Abstract
Pancreatic islet transplantation has the potential to be an effective treatment for type 1 diabetes mellitus. While recent improvements have improved 1-year outcomes, follow-up studies show a persistent loss of graft function/survival over 5 years. One possible cause of islet transplant failure is the immunosuppressant regimen required to prevent alloimmune graft rejection. Although there is evidence from separate studies, mostly in rodents and cell lines, that FK506 (tacrolimus), rapamycin (sirolimus), and mycophenolate mofetil (MMF; CellCept) can damage pancreatic β-cells, there have been few side-by-side, multiparameter comparisons of the effects of these drugs on human islets. In the present study, we show that 24-h exposure to FK506 or MMF impairs glucose-stimulated insulin secretion in human islets. FK506 had acute and direct effects on insulin exocytosis, whereas MMF did not. FK506, but not MMF, impaired human islet graft function in diabetic NOD.scid mice. All of the immunosuppressants tested in vitro increased caspase-3 cleavage and caspase-3 activity, whereas MMF induced ER-stress to the greatest degree. Treating human islets with the GLP-1 agonist exenatide ameliorated the immunosuppressant-induced defects in glucose-stimulated insulin release. Together, our results demonstrate that immunosuppressants impair human β-cell function and survival, and that these defects can be circumvented to a certain extent with exenatide treatment.
Introduction
Diabetes is a chronic disease with devastating complications that place enormous burdens on health care systems. Islet transplant represents a promising experimental treatment for diabetes (47,53), although most grafts eventually fail (43). A number of factors likely contribute to the eventual decline in graft function and survival seen in the majority of transplant cases. It appears likely that both a decline in β-cell mass, perhaps due to a combination of increased β-cell death and impaired compensatory proliferation, and a decline in β-cell function contribute to islet graft deterioration. To be sure, transplanted β-cells face a number of challenges, including suboptimal glucose control, allorejection, and resurgent autoimmunity. There is also considerable evidence that immunosuppressive drugs used to prevent graft rejection have deleterious effects on β-cell survival and/or function (40), leading to posttransplant diabetes mellitus (37,44). Indeed, a number of studies have documented negative effects on insulin secretion, insulin gene expression, β-cell apoptosis, and β-cell proliferation of specific immunosuppressant drugs, FK506 (tacrolimus) (15,31,34,36,38,39,41,49,50), rapamycin (sirolimus) (6,34, 57,58), mycophenolate mofetil (MMF; CellCept) (15,18, 36,38,45), and cyclosporine (2,28,31,36,38).
There are also studies that point to potential protective effects of immunosuppressive drugs (14,30,33). However, many of these studies have focused on rodent islets or β-cell lines and have not employed human islets. Moreover, few studies have compared multiple immunosuppressant drugs side-by-side at clinically relevant doses and examined multiple parameters to determine the relative functional toxicity. Herein, we present a comprehensive comparison of the effects of several immunosuppressant drugs, including FK506, rapamycin, and MMF, on glucose-stimulated insulin release, caspase-3-dependent apoptosis, as well as in vivo graft survival. We demonstrate that the GLP-1 agonist, exenatide, can ameliorate islet dysfunction caused by immunosuppressants. These results support a proof-of-concept clinical pilot study on the feasibility of using perioperative and postoperative exenatide to improve graft survival in patients. We expect these results will eventually contribute to the improvement of clinical outcome in islet transplant patients.
Materials and Methods
Drugs and Solutions
All drugs were obtained from the Vancouver General Hospital Pharmacy unless otherwise indicated. Rapamycin (M.W. 914.2; also known as Sirolimus and Rapamune) was from Wyeth (Montreal QC). FK506 (M.W. 804; also known as tacrolimus, fujimycin, and Prograft) was from Astellas (Markham, ON). Mycophenolate mofetil (M.W. 433.5; also known as CellCept) was from Hoffmann-La Roche (Mississauga, ON). Cyclosporin A (M.W. 1202.61) was purchased from Sigma (St. Louis, MO) and made up in DMSO. Exenatide (Byetta) was from Amylin (Markam ON). Experiments in Figure 3A used nonclinical grade immunosuppressants purchased from Sigma.
