Abstract
BACKGROUND:
Recent studies implicate the effect of vestibular loss on cognitive decline, including hippocampal volume loss. As hippocampal atrophy is an important biomarker of Alzheimer’s disease, exploring vestibular dysfunction as a risk factor for dementia and its role in hippocampal atrophy is of interest.
OBJECTIVE:
To replicate previous literature on whole-brain and hippocampal volume in semicircular canal dysfunction (bilateral vestibulopathy; BV) and explore the association between otolith function and hippocampal volume.
METHODS:
Hippocampal and whole-brain MRI volumes were compared in adults aged between 55 and 83 years. Participants with BV (n = 16) were compared to controls individually matched on age, sex, and hearing status (n = 16). Otolith influence on hippocampal volume in preserved semicircular canal function was evaluated (n = 34).
RESULTS:
Whole-brain and targeted hippocampal approaches using volumetric and surface-based measures yielded no significant differences when comparing BV to controls. Binary support vector machines were unable to classify inner ear health status above chance level. Otolith parameters were not associated with hippocampal volume in preserved semicircular canal function.
CONCLUSIONS:
No significant differences in whole-brain or hippocampal volume were found when comparing BV participants with healthy controls. Saccular parameters in subjects with preserved semicircular canal function were not associated with hippocampal volume changes.
Key points
Recent research suggests an association between vestibular function and cognition. Hippocampal atrophy is an important biomarker of Alzheimer’s disease. Bilateral vestibular loss did not modulate hippocampal or whole-brain volume.
Introduction
Bilateral vestibulopathy (BV) is a severe chronic vestibular disorder of the labyrinth or the eighth cranial nerve characterized by postural imbalance, unsteadiness of gait which worsens in darkness and/or on uneven ground, and oscillopsia during head movements. Symptoms are typically absent under static conditions [48]. Multiple possible etiologies for BV exist, including but not limited to ototoxicity, bilateral Menière’s disease, bilateral vestibular schwannoma, genetic, or infectious causes [32].
There is evolving evidence suggesting that vestibular loss is associated with cognitive impairment and may even contribute to the onset of Alzheimer’s disease [5, 46].
When zooming in on the anatomical level, structural brain changes have been reported in patients with BV over the past twenty years in cross-sectional manual segmentation studies, specifically at the level of the hippocampus [9, 25]. The hippocampus is a seahorse-shaped structure necessary for memory processing (encoding, consolidation, and retrieval) [34, 45] and spatial memory function [35, 38]. These cognitive functions have been identified to be impacted in BV patients [6, 16]. Previous studies have compared hippocampal volumes between subjects with and without BV. T. Brandt et al. [9] observed a significant selective shrinkage of hippocampal volume by 16.9% in people with BV relative to controls. A study by O. Kremmyda et al. [30] described a significant reduction in grey-matter mid-hippocampal and posterior parahippocampal volume in long-standing BV patients compared to healthy controls. On the other hand, other studies observed a lack of hippocampal volumetric differences when comparing patients with BV and healthy controls [17, 43].
A study by R.J. Kamil et al. [29] took a different approach and evaluated hippocampal volume in healthy older adults (≥60 years) from the Baltimore Longitudinal Study of Aging (BLSA). They observed that a larger cervical vestibular-evoked myogenic potential (cVEMP) amplitude was significantly associated with a larger mean hippocampal volume (p = 0.003). They proposed that lower cVEMP amplitude, implying reduced saccular function, is significantly associated with a lower mean volume of the hippocampus. A. Jacob et al. [28] included healthy older adults (≥60 years) from the BLSA cohort. They investigated the relation between vestibular function (using cVEMP) and the volume of structures comprised of or connected to the vestibular cortex. They observed smaller volumes of the hippocampus and entorhinal cortex associated with reduced vestibular function. A review by P.F. Smith [47] supports these findings, stating that reduced saccular function can be associated with poorer spatial memory, Alzheimer’s disease, and reduced hippocampal volume.
There is a high risk of concomitant sensorineural hearing loss (SNHL) in patients with vestibular dysfunction and vice versa [32, 50]. As concomitant hearing loss could exacerbate a potential effect of vestibular dysfunction on brain volume, the hippocampus being of main interest, hearing levels should be included in these analyses. Previously mentioned studies comparing hippocampal volumes between BV patients and healthy controls generally lack a detailed description of hearing performance and did not include hearing performance in their methodological approach to the topic.
