Abstract
Accumulating evidence has shown that lncRNA GAS5 is a novel tumour-promoting RNA that contributes to tumour progression by sponging miRNAs. However, the detailed role of lncRNA GAS5 in B lymphocytic leukaemia is still unclear. A qRT-PCR assay was used to examine the levels of lncRNA GAS5 and miR-222 in leukomonocytes of patients with B lymphocytic leukaemia and in healthy donors. Raji cells were transfected with GAS5 overexpression or shRNA-GAS5 plasmids for
Introduction
Chronic lymphocytic leukaemia, defined as a lymphoproliferative disorder with low immunoglobulin levels and composed of monomorphic round B lymphocytes in peripheral blood, bone marrow and lymphoid organs, is the most common type of adult leukaemia in western countries [1, 2]. Recent studies have indicated that there are approximately 15000 new cases of chronic lymphocytic leukaemia per year, with approximately 4500 deaths, thereby chronic lymphocytic leukaemia and its associated high rate of morbidity and mortality are still a major health problem [3, 4]. In the past few years, chronic lymphocytic leukaemia treatment has evolved from chemotherapy to the use of agents specifically targeting various signalling pathways, but these treatment effects are still very limited [5, 6]. Thus, a detailed understanding of the molecular mechanisms involved in chronic lymphocytic leukaemia requires further investigation.
Dysregulated expression of miRNAs has been found in different human cancers, and the functions of miRNAs in cancer are quite complex; it has been well investigated that miR-222 functions as an oncogene in multiple cancer types, including colorectal cancer [7], cervical cancer [8], glioblastoma [9], osteosarcoma [10], and breast cancer [11]. In chronic lymphocytic leukaemia, Frenquelli et al. reported that enforced expression of miR-222 can modulate G1/S phase cell cycle transition by suppressing p27 expression, which could promote tumour cell proliferation [12].
Long non-coding RNAs (lncRNAs), a type of highly conserved RNA transcripts composed of more than 200 nucleotides in length and lacking protein-coding potential, have been found to be involved in transcriptional and post-transcriptional regulation of gene expression [13]. Accumulating evidence has demonstrated that lncRNAs could play pivotal roles in almost all major physiological processes as well as multiple pathological processes, especially the pathogenesis of different types of malignant tumours [14, 15]. Previous studies have reported that lncRNA GAS5 could play a role as a tumour-suppressor gene in the development of several cancer types [16, 17]. For example, lncRNA GAS5 can suppress ovarian cancer by inducing the formation of the inflammasome [18]; lncRNA GAS5 binds to Y-box binding protein 1 (YBX1) to regulate p21 expression, which arrests the cells at the G1 phase of the cell cycle in stomach cancer [19]; and lncRNA GAS5 inhibits oesophageal squamous cell carcinoma cell proliferation and migration by inactivating the phosphatidylinositol 3-kinase (PI3K)/AKT/ mammalian target of rapamycin (mTOR) signalling pathway [20]. In addition, intensive research in the past two decades has uncovered that lncRNAs can bind to microRNAs (miRNAs) to sequester and control them, such as lncRNA-BGL3 (to miR-17, miR-93, miR-20a, miR-20b, miR-106a and miR-106b) [21], MIAT (to miR-150-5p) [22], Linc-RoR (to miR-145) [23], and lncRNA GAS5 (to miR-222) [24]. Hence, it is necessary to explore whether lncRNA GAS5 could function as a sponge for miR-222 to exert its role during the development of human B lymphocytic leukaemia.