Human and Mouse Islet Culture
All human and mouse islet studies were approved by the local Institutional Review Board and Animal Care Committees, in accordance with national guidelines. Human islets were isolated from consecutive donors over a 2.5-year span. Both males and females were used. Islet purity assessed by dithizone staining was typically 60–90%. Human islets were isolated and cultured using protocols that have been detailed elsewhere (23,26,29,47, 53). Human islets (~2500 IE/2 ml culture) were cultured in media containing 5 mM glucose and were generally studied within 3 days of isolation. All experiments were replicated on islets from at least three donors. Mouse islets were isolated as described (11,23) and cultured in RPMI media (containing 5.5 mM glucose and 100 IU/ml penicillin, 100 μg/ml streptomycin, 10% fetal calf serum; pH 7.4 with NaOH) at 37°C, 5% CO2, and saturated humidity. Mouse islets were studied 2–4 days after isolation. All experiments were conducted on mouse islets from at least three separate isolations.
Hormone Release and Radioimmunoassays
Human islet and mouse islet perifusions were carried out as previously described (29) with the assistance of the In vitro Beta-cell Phenotyping Core of the MSFHR Centre for Human Islet Transplantation and Beta-cell Regeneration (CHITBR). Briefly, islets were superfused at 37°C and 5% CO2/95% O2 using in a multichamber apparatus and an automated fraction collector. Islets were perifused in Kreb's Ringer's buffer containing: 115 mM NaCl, 24 mM NaHCO2, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, and 5 g/L RIA grade albumin, and either 2.8 mM or 20 mM glucose as indicated. Perifused islets were allowed to equilibrate in the chambers for 1 h prior to the start of fraction collection. In some studies, islets were also stimulated with exenatide (1 nM) in the presence of high glucose. Before human islet perifusion experiments, some groups of islets were cultured with low and high doses of specific immunosuppressants for 24 ± 6 h. In one set of studies, islets were cultured in the presence or absence of 1 nM exenatide, then perifused as above in the absence of exenatide. Perifusate was collected for insulin radioimmunoassay (Linco/Millipore, Billerica, MA).
The rationale for choosing specific immunosuppressant doses is as follows. The therapeutic level of FK506 (Tacrolimus) is generally thought to be between 5 and 15 μg/L, although it may reach toxic levels of 20–25 μg/L (51). We chose doses of 10 μg/L (12 nM) and 30 μg/L (37 nM), representing low and high levels. The therapeutic level of rapamycin (Sirolimus) is thought to be 5–15 μg/L, although it may reach levels of 20–25 μg/L (51). We chose 10 μg/L (11 nM) and 30 μg/L (33 nM), representing low and high levels. The therapeutic level of MMF is thought to be 1–4 μg/L, although it may reach levels of 5–10 μg/L (51). We chose to study cells treated with 4 μg/L (9 nM) and 12 μg/L (28 nM), representing low and high levels.
In some experiments, human islet cells were dispersed and cultured for 3 days as described in detail elsewhere (26,29). Cells were washed for 30 min and then incubated as above for 90 min in Kreb's Ringer's buffer (containing, in mM: 115 NaCl, 24 NaHCO2, 5 KCl, 2 CaCl2, 1 MgCl2, and 5 g/L RIA grade albumin), after which the supernatant was removed and frozen for subsequent radioimmunoassay.
Apoptosis and ER Stress Detection
Apoptosis was analyzed in groups of 100 human islets using Western blots and a rabbit monoclonal antibody to cleaved caspase-3 (Cell Signaling, Danvers, MA). ER stress was detected with a monoclonal antibody to the well-established marker, C/EBP homologous protein (CHOP), from Cell Signaling. An antibody to β-actin (Novus Biologicals, Littleton, CO) was used as a loading control. Bands were quantified by densitometry after subtracting background and normalization. Bands were first normalized to the control for that gel, then to the respective loading control value. Our methods for Western blotting have been described in detail (23). Human islets were also collected for caspase-3 activity assay. The Caspase-3 Fluorometric Assay Kit (Bio-vision Research Products, Mountain View, CA) was used according to the manufacturer's instructions.