We are interested in evaluating the impact of semicircular canal dysfunction (in this case: BV) and otolith function (in this case: saccular function) on hippocampal volume. We hypothesize that the effect of BV will not result in significant hippocampal volume differences when compared to controls because we will adjust for hearing level. In addition to hippocampal and whole-brain analyses, we will also perform cortical thickness and sulcus depth analyses as well as surface-based morphometry. A second aim of this study is to delineate otolith (saccular) influence on hippocampal volume in a population with preserved semicircular canal function.
Materials and methods
Participant Characteristics
All participants were recruited from the GECkO-study (Gehoor, Evenwicht, COgnitie), an ongoing prospective longitudinal cohort study of the effect of hearing loss and vestibular decline on cognitive function in older adults [7]. This protocol was approved by the ethical committee of the University Hospital of Antwerp, Belgium (EC number B300201938949) and all participants gave their written informed consent in accordance with the Declaration of Helsinki prior to participation. The study protocol builds upon the Clinical Trials protocol with identifier NCT04385225.
BV population
The diagnosis of BV was made according to the Bárány Society criteria and was defined as (1) a bilaterally pathological horizontal angular VOR gain (< 0.6) measured by the vHIT, and/or (2) reduced horizontal angular VOR gain (< 0.1) upon sinusoidal stimulation on a rotatory chair (0.1 Hz, Vmax = 50°/sec), and/or (3) reduced caloric response (sum of bi-thermal (30°C/44°C) maximum peak SPV on each side < 6°/sec) [48].
Healthy controls
BV participants were matched based on age, sex, and best aided speech audiometry in noise. All participants underwent vHIT to confirm normal vestibular function (bilateral horizontal VOR gain > 0.6).
For all participants (BV and healthy controls) the following inclusion criteria were applied (1) age 55–84 years, (2) Dutch as native language, (3) right-handed as defined by the Edinburgh Handedness Inventory [39], and (4) preserved cognitive function. A neuropsychological exam including a Mini-Mental State Examination (MMSE) and Repeatable Battery for the Assessment of Neuropsychological Status for Hearing impaired individuals (RBANS-H) was performed in all participants [13, 19]. Participants were considered having preserved cognitive function when scoring ≥24/30 on the MMSE [19]. This cut-off is recommended in patients with at least eight years of education, which is the case in the current study [36]. In addition, participants were considered having preserved cognitive function when scoring≥percentile 16 on the RBANS-H total score. Patients with Mild Cognitive Impairment score on cognitive tests generally 1 to 1.5 standard deviations below the mean. Here we apply the less stringent approach of using 1 standard deviation below the mean as cut-off, resulting in a percentile score of 16 [1]. Participants with lower cognitive scores were excluded as cognitive impairment can affect hippocampal volume and confound our results. People with an implanted hearing aid device (e.g., cochlear implant or bone-anchored hearing aid) were also excluded from this study.
MRI volumetry
Acquisition protocol
All subjects were investigated in a clinical 3.0 T scanner (Siemens Magnetom Prisma, Erlangen equipped with a 32-channel receiver head coil, 24 subjects in total, being 11 with BV and 13 healthy controls; Siemens Magnetom Vida, Erlangen equipped with a 64-channel receiver head coil, 8 subjects in total, being 5 with BV and 3 healthy controls). A high-resolution T1-weighted image (GRAPPA sequence, 256 slices, slice thickness = 0.75 mm, voxel size = 0.75×0.75×0.75 mm, TR = 2060 ms, TE = 2.17 ms) was obtained in sagittal orientation.
MRI data processing
Neuroimaging data quality control was performed via MRIQC version 0.15.1 [18]. Structural images were pre-processed and automatically segmented by the Computational Anatomy Toolbox (CAT12 Version 1980) (Fig. 1, Panel A) [21], an extension within the framework of Statistical Parametric Mapping software (SPM12) in MATLAB. Atlas-based segmentation for regions-based morphometry included the entire hippocampus as well as the volume of its substructures (CA1, CA2, CA3, dentate gyrus, and subiculum) taken from the cytoarchitectonic representation in the Julich Brain atlas [3]. In addition, total intracranial volume (TIV) was estimated and used (together with age and scanner type) as a covariate for all the voxel- and region-based, but not for surface-based analyses [26].
