Abstract
Pluripotent stem cells, including human embryonic stem cells and induced pluripotent stem cells, have generated much excitement about their prospects for use in cell transplantation therapies. This is largely attributable to their virtually unlimited growth potential, their ability to be precisely genetically altered in culture, and their utility for forming differentiated cell populations with potential clinical applications. Lysosomal storage diseases such as Sanfilippo syndrome (MPS-IIIA) represent ideal candidate diseases for the evaluation of cell therapies in the central nervous system (CNS). These diseases exhibit widespread pathology yet result from a single gene deficiency, in the case of Sanfilippo syndrome the lysosomal enzyme sulfamidase. The aim of this study was to investigate mouse embryonic stem (ES) cell-derived glial precursor cells as a vehicle for sulfamidase delivery in the MPS-IIIA mouse brain. In this study we have created a mouse ES cell line genetically modified to stably express and secrete high levels of human sulfamidase and a protocol for the in vitro derivation of large numbers glial precursors from ES cells. Differentiation of sulfamidase-expressing ES cells resulted in cell populations with sustained secretion of high levels of sulfamidase, comprised primarily of glial precursor cells with minor contaminants of other neural cell phenotypes but not residual pluripotent cells. CNS implantation studies demonstrated that ES cell-derived glial precursor cells formed using this differentiation method were able to engraft and survive for at least 12 weeks following implantation. The percentage of engraftment was quantified in different regions of the brain in 2-, 4-, and 8-week-old normal and MPS-IIIA mice. No teratomas were observed in any of the cell-transplanted animals. The results of this study support the further investigation of sulfamidase-expressing glial precursor cells as a vehicle for delivery of deficient enzyme into the CNS of MPS-IIIA mice.
Keywords
Introduction
Embryonic stem (ES) cells are a pluripotent cell type derived from the inner cell mass of a preimplantation embryo (6,22,38). Although these cells represent a transient phenotype in the developing embryo, in culture they can be maintained in a pluripotent state and induced to proliferate. Previous studies have shown the ability of pluripotent cells to generate various neuronal and glial cell types for a range of potential applications, including transplantation of neural cells in an ischemic stroke model (40), neuronal cell replacement in Parkinson's disease (36,39), or transplantation of glial cells to play a support role in demyelinating diseases (3,8). The potential of pluripotent cells to generate neuronal and glial cells, coupled with their amenability to precise genetic modification in their undifferentiated form, has generated much excitement about the prospects for their utility as a vehicle for therapeutic gene expression in the brain (8).
Lysosomal storage disorders represent an ideal candidate for cell therapies, as most are caused by a defect in a single lysosomal enzyme. Moreover, many of these enzymes are mannose-6-phosphorylated, allowing efficient cellular uptake from the extracellular environment and targeted distribution to the lysosome. Cell transplantation studies have demonstrated proof of principle for treatment of neuropathology in experimental models of lysosomal storage diseases (16), such as MPS-VII (24,34), Tay-Sach's disease (7), and Sandhoff disease (20). Sandoff disease is a monogenic neurodegenerative lysosomal storage disorder resulting from a deficiency of the enzyme β-hexosaminidase, with consequent accumulation of the ganglioside GM2 within the lysosomes of cells of the central nervous system (CNS). A previous study by Lee and colleagues (20) involved implantation of mouse neural stem cells into the CNS in an experimental mouse model of Sandhoff disease. This study concluded that of the donor-derived engrafted cells, those that had the most effect in treatment of neuropathology were probably not the cells that had differentiated into neurons but the cells that had differentiated to glial phenotypes. The authors speculated that this may be due to their “chaperone-like” properties, their inherent synthesis and secretion of therapeutic proteins (both the deficient lysosomal enzyme and other trophic factors), and/or their ability to participate in the detoxification of the microenvironment (20). Additionally, the migratory nature of glial cells make them an ideal candidate for therapeutic gene expression (3,8,27,33).
Mucopolysaccharidosis type IIIA (MPS-IIIA, also known as Sanfilippo syndrome) is a lysosomal storage disorder caused by a genetic defect in the mannose-6-phosphorylated lysosomal enzyme sulfamidase (SGSH; EC 3.10.1.1). This leads to primary storage of its substrate, heparan sulphate, in the lysosomes of affected individuals, which in turn initiates a cascade of secondary lysosomal storage and neuroinflammatory pathology, resulting in CNS dysfunction. We have a congenic C57BL/6 mouse model of MPS-IIIA, which is characterized by reduced sulfamidase activity and progressive heparan sulfate accumulation in the CNS, along with behavioral changes including motor, cognitive, and sensory deficits (5).
