Abstract
Reliable bone regeneration can be achieved with a pellet culture system using bovine periosteal cells. However, bone regeneration and neovascularization processes in this system have remained unclear. The present study aimed to clarify the extracellular environment and neovascularization process. To detect components of the extracellular matrix secreted by cells and to identify the conditions necessary for bone regeneration in the body, Western blotting and in vivo tests in nude mice were performed. Cells were cultured with or without ascorbic acid and culture supernatant was precipitated. Western blotting showed that culture supernatant contained collagen type I, procollagen type I, and procollagen type I C-terminus when cells were cultured with ascorbic acid. Cells cultured with ascorbic acid formed partial bony tissues at 2 weeks after grafting to nude mice, while bone formation was missing without ascorbic acid. Immunostaining was performed using species-specific vascular endothelial cell markers to ascertain whether vascular endothelial cells were bovine or murine (nude mouse). Immunohistological methods showed vascular endothelial cells in osseous tissue formed in the subcutaneous tissue of nude mice were murine. Extracellular matrix synthesis in vitro and host blood flow in vivo are essential for bone regeneration.
Keywords
Introduction
With existing tissue engineering techniques, regeneration is attempted using cells, scaffolds, and growth factors (5,17,26). However, with tissue engineering, adhesion between cells and scaffolds is uncertain, and the effects of scaffolds on cells and the body have not been clarified (8,15,20,22,23). Conversely, with a pellet culture system or other scaffold-free methods, cells themselves synthesize extracellular matrix to act as a natural scaffold (3,21). Pellet culture systems were initially used to regenerate cartilage, but have been used in recent years to regenerate tissues other than cartilage (2,4,19, 21). However, tissue regeneration by a pellet culture system is not necessarily possible for some cell types (19). With the pellet culture system using bovine periosteal cells that we have reported in the past, reliable bone regeneration was achieved (1). The present study investigated why pellet culture using bovine periosteal cells is efficient.
Self-assembly of extracellular matrix is now in the spotlight as the environment for developing tissues or scaffolds for cell attachment (7,9,14). Ascorbic acid is known to be essential for collagen deposition, while fibroblasts and other kinds of cells have been found to secrete procollagen into the culture medium (11,18). Graham et al. (11) studied procollagen secretion by human intestinal smooth muscle cells in both the cell layer and culture medium, revealing differences in the proportion of procollagen in the cell layer and culture medium. Eleftherriades et al. (9) found that intracellular accumulation of type I procollagen decreased extracellular type I collagen. Intracellular accumulation of type I procollagen does not necessarily clarify the extracellular environment. The present study used a small amount of culture supernatant (1 ml) for each sample, to clarify the extracellular environment.
Neovascularization is also essential for tissue regeneration and research has been conducted to clarify this issue. For neovascularization, techniques such as scaffolds containing endovascular cells and vessel grafts have been used (10,12,24). These vascular endothelial cells and vessel grafts have been shown to bind with host vessels (25). In our previous report, vessels were identified in osseous tissue formed from bovine periosteal cells grafted into the subcutaneous tissue of nude mice, but whether neovascularization occurred because cell pellets included bovine endovascular cells and precursor vascular endothelial cells was unclear (1). Using two vascular endothelial cell markers that stain differently in different animal species, immunostaining was thus performed to ascertain whether vascular endothelial cells were bovine or murine (nude mouse) and to investigate the neovascularization process. The goal of the present study was to clarify the extracellular environment and neovascularization process and to identify the conditions necessary for bone regeneration in the body.
Materials and Methods
Periosteal Cell Culture Conditions
Primary periosteal cells were cultured as described previously (1). Briefly, periosteum was detached from a 30-month-old bovine forelimb, and explant culture was performed using 10% fetal calf serum and antibiotics. Cells were cultured with or without ascorbic acid. Culture solution was exchanged once a week and cells were cultured using tissue culture dishes for up to 5 weeks without subculturing.
