Abstract
Endostatin (ES) is a potent inhibitor of angiogenesis and tumor growth. Continuous ES delivery of ES improves the efficacy and potency of the antitumoral therapy. The TheraCyte® system is a polytetrafluoro-ethylene (PTFE) semipermeable membrane macroencapsulation system for implantation of genetically engineered cells specially designed for the in vivo delivery of therapeutic proteins, such as ES, which circumvents the problem of limited half-life and variation in circulating levels. In order to enable neovascularization at the tissues adjacent to the devices prior to ES secretion by the cells inside them, we designed a scheme in which empty TheraCyte® devices were preimplanted SC into immunodeficient mice. Only after healing (17 days later) were Chinese hamster ovary cells expressing ES injected into the preimplanted devices. In another model for device implantation, the cells expressing ES where loaded into the immunoisolation devices prior to implantation into the animals, and the TheraCyte® were then immediately implanted SC into the mice. Throughout the 2-month study, constant high ES levels of up to 3.7 μg/ml were detected in the plasma of the mice preimplanted with the devices, while lower but also constant levels of ES (up to 2.1 μg/ml plasma) were detected in the mice that had received devices preloaded with the ES-expressing cells. Immunohistochemistry using anti-ES antibody showed reaction within the device and outside it, demonstrating that ES, secreted by the confined recombinant cells, permeated through the membrane and reached the surrounding tissues.
Introduction
Angiogenesis, the formation of new blood vessels from existing capillaries, is required for tumors to expand beyond 1–2 mm3 in size. It is also essential for the growth and persistence of solid tumors and their metastases (7).
Endostatin (ES) is a specific angiogenesis inhibitor that prevents vascular endothelial cells from proliferating and migrating in response to proangiogenic proteins. It can potently inhibit tumor growth without inducing toxicity or acquired drug resistance (2,15). However, injected ES is rapidly cleared from the circulation (10). It has been shown that continuous ES administration by an osmotic pump, which results in sustained systemic ES concentration, leads to increased efficacy as indicated by increased tumor regression and an 8- to 10-fold decrease in the dose required for the achievement of the same antitumor effect through the single daily SC or IP ES administration (12). Also, preclinical studies have indicated that ES exerts cytostatic rather than cytotoxic effects (9), so its long-term administration is necessary for a prolonged therapeutic effect. This feature and the difficulty in synthesizing large amounts of recombinant functional proteins have prompted this study, which has investigated ES delivery by a gene therapy approach.
Encapsulation of recombinant cells expressing therapeutic proteins in semipermeable devices for in vivo delivery has potential application as a sustained and long-term release system of the protein to the systemic circulation. The semipermeable membranes allow exchange of nutrients and oxygen between the implanted cells and the host (13). In addition, the encapsulation technology can protect the cells from rejection by the immune system. Most importantly, this system can circumvent the problems of limited half-lives and variation in circulating levels of proteins with therapeutic capacity.
The treatment of solid tumors using local ES delivery by implantation of alginate microcapsules containing genetically engineered cells that produce ES at the vicinity of the tumor site (10,16) or by transplantation of ES-producer cells into the peritoneal cavity of model animals (18,21,22) has been described. However, there is no possibility of withdrawing the alginate microcapsules containing the genetically engineered cells in the case of undesired side effects. Another possible problem is the rupture of the microcapsules and cell release, thus leading to attack from the host's immune system.
The TheraCyte® immunoisolation device is a poly-tetrafluoroethylene (PTFE) membrane macroencapsulation system comprised of a 0.4-μm pore cell-impermeable membrane, laminated to a 5-mm pore membrane that induces neovascularization at the device-host tissue interface, developed for implantation of cells that produce therapeutic proteins (3). These membranes allow for high density of encapsulated cell packing in a relatively small area, and they have been found to be allo-protective (4). Furthermore, these membranes can be removed altogether with the cells, and new devices can be implanted. In other words, TheraCyte® offers a safe system of cell encapsulation and delivery of the antiangiogenic protein ES, and its withdrawal is possible in the case of undesired side effects.