Human Islet Transplant Into Mice
In vivo human islet graft function was examined with assistance from the In vivo Beta-cell Phenotyping Core of the CHITBR. Five hundred hand-picked human islets (150–400 μM in diameter) were transplanted under one kidney capsule of adult NOD.scid mice made diabetic by an IP injection of 200 mg/kg streptozotocin. Post-transplant, mice were injected (IP) daily with tacrolimus (30 mg/g body weight), mycophenylate (1.5 mg/g body weight), sirolimus (50 mg/g body weight), or saline (control). Blood glucose was monitored regularly for 3–4 weeks. Hyperglycemia was defined as two consecutive blood glucose levels greater than 15 mM. Grafts were removed and assessed by insulin immunostaining using a guinea pig anti-insulin primary antibody from Linco/Millipore and secondary antibodies from Jackson Immunoresearch Laboratories (West Grove, PA). Nephrectomy of the kidney containing the islet transplant in normoglycemic mice resulted in a rapid return to hyperglycemia.
Statistical Analysis
All experiments were performed at least three times on completely independent islet isolations (i.e., different donors). Statistical analyses were performed using Stu-dent's unpaired t-test or one-way ANOVA (followed by Fisher's PLSD post-hoc test), where appropriate. Differences were considered significant when p < 0.05. Results are presented as mean ± SEM. For perifusion studies, area under the curve (AUC) of glucose-stimulated insulin secretion was compared among treatments. The AUC was calculated as a cumulative fold increase over baseline for a 30-min time period 10 min after the application of high glucose. Each percent pretreatment data point was divided by an average baseline value. These values were then added and multiplied by 30 min to get the final number.
Results
Comparison of Immunosuppressant Effects on Insulin Secretion
Secretory function of human islets from 15 separate donors was analyzed using an islet perifusion system. Islets cultured in normal conditions exhibited a robust and rapid increase in insulin secretion when glucose was increased from 2.8 to 20 mM (Fig. 1). The GLP-1 agonist exenatide was able to further increase insulin secretion in the presence of 20 mM glucose. After islets were returned to low glucose, insulin secretion could be stimulated with 30 mM KCl, which increases insulin secretion by directly depolarizing the plasma membrane, by-passing the glucose-sensing machinery of the β-cell. Next, we examined insulin secretion in islets cultured in the presence of three different immunosuppressants (FK506, rapamycin, and MMF) for 24 h, at concentrations approximating the clinically relevant high and low range. Culture in 37 nM FK506 reduced glucose-stimulated insulin secretion by ~50%, and severely blunted the first phase of insulin release (Fig. 1). GLP-1 receptor-mediated potentiation of insulin secretion was also blunted. The response to KCl was also impaired, indicating effects on the voltage-gated Ca2+ channels and/or the exocytotic machinery. A lower dose of FK506 did not significantly impair glucose-stimulated insulin release. Culturing islets with either 9 or 28 nM MMF reduced glucose-, GLP-1-, and KCl-induced insulin secretion. Clinically relevant concentrations of rapamycin did not inhibit glucose-, GLP-1-, or KCl-stimulated insulin secretion in human islets. These experiments demonstrate that longer-term exposure to some, but not all, immunosuppressant drugs impairs insulin secretion from human islets, although these results do not indicate whether the defect is subsequent to a reduction in viability or a direct impairment of β-cell function.