Previous studies in MPS-IIIA mice have demonstrated that repeated delivery of recombinant human sulfamidase to the CNS improves the neuropathological and clinical manifestations of this condition (9,11–13). Direct and repeated injection of sulfamidase into the cerebrospinal fluid, while highly effective, is in its present form not optimal due to the invasive nature of this procedure and the potential associated complications. Accordingly, alternative methods to allow CNS delivery of lysosomal enzymes on a chronic basis are currently being explored. One such therapeutic approach is stem cell therapy.
Cell therapies using pluripotent cells require an appropriate source of cells that are amenable to expansion to therapeutically useful numbers coupled with reproducible protocols for differentiation of cells to a therapeutically useful phenotype. Previous studies have demonstrated the utility of mouse ES cells for construction of lysosomal enzyme-expressing cell lines (19,31). The implantation of early differentiated derivatives of these lines, however, has not resulted in suitable outcomes for therapy (32).
We have developed a more extensive differentiation protocol for the reproducible generation of large numbers of glial precursor cells from mouse ES cells, and used this protocol to generate sulfamidase-expressing glial precursor cells. Implantation studies permitted the analysis of survival, engraftment, and distribution of implanted glial precursor cells, and ongoing expression and delivery of sulfamidase to the MPS-IIIA mouse brain.
Materials and Methods
Construction of Sulfamidase-Expressing C57BL/6 ES Cell Lines
C57BL/6 male mouse ES cells from the line Bruce-4 (17) were cultured in feeder-free conditions on tissue culture grade plasticware (Falcon) pretreated with 0.2% gelatin/PBS (30). Cells were grown in Dulbecco's modified Eagles medium (DMEM; Gibco) supplemented with 15% fetal calf serum (FCS), 1 mM L-glutamine, 0.1 mM β-mercaptoethanol (β-ME), and 1000 units of leukemia inhibitory factor (LIF) at 37°C under 10% CO2 in a humidified incubator (30). Embryonic stem cells were transfected with the vector pCAGrhNS-Ipuro (19). Following selection, clones were isolated and screened for enzyme activity in cell lysates and conditioned media, using a radiolabeled tetrasaccharide assay for sulfamidase (14). High expressing clones were karyotyped, differentiated using the protocol described below, and again screened for sulfamidase activity at various stages of differentiation, to ensure similar in vitro culture characteristics to untransfected cells, along with maintenance of sulfamidase expression.
Differentiation of Glial Precursor Cells From C57BL/6 Mouse ES Cells
The following protocol was developed as an extension of previously published protocols (1,3,29).

Characterization of ES Cell-Derived Glial Cells
Fixed cells were analyzed using immunofluoresence techniques. Cells were washed in PBS and blocked by incubation in 10% normal donkey serum (±0.2% Triton X-100; Sigma), in PBS for 45 min at room temperature. Triton X-100 was omitted from all protocols involving A2B5 or PSA-NCAM immunofluorescence. Cells were incubated in primary antibodies overnight at room temperature diluted in the same solution as for blocking, at the following concentrations: mouse anti-Oct4 1:100 (Octamer-4, a marker for pluripotent cells; Santa Cruz Biotechnology), mouse anti-PSA-NCAM 1:400 (polysialated neural cell adhesion molecule, a marker for neuronal precursors; T. Seki), mouse anti-A2B5 1:400 (a marker for glial precursor cells; Chemicon), rabbit anti-GFAP 1:400 (glial fibrillary acidic protein, a marker for astrocytes; DAKO), mouse anti-RIP 1:3 [2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase), a marker for oligodendrocytes; Developmental Studies Hybridoma Bank], rabbit anti-MAP2 1:400 (microtubule-associated protein 2, a marker for neurons; Chemicon). Cells were washed three times in PBS and incubated with the secondary antibody for a minimum of 4 h (donkey anti-mouse Cy3, 1:400 or donkey anti-rabbit Cy3, 1:300, both from Jackson ImmunoResearch). Following another three washes, cells were mounted in Vectashield (containing 4′,6-diamidino-2-phenylindole; DAPI), and visualized with an Olympus AX70 fluorescent microscope. Proportions of different phenotypes were determined by capturing random fields at low magnification, and counting the number of immunopositive and negative cells per field using Adobe Photoshop. Analyses were performed on biological replicates, to confirm reproducibility of the protocol.
In Vitro Treatment of MPS-IIIA Human Skin Fibroblasts
Human MPS-IIIA (SF4636) and unaffected (SF4976) patient skin fibroblasts were obtained from skin biopsies donated to the Children, Youth and Women's Health Service (CYWHS). Fibroblasts were maintained in Basal Medium Eagle (BME; Gibco) supplemented with 10% heat-inactivated FCS and 1% P/S, at 37°C under 5% CO2 in a humidified incubator (19). Glial precursor cells were generated as described above, from both untransfected ES cells and sulfamidase-expressing NS21 cells. Media added to the differentiating cells on day 21 was collected 72 h later, concentrated in a stirred cell (Millipore) and then passed through a 0.22-μm filter. The media samples were supplemented with 10% heat-inactivated FCS and sulfamidase activity was determined (14). Conditioned media from NS21 glial precursor cells (containing 2000 pmol sulfamidase), conditioned media from untransfected glial precursor cells cells (containing 70 pmol sulfamidase), or unconditioned BME was then transferred onto previously established human MPS-IIIA or unaffected skin fibroblast cultures. Forty-eight hours later, fibroblast lysates were prepared and cellular sulfamidase activity was measured, along with the relative level of a heparan sulfate-derived disaccharide (35).
