polymeric extracellular CPAs may replace current CPAs and the high viability after thawing and nonnecessity of serum ensure that these CPAs may be used in various preservation fields.
Introduction
Cell transplantation is becoming a valuable tool in clinical medical care and many studies have focused on the development of effective methods of long-term preservation of living cells (8, 11, 21, 22, 33). The cryopreservation of cells is an important traditional approach of preservation of tissue for cell transplantation for various diseases with recent advances in tissue engineering (4, 15, 23, 24, 28). Since Polge reported that a glycerol solution could preserve cattle sperm at −79°C in 1949 (25), the rapid improvement of the science of cryobiology led to the discovery of the dimethyl sulfoxide (DMSO) as a cryoprotective agent (CPA) for other types of cells (18). Although many chemicals such as glycerol, ethylene glycol, propylene glycol (PG), and trehalose are known as CPAs, DMSO is the most effective CPA and is used in cell banks around the world. Many kinds of stem cells can be preserved with DMSO-based cryopreservation solution (9, 14, 15, 23, 24). These chemicals penetrate the cell membrane and function by replacing part of the water in the cell (28) and their optimal use requires designing optimal preservation protocols. Furthermore, current CPAs that penetrate the cell membrane are toxic themselves due to the high osmotic pressure at the effective concentrations. DMSO is also toxic and affects the differentiation of many types of cells (12, 31) and needs to be eliminated rapidly after thawing. In contrast, carbohydrate CPAs, such as trehalose and sucrose, cannot penetrate the cell membrane and have an exceptional ability to stabilize and preserve the membrane (3). In addition, some proteins and synthetic polymers have some degree of cryopreservative properties. However, CPAs that cannot penetrate the cell membrane have weak cryoprotective properties and are insufficient for single application. Although many have reported that cryopreservation of stem cells with DMSO, glycerol, or mixtures of CPAs, few studies focused on the development of novel CPAs.
We have revealed that the carboxyl group introduced by ∊-poly-l-lysine (COOH-PLL), which is classified as a polyampholyte, solutions has high cryoprotective properties (20). In this study, the cryoprotective properties of novel PLL derivative CPAs were compared to current CPAs for many types of cells including stem cells, and their mechanism of action is discussed based on the antifreeze protein (AFP)-like properties of these polyampholytes.
Materials and Methods
Poly-l-Lysine Modification
A 25% (w/w) PLL aqueous solution (Chisso Corp., Tokyo, Japan) and succinic anhydride (SA; Wako Pure Chem. Ind. Ltd., Osaka, Japan) in 0–100% mol ratios (SA/PLL amino groups) were mixed and reacted at 50°C for 1 h to convert amino groups into carboxyl groups (Fig. 1A). The amount of amino groups was determined by the 2,4,6-trinitrobenzenesulfonate (TNBS) method (10). Briefly, 0.3 ml of 250 μg/ml sample solution, 1 ml of 1.0 mg/ml TNBS solution, and 2 ml of 40 mg/ml sodium bicarbonate aqueous solution containing 10 mg/ml sodium dodecyl sulfate (pH 9.0) were mixed and incubated at 37°C for 2 h. After the mixture was cooled at 25°C, the absorbance was then measured at 335 nm.

COOH-PLLs synthesis and their cryopreservation properties. (A) Schematic illustration of the reaction of PLL succinylation. (B) Cell viability immediately after thawing of L929 cells cryopreserved with 7.5% (w/w) PLL with different ratios of introduced COOH. Data are expressed as the mean ± SD for five independent experiments.
Cell Culture
L929, MG63, HT1080, B16F1 (American Type Culture Collection, Manassas, VA, USA), Caco2, MC3T3, KB cells, and rat bone marrow mesenchymal stem cells (RMSCs; DS Pharma Biomedical Co., Ltd, Osaka, Japan) were cultured in Dulbecco's modified Eagle's medium (DMEM, Sigma Aldrich, St. Louis, MO, USA) and Jurkat E6–1 (DS Pharma), and Colon26 cells (Riken Bio Resource Center, Tsukuba, Japan) were cultured with RPMI-1640 medium (Sigma), and each was supplemented with 10% fetal bovine serum (FBS). Normal human coronary artery endothelial cells (HCAEC) and normal human umbilical vein endothelial cells (HUVEC) were purchased from Lonza Walkersville, Inc. (Walkersville, MD, USA) and they were cultured with the medium of EGM-2MV and EGM-MV (Lonza) with 10% FBS, respectively. Cell culture was carried out at 37°C under 5% CO2 in a humidified atmosphere. When the cells reached 80% confluence, they were removed by 0.25% (w/v) of trypsin containing 0.02% (w/v) of EDTA in PBS(-) and then were seeded on a new tissue culture plate for subculture. In all experiments, RMSCs, HUVEC, and HCAEC were used at the third or fourth passage.