In the present study, Chinese hamster ovary (CHO) cells engineered to continuously secrete high ES levels were encapsulated in TheraCyte® immunoisolation devices implanted subcutaneously into mice. Two protocols were employed for implantation of the cells into the devices: 1) the devices were preloaded with ES-producer cells and then implanted into the mice; 2) the devices were preimplanted SC into the animals (preimplanted mice). Only after wound healing (17 days) were the ES-producer cells injected into the devices. The ability of the TheraCyte® system containing ES-expressing cells to secrete and maintain ES levels in the systemic circulation was evaluated.
Materials and Methods
Cell Culture and Reagents
Chinese hamster ovary (CHO) cells transformed with the vector pED- endo, expressing 28.8 μg of (his)6-met-endostatin (ES)/106 cells/24 h (5) were maintained in α-minimum essential medium (α-MEM; Gibco, USA) supplemented with 50 U/ml penicilin, 50 μg/ml streptomycin, 1.25 μg/ml amphotericin B, 2 mM glutamine, and 10% heat-inactivated fetal bovine serum. C-PAE cells (ATCC CCL-209) were maintained in Eagle's minimal essential medium (EMEM) supplemented with 20% FBS, 50 U/ml penicillin, 50 μg/ml streptomycin, and 2 mM l-glutamine. The cells were incubated in a humidified environment at 37°C, in the presence of 5% CO2.
Endothelial Cell Proliferation Assay
The ability of the supernatant of CHO cells expressing ES to inhibit the proliferation of cow pulmonary artery endothelial cells (C-PAE) was tested by determination of their [3H]thymidine incorporation. Incubations were performed in 96-well plates, in a final volume of 200 μl/well of EMEM containing 2% FBS, with initial cell concentrations of 5 × 103 cells/well. After incubation for 24 h, the medium was replaced with fresh EMEM containing 2% FBS and 3 ng/ml bFGF with serial dilutions of a supernatant of a culture of CHO cells expressing endostatin, or an undiluted supernatant of a culture of CHO cells transfected with the vector pED without the endostatin gene. The cells were pulsed with 1 μCi [3H]thymidine for 24 h, followed by cell harvesting. Cell-associated radioactivity was determined using a liquid scintillation counter.
Animals
The animal experiments were approved by the IPEN ethics committee for animal research (CEPA—protocol number 18). SCID male mice aged 6–8 weeks and weighing 20–25 g were employed. The animals were acclimated, caged in groups of up to five in a barrier care facility, and fed with animal chow and water ad libitum.
Injection of “Free” ES-Expressing Cells in Mice
Recombinant CHO cells maintained in culture were trypsinized and counted. Cells (106) resuspended in PBS were freely injected SC into the abdominal region of SCID mice.
Immunoisolation Device Preparation and Loading
TheraCyte® capsules (Baxter Healthcare Corp., Round Lake, IL) with a capacity of 20 μl within a poly-tetrafluoroethylene (PTFE) bilayer membrane wall were employed. The outer layer had a 5-μm pore size, which assisted formation of blood vessels. The inner PTFE membrane had a smaller pore size (0.4 μm), to prevent cell-to-cell contact (3). An outer polyester mesh supported the membranes. TheraCyte® capsules were sterilized using 100% and 70% ethanol washes before three 20-min washes with sterile normal saline (0.9% NaCl). The appropriate cell suspension (106 cells) was taken up in a Hamilton syringe with a blunt 26-gauge needle. The needle was inserted into the device port leading into the capsule. Silicon adhesive was then injected (Dow Corning, USA), in order to seal the device port entrance.
Surgical Implantation of the Immunoisolation Devices
Two protocols were tested for implantation of the TheraCyte® devices. In the first protocol, devices preloaded with the ES-expressing cells were implanted into the animals. The mice were intraperitoneally anesthetized with xylazine (65 mg/kg) and ketamine (5 mg/kg) (Rhabiofarma Ind. Pharmaceutic, Hortolândia, Brazil). The dorsal region was opened with a midline incision, and the preloaded device was placed in this site. The incision was closed using surgical sutures. In the second protocol, mice were anesthetized, the dorsal region was opened with a midline incision, and the empty device was preimplanted into this site. The incision was closed. After wound healing (approximately 17 days), the mice were anesthetized again, and a little incision was made to expose the device port, which was cut to withdraw a part of the protruding tube containing the silicon adhesive. The ES-expressing cells were then injected into the device, as described above, and TheraCyte® was once more sealed with silicon adhesive. The incision was closed using surgical sutures. The mice were monitored until recovery from the anesthetic. For the group of animals treated with the preloaded devices (n = 3), the mice were euthanized and the devices were removed for histology at weeks 1, 3, and 11. For the group of animals preimplanted with the device alone, all the animals (n = 3) were euthanized and the devices removed at week 8.