Implantation of ES Cell-Derived Glia Into the Brains of Newborn Mice
Mice were genotyped using established methods (2) and were bred, housed, and maintained in the CYWHS Animal House, with breeding and experimental procedures undertaken with the approval of the CYWHS Animal Ethics Committee (Adelaide, South Australia), with regard to the guidelines of the National Health and Medical Research Council of Australia on the use and care of experimental animals. Newborn C57BL/6J mice were cryoanesthetized and received bilateral lateral ventricle injections of a suspension of 5 × 104 cells (day 24 glial precursors) in 1 μl of DMEM (per hemisphere). Injections were carried out on cryoanesthetized pups using sterile 27-gauge dental needles attached to tubing connected to a 25-μl Hamilton syringe fitted on an injection pump (World Precision Instruments, model SP200iZ; Florida, USA). The dental needles were mounted on the arm of a stereotaxic frame (David Kopf Instruments, model 900, California, USA) and were slowly lowered into the lateral ventricles. The skull was transilluminated using a cold light source (Microlight 150). Pups were warmed after the injection under a warm lamp before being returned to the dam. Animals were sacrificed at 1 week (n = 14 cell implanted, n = 19 control), 2 weeks (n = 13 cell implanted, n = 17 control), 4 weeks (n = 14 cell implanted, n = 16 control), 8 weeks (n = 14 cell implanted, n = 16 control), and 12 weeks (n = 14 cell implanted, n = 17 control) postimplantation by CO2 asphyxi-ation and the brain was fixed by transcardial perfusion of PBS, followed by 4% paraformaldehyde in PBS. Female mouse brains were embedded in paraffin, 5-μm sagittal sections were cut, deparaffinized, and Y chromosome in situ hybridization was performed (10). GFAP immunohistochemistry was subsequently undertaken. Blinded analyses were carried out on cell-implanted and control female brains. A male mouse brain provided a positive control. Imaging was undertaken in all color channels, to eliminate the possibility of false positives attributable to autofluorescent artifact.
Real-Time PCR for Quantification of Cell Engraftment
An additional cohort of newborn mice (n = 11) received 2 × 105 glial precursor cells in 2 μl DMEM, injected into the lateral ventricle, unilaterally. At 2, 4, and 8 weeks after injection mice were euthanized by CO2 and the brain was separated into left and right olfactory bulb, left and right cerebral hemisphere, and midbrain and cerebellum. Homogenized brain samples were treated with RNase A (20 μg/ml) and then digested overnight in proteinase K (100 μg/ml). Genomic DNA was prepared by phenol/chloroform (1:1) extraction.
PCR reactions were performed in a volume of 50 μl and contained 25 μl of Universal PCR Master Mix (Applied Biosystems), 900 nM forward and reverse primers, 250 nM TaqMan probe, and 500 ng of target template. Reactions were incubated at 50°C for 2 min, 95°C for 10 min, and then amplified for 40 cycles. Each cycle was comprised of an incubation step at 95°C for 15 s followed by 60°C for 1 min. Standard curves were generated by serially diluting genomic DNA prepared from NS21 cells into DNA prepared from murine brain. The sequence of the PCR primers and probe used for detection of the human sulfamidase insert were as follows: forward PCR primer, 5′-ccacaatgagctgtgaccat-3′; reverse PCR primer, 5′-gacacgtgtgggatgtgtct-3′; TaqMan probe, 5′-FAM-cctgtgcacacatcccaggca-TAMRA-3′.
Reverse Transcriptase PCR Quantification of Sulfamidase Expression
Reverse transcriptase PCR was carried out to determine whether there was any change in the level of human sulfamidase mRNA expression during the process of differentiation of NS21 ES cells into glial precursors. Cells were harvested on day 0 and day 24 of the differentiation protocol and RNA was extracted using Trizol (Invitrogen). RNA (2 μg) was used in reverse transcription. The PCR mixture was: 1x PCR Master's mix with 1.5 mM MgCl2, 200 μM dNTP mix, 200 nM forward primer, 200 nM reverse primer, 2 U Taq DNA polymerase, 2 μl cDNA from first-strand reaction. The PCR cycles included: 96°C for 2 min, followed by 35 cycles of 94°C for 30 s, 55°C for 30 s, and 72°C for 30 s. The PCR was finished with a 72°C incubation for 5 min. The forward primer sequence for the human sulfamidase mRNA was: 5′-ACCAAGGAGTTGGACTGGTG-3′. The reverse primer sequence for the human sulfamidase mRNA was: 5′-CCAGTGAGGTGGATGGTCTT-3′. The forward primer sequence for the β-actin mRNA was: 5′-GGTCATCACTATTGGCAACG-3′. The reverse primer sequence for the β-actin mRNA was: 5′-ACGGATGT CAACGTCACACT-3′.
