Abstract
Cell transplantation after enclosing in microcapsules has been studied as an alternative approach for treatment of wide variety of diseases. In the present study, we examined the feasibility of using agarose microcapsules, having a cell-enclosing hollow core of 100–150 μm in diameter and agarose gel membrane of about 20 μm in thickness, as a device for the methodology. We enclosed cells that had been genetically engineered to express cytochrome P450 2B1, an enzyme that activates the anticancer prodrug ifosfamide. The enclosed cells were shown to express the enzymatic function in the microcapsules in that they suppressed the growth of tumor cells in medium containing ifosfamide. In addition, a more significant regression of preformed tumors was observed in the nude mice implanted with the cell-enclosing microcapsules compared with those implanted with empty capsules after administration of ifosfamide. Preformed tumors shrank by less than 40% in volume in 6 of the 10 recipients implanted with cell-enclosing microcapsules. In contrast, only 1 in 10 of the preformed tumors in the recipient implanted with empty microcapsules shrank by this amount. These results suggest that agarose microcapsules containing cytochrome P450 2B1 enzyme-expressing cells are feasible devices for improving the chemotherapy of tumors. Thus, agarose microcapsule having hollow cores are generally a good candidate as vehicles for cell-encapsulation approaches to cell therapy.
Introduction
Treatment of diseases by transplanting living cells or tissue fragments enclosed in spherical vehicles with polymeric membranes has been studied since the 1960s (3), and its efficiency was revealed not only in animal studies (1,2,12) but also in several clinical trials (8,14, 15,26). Recent advances in genetic engineering enabling the use of cells as reactors to provide desired proteins have increased the applicability of the methodology to a wider range of diseases (4,9). Advances in the production of spherical devices or microcapsules as protective structures for implanted therapeutic cells also have improved the feasibility of the methodology. Over the past decade, a wide variety of spherical vehicles have been developed using a variety of materials and encapsulation methods for enhancing immunoisolatability, biocompatibility, and mechanical stability (18,27).
Recently, we developed a novel technique for producing microcapsules of about 200 μm in diameter with hollow cores of 100–150 μm in diameter in which enclosed cells grew and formed spheroid-like structures until they almost completely filled the hollow core (21). The size of the microcapsules was smaller than those of conventional cell-enclosing microcapsules or vehicles of 300–1,000 μm in diameter (18,27). Such small vehicles are attractive from the view points of molecular exchangeability, mechanical stability (22), and suppression of foreign body reaction to implanted vehicles (19,25). In addition, smaller vehicles are expected to reduce the surgical trauma for implanting cell-enclosing vehicles because of the reduction in the size of the vehicles' injection device. Moreover, the size of hollow cores was less than the allowable size of 150–200 μm in diameter for multicellular spheroids that does not limit the oxygen supply, resulting in necrotic region formation inside the tissues (5,13).
Microcapsules consisting of an outer shell inside which is a small hollow core containing mammalian cells have not been reported, except for our recent reports (20,21). We demonstrated the efficiency of this technology in creating vehicles for the production of spherical tissues such as embryoid body from embryonic stem cells in vitro. The purpose of this study is to evaluate the feasibility of using agarose microcapsules as cell-enclosing vehicles for cell therapy and we focus especially on the targeted treatment of tumors in this study. Agarose is a natural polysaccharide extracted from the cellular walls of agarophyte seaweed and exhibits temperature-sensitive water solubility. Agarose gels have been widely studied for biomedical applications including spherical vehicles for allotransplantation due to a high degree of biocompatibility (11).
In this study, we enclosed the cells genetically engineered to express cytochrome P450 2B1 (CYP2B1) into the hollow core of our novel agarose microcapsules. By implanting the microcapsules enclosing the cells into or close to tumors, we attempted to develop an alternative site of activation of prodrug, ifosfamide, which normally occurs in the liver. Ifosfamide is a prodrug that is specifically metabolized into two major active compounds, phosphoramide mustard and acrolein, by CYP2B1 in the liver (7). Due to a very short halflife of the activated compounds in plasma, ifosfamide has to be given in relatively high does despite severe side effects associated with such doses. One effective approach for reducing the side effects without lowering response rates involves the establishment of a second site of enzyme conversion near or in the tumor by bringing cells transfected to express CYP2B1 close to tumor (14–16).