Western Blotting Analysis
Serum collected from animals with freely implanted CHO cells expressing ES were analyzed by Western blotting. The samples were submitted to SDS-PAGE under reducing conditions, and the proteins were transferred to a nitrocellulose membrane. The membrane was probed with a 1:150 dilution of polyclonal antibody anti-endostatin (Chemicon, Temecula, USA) diluted in PBS containing 5% skim milk. Reactions were detected with secondary antibody conjugated to horseradish peroxidase (Armeshan Pharmacia Biotech) by means of enhanced chemiluminescence.
Determination of ES Concentration
Serum samples were obtained every week by retro-orbital puncture using capillary tubes with heparin after anesthesia. The serum was collected after blood centrifugation at 227 × g for 5 min. ES concentrations were determined by a commercial ELISA kit (CYT 160, Chemicon, USA) following the manufacturer's protocols. A Student's t-test, with Bonferroni correction as appropriate, was applied for analysis of the significance of the difference among the values of endostatin concentration in the serum of the treated and control mice.
Histological Analysis
The animals were killed at the end of the experiment. The devices were excised, washed in PBS, fixed in PBS-buffered 3.7% formalin for 24 h, and processed. Histological analysis was performed in 4-μm sections stained with hematoxylin and eosin (H&E). For ES immunohistochemical detection, sections of the devices were processed, subjected to the antigen retrieval method using anti-endostatin antibody (anti-rabbit AB1880, Chemicon, EUA), and developed using a diaminobenzidine-based detection system (SuperPicture Poly HRP conjugated, Zymed, CA). For the control reaction, the antibody was replaced with BSA.
Results
ES Biological Activity
The biological activity of ES secreted by CHO cells transfected with the expression vector containing the ES gene was determined in the conditioned medium of CHO cells by means of its inhibitory activity on endothelial cell proliferation. The inhibitory effect of ES on [3H]thymidine incorporation is shown in Figure 1. The percentages of [3H]thymidine incorporation, which indicates cellular viability, were directly proportional to the factor of dilution of the supernatants of the CHO cells expressing endostatin, demonstrating that ES inhibited the replication of bovine endothelial cells in a dose-dependent fashion.
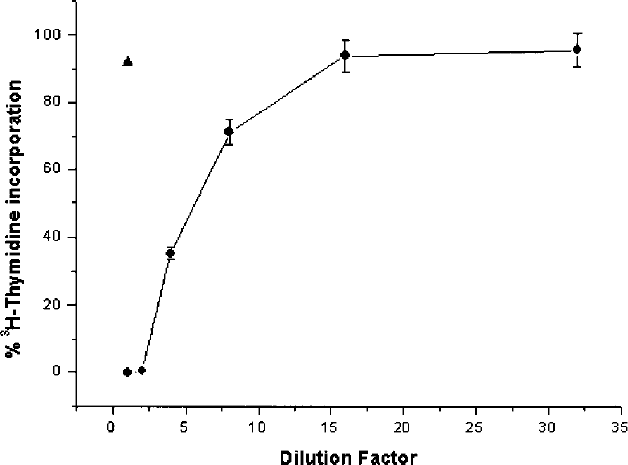
Endothelial cell proliferation assay. Recombinant mouse ES (circle) expressed in conditioned culture medium from CHO-transfected cells was tested for its ability to inhibit [3H]thymidine incorporation into C-PAE cells. Conditioned medium from CHO cells transfected with the pED vector without endostatin gene (triangle) was used as a control.