Cryopreservation Protocol and Evaluation of Survival
The cryopreservation solutions were prepared as follows: DMSO, glycerol, PG, polyethylene glycol (PEG, MW 3000), bovine serum albumin (BSA), gelatin, trehalose, poly(acrylic acid; PAAc, MW 5000; Wako), and PLL derivatives were dissolved in DMEM without FBS at the concentration of 1–50% (w/w) and pH was adjusted to 7.4 using HCl or NaOH. Osmotic pressure was measured by an Osmometer (Osmometer 5520, Wescor, Inc. UT, USA) and adjusted to about 600 mOsm in the cryopreservation solution of PLL derivatives using sodium chloride solution. The cells were counted and resuspended in 1 ml of cryopreservation solution without FBS at a density of 1 × 106 cells/ml in 1.9 ml cryovials (Nalgene, Rochester, NY) and stored in a −80°C freezer overnight. It was then transferred into liquid nitrogen until thawing. Individual vials were thawed at 37°C in a water bath with gentle shaking, and the thawed cells were diluted in DMEM. After centrifugation, the supernatant was removed and the cells were resuspended in a 5 ml medium. The cells were plated in six-well culture plates at a cell density of 1 × 104/cm2 (n = 5). To determine cell survival, the medium was collected and attached cells were trypsinized. All the cells were counted using a hemocytometer with a trypan blue staining method at 0 (immediately postthawing) and 6-h thawing. The reported values are the ratios of living cells to total cell numbers. To evaluate cell proliferation after thawing, the cells were seeded onto 24-well culture plates at a cell density of 1.0 × 105/cm2 with 2 ml of DMEM (n = 3) and the cell number was counted up to 72 h after thawing.
Ice Recrystallization Inhibition
Four microliters of each sample solution containing 30% (w/w) of sucrose was placed on a slide, covered, loaded on a cooling stage (Linkam 10002L Cooling Stage, Japan High Tech., Fukuoka Japan), which was programmed to quench cool the slide to −40°C and then warm it to −9°C, with the temperature held for 30 min. Ice crystal images were then acquired using a mounted photomicroscope (Olympus BX51).
Induction of Differentiation of RMSCs
Unfrozen and cryopreserved RMSCs were seeded at a cell density of 1 × 104/cm2 per well in 12-well culture plates. After confluence, the RMSCs were induced to differentiate into osteoblasts and adipocytes for 14 days in osteogenic and adipogenic media, respectively (16). Osteogenic medium contained 0.1 μM dexamethasone, 10 mM β-glycerophosphate disodium salt, 0.07 mM l-ascorbic acid 2-phosphate magnesium salt n-hydrate (Sigma), 10% FBS, and 1% antibiotic/antimycotic in the DMEM. Adipogenic medium contained 0.5 mM isobutyl methylxantine, 0.1 mg/ml insulin, 0.2 mM indomethacin, 1 μM dexamethasone, 10% FBS, and 1% antibiotic/antimycotic in the DMEM. The culture medium was changed three times per week. Chondrogenic differentiation was performed in a pellet culture (19). Approximately 2.5 × 105 of unfrozen and cryopreserved RMSCs were centrifuged at 300 × g for 4 min in a conical tube to produce a mass pellet. The pellet was cultured at 37°C with CO2 for 14 days in 500 μl of chondrogenic differentiation medium containing 6.25 μg/ml insulin, 6.25 μg/ml transferrin, 6.25 μg/ml selenious acid, 1.25 mg/ml bovine serum albumin, 5.35 mg/ml linoleic acid, 0.07 mM l-ascorbic acid 2-phosphate magnesium salt n-hydrate, 0.1 μM dexamethasone, 10 ng/ml tumor growth factor-β1 (TGF-β1), 10% FBS, and 1% antibiotic/antimycotic in DMEM. The medium was replaced every 3 days. For all negative controls, the cells were maintained in DMEM containing 10% FBS.