Results
Characterization of Sulfamidase-Expressing NS21 ES Cells
C57BL/6 ES cells were transfected with the vector pCAGrhNSIpuro (19). Antibiotic-resistant clones were propagated and characterized to identify stably transfected clones. When the level of sulfamidase activity from each of the transfected ES cell clones was examined, a range of activity levels was observed from 7.4-fold to 41.1-fold increased levels in cell lysates and from 4.5fold to 16.4-fold increased levels in conditioned medium (Fig. 1). The highest expressing clones were differentiated to ensure in vitro culture behavior and differentiation equivalent to untransfected cells. Clones exhibiting normal in vitro characteristics were then karyotyped. Clone NS21 was selected for further characterization; although this karyotypically normal clone expressed only moderately high levels of sulfamidase (11.9-fold untransfected levels in cell lysate and 6.1-fold untransfected levels in conditioned media), all other higher expressing clones examined were found to be karyotypically abnormal and/or they responded abnormally during in vitro differentiation (e.g., cells did not form embryoid bodies in a similar fashion to untransfected cells or failed to produce outgrowths of mature neural cells). Cultures derived from untransfected cells (Fig. 2A) were found to differentiate in a similar fashion to those derived from NS21 cells (Fig. 2B).
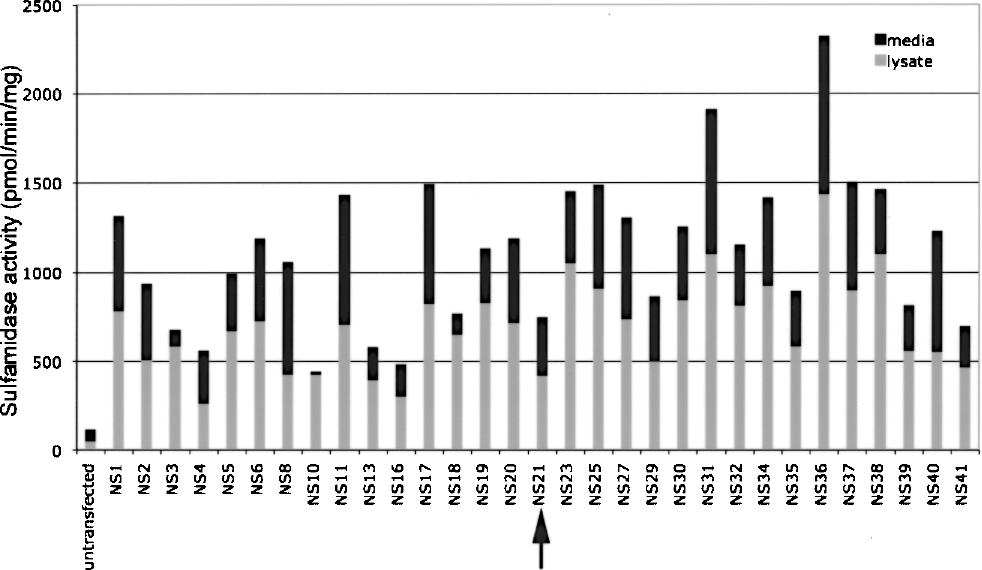
Sulfamidase activity in conditioned media and cell lysates from C57BL/6 mouse ES cell clones transfected with human sulfamidase cDNA. Activity is expressed as pmol/min/mg of total cell protein. Arrow indicates the highest expressing karyotypically normal clone (NS21) that was selected for further studies.

Phase-contrast microscopy images of untransfected (A) and sulfamidase-expressing (B) mouse ES cells (clone NS21) at various stages of the differentiation protocol. With the exception of day 5, which depicts embryoid bodies in suspension culture, all other images depict adherent cultures. Scale bars: 100 μm.
Human sulfamidase mRNA expression in NS21 cells was sustained during the differentiation protocol from day 0 to day 24 (Fig. 3A). A minor band of a similar size to the PCR product from human sulfamidase mRNA was observed in untransfected cells on days 0 and 24. NS21 cells were analyzed on days 0, 5, 12, and 24 of differentiation and maintained their high level of sulfamidase secretion, with 22-fold and 47.8-fold untransfected levels of sulfamidase activity recorded in the media and lysates on day 24, respectively (Fig. 3B, C).