Injection of Free ES-Expressing Cells
Previous tests indicated that the subcutaneous injection of 106 free ES-expressing cells led to higher ES systemic levels than those obtained by the IP injection of the same amount of cells (data not shown). Three different clones of CHO cells expressing ES were injected into the preimplanted immunoisolation TheraCyte® devices. The clone that produced the highest ES level in the serum of the animals was selected for further assays (data not shown). Serum samples of mice whose blood had been removed at days 7, 15, and 23 after the SC injection of 106 free CHO cells expressing ES were analyzed by Western blotting (Fig. 2). Although no band could be seen in lanes 2 and 3, which correspond to the serum of the animal that had been injected with control cells and to the serum of the animal implanted with ES-expressing cells at day 7, respectively, strong ES bands (20.1 kDa) could be seen in the columns corresponding to the serum of animals injected with 106 ES-expressing cells at day 15 (lanes 4 and 6). Fainter bands could be seen in the columns corresponding to the serum withdrawn at day 23 (lanes 5 and 7).

Western blotting of the serum of mice treated with 106 CHO cells. Lane 1, ES positive control; lane 2, serum withdrawn from a mouse at day 7 after injection of cells that do not express ES; lane 3, serum withdrawn from mouse No. 1 at day 7 after injection of ES-expressing cells; lane 4, serum withdrawn from mouse No. 1 at day 15 after injection of ES-expressing cells; lane 5, serum withdrawn from mouse No. 1 at day 23 after injection of ES-expressing cells; lane 6, serum withdrawn from mouse No. 2 at day 15 after injection of ES-expressing cells; lane 7, serum withdrawn from mouse No. 2 at day 23 after injection of ES-expressing cells.
According to data obtained by ELISA for murine ES, high ES levels were obtained in the serum of the mice injected with 106 free CHO cells expressing this protein. The mean ES concentration (n = 3 animals) was 5.65 μg/ml at week 2. The serum of mice injected with CHO cells transfected with the vector without the ES-gene presented 13 times lower mean ES level (Table 1).
ES Levels in the Serum of the Mice Injected With “Free” CHO Cells

In Vitro Cultivation of Immunoisolation Devices Containing ES-Expressing Cells
Conditioned medium of CHO cells expressing ES cultivated in vitro were applied to an SDS-PAGE and an immunoblot using anti-endostatin antibody. Figure 3 shows bands corresponding to ES in both the supernatant of cells cultivated in a T75 culture flask and the same amount (106) of cells cultivated in a TheraCyte® immunoisolation device, during a 4-day period. A stronger ES band could be seen in the supernatant of the flask-cultivated cells, probably due to the smaller space for cell adherence inside the immunoisolation device.

Western blotting of conditioned medium of 106 ES-expressing CHO cells cultivated in vitro. Lane 1, ES-positive control; lane 2, supernatant cells cultivated in a T75 culture flask; lane 3, supernatant cells cultivated in the TheraCyte® immunoisolation device.
In Vivo Implants of Immunoisolation Devices
High ES levels were obtained in the serum of the mice treated with immunoisolation devices containing ES-producer cells using both implantation protocols. When the cells were injected into the preimplanted devices, lower ES levels were obtained 1 and 2 weeks after transplantation of the ES-expressing cells (Table 2). These levels increased continuously, reaching up to 3.67 μg ES/ml at week 6 after cell loading into the device. When preloaded devices (i.e., devices already containing the ES-expressing cells) were implanted, high ES levels were obtained from the very first week, reaching up to 2.10 μg ES/ml at week 11 (data not shown). Apparently, there was net cell growth after cells were introduced into the preimplanted devices, but no net cell growth occurred in the case of the preloaded devices. Considering that in the case of the preimplanted devices the vascularization at the tissues adjacent to the device could occur before the injection of the ES-expressing cells, and that in the case of the implantation of the preloaded devices vascularization would possibly be impaired by the ES secreted by the cells inside the devices, it is likely that the higher ES levels achieved in the serum of the animals in which the devices were preimplanted reflects the higher degree of vascularization, which allows for the proliferation of ES-expressing cells.
ES Levels in the Serum of the Mice Implanted With TheraCyte® Immunoisolation Devices Containing CHO Cells Expressing ES

A Student's t-test, with Bonferroni correction as appropriate, was pplied for analysis of the significance of the difference among the values of endostatin concentration in the serum of the treated and control mice (without treatment).