Materials and methods
Patients and cell lines
Blood specimens were obtained from 30 human B lymphocytic leukaemia cell patients from November 2017 to March 2018 in the Qingdao Hospital of Traditional Chinese Medicine. In addition, 30 normal blood specimens from healthy donors were collected as control samples. Moreover, all samples were frozen in liquid nitrogen and maintained at
RNA extraction and quantitative RT-PCR (qRT-PCR) assay
Total RNA from clinical blood samples or cultured cells was extracted using TRIzol reagent (Invitrogen, USA) according to the provided protocol. Briefly, 10
Cell transfection
To overexpress and knock down lncRNA GAS5 in cell lines, lncRNA GAS5-overexpression (GAS5-pcDNA3.1), lncRNA GAS5-knockdown (GAS5 shR-NA) and negative control (NC) plasmids were synthesized and purchased from Genepharma Inc. (China). Then, Raji cells were seeded in 6-well plates at a density of 5
Cell proliferation assay
Cell proliferation was assessed using a cell counting kit-8 (CCK-8) assay (Dojindo, Japan). A total of 2
Flow cytometry analysis
Flow cytometry for cell apoptosis detection was carried out using an Annexin V-FITC Apoptosis Detection Kit (Beyotime Biotechnology, China) in accordance with the manufacturer’s recommendations. Cells were harvested during the logarithmic growth phase and then successively stained with 250
To determine the cell cycle, cell suspensions with a density of 4
In vitro invasion assays
The 24-well Transwell insert chambers coated with Matrigel (BD Biosciences, USA) were utilized for the cell invasion assay. The transfected Raji cells were resuspended in 100
Dual luciferase reporter assay
To identify the direct interaction between lncRNA GAS5 and miR-222, 293T cells seeded in 24-well plates (5
qRT-PCR analysis of lncRNA GAS5 and miR-222 expressions in human B lymphocytic leukemia cells. (A) The levels of lncRNA GAS5 in leukomonocytes of 30 patients with B lymphocytic leukemia and in 30 heathy donors. (B) The levels of miR-222 in leukomonocytes of 30 patients with B lymphocytic leukemia and in 30 heathy donors. (C) The levels of lncRNA GAS5 in B lymphocytic leukemia cell lines (RAMOS, ST486, Raji, Farage) and normal B lymphocytic cell line IM9. * indicates 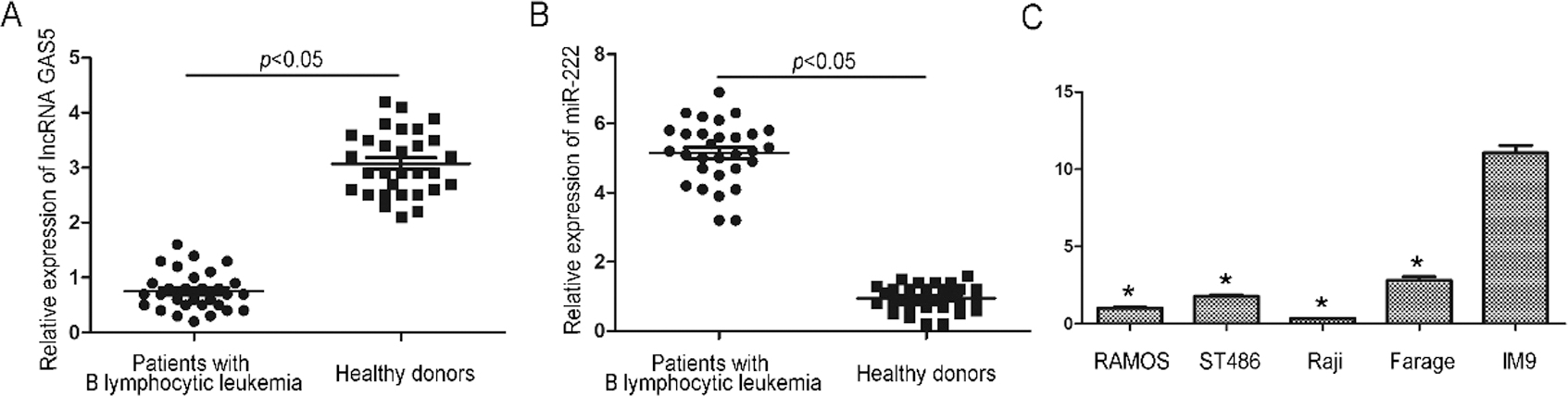
SPSS 16.0 statistical software was used to perform statistical analyses, and GraphPad Prism 7 was applied for graphical data representation. Data are shown as the mean
Results
Expression pattern of lncRNA GAS5 and miR-222 in leukomonocytes of human B lymphocytic leukaemia patients