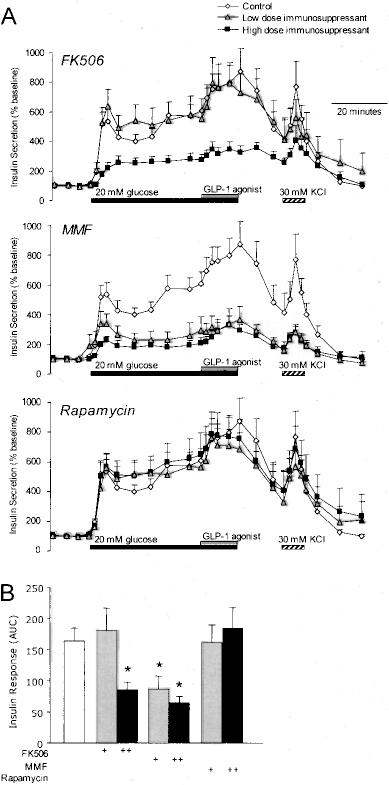
Comparison of the effects of immunosuppressant culture on insulin secretion from human islets. (A) Groups of 100–200 human islets were perifused with a baseline glucose of 2.8 mM, then 20 mM glucose (supplemented for the last 15 min with GLP-1 agonist), followed by 30 mM KCl, in the presence or absence of immunosuppressant drugs. Islets were cultured for 24 h in the presence of 37 or 12 nM FK506, 28 or 9 nM MMF, or 33 or 11 nM rapamycin. (B) Quantification of (A) (n = 15 consecutive, independent human islet isolations).
To determine whether FK506 acutely impairs insulin secretion, dispersed human islets were examined in a short-term static incubation. Whereas untreated cells responded to 15 mM glucose with a doubling of insulin secretion in 90 min, concurrent exposure to a wide range of FK506 doses dramatically inhibited glucose-stimulated insulin secretion (Fig. 2A. A glucose-independent stimulation of basal insulin release was also evident at high doses of the drug. Again, rapamycin had no effect on glucose-stimulated insulin secretion (Fig. 2B). Although these experiments do not have the temporal resolution of the perifusion data, these data suggest that FK506 can have acute effects on in vitro β-cell function, even at very low concentrations.

Dose–response profile of the acute effects of selected immunosuppressants on insulin secretion from dispersed human islets. (A) Dispersed human islets were incubated for 90 min in 3 or 15 mM glucose in the presence or absence of various concentrations of FK506 (n = 3). (B) Dispersed human islets were incubated for 90 min in 3 or 15 mM glucose in the presence or absence of various concentrations of rapamycin (n = 3). *Statistical difference at each concentration of drug compared with 3 mM glucose.
Next we examined the acute effects of multiple immunosuppressant drugs on isolated mouse islets, which tend to have more robust glucose responsiveness due to the homogeneity of the isolation process. In these experiments, immunosuppressant drugs were added 20 min prior to the glucose challenge. A high concentration of FK506 inhibited glucose- and KCl-stimulated insulin secretion by ~50%, similar to the effect seen in human islets exposed to this drug for 24 h (Fig. 3A). FK506 at a lower dose of 10 nM also reduced glucose-stimulated insulin secretion in mouse islets (not shown). Neither rapamycin nor cyclosporine inhibited insulin secretion in these experiments. We also examined mouse islets acutely exposed to MMF and found that insulin secretion in response to glucose, GLP-1 agonist, or KCl was completely intact. Together, these data suggest that FK506 can have acute effects on islet function, whereas MMF may reduce insulin secretion indirectly by reducing cell viability.
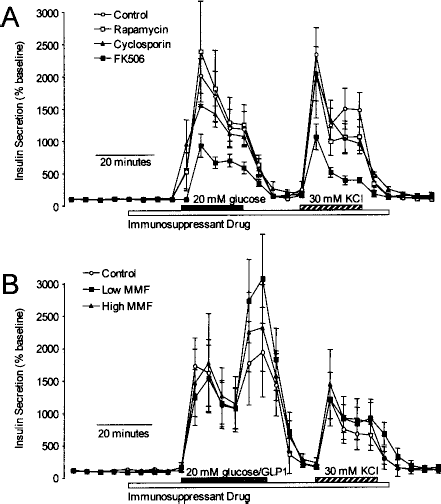
Comparison of acute effects of immunosuppressant on insulin secretion from mouse islets. (A) Groups of 100–120 mouse islets were perifused with a 3 mM glucose baseline, then stepped to 20 mM glucose, followed by a depolarization with 30 mM KCl. This was done in the absence (control) or presence of 1 μM rapamycin, 10 μM FK506, or 10 μM cyclosporine (n = 8). (B) A similar study was conducted with 10 or 30 μM MMF. In this study, 1 nM GLP-1 was added to the last 10 min of the high glucose challenge (n = 6).