Histology of the Devices
We had some difficulty performing the histological slices because the TheraCyte® devices are made with PTFE, a polymer that is considerably more consistent than the biological tissue, despite being flexible. The devices removed from the animals did not show any sign of rupture. The histology of the devices removed from the mice in which the cells had been injected 17 days after preimplantation of the TheraCyte® device is shown in Figure 4A-C. Figure 4D-F depicts the histology of the devices withdrawn from mice that were implanted using the other implantation protocol (preloaded TheraCyte®), stained with H&E. The presence of defined cellular nucleus could be observed, indicating cellular viability. The presence of cells adhered to the internal membrane of the immunoisolation devices withdrawn at weeks 4 (Fig. 4A) and 6 (Fig. 4C, D) after implantation of ES-expressing cells was also evident. The presence of cells adhered to the internal face of the device is an indication that these cells are viable, and that the oxygenation in this area, closer to the external healing, is probably more effective, which would possibly lead to larger ES release to the external tissues and circulation. More viable cells were observed when the devices were preimplanted into the mice before injection of the ES-expressing cells compared with the implantation of the preloaded devices. The presence of blood vessels in the neighborhood of the devices reveals that the larger (5 μm) PTFE pores of the devices stimulated neovascularization at the vicinity of the device. The presence of vessels is important for cell viability inside the device as well as for the transport of the ES secreted by the cells inside the device into the mouse systemic circulation.
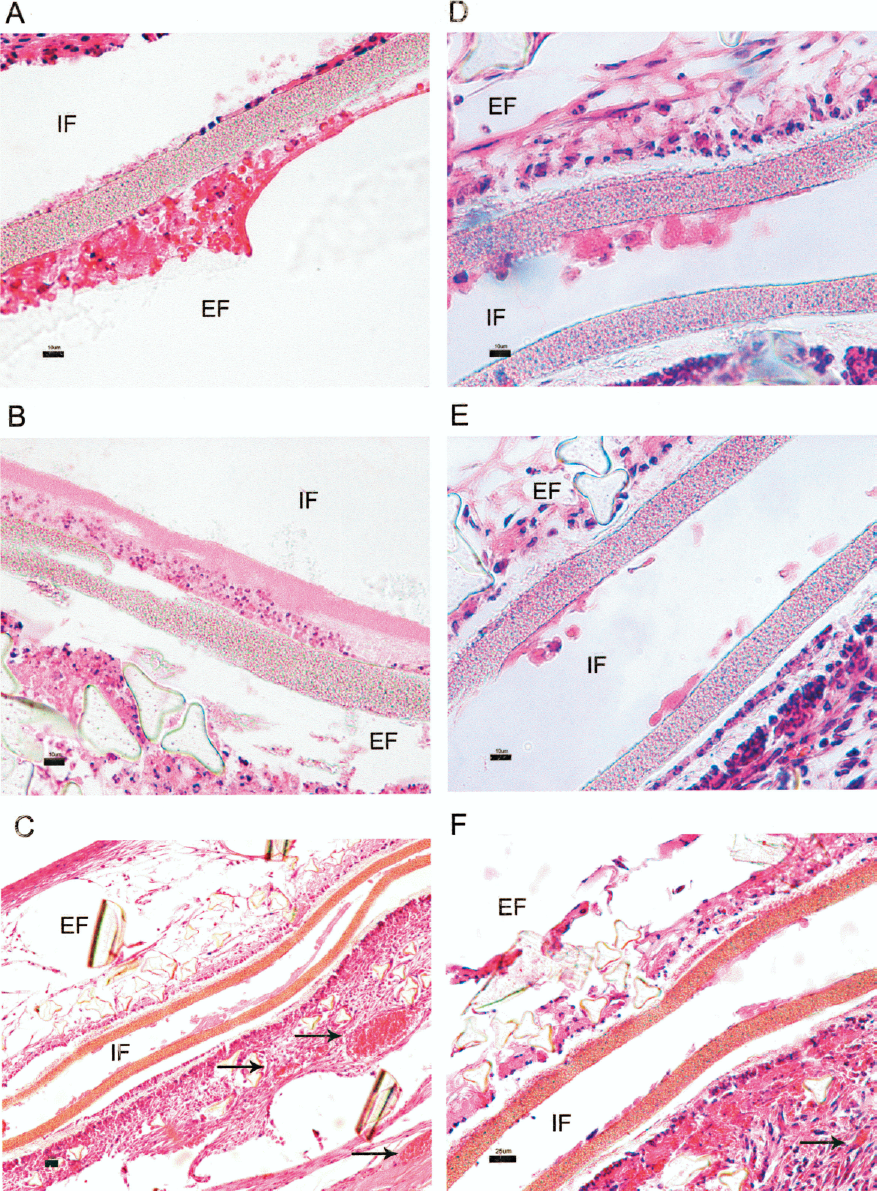
Histological analysis staining H&E of the TheraCyte® devices. (A-C) ES-expressing cells were injected 17 days after preimplantation of the device. (D-F) Devices preloaded with ES-expressing cells were injected into mice. Note that the cells are apparently viable inside the devices, with the presence of intact cellular nucleus and cells adhered to the internal membrane of the devices, indicating cellular viability. The arrows in (C) and (F) indicate the location of the blood vessels. IF, internal face; EF, external face. Scale bars: (A, B, D, E) 10 μm (original magnification 40×); (C, F) 25 μm (original magnification 20×).