(A) Reverse-transcriptase PCR analysis of human sulfamidase expression in transfected (clone NS21) and untransfected cells, at ES cell stage (day 0) and glial precursor cell stage (day 24 of the differentiation protocol). β-Actin expression in the same cell populations is shown for comparison. Sulfamidase activity in conditioned media (B) and cell lysate (C) expressed by untransfected C57BL/6 ES cells and sulfamidase-overexpressing clone NS21, at various stages of the differentiation protocol. Activity is expressed as pmol/min/mg total cell protein.
In Vitro Treatment of MPS-IIIA Human Skin Fibroblasts
MPS-IIIA and normal human skin fibroblasts were cultured in the presence of media conditioned by untransfected or NS21-derived glial precursors, or BME (control media). NS21-conditioned media (containing 2000 pmol of sulfamidase activity) increased the sulfamidase activity measured in MPS-IIIA skin fibroblasts to more than half the level observed in normal human skin fibroblasts (61.1% of normal levels) (Fig. 4A) and reduced the storage of a heparan sulfate-derived oligosaccharide (HNS-UA) in these cells by 78% (Fig. 4B). Culture of MPS-IIIA skin fibroblasts in media conditioned by untransfected glial precursors (containing 70 pmol of sulfamidase activity) increased the intracellular sulfamidase activity to a lesser degree (18.6% of normal) and resulted in a 47% decrease in HNS-UA levels in the MPS-IIIA cells.

Sulfamidase activity (A) and relative level of the heparan sulphate-derived disaccharide HNS-UA (B) in MPS-IIIA skin fibroblast lysates. The cells were cultured in the presence of media conditioned by either untransfected C57BL/6 ES cells or sulfamidase-overexpressing (clone NS21) ES cells or unconditioned BME medium. Normal: normal human skin fibroblasts; MPS control: fibroblasts from an MPS-IIIA patient grown in unconditioned BME medium; MPS untransfected: MPS-IIIA skin fibroblasts grown in media conditioned by untransfected C57BL/6 glial precursor cells; MPS NS21: MPS-IIIA skin fibroblasts grown in media conditioned by NS21-derived glial precursor cells. Duplicate cultures were performed and each data point is plotted (shown as an individual dot), with the exception of one MPS control replicate for which mass spectrometry data were not available. Lines indicate mean values for each experimental group.
Phenotypic Characterization and Implantation of Sulfamidase-Expressing Glial Precursor Cells Into the Brains of Newborn Mice
Prior to use of these cells in transplantation studies, we characterized the cell types present at day 24 of the differentiation protocol to ensure no pluripotent cells remained. No significant differences were observed in the differentiation potential of untransfected and NS21 cell lines or the phenotypic ratios of cells formed during differentiation. The vast majority of cells generated were glial cells, either A2B5-positive glial precursor cells (67.5 ± 4.5% and 70.7 ± 6.6% for untransfected and NS21 cell lines, respectively) (Fig. 5A) or GFAP-positive astrocytes (37.3 ± 2.7% and 35.3 ± 8.5%) (Fig. 5B). There were 28.7 ± 5.4% and 30.6 ± 13.9% that expressed PSA-NCAM, a neuronal precursor marker (Fig. 5C), 25.8 ± 8.7% and 24.2 ± 11.0% that expressed MAP2, a neuronal marker (Fig. 5D), and 2.8 ± 0.4% and 3.3 ± 3.7% that expressed RIP, an oligodendrocyte marker (Fig. 5E). Only 0.8 ± 1.0% and 0.4 ± 0.2% of the untransfected and NS21 cells examined were observed to have a positive signal following Oct4 immunostaining, a marker of pluripotency (Fig. 5F). The Oct4 staining pattern was far less pronounced in these cells than that seen in undifferentiated ES cells, which is consistent with previous observations that Oct4 expression continues at a low level after differentiation into neural lineages (29).

Fluorescent images of various phenotypic markers expressed by day 24 NS21 cells. Differentiated cell populations consisted of (in order of prevalence) glial precursors (A2B5; A), as well as mature astrocytes (GFAP; B), and express markers for neuronal precursors (PSA-NCAM; C), neurons (MAP2; D), and oligodendrocytes (RIP; E). Differentiated cell populations lost pluripotency, as demonstrated by a lack of Oct4 expression (F). Inset graphs show the percentage of cells that stained positive for each phenotypic marker at day 24 of the differentiation protocol, either from untransfected ES cells (blue bars) or sulfamidase-overexpressing NS21 ES cells (yellow bars) (±1 SD). (G–J) Implanted sulfamidase-expressing glial precursor cells detected in the brains of female mice using Y chromosome in situ hybridization, following bilateral implantation of 5 × 104 cells per hemisphere, into the lateral ventricles of newborn (day 0–1) mice. Implanted cells detected at 1 week in the cerebral lateral ventricle (G), 2 weeks in the choroid plexus (H; indicated by arrows), 12 weeks in the olfactory bulb (I), and at 12 weeks below the ventricular wall (J).
