Abstract
Obtaining accurately staged rat embryos can be difficult because of the variety of breeding protocols employed and because precise staging cannot be confirmed until excision of the embryos from the dam. The detection of estrus, pairing of animals, and confirmation of pregnancies is generally left to commercial suppliers, as in-house breeding can be laborious and unpredictable. Here we describe a simple, reliable in-house breeding protocol for the generation of accurately staged embryos as assessed by measurements of average crown to rump length (CRL).
Introduction
Recent work in a rat model of Parkinson's disease and repair has emphasized the crucial role that donor age can play in optimizing survival and integration of donor tissue (8,14,15,17,22,23). Other lines of research, for instance investigating the timing of gene induction (9,18,20) or cell interactions (3,11,19) during embryonic development, also rely on accurately staged embryos. A factor hampering the reproducibility of thus obtained results is the lack of details in many published reports as to how the embryos of the desired age or stage of development were obtained.
Both animal suppliers and researchers carrying out their own in-house breeding employ a variety of methods in the routine production of pregnant rat dams. For instance, there does not appear to be a consensus on the detection of female eligibility; both vaginal smears (1,12,13,21) and vaginal impedance measurements (2, 21) are used. Similarly, there is no standard on the duration of pairings or on the average number of pregnancies obtained from a given number of pairings. A further important issue in embryo staging is the discrepancy between laboratories in assigning embryonic day zero (E0) (5,24). Some laboratories assign E0 to the day of pairing; others register the day following overnight pairing as E0. Hence, embryos produced according to these differing standards may be as much as a whole day apart in their development (24).
Recent work in our laboratory (24) established a staging scale based on crown–rump length (CRL) of rat embryos for donor ages E11 to E17. Furthermore, it outlined the “Charles River Cardiff Protocol” established in collaboration with the animal supplier Charles River, which allowed the procurement of accurately staged embryos (e.g., CRL 5.89 ± 0.21 mm at E12, 10.52 ± 0.38 mm for E14). In order to increase the level of control and cost-effectiveness for the experimenters, the aim of the current work was to replicate this commercial protocol in-house and to devise an improved and user-friendly method of breeding rats, using a short pairing time, that produces reliable pregnancies and accurately staged embryos. Here estrus was determined using vaginal lavage (1,4,7,12,21); pregnancy was confirmed by anesthetization of the dam and palpation of the abdomen 11 or 12 days postmating (6,16). Embryos were harvested at the same time as pairing had occurred (1000–1300 h) 11 to 14 days (E11–E14) postmating. Here we report data collected over 31 breeding sessions, collected in the course of breeding embryos for transplant donations for experiments to be reported elsewhere.
Materials and Methods
Animals
All animals were housed and killed in accordance with the UK Animals (Scientific Procedures) Act 1986 and with current Home Office regulations. Our breeding colony tends to include 6 male and up to 30 female Sprague-Dawley rats (Charles River, UK) at any one time, which are restocked as required. All animals were housed in either normal 12-h light/12-h dark (lights on 0700 to 1900 h) or reversed light cycle rooms (lights on 1900 to 0700 h) with ad libitum supply of food (Harlan, UK) and water. Sires were kept individually while dams shared cages in groups of four. Thirty-seven pregnant dams were used in this study. Thirty-eight embryos from three litters were measured at E11; 24 (two litters) at E11.5; 335 (26 litters) at E12; 14 (one litter) at E13; and 63 embryos (five litters) at E14.