Next, immunohistochemistry on paraffin sections of TheraCyte® devices withdrawn from mice 6 weeks after the start of the treatment was performed. This procedure was carried out using the polyclonal antibody against mouse ES, in order to confirm the presence of this protein inside or at the vicinity of the device. A strong staining was found in the internal face of the device. It was clearly visible that the staining in the external face adjacent to the device became fainter in the tissues that are more distant from the membrane (Fig. 5B). This indicates that the ES produced by the cells inside the device diffused, reaching the contiguous tissues. No specific immunostaining was observed in the control sections (Fig. 5A, C, D).
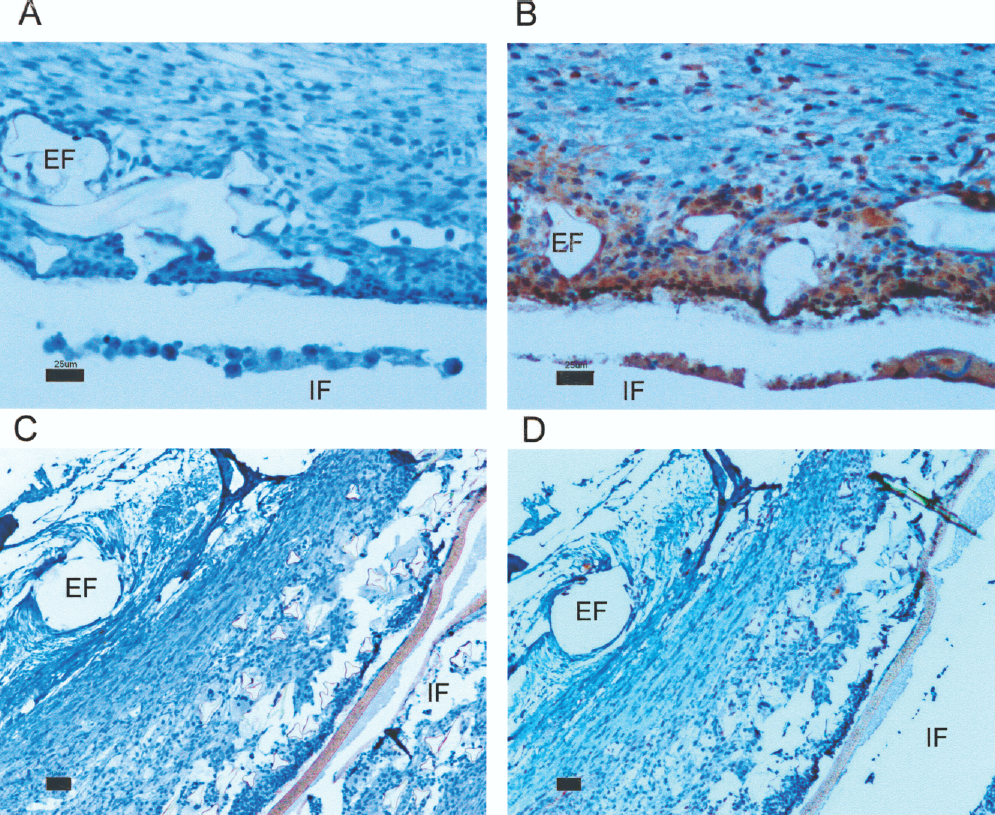
Immunohistochemistry of the preloaded TheraCyte® devices withdrawn from mice at week 6. (A, B) CHO cells expressing ES. (C, D) CHO cells transfected with the vector without the ES gene. (A, C) Control of the immunohistochemistry reaction. (B, D) Reaction using anti-endostatin antibody. IF, internal face; EF, external face of TheraCyte® devices. Scale bars: (A, B) 25 μm (original magnification 20×); (C, D) 10 μm (original magnification 40×).