Materials and Methods
Materials
Agarose with a low gelling temperature (26–30°C at 1.5%) was purchased from Cambrex Bio Science Rockland (Rockland, ME, USA). Alginate containing 2.8 hydroxyl phenolic moieties per 100 repeat units of uronic acid (Alg-Ph) was synthesized as reported previously (24). Aqueous H2O2 solution (31%, w/w) and liquid paraffin were obtained form Kanto Kagaku (Tokyo, Japan). Lecithin from soybean and horse radish peroxidase (HRP; 170 U/mg) were obtained form Wako Chemicals (Osaka, Japan).
Animals and Cells
Male nude mice (BALB/cA Jcl-nu, 8 weeks old) were purchased from Kyudo (Saga, Japan). The animals were housed in an isolator at 20°C. All the protocols using the animals were according to the recommendations of Kyushu University entitled Guide for the Care and Use of Laboratory Animals.
Feline kidney cells engineered to produce the liver active enzyme CYP2B1 (16), an ifosfamide-activating cytochrome (CYP2B1 cell), were grown in Dulbecco's modified Eagle's medium (DMEM, Sigma) containing 10% fetal bovine serum (FBS), 400 mg/dl glucose, 75 mg/L penicillin, and 50 mg/L streptomycin. They were encapsulated in agarose microcapsules with hollow cores. Human tongue squamous carcinoma cell line SAS was used to establish tumors in nude mice. The cells were grown in RPMI-1640 medium containing the same quantities of FBS, glucose, penicillin, and streptomycin as those in the medium for CYP2B1 cells, respectively. Both cells were cultured in a humidified atmosphere at 37°C with 5% CO2.
Cell Encapsulation
Agarose microcapsules with hollow cores of about 100 μm in diameter and capsule membrane of about 20 μm in thickness were prepared based on a previously described method that has negligible effects on the viability of mammalian cells and results in microcapsules with a single hollow core (21). Briefly, as a template of the hollow core of microcapsules, CYP2B1 cell-enclosing spherical Alg-Ph particles of about 100 μm in diameter were prepared via HRP-catalyzed enzymatic reaction by extruding 1.5% (w/v) Alg-Ph dissolved in calcium-free Krebs Ringer HEPES-buffered solution (KRH, pH 7.4) into a laminar flow of liquid paraffin dissolving H2O2 (0.82 mmol/L) and lecithin (3.0%, w/w). The extruded Alg-Ph solution contained HRP and CRFK cells at 1.7 U/ml and 1.5 × 107 cells/ml, respectively. The emulsion system was collected in a 50-ml plastic tube and incubated for further 10 min to allow enzymatic gelation. After being rinsed using KRH, resultant cell-enclosing Alg-Ph microparticles were suspended in 4% (w/w) agarose in KRH maintained at 38°C. The suspension was extruded into a coflowing stream of liquid paraffin containing lecithin (3.0%, w/w) but not H2O2. The resultant emulsion of ungelated agarose droplets containing Alg-Ph microparticles was collected in a 50-ml plastic tube and then cooled for 10 min in an ice bath for gelation of the agarose solution. The resultant microparticles were collected in KRH and rinsed several times with medium. Next, the microparticles were incubated in medium containing alginate lyase (0.2 mg/ml) for 1 h to liquefy the enclosed alginate microparticles by enzymatic degradation. Growth profiles of the enclosed cells were estimated by increases in the amount of a water-soluble formazan dye, which was derived from a tetrazolium salt dissolved in medium containing suspended cell-enclosing microcapsules, using a colorimetric assay kit (Cell Counting Kit-8; Dojindo, Kumamoto, Japan).