Comparison of Immunosuppressant Effects on Islet Apoptosis and ER Stress
We addressed the effects of three immunosuppressants on human islet apoptosis using two independent approaches. Increased cleaved caspase-3 protein levels are a hallmark of β-cell apoptosis. Although basal rates of apoptosis appeared relatively high in isolated human islets, an increase in caspase-3 cleavage was observed upon 24-h exposure to some immunosuppressants (Fig. 4A). Among the immunosuppressants tested, rapamycin induced the greatest increase in cleaved caspase-3, whereas the effects of FK506 and MMF were minimal. The activation of caspase-3-like proteolytic activity was similarly increased with exposure to immunosuppressants (Fig. 4B). One mechanism of cell death that is particularly important for pancreatic β-cells is ER stress (13). CHOP is a transcription factor that plays an essential role in β-cell ER stress (35). Rapamycin and MMF induced significant CHOP expression (Fig. 4C). Together, these data suggest that immunosuppressants have significant effects on β-cell survival pathways.
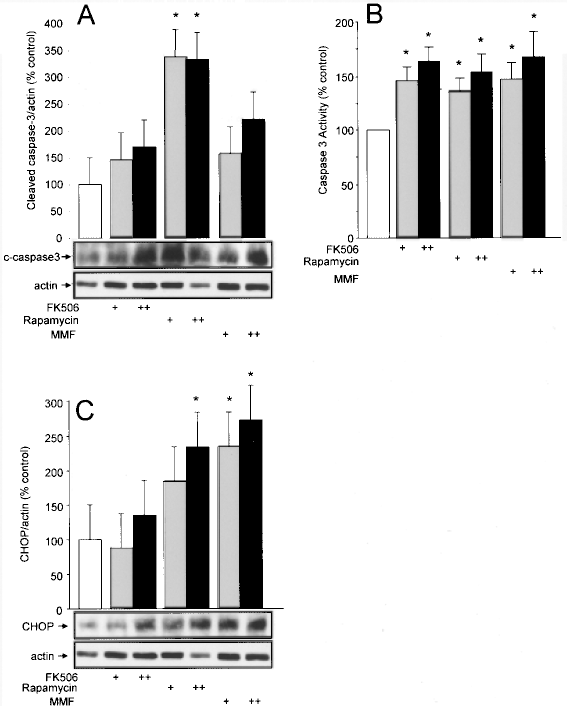
Immunosuppressants increase islet cell apoptosis and ER stress. (A) Western blot of cleaved caspase-3 protein levels in human islets treated as indicated (n = 3). (B) Quantification of caspase-3 activity in human islets treated as indicated (n = 9). (C) Western blot of CHOP protein levels in human islets treated as indicated (n = 3).
Comparison of Immunosuppressant Effects on In Vivo Graft Survival
Next we examined the survival and function (functional engraftment) of human islet xenografts in NOD. scid mice made diabetic with streptozotocin. Transplant of human islets into NOD.scid mice resulted in a rapid (<5 days) return to normoglycemia (Fig. 5A). Administration of FK506 resulted in graft failure and return of hyperglycemia, whereas mice injected with saline or MMF remained normoglycemic. Treatment with FK506 was halted at around 20 days posttransplant when mice became ill and rapidly lost weight. A similar experiment was conducted to assess the effects of rapamycin on human islet graft function in NOD.scid mice wherein no differences were seen (data not shown). At the end of the experiments, islet grafts were collected from the kidney capsule and assessed histologically. Human islet grafts from FK506-treated mice showed a dramatic loss of insulin staining, whereas insulin staining was not decreased in MMF-treated mice relative to saline-treated controls (Fig. 5B). Together, these results imply that FK506, but not MMF, induces sustained β-cell defects severe enough to dramatically impair the in vivo function of transplanted human islets.