Discussion
We have described a protocol for ES delivery to the systemic circulation of SCID mice using recombinant xenogenic cells expressing high ES levels, encapsulated in bioisolating devices. We used immunodeficient mice, considering that it has been shown that allogeneic tissues can survive within TheraCyte® devices for long periods, but xenogenically tissues, such as the case of cells from hamster origin (CHO cells) in mice models, can be destroyed within few weeks (4). Therefore, in the case of a possible treatment in humans, cells of human origin should be used.
Two methods were analyzed for implantation of the immunoisolating devices for ES delivery. In the first protocol, the devices were preimplanted into the mice and, after healing of the wound and neovascularization in the vicinity of the device, the ES-expressing CHO cells were inoculated into the device. An ES effect on neovascularization, namely formation of a higher percentage of nonfunctional vessels, had been demonstrated (1). Device implantation prior to the injection of ES-expressing cells aimed at improving vascularization at the membrane of the device/host tissue interface before implantation of the ES-secreting cells, which would possibly interfere negatively with the formation of blood vessels and cell viability. It has been shown that preimplantation significantly lowers the curative dose of macroencapsulated pancreatic islets for the treatment of diabetic athymic mice (17). High and increasing ES systemic levels were observed in the animals treated with preimplanted devices. The expression ranged from 2.1 to 3.67 μg ES/ml at week 6 of the assay, with levels still rising 2 months after the beginning of the treatment. In another protocol, we implanted preloaded devices into the animals, considering that cellular viability has also been described using this scheme (8,14,19). Levels of up to 2.1 μg ES/ml at week 11 were obtained, whereas the mean ES concentration in the serum of untreated mice was 0.45 μg ES/ml. The presence of functional blood vessels in the vicinity of the devices implanted via both protocols of ES-expressing CHO cell transplantation was confirmed by microscopic analysis of the microscopy slides stained with H&E.
The use of TheraCyte® bioisolation system for correction of diabetes in animal models has been described (6,17,20). The delivery of therapeutic proteins from recombinant cells has also been previously shown (11,19). Systemic levels of the heterologous proteins of the human growth hormone (0.002 μg/ml) in NIH nude mice serum (11), and factor IX (0.2 μg/ml) in HSD athymic nude mice serum (4) were relatively low compared with the ES systemic levels described herein. The high levels obtained by us are probably related to three factors: the high level of ES secretion by recombinant CHO cells (28.8 μg/106 cells/24 h), the fact that the therapeutic protein is homologous and thus more stable, and the fact that there are relatively high basal ES levels in untreated animals (up to 0.56 ng/ml serum).
The implantation of ES-expressing cells encapsulated in alginate microcapsules into the peritoneal cavity of mice has also been described. The articles reported lower levels of heterologous ES, namely 0.257 μg/ml (22), 0.194 μg/ml (21), and 0.017 μg/ml (10), compared with the levels of up to 2.98 μg/ml described here.
The system described by us is therefore a simple and safe sustained gene delivery system that allows for the removal and implantation of new devices. Despite not having been directly tested in our assays, this system does not seem to require immunosuppression of the host for antitumoral treatment because it has been shown that allogeneic tissues are able to survive within TheraCyte® devices (4,19).
The devices can be conveniently removed following a term of treatment, which would be desirable for therapies intended for only a certain period of time. All the cells can be removed, unlike free cell implants, viral infusions, or microencapsulated cells, in which complete withdrawal of the implanted cells may be problematic. A system in which universally implantable cell lines from human origin could be developed for therapies and uniquely engineered for a specific therapeutic approach would be considerably less demanding than therapies based on autologous ex vivo cell manipulation.
The studies using this system can be applied to similar systems, using recombinant eukaryotic cells for secretion of other proteins with therapeutic properties.
Footnotes
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Project 00/04658-0 and by Conselho Nacional de Pesquisa (CNPq).