Determination of Stage of Estrus Using Vaginal Lavage
After an acclimatization period of at least 1 week after arrival from the supplier (Charles River, UK), a timetable of estrus cycles was established using the vaginal smear method (12). In short, the dam was restrained lightly in one hand and turned on her back while approximately 100–200 μl of sterile saline (Fisher, UK) was flushed into the vagina via a clean, fire-polished, glass pipette, with a rubber bulb, sucked back into the pipette, and discharged onto a microscope slide to be analyzed under a light microscope (Leica, Germany) at 10× magnification. This was achieved with apparently minimal discomfort for the animal and with good between-user reliability. To view the collected cells, it was not necessary to apply a stain; simply closing the condenser diaphragm introduced enough contrast to make the cells discernible. Depending on the relative proportions of three different cell types (nucleated epithelial cells, cornified epithelial cells, and inflammatory leucocytes) in the sample, smears were assigned to one of four stages, namely: proestrus, estrus, metestrus, and diestrus (Fig. 1) (4,12). Most animals (1,12,21,25) seemed to cycle through these phases in the course of 4 days, while others did so with certain irregularities as has been described previously (1,25). With animals living in a normal 12-h light/dark cycle room, eligible females in estrus were established by lavage in the morning of the intended pairing. Females in full estrus were selected for breeding and paired with an experienced male, for a maximum of 3 h, typically between 1000 and 1300 h. This approach provided a satisfactory balance between impregnation success and precise predictability of the sizes of the embryos produced. Dams were paired in a 1:1 ratio with a male in his home cage. Females were then returned to their home cages and day of mating was registered as E0.
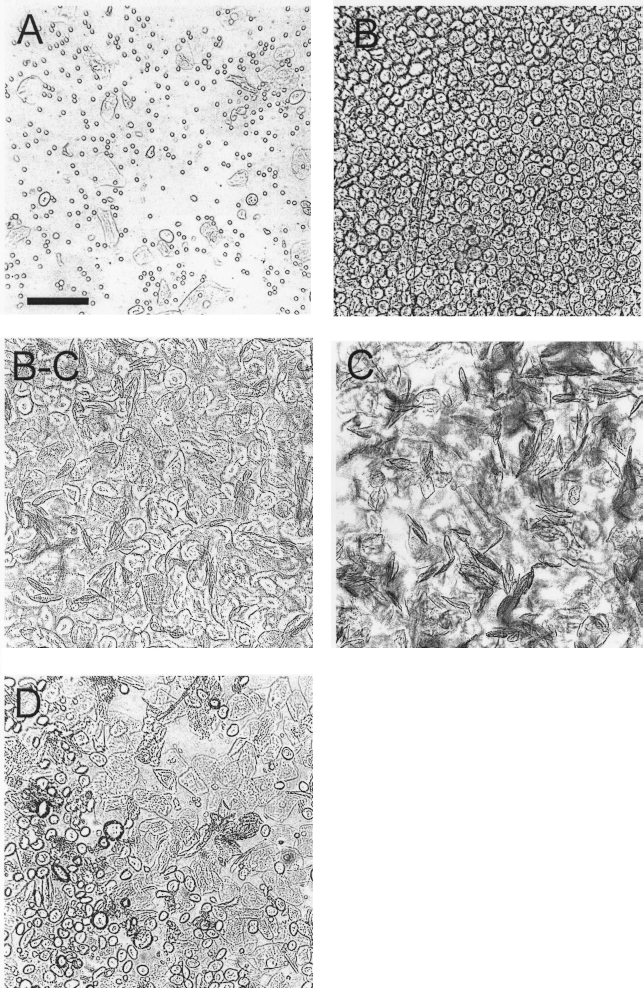
Microphotographs of vaginal smears taken at diestrus (A), proestrus (B), the transitional period from proestrus to estrus (B-C), estrus (C), and metestrus (D) using light microscopy with a closed condenser diaphragm at 10× magnification. Scale bar: 100 μm.
Confirmation of Pregnancy by Palpation
Pregnancy was confirmed 11–12 days after pairing (E11 or E12) as follows. Rats were anesthetized with 1–3% Isoflurane (Baxter) and 4% NO2 delivered using oxygen at 0.8 L/min. Once anesthetized the abdomen of each dam was palpated according to previously described protocols (6,16). Pregnancy was confirmed if the uterine horns displayed several separate swellings ~6 mm in diameter.