Subsequently, newborn mice (day 0 or 1) received bilateral injections of 5 × 104 NS21 cells or vehicle only (DMEM) into the lateral ventricles. Cohorts of mice were sacrificed at 1, 2, 4, 8, and 12 weeks postimplantation and no adverse events were observed at any time during the experimental period. At postmortem, there were no macroscopic signs of teratoma formation in any of the brains analyzed, with subsequent histological assessment confirming this finding. Y chromosome in situ hybridization analysis of female brains was undertaken, revealing transplanted syngeneic cells to be most frequently engrafted in the walls of the lateral ventricle or associated with the choroid plexus. Transplanted cells were also found in more distal areas such as the corpus callosum, rostral migratory stream, and the anterior cortex at 4 weeks, the thalamus at 8 weeks, and the olfactory bulb and deeper within the ventricular wall at 12 weeks postimplantation (Fig. 5G–J).
In order to quantify the level of engraftment and extent of distribution of implanted cells, an additional cohort of newborn mice received a unilateral lateral ventricular injection of 2 × 105 NS21 glial precursor cells. These mice were sacrificed at 2, 4, or 8 weeks postimplantation and brain samples were prepared for real-time PCR analysis. The slope of standard curve was 3.3, indicating that the efficiency of PCR reaction was 100% and the PCR assay was linear over the 0.001% to 100% range of template, in 500 ng of recipient background DNA. Thus, values below 0.001% were regarded as undetectable in this study. The percentage of NS21 genomic DNA in total DNA was converted to a percentage of donor cells (NS21 cells) in total cells, based on the assumption that each NS21 cell and host brain cell contain the same amount of genomic DNA.
The level of cell engraftment was higher in the (left side) injection hemisphere compared to the contralateral hemisphere and other parts of the brain examined (Fig. 6). Some injected cells were found in brain regions distal to the injection site. A maximum of 0.051% of total brain cells in the left hemisphere of a 4-week-old normal mouse were found to contain the human sulfamidase sequence. In an 8-week-old MPS-IIIA mouse brain, the exogenous cells represented 0.011% of total number of cells in the left hemisphere. In the injection hemisphere in normal mice, the engraftment ranged from 0.004% to 0.051%, whereas in MPS-IIIA mice engraftment ranged from 0.005% to 0.015%.
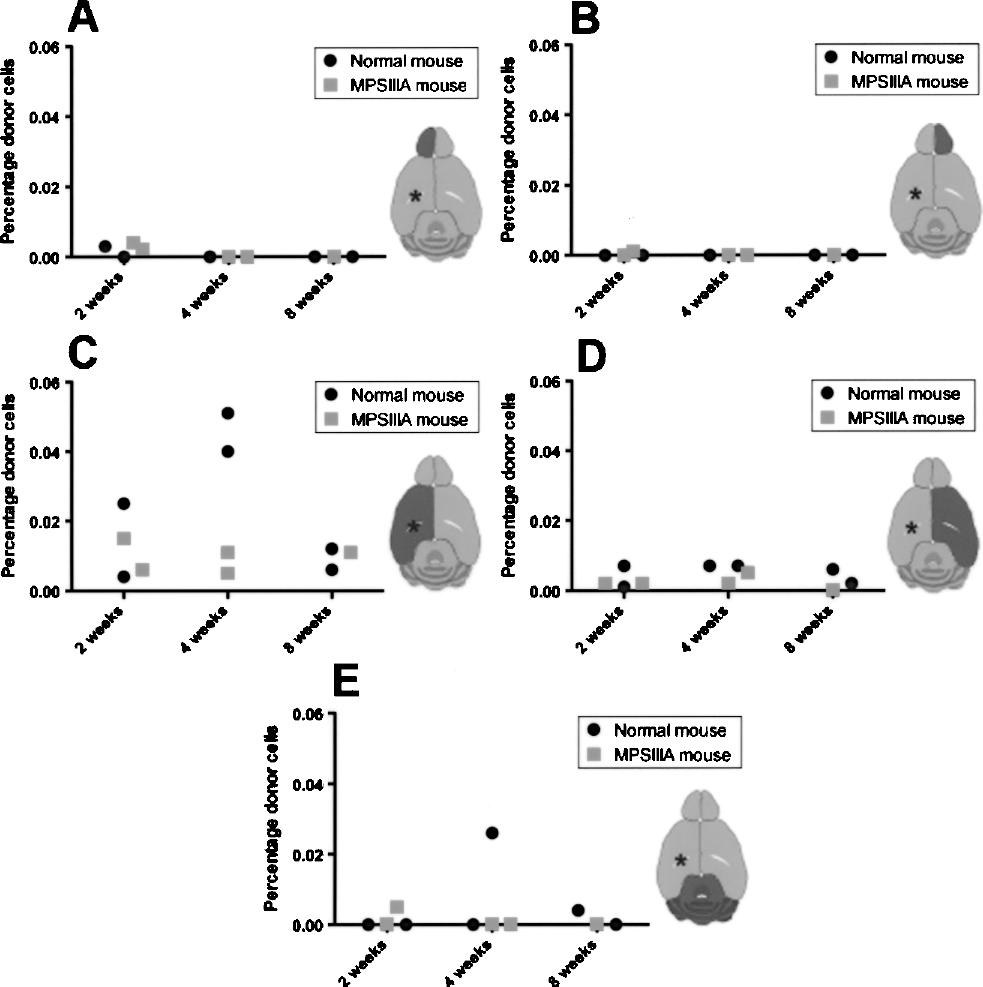
Quantification of implanted glial precursor cells in unaffected and MPS-IIIA brains following implantation of 2 × 105 NS21 glial precursor cells into the lateral ventricle of newborn mice. The percentage of donor cells (as a percentage of total brain cells) in the left olfactory bulb (A), right olfactory bulb (B), left cerebral hemisphere (C), right cerebral hemisphere (D), and midbrain/hindbrain/cerebellum (E) were quantified at 2, 4, and 8 weeks postimplantation by real-time PCR analysis of sulfamidase cDNA insert.
Discussion