Effects of various immunosuppressants on the function and survival of transplanted human islets in mice. (A) NOD.scid mice were made diabetic with streptozotocin (arrow), then transplanted with 500 human islets (two separate donors are represented in the data). Mice were subsequently injected intraperitoneally with saline (n = 2), 30 mg/g FK506 (n = 4), or 1.5 mg/g MMF (n = 3) each day. Nx, removal of islet graft-bearing kidney. (B) Representative images showing insulin (red) in grafts isolated from mice shown in (A). To top row shows the whole graft (10X objective) and the field of view is 950 μm wide in each image. The bottom row shows a close-up image (40x objective; 230 μm field of view) and the border of this part of the graft is delineated with a fuchsia line. The acquisition parameters were identical for each image in each row.
Exenatide Protects Islets From Immunosuppressant-Induced Dysfunction
Having shown that human islets are susceptible to the deleterious effects of clinically important immunosuppressants, we next set out to improve islet function and survival. It is established that GLP-1 and its agonists potently increase insulin secretion and protect islets from apoptosis and ER stress (12,56). Indeed, culturing human islets with the clinically approved GLP-1 agonist, exenatide, ameliorated the negative effects of immunosuppressants on glucose-stimulated insulin secretion (Fig. 6). The deleterious effects of FK506 on insulin exocytosis were completely prevented. On the other hand, exenatide only partially restored glucose-stimulated insulin release in islets cultured with MMF. Exenatide also tended to decrease caspase-3 activity induced by immunosuppressants, although the trend was not statistically significant (Fig. 7). Together these data suggest that exenatide can protect human islets from immunosuppressant-induced dysfunction, although the extent varied depending the mechanism by which the immunosuppressant altered β-cell function.
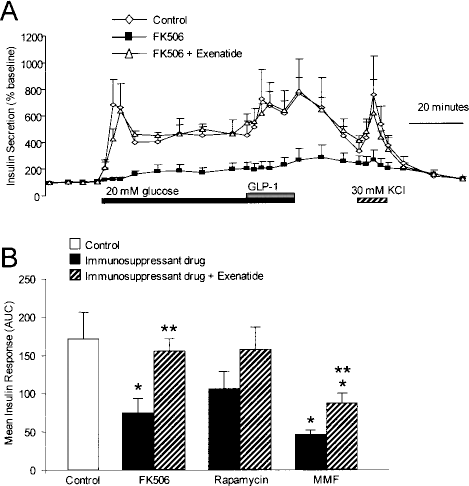
Exenatide protects human islets from impaired glucose-stimulated insulin release in the presence of immunosuppressants. (A) Human islets were cultured in media containing 37 nM FK506 with or without 1 nM exenatide. Exenatide was not included during the perifusion, except where indicated. (B) Quantification of the area under the curve for glucose-stimulated insulin release from human islets exposed to 37 nM FK506, 33 nM rapamycin, or 28 nM MMF as in Figure 1, in the presence or absence of 1 nM exenatide. *Significant difference from untreated control. **Significant difference between exantide-treated and untreated islets (n = 11).

Effect of exenatide and immunosuppressants on human islet apoptosis. Caspase-3 activity was measured after 24-h culture with 37 nM FK506, 33 nM rapamycin, or 28 nM MMF in the presence or absence of 1 nM exenatide (n = 11).
Discussion
The major hurdle in human islet transplantation is chronic deterioration of graft function. The objective of the present study was to examine the effects of multiple immunosuppressant drugs on human islets, in vitro and in vivo. Our study has two major findings. First, we demonstrated that 24-h treatment with FK506 or MFF significantly reduced glucose-stimulated insulin section, although through different means. Second, we showed that activating GLP-1 receptors restored the ability of human islet cells to respond to glucose in the presence of immunosuppressants. Considering that the intrahepatic levels of immunosuppressive drugs that transplantated islets could be exposed to may much higher than peripheral level (10,46), combating immunosuppressant toxicity is clinically important. We present evidence that using exenatide in islet cultures and postoperatively may be a feasible approach in the clinic.