Evaluation of Differentiation
Monolayer cultures treated with an osteogenic differentiation medium were stained with 1% Alizarin Red S for 15 min after fixation with 4% neutral buffered formalin for mineralized matrix deposition and the alkaline phosphatase activity was also examined by the modified Kind-King method (13) using a commercially available kit (ALP Kainos kit, Kainos Lab., Inc., Tokyo, Japan). Monolayer cultures treated with adipogenic differentiation medium were stained with oil Red O solution to visualize the formation of fat vacuoles. Briefly, the cells were fixed with 4% neutral buffered formalin and incubated for 20 min with 0.2% oil Red O isopropanol solution. In addition, glycerol-3-phosphate dehydrogenase (GPDH) activity was determined for the differentiation of adypocytes using a GPDH assay kit (GPDH assay kit, Primary Cell Co., Ltd., Hokkaido, Japan). Cell pellets for chondrogenic differentiation were fixed in 4% neutral buffered formalin for 1 h, embedded in paraffin, cut into 3–5-μm sections, and stained with collagen type II using antibody (anti-collagen type II antibody, sc-52658, Santa Cruz Biotechnology, Inc., CA, USA). In addition, the glycosaminoglycan (GAG) contents in the cell pellets were determined using a dimethylmethylene blue (DMMB) dye binding assay kit (Blyscan kit; Biocolor Ltd., Newtownabbey, Northern Ireland) according to the manufacturer's instructions.
Statistical Analysis
All data are expressed as the mean ± SD. Data among more than three groups were compared by the Tukey-Kramer test (one-tailed). Data between two groups were compared by Student's t-test. A value of p < 0.05 was considered to be statistically significant.
Results
Cryopreservation of L929 Cells
The carboxyl groups were introduced into PLL by treatment with SA (Fig. 1A), which reacts with amino groups. The ratio of carboxylation was well controlled by the reaction with SA. The ratio of carboxylation shown in parentheses [e.g., PLL (0.65)] indicates that 65% of the α-amino groups have been converted into carboxyl groups by SA addition. Figure 1B shows L929 cell viability immediately after thawing with various amounts of carboxyl groups introduced in PLL solution at 7.5%. This clearly demonstrates that COOH-PLLs with a ratio of carboxylation from 0.5 to 0.8 have high cryopreservation efficiency and the cells show >90% viability after thawing. The viability immediately after thawing of L929 cells cryopreserved with various CPA solutions is shown in Figure 2. The appropriate concentration of DMSO, glycerol, PG, and trehalose was 10% and concentrations greater than 20% have high osmotic pressure and can induce lethal stress in cells. Polymeric CPAs such as BSA, gelatin, PEG, and PAAc have some degree of cryoprotective ability but the cell viability was reduced in comparison to that of DMSO. In contrast, when PLL (0.65) was used for cryopreservation, concentrations of more than 5% resulted in retained viability and a concentration of 7% or more of PLL (0.65) resulted in a higher viability than with other CPAs.

Cell viability immediately after thawing of L929 cells cryopreserved with various concentrations of various CPAs. (A) DMSO, (B) glycerol, (C) propylene glycol, (D) trehalose, (E) bovine serum albumin, (F) gelatin, (G) polyethylene glycol, (H) poly(acrylic acid), (I) PLL (0.65). Data are expressed as the mean ± SD for five independent experiments.
Cryopreservation of Rat Mesenchymal Stem Cells
The viability of RMSCs cryopreserved with various carboxylated PLLs solutions at 7.5% at immediately and 6 h after thawing is shown in Figure 3A. The cell viability increased with the percentage of introduced carboxyl groups up to 65% and the viability at 6 h after thawing of cells cryopreserved with PLL (0.65) was significantly higher than that of 10% DMSO. Cell growth curves up to 72 h after thawing are shown in Figure 3B. The proliferation rate of RMSCs cryopreserved with PLL (0.65) and PLL (0.76) was significantly higher than that with 10% DMSO. The cell morphology at 24 h after thawing is shown in Figure 3C. RMSCs cryopreserved with PLL (0.65) attached and spread similar to those with DMSO, but most of the cells with PLL (0.20) could not attach and underwent cell death.