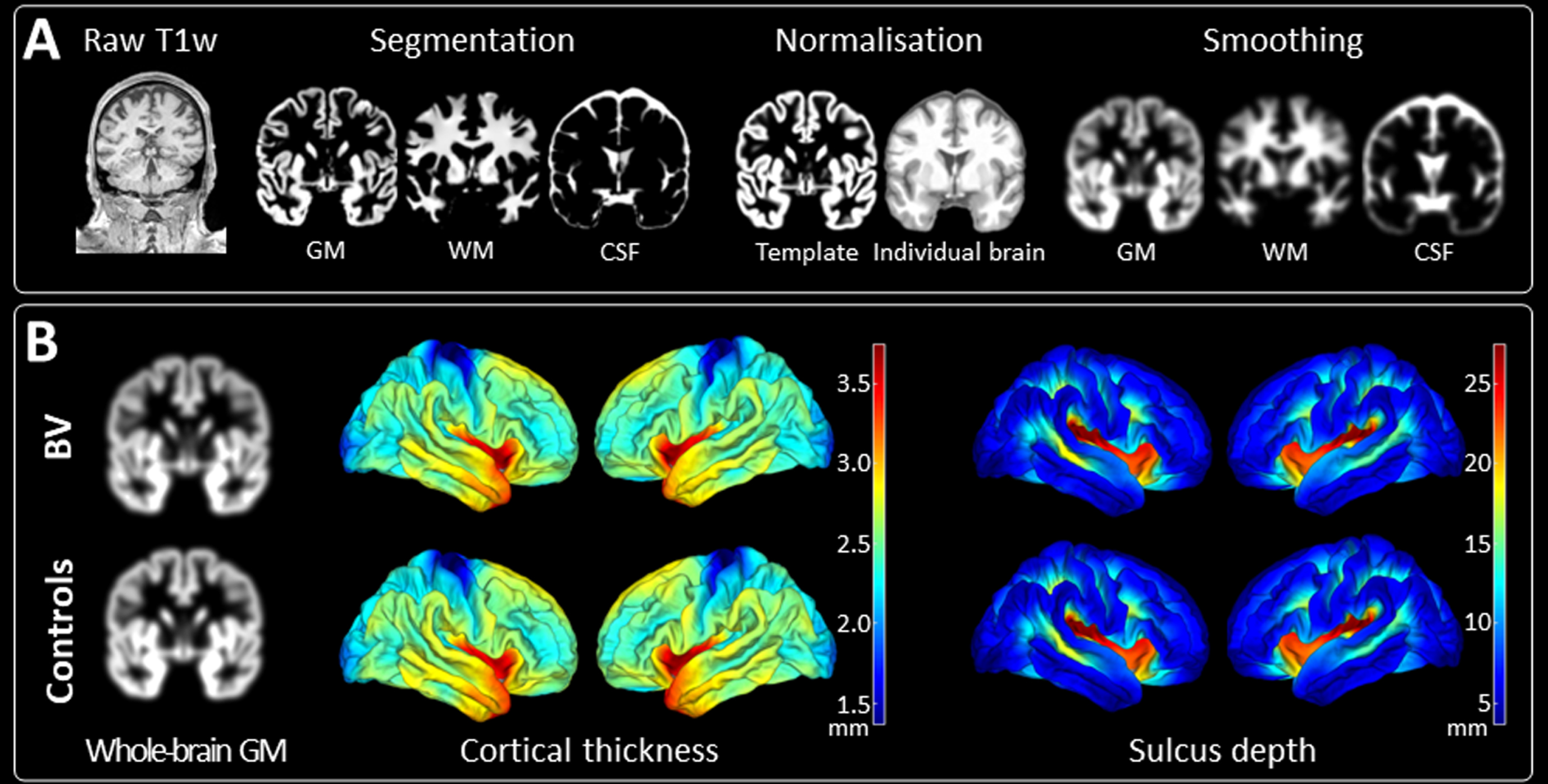
(A) Flowchart of the structural MRI preprocessing pipeline. All presented images are derived from the same control participant. The MNI152 NLIN 2009c 1 mm template is used for normalisation. A smoothing kernel of 6 mm full width at half maximum is applied. (B) Results of whole-brain comparisons between patients with BV (n = 16) and their matched controls (n = 16). Whole-brain comparisons encompassed whole-brain grey matter volumetric analyses and surface-based measures including cortical thickness and sulcus depth analyses. No significant differences were found in any of the comparisons. GM, grey matter; WM, white matter; CSF, cerebrospinal fluid; BV, bilateral vestibulopathy.