Western Blotting
Cells were cultured with or without ascorbic acid to determine the components of extracellular matrix secreted by periosteal cells in the culture solution, and culture supernatant was recovered. No cells were present at the start of incubation, and the number of cells was low even after 2 weeks of incubation. Culture supernatant was thus recovered after 3, 4, and 5 weeks. All proteins in culture supernatant were precipitated using a Proteo-Extract Protein Precipitation Kit (Calbiochem, Darmstadt, Germany) and dissolved in Laemlli sample buffer solution. Pre-electrophoresis conditions varied depending on antibodies used, with heating for 3 min using β-mercaptoethanol when using mouse monoclonal antibovine collagen type I antibody (Abcam, Cambridge, UK), and no reduction or heating when using mouse monoclonal anti-human procollagen type I C-terminus antibody (TaKaRa Bio, Shiga, Japan). For each lane, 20 μg of protein was subjected to electrophoresis in 7.5% polyacrylamide gel and transferred to a membrane. As the primary antibody, collagen type I antibody with 1,000-fold dilution and procollagen C-terminus antibody with 1,000-fold dilution were used overnight. As secondary antibody, Envision polymer with 50-fold dilution (DAKO Cytomation, Carpinteria, CA) was used, and bands were detected by chemical luminescence. Independent experiments were repeated three times. Also, to sever the C-terminus of procollagen, pepsin was added to the culture supernatant for 2 h at room temperature. After neutralizing with Tris buffer, concentration was performed in the same manner, and Western blotting was performed using collagen type I antibody.
Grafting to Subcutaneous Tissue of Nude Mice
With or without ascorbic acid, cells were cultured at 37°C with 5% CO2 for 5 weeks. As reported in the previous study, a pellet was made by centrifuging cells, and without a scaffold, the pellet was grafted subcutaneously to the back of a nude mouse under anesthesia (1). Cells cultured with ascorbic acid were grafted for 2 weeks (n = 4). As cells cultured without ascorbic acid disappeared from the grafted area, pellets grafted from 5 days to 2 weeks (n = 4) were used for histological analyses. Guidelines for animal experimentation at Osaka Dental University (No. 07-02032) were followed for all animal experiments.
Neovascularization
OCT compound (Miles, IN, USA) and a cryostat were used to prepare frozen sections of nude mouse tissue, bovine tissue, bovine vascular tissue grafted to nude mice for 2 weeks, and bovine periosteal cell pellets grafted to nude mice for 2 weeks. Immunostaining was performed by targeting von Willebrand factor (polyclonal rabbit anti-human von Willebrand factor, #A0082; DAKO Cytomation) and CD31 [rat anti-mouse CD31, #732116 (clone 390); Beckman Coulter, CA, USA]. As reported by the manufacturer, von Willebrand factor antibody cross-reacts with cow, horse, mouse, swine, chicken, dog, and rat, so the antibody is presumed to be expressed in both bovine and murine vascular endothelial cells. CD31 antibody is expressed weakly, so the antibody reacts only on frozen sections and cross-reaction is uncertain in animals other than mouse. Bovine periosteal cells grafted in this experiment were cultured for 5 weeks in vitro with ascorbic acid. Using von Willebrand factor primary antibody with 2,000-fold dilution, Envision Polymer was used to incubate sections. After using CD31 primary antibody with 300-fold dilution, mouse anti-rat secondary antibody [#732695 (clone 2a8F4); Beckman Coulter] with 1,000-fold dilution was used. Sections were stained using 3,3′-diaminobenzidine (DAB) and contrast staining was performed using hematoxylin.
Results
Components of the Extracellular Matrix Secreted by Periosteal Cells
Using collagen type I antibody, two bands were seen for samples from weeks 3, 4, and 5 with ascorbic acid, but without ascorbic acid, no bands were seen (Fig. 1a). When using procollagen C-terminus antibody, a single band was seen with samples from weeks 3, 4, and 5 with ascorbic acid, but an obscure band was observed without ascorbic acid (Fig. 1b). Also, the terminus of procollagen was severed by adding pepsin to the culture supernatant, and when collagen type I antibody was used for band detection, a single band was seen for the samples at weeks 3, 4, and 5 with ascorbic acid (Fig. 2).