Procurement of Embryonic Tissue
On the day of the desired embryonic age, harvesting took place at 1000 h. Pregnant dams were terminally anesthetized using 300 mg/kg sodium pentobarbital (1 ml/kg; Euthathal, Park Vet Group, UK) and subsequently killed by dislocation of the neck and severing of the spinal cord. Embryos were carefully excised from the uterine horns, and immersed in Hanks balanced saline solution (HBSS, Invitrogen, UK) for transfer to the laboratory. Measurements of CRL were carried out using a binocular, zoom microscope (Leica UK), with the embryos lying on one side, submerged in HBSS, as described previously (24).
Statistical Analysis
Where appropriate, the data collected over several breeding sessions were subjected to Kruskal-Wallis and Dunn's Multiple Comparison tests.
Results
As shown in Table 1, our in-house protocol reliably generates 5.86 ± 0.20 mm (mean ± SD) embryos at E12, which is the accurate rat embryo size as established using Charles River's time-mating method (24). Variability within and between litters as well as litter sizes were comparable between protocols. When rats were housed in a reversed light cycle facility (lights on 1900 to 0700 h), dams paired using the same protocol produced litters that were reliably at sizes intermediate to those predicted for their age by the staging scale obtained using Charles River's time-mating method (24) (unreported data).
Comparison of Time-Mated Litters

Litters were obtained from the animal supplier Charles River, as described in Torres et al. (24), and in-house generated time-mated embryos (E12: embryonic day 12; CRL: crown to rump length).
Estrus stages were identified by two blind raters. While discerning metestrus from diestrus was at chance level, the experimenters agreed on proestrus and estrus in all observed cases. Throughout the entire stock, we found that 18 out of 26 dams (69%) cycled through these stages within a 4-day period while we were not able to detect the same degree of regularity in the remaining 8 animals (30%). These cycles were not necessarily always synchronized throughout the stock.
Detecting dams in estrus and pairing them immediately for 3 h with a proven fertile sire (Fig. 2, C 3h) yielded a mean of 53.18 ± 12.75% pregnancies per breeding session (n = 17 mating sessions). Pairing animals for a longer duration (e.g., over 3 days; n = 4 mating sessions; pregnancy rate per session: 50.00 ± 0.00%) or pairing animals overnight that were in proestrus (n = 4 mating sessions; pregnancy rate: 54.00 ± 8.00%) did not affect a change in the number of pregnancies per pairing. A Kruskal-Wallis test revealed no significant differences between the strategies (H = 0.46, df = 24, p = 0.79).

Percentage of pregnant females per pairing session. C 3h: females in estrus mated for 3 h with a stud male (17 sessions); 72 h: nonestrous females paired with male for 72 h and embryonic day 0 (E0) assigned to the day they were predicted to reach estrus (4 sessions); B-C 24 h: females in the transitory stage between proestrus and estrus paired with a male for 24 h (4 sessions).
Most importantly, however, a Kruskal-Wallis test for nonparametric data showed a highly significant difference between the strategies in respect to the production of accurately staged embryos (H = 13.41, df = 24, p = 0.0012) (Fig. 3). When dams were paired during estrus for 3h (C 3h), the resulting embryos were of the expected CRL (5.86 ± 0.20 mm) at E12 in 94.12 ± 24.25% of breeding sessions. The other strategies yielded accurately staged embryos in a significantly lower number of mating sessions (33.33 ± 51.64% for 72 h and 20.00 ± 44.72% for B-C 24 h).