Deleterious effects of immunosuppressive agents on β-cells have been documented primarily in immortalized cell lines and rodent islets, but has not been well defined in humans. In our study, we observed different effects in the case of each drug. The differential effect of each of the immunosuppressants examined in this study likely reflects the nonoverlapping nature of their molecular targets. The immunosuppressive actions of FK506 have been attributed to its ability to bind FKBP12 and subsequently block calcineurin, a Ca2+-dependent phosphatase that plays a critical role in T-cell activation (5). However, calcineurin is also present in pancreatic β-cells where it is thought to regulate insulin secretion (4) and β-cell growth (22). FK506 directly, or indirectly via calcineurin, may also modulate critical β-cell ion channels. FK506 can bind FKBP12 and related proteins that act as subunits for intracellular Ca2+ release channels such as the ryanodine receptor (1,7). We have shown that ryanodine receptors regulate β-cell survival (24), and to a lesser extent suppress insulin secretion in human islets (25). FK506 is known to be diabetogenic in animals and humans via direct inhibition of insulin secretion and insulin synthesis human islets and other β-cell models (9, 34,58). For example, low doses of FK506, but not rapamycin, reduced insulin mRNA in long-term human pancreatic cultures (15). Clinical concentrations of FK506 or MMF, but not rapamycin or cyclosporin A, significantly reduced glucose-stimulated insulin release from rat islets after a 24-h incubation (45). In a previous study using perifused human islets, 5-day culture with 1 nM FK506 caused a modest defect in glucose-stimulated insulin secretion relative to an increased basal secretion, whereas 100 nM MMF or 100 nM cyclosporin A were without effect (38). This contrasts with our observation that culture with either FK506 or MMF inhibited insulin secretion in response to high glucose. Our short-term study on dispersed human islets noted an increase in basal insulin secretion in the presence of FK506. Perifusions designed to assay “responsiveness” were normalized to the basal insulin secretion rate to control for possible differences in islet loading. Together our studies and those described above strongly suggest that FK506 can inhibit glucose-stimulated insulin release.
Another calcineurin inhibitor, cyclosporin A, has also been shown to inhibit β-cell function in a number of studies (2,28,31,36,38). Although we did not thoroughly examine cyclosporin A in human islets, as it is not currently used in clinical islet transplant protocols, preliminary studies in human islet cells indicated that it was not as toxic as FK506 (not shown). Our limited studies in mouse islets suggest that it does not directly block glucose-stimulated insulin section.
The immunosuppressant actions of rapamycin are mediated by its actions on mTOR (54). In addition to its role in T-cell proliferation, mTOR is involved in nutrient sensing, insulin signaling, and protein synthesis in β-cells (21,32,42). Rapamycin has been shown in some studies to have deleterious effects on β-cell function and growth (9,34,58). Nevertheless, it seems that the doses of rapamycin used in our studies have relatively modest effects on β-cell function and survival.
Case reports document the switch from FK506 to MMF in clinical islet transplantation to reduce β-cell toxicity (15). However, there have also been studies in islets suggesting toxic effects of this inosine 5′-mono-phosphate dehydrogenase inhibitor. For example, 6-day culture with MMF impaired glucose-stimulated insulin release in isolated rat islets (45), and MMF had antiproliferative effects on pancreatic ductal cells in vitro (18). Our experiments indicated that MMF inhibits insulin secretion from human islets via a mechanism involving ER stress. Previous studies have shown that MMF can induce apoptosis in other cell types (3,48,52), but to our knowledge we are the first to show an apoptotic effect of MMF in β-cells or to implicate ER stress as a mechanism. This is perhaps not surprising, because the chronic depletion of guanine nucleotides induced by MMF might be expected to impair ER Ca2+ homeostasis, ER protein folding, and perhaps insulin secretion (8,20,27). Our experiments on mouse islets suggested that MMF does not have acute effects on insulin secretion, unlike FK506. While all of the immunosuppressants increased human islet apoptosis to some degree, only FK506, with its substantial effects on β-cell secretory function, was associated with graft failure in mice. Grafts from MMF-treated mice appeared to have very robust insulin staining.