In this study we have created and characterized C57BL/6 ES cell lines genetically modified to express and secrete high levels of human sulfamidase. To enable these cells to be effectively used as a vector to deliver sulfamidase activity to the brain, we have also developed a coculture-free differentiation protocol for deriving cell populations significantly enriched in sulfamidase-expressing glial precursor cells from these ES cell lines. This protocol allows initial neural induction and rapid expansion of cell populations as embryoid bodies, followed by selective expansion of precursor cells in an adherent culture, and further enrichment of these precursor cells using a collagenase-based passage protocol to maximize cell viability and remove remaining clumps of embryoid bodies. Phenotypic characterization of cell populations generated using this protocol showed the majority of cells were glial precursor cells, which maintain sulfamidase expression and secretion following differentiation, and additionally lack contaminating pluripotent cells. When applied to MPS-IIIA patient skin fibroblasts in vitro, sulfamidase secreted by these glial precursor cells was able to be taken up by MPS-IIIA fibroblasts and reduce the storage of a heparan sulfate-derived oligosaccharide. These cell lines and differentiation protocol have allowed the derivation of large numbers of sulfamidase-overexpressing glial precursor cells, enabling preliminary evaluation of a cell therapy approach for treatment of CNS pathology in MPS-IIIA.
The NS21 cell line was karyotypically normal when analyzed in vitro. However, many of the sulfamidase-overexpressing clonal cell lines, produced after transfection and selection, either behaved abnormally in culture or were found to be karyotypically abnormal on further analysis. This finding is consistent with previous reports demonstrating that Bruce-4 C57BL/6 ES cell line-derived clones display a higher percentage of karyotypic abnormalities than ES cell lines generated from other mouse strains (15). Bruce-4 C57BL/6 cells were chosen for this study as they are syngeneic with the Sanfilippo mouse model (5) and will simplify the analysis of therapeutic action in the future. A similar level of abnormality would not be expected to occur during manipulation and clonal expansion of other ES cell lines; however, this experience does underline the importance of screening transgenic lines for culture and differentiation characteristics prior to undertaking in vivo studies.
Human sulfamidase expression was sustained during the differentiation of NS21 ES (day 0) cells into glial precursors (day 24), demonstrated by analysis of mRNA expression and sulfamidase activity in cell lysates and conditioned media. The band detected in untransfected C57BL/6 ES cells when using human sulfamidase primers is likely to arise from inefficient amplification of the mouse sulfamidase mRNA, which shares homology with the human sequence. The expression of transgenes in ES cells has frequently been reported to be unstable (21), possibly due to gene silencing during growth and differentiation or as a result of variable promoter activity in different differentiated cells (4,31). Transfection methods have also been shown to influence gene expression in stem cells (18). Previously, sulfamidase gene overexpression was studied in D3 ES cells, a line derived from 129/Sv mice (19). In this previous study, which used the same vector construct, expression of human sulfamidase decreased after 16 days of differentiation; despite this, sulfamidase expression remained eight times higher than untransfected controls. These data are consistent with other reports suggesting that individual promoters have different activities in different cell lines (4). The sustained expression during differentiation in the present study suggests that the chicken β-actin promoter used in the vector pCAGrhNSIpuro effectively achieved sulfamidase gene overexpression in Bruce-4 C57BL/6 ES cells and differentiated derivatives over time.