Saccular function was investigated via the vestibulocollic reflex (VCR) using cVEMP with the validated Neuro-Audio device incorporating electromyography feedback (Neurosoft, DIFRA). While participants lay in a supine position, they lifted and rotated their head to one side, contracting the sternocleidomastoid (SCM) muscle. Short 500 Hz tone bursts were presented in the contralateral ear at suprathreshold level (95 dB nHL). Present responses were biphasic and had two distinctive peaks (p13 and n23). Normative ranges were applied, with the p13 occurring 11.81–15.59 ms after stimulus onset, and with the n23 occurring 18.15–25.64 ms after stimulus onset [31]. Intact responses needed to be elicited at least twice to confirm presence of the VCR. Outcome measures included presence of intact responses (0, 1 ear, or both ears), and for each present response outcome measures included p13 latency (ms), n23 latency (ms), P-N amplitude (μV), rectified amplitude (μV), and SCM muscle contraction level (mean rectified voltage, MRV, μV).
Hearing assessment
Unaided pure-tone audiometry was measured over a frequency range from 125 Hz to 8 kHz (specifically 0.125, 0.25, 0.5, 1, 2, 3, 4, 6, 8 kHz). Hearing thresholds were measured separately for each ear using a 2-channel Interacoustics AC-40 audiometer with insert earphones. Speech audiometry in noise (speech-in-noise; SPIN) was evaluated by the Leuven Intelligibility Sentences Test (LIST) with an adaptive procedure [49] in free field using a loudspeaker at a distance of 1 meter at 0° azimuth. The noise level was constant at 65 dB sound pressure level (SPL) while the speech level was adapted according to a correct (decreased speech level of 2 dB SPL) or incorrect (increased speech level of 2 dB SPL) response. Two lists of ten sentences each were conducted to acquire the speech reception threshold (SRT in dB SNR; averaged speech levels of the last five sentences and the imaginary 11th sentence), both in an unaided and aided condition. The mean value of the best aided condition was used for analyses.
Statistical analysis
For demographic and region of interest (ROI) based analyses (by use of the Julich-Brain atlas [2]), JMP Pro 15 (Medmenham, UK) was used. Levene’s tests and visualization of data using histograms confirmed equal variances and the normality of reported data. However, because of the small sample size, nonparametric tests with the median and range are reported. Continuous patient characteristics were compared using Kruskal-Wallis ANOVA, for nominal patient characteristics, the Pearson Chi-squared statistic was used. For voxel-based morphometry analyses, the CAT12 toolbox and SPM12 were used. For each aim, a two-sample t-test was performed. Whole-brain changes were investigated by an F-contrast, with age, TIV, and scanner type as covariates. Similar statistics were performed for surface analyses (cortical thickness and sulcus depth), with only age and scanner type as covariates. Regarding p-value adjustment, the Monte-Carlo method for permutation testing (10.000 permutations) was applied using the TFCE toolbox (Version 224), with correction for multiple comparisons via false discovery rate (p < 0.05). In addition, machine learning in the form of multi-voxel pattern analysis is performed to increase the sensitivity to detect differences in each pairwise comparison by use of the Pattern Recognition for Neuroimaging Toolbox v3.0 (PRoNTo) [44]. Classification was performed using a binary support vector machine (SVM) with one subject per class left out as the cross-validation scheme and 10.000 permutations. A Spearman correlation (and its 95% confidence interval) was performed for saccular analyses. P-values are reported, as well as eta squared (η2) indicating the effect size. The Pearson Chi-squared statistic was used for ordinal parameters, with w indicating its effect size. Between-scanner type differences were examined by a two-sample t-test of quality control parameters derived from MRIQC.
Results
Patient Characteristics
Demographic and clinical details as well as neuroimaging data quality of included participants can be found in Table 1. The median [range] disease duration for the BV population was 8 years [2, 22]. Among the etiologies of BV, 6 patients had a genetic risk (DFNA9), 1 patient autoimmune, 2 patients infectious (meningitis, varicella zoster), 1 patient ototoxic, 2 patients due to trauma, 1 patient with unknown etiology, and 3 patients idiopathic [51, 52]. All patients with idiopathic etiology had undergone an MRI internal auditory canal, tonal audiometry, and (hetero)anamnesis to exclude other causes, whereas in patients with unknown etiology this diagnostic process has not been completed yet. To confirm the diagnosis of BV, patients must meet at least one out of three of the Bárány Society criteria [48]. All three criteria (bilaterally reduced vHIT response, rotatory chair, and caloric testing) were met by 25% (n = 4) of people with vestibular loss. In 37.5% (n = 6), two out of three criteria were fulfilled, and the remaining 37.5% (n = 6) of people met one criterion. Based on the unaided tonal audiometry of the best hearing ear, 6 subjects with BV demonstrated age-normal hearing function (≤40 dB HL), 4 had moderate SNHL (41–60 dB HL), and 6 had severe SNHL (≥60 dB HL) [27].