The results of this study demonstrate that long-term exposure to FK506 or MMF is toxic to cultured human islets, resulting in reduced glucose-stimulated insulin secretion and increased apoptosis. We asked whether drug toxicity may be reduced or prevented by treatment with the GLP-1 agonist, exenatide. This GLP-1 receptor agonist can protect β-cells against a wide array of insults due to its simultaneous actions on survival and function (12,55). In our study, it is likely that exenatide's action on insulin secretion may be more important for two reasons. First, exenatide did not significantly reverse the increase in caspase-3 activity induced by the immune-suppressants. Second, the secretory defect induced by FK506 was completely resolved, whereas the effects of MMF were only partially abrogated. In any case, our results suggest perioperative treatment of islet cultures with exenatide may be beneficial for islet transplantation. Indeed, the clinical applicability of these results is being tested with a small proof-of-concept clinical pilot study in which seven patients, each whom had been given a previous islet transplant, were given a second transplanted using a modified protocol that included supplementing islet cultures with exenatide prior to transplantation and adding exenatide injections to their medication posttransplant (unpublished data). Thus, a single islet supplement (retransplant) with the GLP-1 agonist exenatide after partial graft failure may be able to restore endogenous insulin production, euglycemia, and insulin independence in patients (19). We did note known side effects of exenatide in our patients, including nausea, resulting in reduced compliance. This limited set of cases, though lacking a parallel set of control subjects, at least suggests that this islet transplantation protocol can be effective as a second transplant. Similar results have been observed by other groups (16,17). We look forward to more extensive and varied trials. The lack of an exendin-only group in our in vitro experiments was a limitation of the current study, which prevented us from evaluating potential effects of GLP-1 receptor signaling on “basal apoptosis” and “baseline function.” In future studies, it will be important to assess the simultaneous effects of multiple immunosuppressants. It will also be important to further characterize the mechanisms by which GLP-1 agonists protect against immunosuppressants and to determine whether this can improve long-term graft function in mice exposed to the immunosuppressant drugs. As mentioned above, such experiments are likely to be translatable to the clinical situation.
In conclusion, we have compared the effects of several immunosuppressants on β-cell function and apoptosis, in vitro and in vivo, at clinically relevant doses. Our results demonstrate that acute FK506 exposure directly inhibits glucose-stimulated insulin release, whereas MMF exerts indirect effects on insulin secretion likely by increasing β-cell ER stress. Rapamycin had milder effects on insulin secretion. FK506, but not MMF or rapamycin, caused graft failure when human islets were transplanted into NOD.SCID mice. Our study also demonstrated that insulin secretory defects induced by immunosuppressants can be ameliorated by exenatide in vitro. The feasibility of applying this approach in vivo is supported by our experience and the results of others (16,17,19).
Footnotes
Acknowledgments
Work was supported by grants from the Canadian Institutes of Health Research (CIHR) (MOP-79414 to G.L.W.; MOP-86559 to J.D.J.; MOP-64427 to C.B.V.), the JDRF (to G.L.W. and J.D.J), and the Canadian Diabetes Association (to C.B.V.). J.D.J. is a Scholar of the Michael Smith Foundation for Health Research (MSFHR) and C.B.V. is a Senior Scholar of the MSHFR. A.K. was supported by a Doctoral Research Award from the Canadian Diabetes Association and K.P. by a studentship from the CIHR RxD program. This study was supported by the British Columbia Transplant Society (BCTS), Vancouver General Hospital, the Vancouver Coastal Health Research Institute, the Woodward Foundation, and the CIHR/MSFHR Training Program in Transplantation Research. Human islet isolation and transplantation were made possible by the I.K. Barber Human Isolation Laboratory at the Vancouver General Hospital. We thank the Michael Smith Foundation for Health Research for funding the Centre for Human Islet Transplant and Beta-cell Regeneration and its Cores that supported many aspects of the work.