RMSCs cryopreservation with COOH-PLLs. (A) RMSCs were cryopreserved with 10% DMSO and 7.5% (w/w) PLL with different ratios of introduced COOH. Cell viability immediately (white bars), and 6 h (gray bars) after thawing at 37°C. Data are expressed as the mean± SD for five independent experiments. ∗∗p < 0.01 versus 10% DMSO. (B) Growth curves of RMSCs cryopreserved with 10% DMSO (open circles), 7.5% intact PLL (closed circles), 7.5% PLL (0.20; open triangles), 7.5% PLL (0.46; closed triangles), 7.5% PLL (0.50; open squares), 7.5% PLL (0.65; closed squares), and 7.5% PLL (0.76; open rhombus) for 72 h after thawing. Data are expressed as the mean ± SD for three independent experiments. ∗∗p < 0.01 versus 10% DMSO. (C) Micro-photographs of RMSCs cryopreserved with (left) 10% DMSO, (middle) 7.5% PLL (0.20), and (right) 7.5% PLL (0.65) at 24 h after thawing. Scale bar: 100 μm.
The results of the evaluation of the differentiation potency after cryopreservation with PLL (0.65) are shown in Figure 4. The histological evaluation revealed that RMSCs cryopreserved with 7.5% PLL (0.65) were well differentiated into osteoblasts (Fig. 4A), adipocytes (Fig. 4B), and chondrocytes (Fig. 4C). Furthermore, quantitative analyses confirmed that RMSCs cryopreserved with PLL (0.65) maintained their differentiation properties. The alkaline phosphatase activity was measured for osteoblastic differentiation (Fig. 4G); glycerol-3-phosphate dehydrogenase activity for adipose differentiation (Fig. 4H); and the amount of glycosaminoglycan for chondrogenic differentiation (Fig. 4I). These results showed that each type of differentiation depended specifically on each induction and that no inappropriate differentiation occurred in cryopreservation with PLL (0.65; Fig. 4D–F).

Differentiation of RMSCs after cryopreservation. (A–F) Histological evaluation of the differentiation of RMSCs cryopreserved with PLL (0.65). Scale bars: 100 μm. (A) Alizarin red staining for osteoblast differentiation after induction culture and (D) normal culture, (B) oil red O staining for adipocyte differentiation after induction culture and (E) normal culture, and (C) collagen type II immunostaining for chondrocyte differentiation after induction culture and (F) normal culture. (G–I) Quantitative analyses of differentiation of RMSCs. (G) Alkaline phosphatase activities, (H) GPDH activities, and (I) amounts of GAG in induced and uninduced cultures of unfrozen and cryopreserved RMSCs. Data are expressed as the mean ± SD for five independent experiments. ∗∗p < 0.01, ∗∗∗p < 0.001.
Cryopreservation of other Mammalian Cells
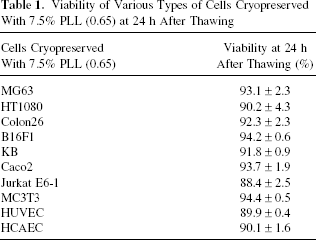
Various cells derived from mice and human were cryopreserved with 7.5% solution of PLL (0.65) and the viability at 24 h after thawing is summarized in Table 1. Almost 90% viability was obtained for all kinds of cells.
Viability of Various Types of Cells Cryopreserved With 7.5% PLL (0.65) at 24 h After Thawing
Ice Recrystallization Inhibition Activity
The results of the ice recrystallization inhibition assay of various polymeric CPAs solutions at the most effective concentration for cryopreservation are shown in Figure 5. The growth of ice crystals in 30% (w/w) sucrose solution was not inhibited by BSA, PEG, PAAc, and intact PLL; in contrast, it was highly inhibited by PLL (0.65).

Ice recrystallization inhibition activity of polymeric CPAs. Inhibition of ice recrystallization of (A) 30% sucrose control and in the presence of (B) 30% bovine serum albumin, (C) 30% PEG, (D) 15% PAAc, (E) 7.5% intact PLL, and (F) 7.5% PLL (0.65) with 30% (w/w) sucrose solution. Scale bar: 100 μm.
Discussion
The focus of this study was to develop an alternative to the conventional DSMO- or glycerol-based cryopreservation solutions for living cell storage. Polyampholytes such as COOH-PLLs were shown to be more effective for cryopreservation of cells than that of any other CPAs and allows the pursuit of a new cell transplantation regime that can allow cell preservation with higher viability without the use of any proteins and toxic organic compounds.