Function of Enclosed CYP2B1 Cells In Vitro
The expression of biologically active CYP2B1 in the cells enclosed in hollow cores of agarose microcapsules was determined using a transwell system. SAS cells suspended in medium were seeded at 2.5 × 104 cells/well in 12-well transwell dishes. After 4 h of incubation, the medium was changed to the medium containing ifosfamide at 1 mM and CYP2B1 cell-enclosing agarose microcapsules were placed into a filter insert at 2.5 × 103 capsules/well. The medium containing ifosfamide was changed to the fresh one containing ifosfamide at 1 mM at 24 h of incubation. The number of SAS cells was counted further at 24 h of culture using a hemocytometer.
Treatment of Tumors
SAS cells suspended in saline was injected subcutaneous into the backs of nude mice (3.0 × 106 cells suspended in 0.25 ml saline/animal) using a 26-gauge syringe. After 11 days of growth, the mice with tumors of >350 mm3 were divided into two groups consisting of 10 animals each. The volumes of tumors were calculated based on the measurement of the greatest, a, and least, b, diameters of the tumors using the following standard formula: volume = (a x b2) x 0.5. The recipients of one group received CYP2B1 cells enclosed in agarose microcapsules (containing 3.0 × 105 cells) and the recipients of the other group served as a control and were implanted with the same number of microcapsules (1.0 × 104 microcapsules per recipient). The microcapsules were injected into each tumor after suspending in saline using a 22-gauge syringe. One day after the implantation, ifosfamide dissolved in saline was injected into the peritoneal cavities of the animals at 100 mg/kg body weight. The injection was done every 3 days for the first 9 days and then every 6 days until day 28. After the period, the mice were euthanized by an overdose of diethyl ether.
Statistical Analysis
Comparisons among groups were carried out using one-way ANOVA and the Bonferroni-corrected t-test. Values of p < 0.05 were considered to be significant.
Results
Cellular Growth in Microcapsules
To determine the growth of the CYP2B1 cells in the hollow core of agarose microcapsules of about 200 μm in diameter, we measured transition of mitochondrial activity of cells per microcapsules for estimating growth of enclosed cells and observed changes in their morphology. The mitochondrial activity gradually increased with prolonged culturing (Fig. 1), where 46.7-fold increase in activity was achieved after 3 weeks of culturing compared to day 1. The area that the cells occupied in the microcapsules increased with increasing culture period (Fig. 2a, b). The cells existed individually in the microcapsules immediately after preparation of microcapsules (Fig. 2a) self-aggregated and formed cellular aggregates in hollow cores (Fig. 2b).
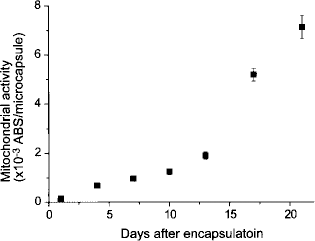
Transition of mitochondrial activities of cells enclosed in microcapsules. Error bars represent mean ± SD (n = 3).

Microphotographs of CYP2B1 cells: (a) immediately after encapsulation, (b) incubated in vitro for 6 days, and (c) recovered from in vivo after 6 days of implantation. Scale bars: 150 μm.
We also studied the potential ability of the enclosed cells to grow in vivo by implanting the CYP2B1 cell-containing agarose microcapsules into the peritoneal cavities of nude mice. The microcapsules containing the cells were recovered after 6 days of implantation by a peritoneal lavage with saline after the animals were sacrificed under deep anesthesia with ether. As can be seen in Figure 2c, the cores of these capsules were filled with cells. A comparison of the cells in the hollow core of microcapsules grown in cell culture medium (Fig. 2b) with those explanted from mice (Fig. 2c) revealed that the latter grew faster.