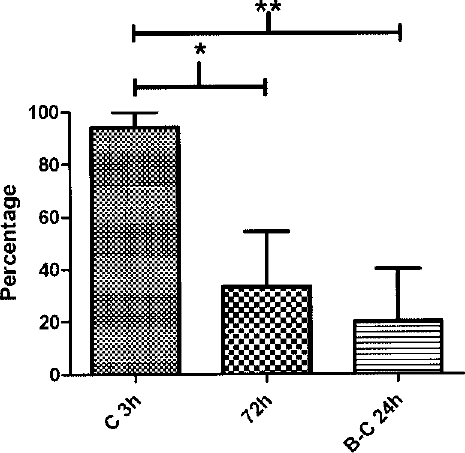
Percentage of litters, between breeding strategies and per pairing session, whose embryo size range fell within that specified by Torres et al. (24) as appropriate for the corresponding embryonic age (e.g., 5.89 ± 0.2 mm at E12). ∗∗p < 0.01; ∗p < 0.05.
Interestingly, we have not observed a single case in which a female became pregnant after having failed to do so twice before when mated during estrus. Hence, overall we observed a scenario in which 51.6% of female rats in estrus became pregnant at the first mating, 9.7% at the second one, when the first pairing was unsuccessful, and 38.7% of females in our breeding colony never became pregnant despite repeated pairings (Fig. 4).
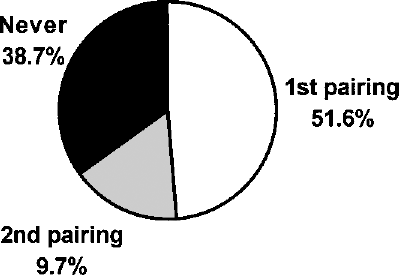
Successful pregnancies by number of pairings. Blank segment: percentage of estrous females that became pregnant at the first paring with a stud male; gray segment: percentage of dams impregnated at the second pairing, when the first one was unsuccessful; full segment: percentage of eligible females that failed to become pregnant despite repeated pairings.
Discussion
In this article we have sought to reproduce in-house the time-mating method first carried out for us within the Charles River commercial breeding laboratory (24). Our most important criterion has been to produce E12 embryos at the same stage of development as obtained previously, namely with a CRL of 5.89 ± 0.21 mm (24). Our results show that the mean litter sizes and CRL obtained at E12 are directly comparable to the standards established using the Charles River's method as described previously (24). The range of embryo sizes obtained here for E12 embryos (5.5–6.5 mm) was slightly larger than that published by Torres et al. (24) (5.8–6.1 mm). This discrepancy may be accounted for by the different measuring techniques used. While Torres et al. took photographs of each embryo and subsequently measured the specimen using computer-based image analysis, the ranges described here were obtained with a standard millimeter scale at the time of dissection, which does not allow for the same degree of accuracy. However, the latter method is more likely to be used by other workers harvesting embryos in this way. Most importantly, the mean sizes of the embryos obtained were directly comparable between the two protocols.
The breeding strategy described above has a number of advantages. The vaginal lavage technique is easy to carry out without causing any distress to the animal. With the references provided, the stages of the estrus cycle are easily identifiable, and the minimal amount of equipment required should be readily available in most laboratories. There was some variability in litter sizes and mean CRL using this breeding protocol; however, the variation observed was no greater than that found in litters from dams purchased from commercial animal suppliers. Moreover, given the accuracy of the timing between copulation and excision of the embryos (within a 3-h time window), this variability probably represents natural variation in embryonic development between animals.
Using the vaginal lavage method has proven to be the most effective and easiest way to determine estrus in rats (1,10,12,13,21). Using two independent raters, we were able to establish a 100% concordance in the detection of estrus (C) and proestrus (B). Agreement on metestrus (D) and diestrus (A) was at chance level; however, this inconsistency is of little relevance as only rats in estrus need to be detected using this method. We have also tried investigating smears for sperm at E1, yet this practice was terminated when several dams that had smeared positive for sperm did not subsequently become pregnant and worries arose about the safety of potential embryos. Because rats were housed on sawdust, it was not possible to find vaginal plugs on E1. Hence, we deemed palpation at the day of excision to be a more reliable indicator of pregnancy.