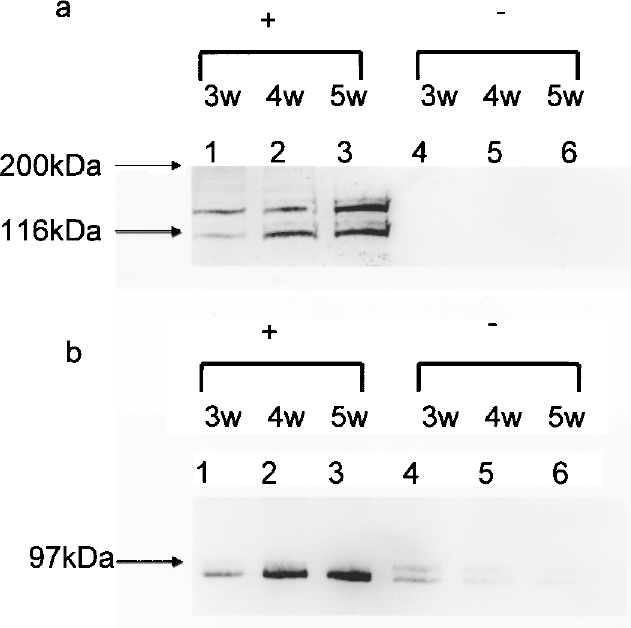
Representative Western blot of (a) collagen type I and (b) procollagen type I C-terminus expression. Cells were cultured up to 5 weeks with ascorbic acid (+, lanes 1, 2, 3) or without ascorbic acid (–, 4, 5, 6). Proteins in culture supernatant were precipitated.

Representative Western blot of collagen type I. Cells were cultured up to 5 weeks with ascorbic acid (+, lanes 1, 2, 3). To sever the C-terminus of procollagen, pepsin was added to the culture supernatant for 2 h at room temperature, then proteins in culture supernatant were precipitated.
Essential Elements for Bone Regeneration
When cells that were cultured for 5 weeks with ascorbic acid were grafted to the subcutaneous tissue of nude mice, partial bone formation was seen at 2 weeks after grafting (Fig. 3a, b). Numbers of red blood cells were observed in the grafted tissue (Fig. 3c, d). When cell pellets incubated without ascorbic acid were grafted to nude mice, pellets shrank and disappeared with time, so tissue sections could not be prepared more than 2 weeks after grafting (Fig. 4a). Preliminary examination revealed that pellets shrank after approximately 1–2 weeks during observation over mouse skin, with no cells remaining after 6 weeks. In this study, three cell pellets were implanted for 5 days and one pellet for 2 weeks to avoid disappearance. Figure 4a–c shows images 2 weeks after grafting. When incubated without ascorbic acid, bone formation was missing and only some fibroblast-like cells and inflammatory cell infiltration with cells such as neutrophils or lymphocytes were present (Fig. 4b, c). Addition of ascorbic acid to culture solutions was associated with bone regeneration for a pellet culture system using bovine periosteal cells.