We first determined the expression of lncRNA GAS5 and miR-222 in leukomonocytes of 30 B lymphocytic leukaemia patients and 30 healthy donors using qRT-PCR assay. It was found that the expression of lncRNA GAS5 was significantly decreased in patients with B lymphocytic leukaemia compared with the healthy group (
The role of overexpression and knockdown of lncRNA GAS5 on B lymphocytic leukemia cell proliferation was evaluated by CCK-8 assay at days 0, 1, 2, 3. * and # indicate 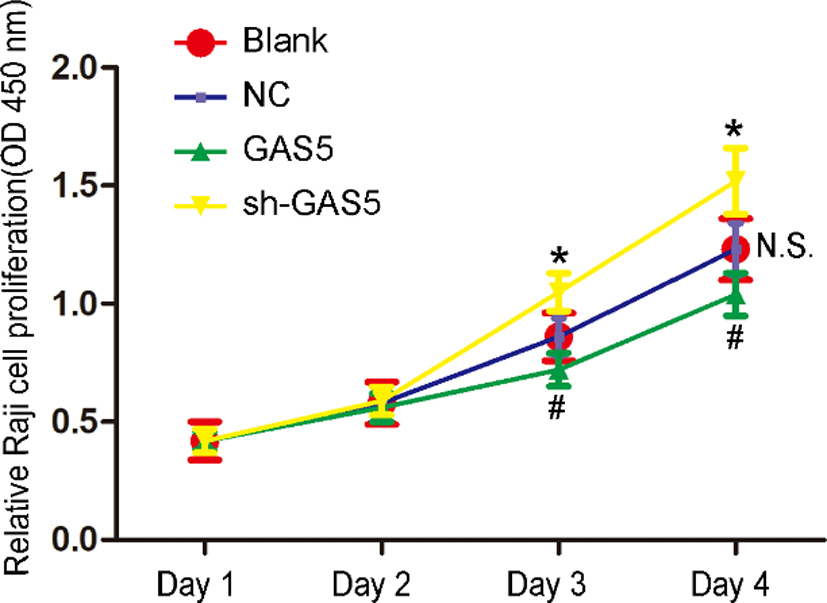
The effect of overexpression and knockdown of lncRNA GAS5 on B lymphocytic leukemia cell apoptosis was examined by flow cytometry. *indicates 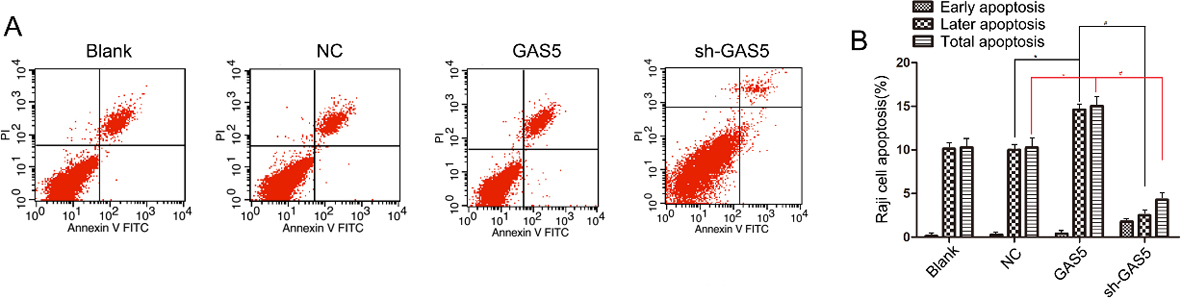
The regulation of overexpression and knockdown of lncRNA GAS5 on B lymphocytic leukemia cell cycle was assessed by flow cytometry. * indicates 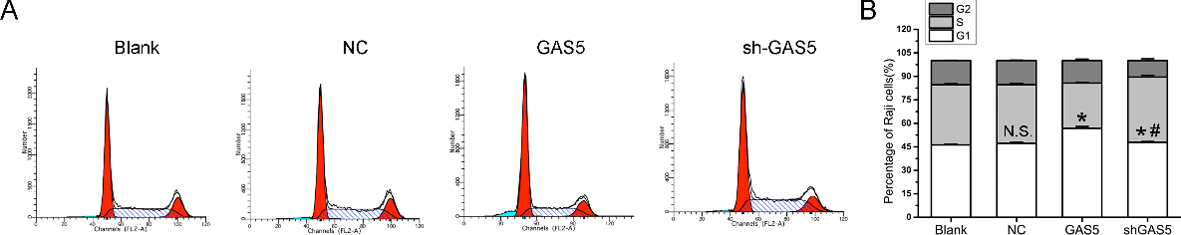
Overexpression and knockdown of lncRNA GAS5 modulates the invasion of human B lymphocytic leukemia cells. * indicate 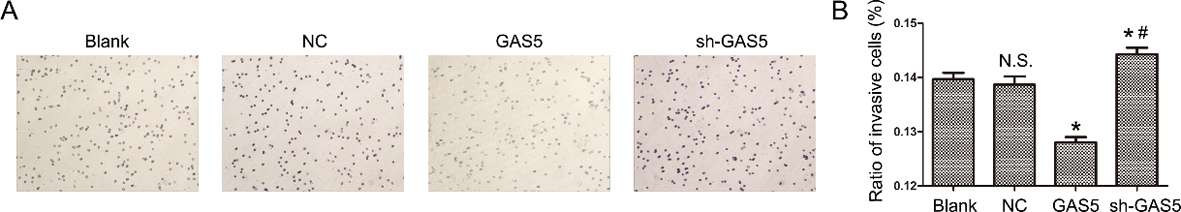
To investigate the functions of lncRNA GAS5, lncRNA GAS5-overexpression and lncRNA GAS5-knockdown plasmids were transfected into Raji cells for multiple cell culture experiments As shown in Fig. 1c, lncRNA GAS5 expression was sharply upregulated in Raji cells transfected with the lncRNA GAS5-overexpression plasmid and dramatically downregulated in cells transfected with the lncRNA GAS5-knockdown plasmid (
MiR-222 is a direct target of lncRNA GAS5. (A) The putative binding sites of lncRNA GAS5 and miR-222 are presented. (B) Dual-luciferase assay was applied to determine the targeted interaction of lncRNA GAS5 and miR-222. (C) Regression analyses of GAS5 expression level and miR-222 expression level in 30 patients with B lymphocytic leukemia. * indicates 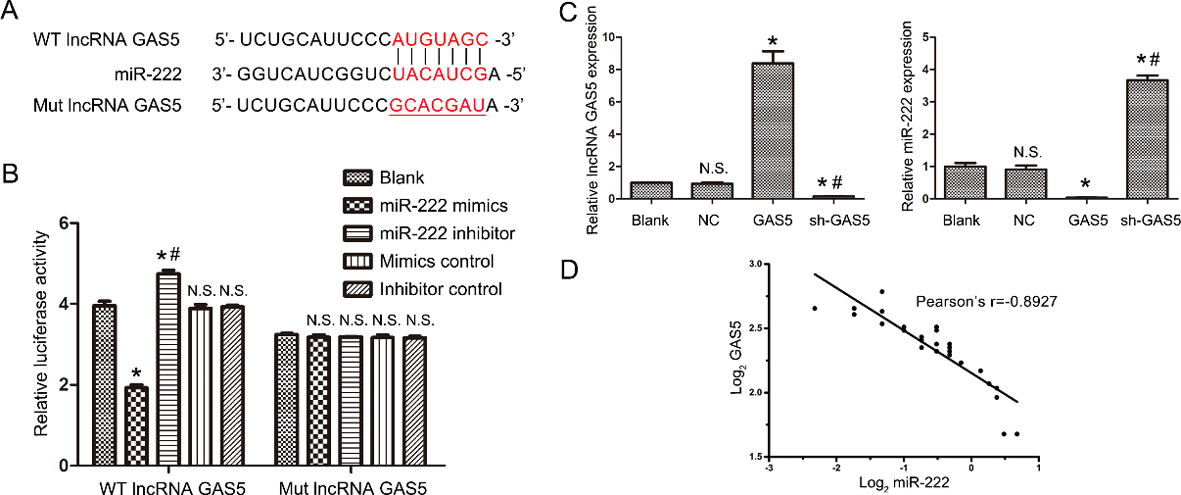
Subsequently, a series of cell function experiments was performed. The CCK-8 assay revealed that the cells in the different groups gradually proliferated with time, but the rate of Raji cell proliferation in the lncRNA GAS5-overexpression group was obviously lower than that in the Raji or NC groups, while the rate of Raji cell proliferation in the lncRNA GAS5-knockdown group was obviously higher than that in the Raji or NC groups (
Previous studies have demonstrated that many lncRNAs function as ceRNAs that regulate target genes by competitively binding miRNAs. In this study, bioinformatics software analysis (StarBase 2.0 programme,
Discussion