Pairing dams in estrus with a stud male from about 1000 h (or planned time of harvest) for a maximum of 3 h and denoting the day of mating as E0 was shown to be the most successful breeding strategy. With this relatively short paring time we have achieved a consistent pregnancy success rate of approximately 50%. While shorter pairings may be as successful, they may necessitate observation of coitus. In an attempt to test the reliability of estrus cycling and to increase the number of pregnancies per mating session, we determined the estrus phase of females and paired them with males in a 1:1 ratio for 72 h. From the stage determined before pairing, we predicted when the dam would come into estrus and counted that day as E0. The results indicate that the number of pregnancies did not improve while the variability of embryo sizes increased significantly (Figs. 2 and 3). Similarly, based on reports that estrus tends to last for approximately 12 h (25), we reasoned that some females may have already passed estrus by the time they encounter the male. So we tried to pair females whose smears indicated they were in the transitional period between proestrus and estrus (B-C) and left them overnight. The success rate of pregnancies did not change while the most important figure, namely correctly staged embryos, decreased. Similarly, using a reversed light cycle facility delayed the time of 6-mm embryos to E12.5, necessitating harvest in the early evening (unreported data). The lower numbers of pairing sessions for the 72 h and B-C 24 h strategies reflect early discontinuation of the practice as soon as the unreliability was demonstrated convincingly.
A number of factors may be considered to affect both the number of successful pregnancies obtained and the developmental stage of the embryos. The age of dams has been suggested to be a critical factor in fertility (7,10,13,21). First estrus has been reported to occur at about 6 weeks of age (13,25) and even as young as 30 days in other strains (1), but a delay of sexual maturity has been noted for inbred strains (25). Holehan and Merry (7) report that younger rats have larger litters and that maximum fertility drops to 30% in intermittently bred females over 10 months of age. In contrast, in our experience very young females tend to have small litters (unpublished observation). Most importantly, however, we have not seen a negative effect of maternal age on the predictability of embryonic size.
It is, however, difficult to assess the ability of younger versus older females to become pregnant from a single 3-h pairing with a male because fertile animals are continually removed from the breeding stock when pregnant dams are killed to harvest the embryos. Although Aoyama and coworkers (1) found a fertility rate of 99.2% (n = 120) in outbred rats, we recommend using young (180 ± 300 g) females. Using our short mating window (3 h), 51.6% of dams in estrus became pregnant after the first pairing with a stud male while all other pregnancies (9.7%) occurred in females mated for the second time, when the first pairing was unsuccessful. Interestingly, the remaining 38.7% of our frequently restocked colony failed to ever become pregnant despite repeated pairings. Hence, in order to ensure high efficiency, we suggest removing those females from the time-mating breeding stock after two failed attempts with two different males. While these dams may not necessarily be infertile, estrus cycling may be irregular or they may need more than 3 h to accomplish coitus. By contrast, if a sire had been successful at impregnating a female in the past, his age was of lesser importance to us. We have kept successful stud males of more than 18 months of age.
Based on reports that the rat estrus cycle lasts approximately 4 days (1,12,21,25) and that estrus cycles of cage mates may not necessarily be synchronized, we hypothesized that at any given time one in four females may be in estrus and ready for mating. A colony of about 25 females and 4 males was thus deemed sufficient to produce a regular supply of one to two pregnant dams per week. Considering our consistent pregnancy rate of 50% and animal holding space permitting, we have found it practical to pair twice the number of dams in each session than the number of litters required. This method is easily reproducible, cheap, and timed for harvesting embryos at the beginning of the working day.
Footnotes
Acknowledgments
The authors would like to thank Anna Papazoglou from the Laboratory of Molecular Neurosurgery, University of Freiburg, for her kind help and suggestions. This study was funded by grants from the MRC and The German Academic Merit Foundation.