Cryopreservation is used routinely for the maintenance of cell culture, reproductive medicine, and cell transplantation and the enhancement of the efficiency has been studied from the aspect of the freezing and thawing temperature conditions using programmable freezers, the optimization of the concentration of CPAs, and the development of novel highly effective CPAs. The major problems with the current cryopreservation methods are toxicity of CPAs and low survival rate after thawing of some types of cells (7, 32). Small CPAs such as DMSO, glycerol, and PG penetrate the cell membrane to protect from freezing; therefore, they show cytotoxicity (5). In addition, when the concentration was raised to increase cryoprotective efficiency, increased osmotic pressure eventually causes cell death (Fig. 2A–C). Furthermore, these CPAs require the addition of FBS or albumin to reduce the toxicity. However, these animal derived proteins should be avoided because of the risk of infection in clinical use. Nontoxic CPAs such as trehalose and polymers clearly have lower cell protective properties than DMSO (Fig. 2). This study focused on the development of low toxic and non-animal-derived polymeric CPAs. PLL is an l-lysine homopolymer biosynthesized by Streptomyces species (30). It is used as a food additive due to its antimicrobial activities ascribed to the cationic charge density of its side chain α-amino groups and its specificity to microorganisms. The COOH-PLLs have CPA properties and the appropriate ratio of carboxyl groups to amino groups of COOH-PLL is crucial for cell cryopreservation (Fig. 1B). COOH-PLLs showed relatively lower osmotic pressure than the lethal value for cells even at a concentration of 20% because of their high molecular weight. The current results clearly demonstrated that COOH-PLL, which has cryoprotectant properties, inhibited ice recrystallization better than other polymeric CPAs (Fig. 5). Larger ice crystals cause severe dehydration and physical damage within freezing cells. Ice recrystallization inhibition may represent the physiological function of AFPs in freezing-tolerant organisms (6). Some AFP-like proteins have membrane protective properties (1, 29). The current results suggest that COOH-PLL could work in membrane protection in a manner similar to that of AFPs.
Mesenchymal stem cells have a multipotent capability to differentiate into various functional cell types of mesodermal tissues (2, 26). In this study, RMSCs were cryopreserved with a simple method of cryopreservation by slow cooling with COOH-PLL as a CPA, without any other CPAs and animal-derived proteins and the results showed that these novel CPAs did not alter the phenotype characteristics of RMSCs, and preserved their viability and proliferative ability after thawing (Fig. 3) and differentiation properties (Fig. 4). Bone marrow-and adipose-derived stem cells that are cryopreserved using DMSO retain their viability and differentiation properties (9, 14, 15, 17, 23, 24). However, most of these studies used FBS. FBS might play an important role as a buffer of osmotic pressure and as a cell membrane protector, thus reducing the risks of damage by ice recrystallization during freezing and thawing (27). The COOH-PLL CPAs in the current study did not require any proteins for cryopreservation and showed the highest viability after thawing among the current CPA solutions (Fig. 2). It was likely that the AFP properties of COOH-PLLs might protect cell membranes during freezing and thawing.
The next stage of this study was to investigate whether the most effective COOH-PLL, PLL (0.65), could be used for the cryopreservation of various cell types. Table 1 clearly shows that PLL (0.65) solution demonstrates efficient cryopreservation in multiple cell types from humans and mice, thus including adhesive cells and floating cells (Jurkat), primary cells (HCAEC and HUVEC), and established cell lines. This general versatility suggests that the novel CPAs might be used for not only stem cell preservation for cell transplantation but also for cell banking of various cell lines and for preserving those cells not well cryopreserved by using current CPAs (7, 32).
In conclusion, novel highly effective CPAs based on specific polymeric compounds were developed. Cell cryopreservation processes using these polyampholytes can replace the conventional system of using DMSO and glycerol solution supplemented serum. These CPAs provide of high viability after thawing and do not require serum, and therefore these cryopreserving solutions are expected to find widespread use in cell preservation for cell transplantation.
Footnotes
Acknowledgments
We thank Dr. N. Nakajima, Kyoto University, for his critical comments and Dr. K. Nishida and Dr. T. Kanaya, Kyoto University, for assistance with data analyses of the ice recrystallization inhibition assay. This study was supported in part by a Grant-in-Aid (21700488) for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