CYP2B1 Activity Expression in Microcapsules
Maintenance of expression of the enzymatic function of CYP2B1 cells after encapsulation in the agarose microcapsules with hollow cores was determined by studying the inhibition of growth of the SAS cells in the medium containing either ifosfamide or CYP2B1 cell-enclosing microcapsules. The number of SAS cells cultured in the medium containing either of them increased to about 4.5-fold after 48 h (Fig. 3a), indicating that there were no significant differences in the number of cells cultured in the media containing either ifosfamide or cell-enclosing microcapsules (p > 0.05). In contrast, the cells cultured in the medium containing both ifosfamide and CYP2B1 cell-enclosing microcapsules showed a significant suppression of growth of the SAS cells (p < 0.05). The number of cells after 48 h of culture with both microcapsules and ifosfamide was 30% of those in the medium containing either of them alone (p < 0.05). Morphological differences between the cells (Fig. 3b–d) supported theses results. We could not find morphological differences between the cells in the media except for that containing both ifosfamide and cell-enclosing capsules where there were fewer cells attached to the culture dish and correspondingly more cell death.

(a) Numbers and (b–e) morphology of SAS cells after 48 h of culture in 12-well cell culture dish. Wells containing (b) SAS cells (SAS), (c) SAS cells + 1 mM ifosfamide (SAS+ifo.), (d) SAS + CYP2B1 cell-enclosing capsules (SAS+cap.), and (e) SAS cells + 1 mM ifosfamide + CYP2B1 cell-enclosing capsules (SAS+ifo.+cap.). SAS cells were seeded at 2 × 104 cells/well. Error bars in (a) represent mean ± SD (n = 3). +p > 0.05 versus the groups except for SAS+ifo.+cap. *p < 0.05 versus all other groups. Scale bars (b–e): 300 μm.
Preformed Tumor Regression After Implanting Microcapsules
To investigate the effects of CYP2B1 expression from the enclosed cells in agarose microcapsules on ifosfamide treatment of preformed tumors, time course changes in volumes of the preformed tumors were measured. The tumor volumes in the recipients of cell-enclosing microcapsules decreased more significantly than those in the recipient of empty microcapsules (Fig. 4). In the former group, the preformed tumors shrank by less than 20% volume in one recipient on day 6. The number of the recipients with tumors of less than 20% volume of the initial preformed tumors increased with increasing experimental period by day 15. On day 15, only one mouse had tumor of more than 60% volume of that of the preformed tumor. At the end of the experiment (i.e., at 28 days), 4 of 10 recipients implanted with cell-enclosing microcapsules had tumors of less than 20% volume of preformed tumors. In contrast, the degree of tumor regression was slower in the recipients implanted with empty microcapsules compared with those implanted with cell-enclosing microcapsules. On day 15, five recipients of empty microcapsules had tumors of more than 60% volume of preformed tumors. At the end of experiment, there were no recipients having the tumors less than 20% volume of preformed tumors.

Photographs of mouse subcutaneously xenotransplanted human tumor: (a) just before injection of the CYP2B1 cell-enclosing microcapsules, and (b) 21 days after treatment with ifosfamide. (c) Preformed tumor size changes of the recipients implanted empty and CYP2B1 cells-enclosing capsules. Ten nude mice each were used as recipients of empty and cell-enclosing capsules. Tumor size just before capsules implantation was set to 1 and each block represents the proportional size of the original tumor.
Discussion
The focus of our research was to evaluate the feasibility of the agarose microcapsules smaller than conventional cell-enclosing spherical vehicles and with liquefied hollow cores of 100–150 μm in diameter as vehicles for treatment of diseases using the functions of enclosed cells. In this study, we examined the treatment of tumors using small microcapsules. To accomplish our objective we encapsulated cells that had been genetically engineered to stably express CYP2B1. Implantation of the microcapsules into or close to tumor was expected to allow us to establish a second site for activating prodrugs for tumors that normally occurs in the liver, which is a major site for the expression of CYP2B1 as well as other cytochrome P450s (14). In a previous study, we implanted CYP2B1 cells enclosed in agarose microparticles smaller than conventional cell-enclosing spherical vehicles but with solid cores to the mice with subcutaneous preformed tumors (23). Drawbacks to the use of these solid core agarose microparticles included hindrance of cellular growth and occurrence of drastic reduction of viability of the enclosed cells after 1 week of encapsulation, due to microscopic stress forces from the surrounding agarose gel (10). Orive and coworkers demonstrated the effectiveness of liquefaction of spherical vehicles' core for improving viability and antibody production of enclosed cells (17). In our system, the cells initially enclosed in Alg-Ph microparticles were relieved from the stresses from surrounding Alg-Ph gel by enzymatically liquefying the gel after the formation of agarose gel layer on the microparticles.