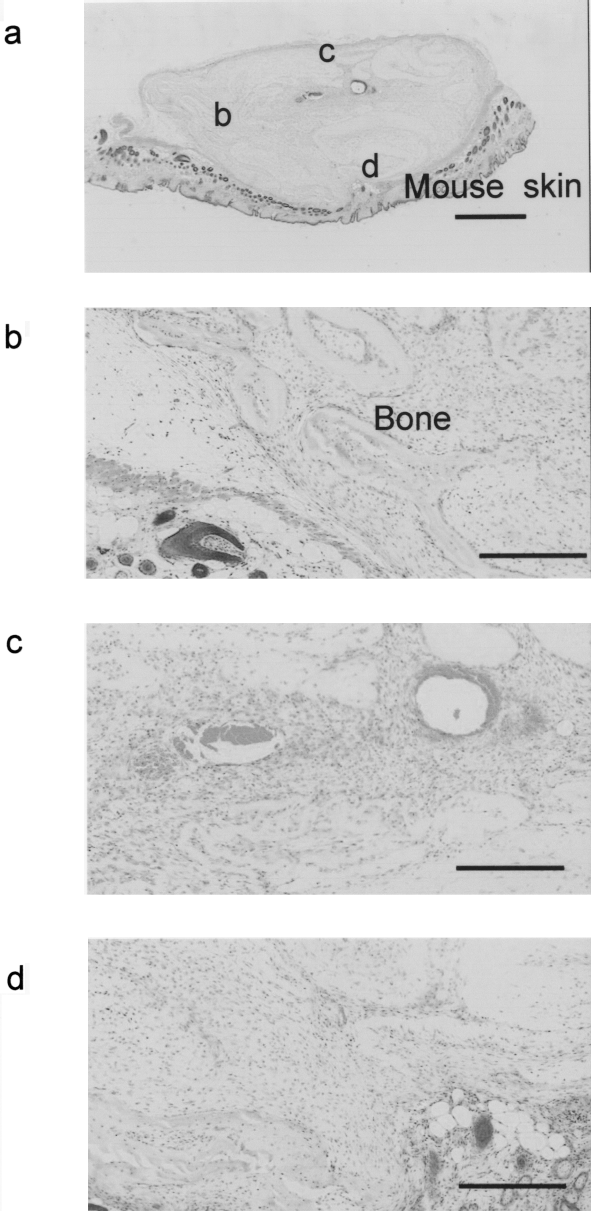
Hematoxylin and eosin-stained (a) implants of bovine periosteal cells cultured with ascorbic acid (+). (b–d) High-power images of the area indicated in (a). Scale bar: (a) 1 mm; (b–d) 300 μm. Images were taken 2 weeks after grafting.

Hematoxylin and eosin-stained (a) implants of bovine periosteal cells cultured without ascorbic acid (–). (b) High-power image of the area indicated in (a). (c) High-power image of the area indicated in (b). Scale bar: (a) 1 mm; (b) 300 μm; (c) 100 μm. Images were taken 2 weeks after grafting.
Staining of Vascular Endothelial Cells Between Two Animal Species
As reported by the manufacturer, von Willebrand factor antibody cross-reacts with both murine and bovine tissue and CD31 antibody reacts with products from mice, but reaction with bovine material is uncertain. In the present study, von Willebrand factor was expressed in nude mouse tissue, bovine tissue, and bovine vascular tissue grafted to nude mice tissue, but CD31 antibody was only expressed by nude mice tissue, not in bovine tissue or bovine vascular tissue grafted in nude mice (Figs. 5a–d, 6a, b). The present results show that nude mouse vascular endothelial cells were stained by both antibodies, while bovine vascular endothelial cells were stained only by von Willebrand factor antibody, clarifying that subcutaneous grafts into nude mice were stained by both von Willebrand factor and CD31 antibodies (Fig. 6c, d).