In vivo studies showed that glial cells derived from the NS21 ES cell line engrafted within various structures in the brain, surviving for at least 3 months after injection into the lateral ventricles of newborn mice. None of the mice in this study formed teratomas following cell implantation. Quantification of the total number of cells engrafted in different parts of the brain by real-time PCR suggested that although a proportion of the injected cells are able to reach areas such as the olfactory bulb and the noninjected side of the brain, the majority of cells remained in the injection hemisphere. Real-time PCR for analysis of the human sulfamidase sequence proved to be a quick and effective method for quantification of engraftment, and allowed examination of donor cells in both male and female recipients.
Lysosomal storage diseases such as MPS-IIIA are characterized by “global” CNS pathology. The lack of brain-wide engraftment of glial precursor cells following implantation into the lateral ventricles in this study suggests that multiple parenchymal injection sites will be needed to achieve a therapeutic outcome. As future studies into the pathogenesis of MPS-IIIA further dissect and illuminate individual aspects of neuropathology, including the role played by specific brain regions and their connections in the complex melange of CNS disease, certain implantation sites might become higher priority targets for achieving maximum clinical benefit. Although it has not been determined what dose of cells and delivered enzyme will be therapeutically optimal, the observation that neuropathology in MPS-IIIA is correctable in a dose-dependent fashion (11) suggests that experimental strategies should focus on maximizing enzyme delivery. One way this might be achieved for cell therapies is by maximizing the survival and engraftment of implanted cells.
Along with optimizing the delivery method for maximizing the viability of implanted cells (26), understanding the dynamics of migration, proliferation, and apoptosis, and the nature of the inflammatory response following implantation of cells into different brain regions, will be important for achieving maximum long-term engraftment and wide-spread therapeutic enzyme delivery with minimal side effects. Additionally, further studies could also be carried out to investigate the migratory potential of various subpopulations of glial precursor cells expressing different receptors and neural adhesion proteins, in a similar approach to that suggested in one study looking at intracerebral implantation of mesenchymal stem cells (28). Such studies could help guide the development of future differentiation protocols designed to provide cells with maximum migratory potential, and thus widest engraftment and enzyme distribution.
The majority of cell implantation studies conducted thus far using stem cells to treat CNS pathology in LSD have also involved implantation of neonatal mice (7,20,24,34). The age of the host at implantation can affect the level of engraftment in the long term (28). While studies in neonatal mice provide important proof-of-principle data, it will be imperative to examine outcomes in older animals, in order to model more clinically realistic situations.
The development of differentiation protocols for deriving specific cell phenotypes from pluripotent cells also raises the possibility of deriving these therapeutically useful cells from an increasing range of sources. Recently, the derivation of “induced pluripotent stem” (iPS) cells from somatic cells has provided an alternative source of pluripotent stem cells (23,25,37,41). These cells may provide opportunities to develop patient-specific pluripotent cell lines, which could overcome issues of rejection following transplantation. Additionally, iPS cell technology promises to potentially circumvent the controversial ethical and logistic issues associated with obtaining human eggs for generation of cell lines by SCNT. The ever-expanding range of techniques for generating pluripotent cells from various sources, and research into generation and manipulation of pluripotent cells will hopefully further advance prospects for the utility of these cells for clinical application to a range of different conditions.
In summary, this study describes the creation and in vitro characterization of a pluripotent cell line for high-level expression of the lysosomal enzyme, sulfamidase. We have developed and characterized a protocol for the manipulation of these pluripotent cells that predominantly produces glial precursors, suitable for implantation studies. The survival of engrafted glial precursor cells in the MPS-IIIA mouse brain into adulthood supports the use of these cells as a vehicle for delivery of deficient enzyme to the CNS in MPS-IIIA.
Footnotes
Acknowledgments
The authors could like to thank Professor Ed Stanley for the Bruce-4 ES cells, Professor Diane Krause for the Y chromosome probe, and Dr. Tatsunori Seki for the PSA-NCAM antibody. Additionally, we would like to thank Dr. Allison Crawley, Ms. Suzanne Brody, and Ms. Amanda Luck for their assistance with the mouse colony, along with Ms. Barbara King and Dr. Maria Fuller for their assistance with mass spectrometry. Dr. Rowani Mohd Rawi and Ms. Helen Beard are acknowledged for their helpful advice on the real-time PCR and histology studies, respectively. This work would not have been possible without the tireless efforts of Randall and Elizabeth Linton and the generous funding support of the Sanfilippo Children's Research Foundation. An international patent is held by J.J.H. and others for mammalian sulfamidase and genetic sequences encoding it, for use in the investigation, diagnosis, and treatment of subjects suspected of suffering from sulfamidase deficiency (US Patent # 5,863,782).