Demographic characteristics of people with BV and its age-, sex-, and hearing-matched controls
Demographic characteristics of people with BV and its age-, sex-, and hearing-matched controls
Education level indicates the number of years spent in school, starting from 6 years old. NA indicates the amount of missing data. SD, standard deviation; FIhigh, Fletcher index high (mean 1 –2 –4 kHz); dB HL, decibel hearing level; SPIN, speech-in-noise; SRT, speech reception threshold; BMI, body mass index; SNRd, Dietrich’s signal-to-noise ratio; EFC, entropy focus criterion; CJV, coefficient of joint variation.
Age, sex, hearing level, education level, obesity, smoking status, tinnitus presence, and depression may affect hippocampal volumes [10, 41]. Therefore, age, sex, Fletcher index high (FIhigh; average threshold of 1 kHz, 2 kHz, and 4 kHz), SPIN, hearing aid ownership, years of education (number of years spent in school, starting from the age of 6 years old), body mass index (BMI), smoking status, tinnitus presence, and the total score of the Beck Depression Inventory were included in the demographic characteristics. No significant demographic or patient characteristic differences were observed (Table 1).
Neuroimaging data quality control encompassed image quality metrics for structural images including Dietrich’s signal-to-noise ratio (SNRd) [14], entropy focus criterion (EFC) [4], and coefficient of joint variation (CJV) [20]. Neuroimaging data quality control was blinded for diagnostic categories and afterwards tested for group differences. The parameters EFC and CJV were included to control for the potential head motion differences between the groups during structural neuroimaging. None of the pairwise comparisons resulted in a significant difference on any of the image quality metrics (Table 1).
To evaluate the effect of semicircular canal dysfunction on brain tissue compartments and to exclude a potential confounding effect of concomitant hearing loss, modulated grey and white matter tissue volumes of people with BV were compared with matched healthy controls. Whole-brain grey matter comparisons yielded no significant differences between these two groups (p > 0.05) (Fig. 1 Panel B). A ROI analysis of the hippocampus proper found no significant morphometric changes between these two groups (total hippocampus proper: p = 0.7806; left hippocampus proper: p = 0.7200; right hippocampus proper: p = 0.8958; see Table 2; Fig. 2). Surface-based analyses (cortical thickness and sulcus depth) also gave no significant differences between these two groups (p > 0.05) (Fig. 1 Panel B). The SVM model resulted in an area under the ROC curve value of 0 (p = 1, total accuracy of 40.62%), reflecting at random classification of people with BV versus their matched healthy controls.