Over the past decades, a large number of innovative targeted therapies, including monoclonal antibodies or bispecific T-cell engagers, personalized vaccines, and immunocellular therapy, have been widely reported [25, 26]. Although the survival outcome of patients with B lymphocytic leukaemia has dramatically improved, the subgroup of patients with relapsed/refractory B lymphocytic leukaemia still has a dismal prognosis [27]. This may be because the molecular mechanism of B lymphocytic leukaemia has not been clearly understood. lncRNAs, a novel class of RNA transcripts longer than 200 nucleotides that lack protein-coding potential, are dysregulated in many disease states, particularly in tumours [14, 28]. For example, lncRNA FBXl19-AS1 regulates osteosarcoma cell progression by sponging miR-346 [29]; lncRNA H19 promotes colorectal cancer cell formation by regulating the miR-29b-3p/PGRN-mediated Wnt signalling pathway [30]; and lncRNA MEG3 inhibits the progression of prostate cancer by modulating the miR-9-5p/QKI-5 axis [31]. Thus, these studies have demonstrated that lncRNAs can function as either carcinogens or tumour suppressors in diverse cancers. Moreover, the complex regulation between lncRNAs and miRNAs is thought to be one of the critical features of lncRNAs [32]. Additionally, lncRNA GAS5 has typically been considered a tumour suppressor and presents a declining expression trend in many cancers, such as cervical cancer, breast cancer, and stomach cancer [17, 33]. In the current study, it was also found that lncRNA GAS5 expression was significantly reduced both in leukomonocytes of patients with B lymphocytic leukaemia and B lymphocytic leukaemia cell lines. Then, the role of lncRNA GAS5 in B lymphocytic leukaemia cells was explored. Our results revealed that lncRNA GAS5 overexpression decreased B lymphocytic leukaemia cell proliferation, promoted B lymphocytic leukaemia cell apoptosis, arrested B lymphocytic leukaemia cell cycle in G1 phase, and inhibited B lymphocytic leukaemia cell invasion. Moreover, when lncRNA GAS5 expression was knocked down in B lymphocytic leukaemia cells, the opposite cell phenotype changes were observed. Therefore, these findings implied that lncRNA GAS5 might play an important role in the development of B lymphocytic leukaemia. The lncRNA GAS5 gene, first reported by Coccia et al. in 1992, is located on the human chromosome 1q25. Previous studies have demonstrated that lncRNA GAS5 is involved in the development of various cancers [17, 33]. For instance, lncRNA GAS5 regulates