In this study, we first studied the growth of CYP2B1 cells enclosed in the hollow core of agarose microcapsules. As we have previously observed with the mouse embryonic stem cells enclosed in hollow cores of agarose microcapsules (21), we could detected growth of the encapsulated CYP2B1 cells in vitro (Figs. 1, 2a, b). In addition, their growth was also demonstrated in vivo by collecting the implanted microcapsules from peritoneal cavity of animals (Fig. 2c). Comparing the cells at the same time point after encapsulation (i.e., 6 days), the cells in the microcapsules implanted in vivo grew faster than those cultured in vitro (Fig. 2b, c). The faster growth of cells in vivo was interpreted as a consequence of the difference in the kind of nutrients and growth factors available in continuous supply to the enclosed cells in body fluids compared with cell culture medium, which become depleted. The cellular growth in the hollow cores of implanted microcapsules indicates the usefulness of the agarose microcapsules not only from the view points of biocompatibility and molecular exchangeability between the enclosed cells and surrounding environment resulting from smaller diameter compared with conventional cell-enclosing capsules but also prolongation of therapeutic effects in vivo.
Next, we studied the expression of CYP2B1 after encapsulation. The significant suppression of growth of SAS cells only in the medium containing both ifosfamide and cell-enclosing agarose microcapsules (Fig. 3) clearly showed the expression of CYP2B1 in the cells enclosed in the microcapsules. We thus performed tumor regression study by implanting CYP2B1 cell-enclosing microcapsules into the mice with preformed tumors. The faster reduction of tumor volumes in the recipients implanted with CYP2B1 cell-enclosing microcapsules compared to those implanted with empty microcapsules under dosing ifosfamide (Fig. 4c) showed that even in vivo the enclosed cells expressed CYP2B1, acting as an alternative site for activating ifosfamide. The slower but measurable reduction of tumor volumes even in the recipients of empty microcapsules can be explained by the endogenous activation of ifosfamide by the liver of these mice (7).
There have been reports about the successful treatment of diseases using cell-enclosing spherical vehicles in clinical trials including treatment of tumors (8,14,15, 26). In the report for treatment of pancreatic tumor (14,15), cell-enclosing vehicles of 800 μm in diameter were placed into a suitable artery feeding a primary tumor. In contrast, the size of the microcapsules used in this study is smaller than the allowable size of multicellular spheroids that does not limit the oxygen supply, resulting in necrotic region formation inside the tissues (5,13). Formation of a necrotic region inside spherical tissues enclosed in microcapsules resulting in decreased functionality has been one of obstacles of the success of transplantation of enclosed cells (6). Thus, the agarose microcapsules used in this study with hollow cores would appear to be promising as vehicles for cell therapy from the view point of suppression of necrotic region formation resulting from limited oxygen supply compared with conventional-sized spherical vehicles.
In summary, the feasibility of using agarose microcapsules with hollow cores of 100–150 μm in diameter and about 20 μm in agarose gel membrane layer for treatment of tumor was shown by enclosing CYP2B1 cells in the hollow core and implanted into tumors.
Footnotes
Acknowledgments
This work was supported by a Grant-in-Aid from the Global COE Program, “Science for Future Molecular Systems,” from the Ministry of Education, Culture, Science, Sports, Japan.