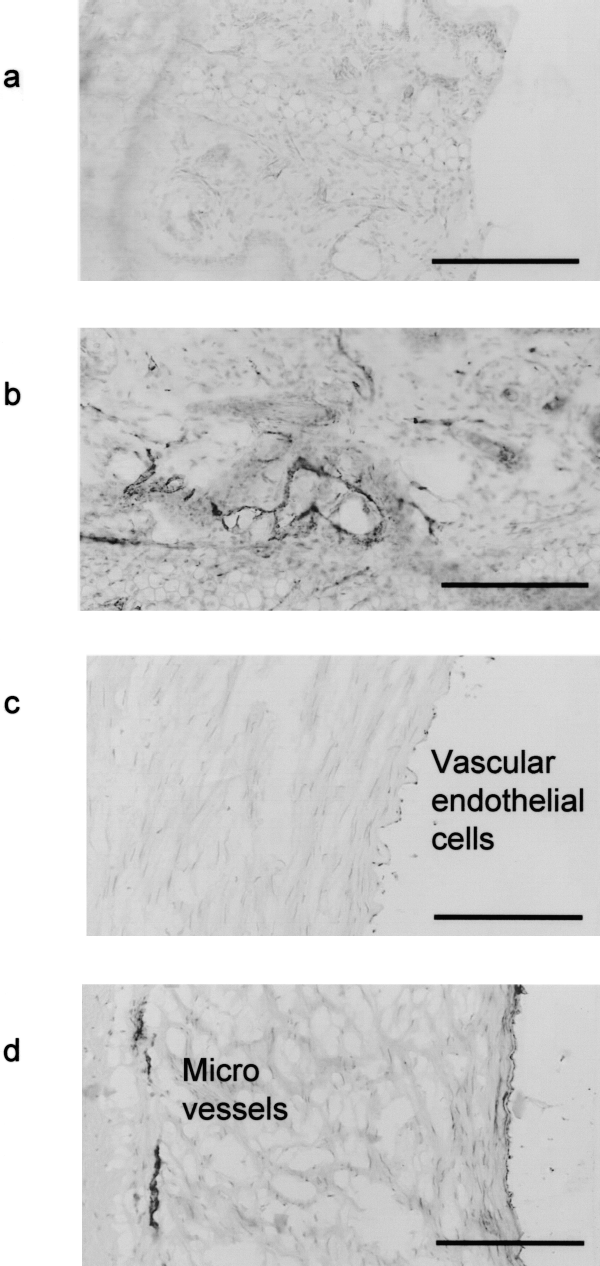
Immunohistochemistry. (a) Anti-CD31 staining of mouse ear; (b) anti-von Willebrand factor staining of mouse ear; (c) anti-CD31 factor staining of bovine vascular tissue; and (d) anti-von Willebrand factor staining of bovine vascular tissue. Scale bar: 200 μm.
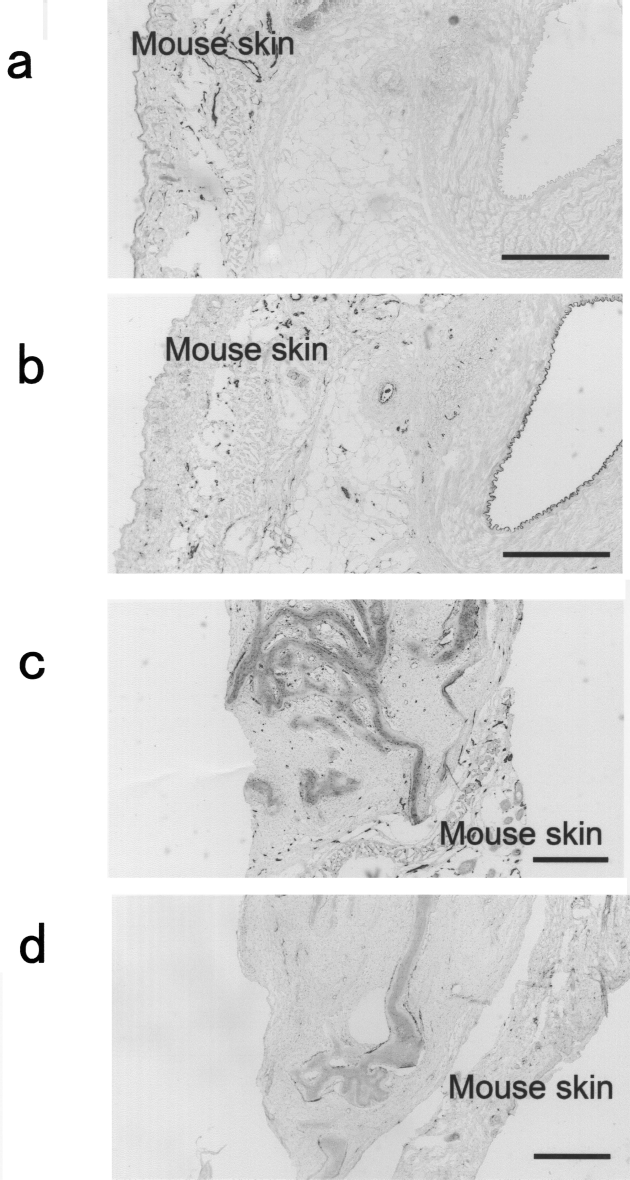
Immunohistochemistry. (a) Anti-CD31 staining of bovine vascular tissue grafted to nude mice; (b) anti-von Willebrand factor staining of bovine vascular tissue grafted to nude mice; (c) anti-CD31 staining of bovine periosteal cells grafted to nude mice; (d) anti-von Willebrand factor staining of bovine periosteal cells grafted to nude mice. Scale bar: (a, b) 300 μm; (c, d) 500 μm. Images were taken 2 weeks after grafting.