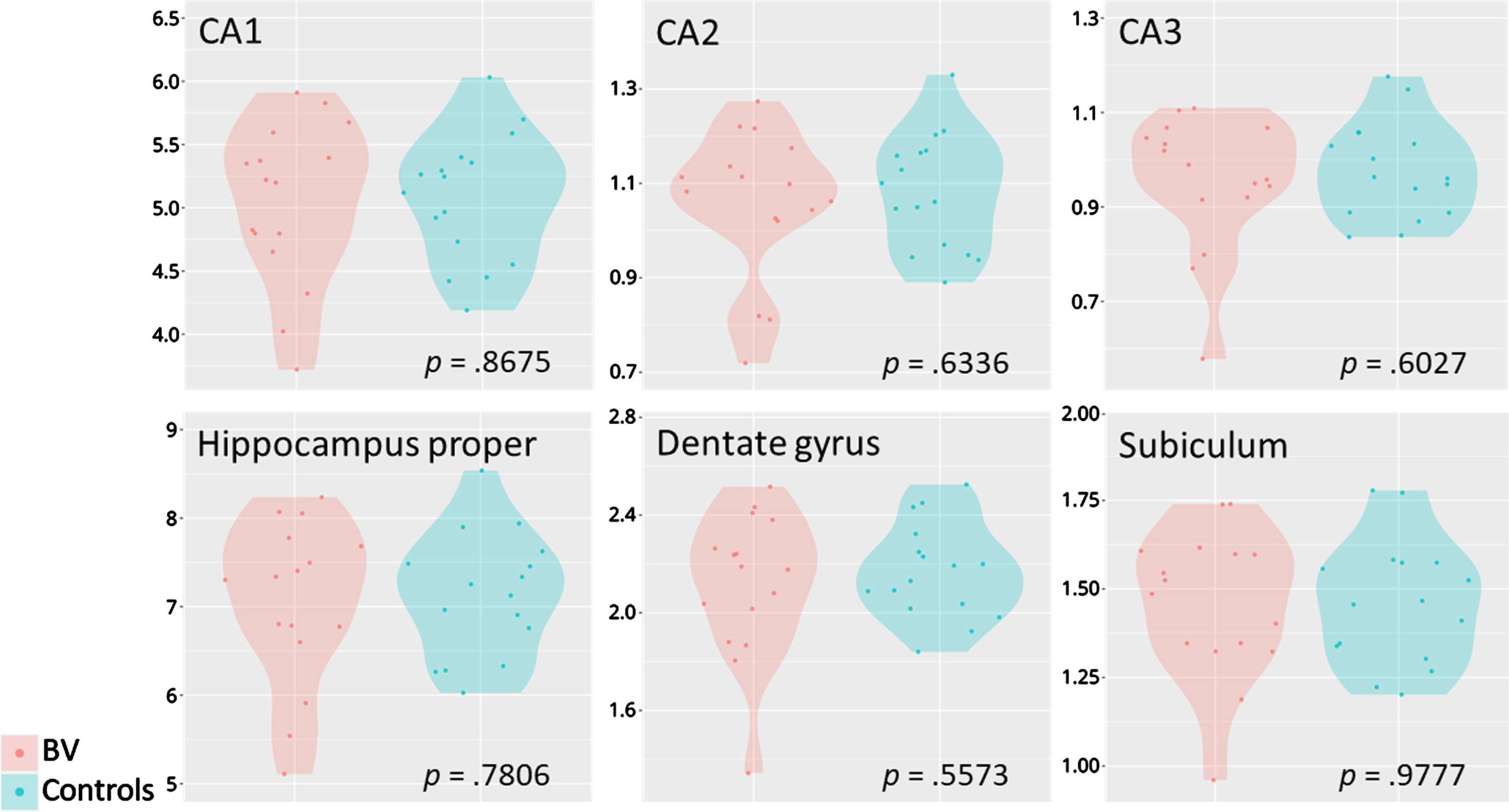
Targeted hippocampal volumetric measurements. Violin plots of the hippocampal subfields (in ml) of patients with BV (n = 16) in comparison with their matched controls (n = 16). The hippocampus proper is calculated as the sum of CA1, CA2, and CA3. BV, bilateral vestibulopathy; CA, cornu ammonis.
ROI volumes of the hippocampus proper and its subdomains
BV, bilateral vestibulopathy; CA, cornu ammonis.
To explore whether hippocampal volume correlates with saccular function in a population with preserved vestibular function, cVEMP parameters of participants without BV were analysed (Table 3). These analyses included a total of 34 participants (15 with sensorineural hearing loss and 19 controls with preserved hearing). Out of all 68 ears, 43 ears demonstrated an intact saccular response. However, the presence of intact responses was not significantly associated with the volume of the hippocampus proper (X2(2, N = 34) = 0.0804, p = 0.9606). Of the ears with intact responses, P-N amplitude, rectified amplitude, and n23 latency demonstrated no significant nor clinically meaningful effect (r(1) = –0.07, p = 0.643; r(1) = 0.01, p = 0.966; r(1) = 0.11, p = 0.472; respectively). Muscle tension of the SCM as measured by MRV also demonstrated no significant effect (r(1) = 0.16, p = 0.304). P13 latency on the other hand was significantly associated with hippocampal volume (r(1) = 0.34, p = 0.028) with a medium effect (η2 = 0.1129). Even though cVEMP testing does not depend on hearing level but to correct for SNHL, p13 latency was correlated with unaided FIhigh-values of the best hearing ear [42]. As expected, this correlation was not significant (r(1) = –0.001, p = 0.995) with a trivial effect size (η2 < 0.001). There are heterogeneous results on the effect of age on p13 latency, but p13 latency is generally known to be associated with age [33]. Indeed, when including age and p13 latency as independent variables with total hippocampal volume as the dependent variable, this model was significant (F(2, 40) = 5.8485, p = 0.006). Parameter estimates were p = 0.020 for age and p = 0.107 for p13 latency. When removing p13 latency from this model, thus resulting in the correlation between total hippocampal volume and age, this model was significant (r(1) = –0.310, p = 0.010).