the proliferation and invasion of hepatoma cells by regulating Vimentin [34]; lncRNA GAS5 expression is notably decreased in colorectal cancer tissue samples, and its expression level could be an independent risk factor or a predictor of prognosis for colorectal cancer [35]; and lncRNA GAS5 has also been discovered to impede the viability, migration and invasion of pancreatic cancer cells [36, 37]. The roles of the lncRNA GAS5/miR-222 regulatory axis in cancer development have been illustrated by several studies. Xihe Zhao et al. showed that lncRNA GAS5 suppresses glioma tumour malignancy by inhibiting miR-222 [38]. Yanhua Li et al. indicated that the lncRNA GAS5/miR-222 axis regulates gastric cancer cell proliferation through the PTEN/Akt/mTOR pathway [24]. XF Zhang et al. have reported that GAS5 induces the suppression of papillary thyroid carcinoma cell proliferation by regulating miR-222 [39]. Our results in the present study were consistent with these previous findings, which uncovered the characteristics of the lncRNA GAS5/miR-222 axis in tumours.
Furthermore, miR-222 exhibited markedly increased expression in lncRNA GAS5-knockdown B lymphocytic leukaemia cells and decreased expression in lncRNA-overexpressing B lymphocytic leukaemia cel-ls. To further pinpoint the relationship between lnc-RNA GAS5 and miR-222, the dual luciferase reporter assay was performed, and the results clearly indicated that lncRNA GAS5 is a direct target of miR-222. A previous study showed that miR-222 could regulate p27 and form a miR-222/p27 regulatory loop to maintain chronic lymphocytic leukaemia cells in a resting state [12]. Therefore, the upregulation of miR-222 due to the absence of lncRNA GAS5 in chronic lymphocytic leukaemia cells may promote the proliferation of tumour cells by inhibiting p27 expression.
In summary, we first identified the regulatory role of the lncRNA GAS5/miR-222 axis in the cell proliferation, cell apoptosis, cell cycle and cell invasion of B lymphocytic leukaemia. The loss of lncRNA GAS5 expression in B lymphocytic leukaemia cells might be likely to stimulate the progression of B lymphocytic leukaemia. Moreover, the direct target interaction of lncRNA GAS5 and miR-222 was also verified. Hence, these data demonstrated that lncRNA GAS5 could effectively sponge miR-222 to modulate human B lymphocytic leukaemia cell tumourigenesis and metastasis. These results could greatly aid in exploring novel diagnostic biomarkers and treatment targets for B lymphocytic leukaemia in the future.