Discussion
Essential Factor for Bone Regeneration
Western blotting was performed to detect proteins in culture supernatant using collagen type I antibody and procollagen type I C-terminus antibody. When ascorbic acid was added, both collagen type I and procollagen type I C-terminus were detected. Using collagen type I antibody, two bands were detected because collagen type I antibody cross-reacted with the chain region of procollagen. Therefore, of the two bands, the upper band (<200 kDa) indicated procollagen, while the lower band (approximately 116 kDa) represented collagen. In 1995, Graham et al. (11) determined procollagen synthesis and secretion by the incubation of cells with L-[5-3H]proline, but did not detect collagen type I. The present study detected both collagen and procollagen. With tissue engineering, scaffolds are essential. However, the scaffold-free pellet culture system resulted in bone regeneration in this study. This is because collagen type I secreted by bovine periosteal cells organized extracellular matrix. After pepsin treatment, only a single band for collagen was seen by severing the C-terminus from procollagen. These results clarified that both procollagen and collagen exist in the culture solution after 3 weeks. Ascorbic acid is necessary to catalyze the posttranslational hydroxylation of procollagen (11) and is involved in collagen biosynthesis. Collagen synthesized by cells was then deposited to function as extracellular matrix, not only facilitating pellet culture, but also promoting bone regeneration and neovascularization in vivo. In a preliminary study, when Western blotting was performed by directly mixing culture supernatant with Laemlli, fluctuations in bands were seen (data not shown), but using a protein concentration kit, culture solution components were removed and clear bands could be detected. The protein concentration kit did not cause target proteins in the present study to degrade. When analyzing culture supernatant by sandwich enzyme-linked immunosorbent assay (ELISA), background noise by fetal calf serum makes accurate measurements impossible. However, with Western blotting in the present study, the effects of fetal calf serum were minimized thanks to membrane blocking and molecular weight separation by electrophoresis. When using procollagen type I C-terminus antibody, samples were not reduced or heated, to avoid destroying the bridging structure of the C-terminus trimer. Without reducing and heating, a clear band was detected for procollagen type I C-terminus.
Screening of Culture Media for Regeneration
In the present study, cells were cultured with or without ascorbic acid. When cells were cultured with ascorbic acid, procollagen and collagen existed in the culture supernatant (in vitro) and osseous tissue was regenerated when grafted subcutaneously to nude mice (in vivo). Moreover, bone formation began at 2 weeks after grafting. The results were reversed without ascorbic acid. These findings suggest that with pellet culture systems, ascertaining the existence of extracellular matrix in the culture supernatant, is useful for predicting the success of tissue regeneration in the body. In 2001, Cecconi et al. (6) reported that screening of conditioned media might provide more reliable parameters. They concluded that electrophoretic protein patterns of conditioned media by cumulus-corona cells are useful to predict fertilization and pregnancy, while the presence of a 31-kDa band is associated with failed implantation. However, useful methods to predict in vivo tissue regeneration do not appear to have been reported. Screening of 1 ml of culture supernatant by bovine periosteal cells may reliably predict bone regeneration. Without ascorbic acid, creating a cell pellet is difficult due to the loose cell-to-cell attachment. Subsequent grafting to nude mice thus results in absence of bone formation. Thus, 5-week culture periods determined the results of in vivo testing. In the present study, to ascertain extracellular environments, concentrated culture supernatant precipitates were used as protein samples. Intracellular protein changes were thus unclear, but the in vivo results appear to correlate to extracellular environments. Only primary bovine periosteal cells were used in this study, but in the future, culture supernatant of subcultured cells, cells of animals other than cows and cells derived from other tissues will need to be examined to ascertain whether in vitro results are indicative of in vivo findings. In our past study, hematoxylin and eosin and von Kossa staining and osteocalcin and type I collagen immunostaining were performed to confirm that tissue growth actually represented bone (1). The results of these assays agreed. Staining with hematoxylin and eosin was used for representative results in this study.
Neovascularization Process
Periosteal cells do not represent a homogenous cell population. Using von Willebrand factor and CD31 antibodies, whether vascular endothelial cells were bovine or murine was ascertained, and the results showed that vascular endothelial cells with bone regeneration and neovascularization in the subcutaneous tissue of nude mice were host nude mouse cells (Fig. 6c, d). Neovascularization might be caused by periosteal cells through various mechanisms. First, as reported by Ishikawa et al. (13), the collagen-binding protein might promote neovascularization in situ. Second, cell pellets made by centrifuging cells might display openings for the development of a vascular supply (1). In 1997, Lehr et al. (16) used mouse-specific (i.e., host) or human-specific CD31 antibody to investigate the host origin of endothelium in human tumor xenografts in athymic nude mice. They demonstrated that the endothelium of the vasculature in human xenografts expressed mouse-specific (i.e., host) CD31. Those results agree with the present findings. Sieminski et al. (25) described bovine aortic endothelial cells incorporated into the host vasculature within the implant. However, the results of the present study suggest that even when vascular endothelial cells or vessel grafts are absent on the implant side, host blood flow alone results in neovascularization. This is because rich synthesized extracellular matrix produced by cultured cells serves as a good platform for neovascularization. Another explanation is that, as Sieminski et al. (25) described, nonendothelial cells such as fibroblasts modulate microvascular networks in vivo. CD31 antibody was only expressed by frozen sections of mouse tissue, not in bovine tissue. Accordingly, to test CD31 antibody for species-specific vascular endothelial cell markers, bovine vascular tissue was grafted into nude mice for 2 weeks. As a result, CD31 antibody was not expressed in grafted bovine vascular tissue. Mouse tissue thus appears to react to both von Willebrand factor and CD31 antibody, while bovine tissue reacts to von Willebrand factor antibody only. The present study confirmed the existence of vessels in cell pellets at 2 weeks after grafting. Also, in past studies, vessels existed in cell pellets with diameters of approximately 1 cm, and necrotic cells were not seen in the center (1). In the absence of capillaries, implants with cells have been accepted as able to avoid necrosis if thickness is less than 2 mm (12). However, in the present experiment model, even when relatively large cell pellets were grafted, cells did not become necrotic and neovascularization was seen relatively early. Thus, due to early formation of vessels, almost all cells in the subcutaneous pellet were thought to survive and participate in bone regeneration (Fig. 3a).
Conclusions
Western blotting showed that the culture supernatant of bovine periosteal cells contained collagen type I, procollagen type I, and procollagen type I C-terminus. Immunohistological analyses showed that vascular endothelial cells in osseous tissue formed in the subcutaneous tissue of nude mice were murine. Extracellular matrix synthesis in vitro and host blood flow in vivo are essential for bone regeneration.
Footnotes
Acknowledgments
The authors would like to thank Kobe Chuo Chikusan for the bovine legs. This work was supported by a Grant-in-Aid for Young Scientists (B) No. 18791465 from the Ministry of Education, Culture, Sports, Science and Technology (MEXT). This study was performed using Institute of Dental Research, Osaka Dental University (Photograph-Processing Facilities, Morphological Research Facilities, Dental Bioscience Facilities, Laboratory Animal Facilities).