Saccular characteristics and their association with volume of the hippocampus proper
Saccular characteristics and their association with volume of the hippocampus proper
Latencies are expressed in milliseconds, amplitude and muscle tension are expressed in microvolts. Significant results are indicated with an asterisk (*: p < 0.05). p-Values and effect sizes (uncorrected) are presented together with p-values and effect sizes corrected for age as a confounder. cVEMP, cervical vestibular-evoked myogenic potentials; MRV, mean rectified voltage.
This study aimed to evaluate the impact of semicircular canal and otolith function on hippocampal volume. As such, this study evaluated hippocampal and whole-brain volumetric differences when comparing BV participants with healthy controls whilst adjusting for hearing level, as previous studies on this inner ear topic did not control for the confounding effects of altered hearing levels. However, we were unable to find any structural differences: neither using whole-brain grey matter analyses, nor using an ROI analysis of the hippocampus proper, nor using surface-based analyses, nor using the SVM model as a more sensitive machine learning technique.
In addition, we aimed to delineate otolith influence on hippocampal volume in a population with preserved semicircular canal function. An intact cVEMP response was elicited in at least one ear in 82% of the cases. The p13 latency was positively correlated with hippocampal volume, where longer latencies within normal ranges indicated larger hippocampal volumes. However, when correcting for age, this significant correlation disappeared and could thus be explained by age as a confounding variable. Other saccular parameters at suprathreshold level (95 dB nHL) including the number of intact responses, P-N amplitude, rectified amplitude, n23 latency, and MRV did not demonstrate a significant correlation with the volume of the hippocampus proper.
This study used the normative ranges of C. Li et al. [31] to indicate the presence of intact cVEMP responses (p13:11.81–15.59 ms; n23:18.15– 25.64 ms). However, different latencies can be observed in the literature, with some diverging from the normative ranges of C. Li et al. [31] (for a recent systematic review with meta-analysis, see Y. Macambira et al. [33]). For transparency reasons, an overview per subject of saccular parameters and additional relevant data can be found in Appendix A.
The emerging theory of the association between vestibular loss and cognitive decline would be supported by associated hippocampal atrophy in BV. As such, positive studies by T. Brandt et al. [9] and O. Kremmyda et al. [30] are often cited exclusively to substantiate this hypothesis. However, the role of the replication crisis should not be underestimated and these current null findings, together with those observed by M. Dordevic et al. [17], M. Göttlich et al. [23], and C.G. Schöne et al. [43] need to be taken into account to correct earlier underpowered findings using less reliable segmentation approaches to avoid future false understandings of this association. However, one can question whether the present study’s absence of significant findings can completely disprove the association between hippocampal atrophy and BV? Not necessarily. First of all, BV is a broad and heterogeneous condition. Therefore, one might consider subdividing the BV population by etiology or duration since onset. Second, multiple tests exist to assess peripheral vestibular end-organ functioning. The current study included older adults diagnosed with BV. Diagnostic criteria for this condition all rely on semicircular canal function. However, measurements of otolithic organs may be of added value. They may provide interesting new insights because of their association with spatial learning and memory [47]. Therefore, this study included saccular characteristics and their association with hippocampal volume. Even though no association between saccular function and brain volumetry was observed, a previous systematic review described longer p13 latencies and smaller VEMP amplitudes with increasing cognitive decline along the Alzheimer’s disease continuum [8]. It appears that the association between vestibular dysfunction and an increased risk of cognitive dysfunction may remain on a behavioral level and may not be expressed at the anatomical level.
One thing that must be kept in mind is the sample size. Our research included 16 participants with BV and 16 healthy controls. Although as a rule of thumb, it is recommended that each subgroup should include at least 20 participants [22]. However, we believe that the obtained data quality and stringency of the employed processing pipeline together with the application of full permutation testing makes our findings robust.
A minor limitation is the difference in disease duration for the current BV population. Our study’s median [range] disease duration was 8 [2–22] years. Comparable studies have a variable disease duration of 5–10 years [9], 13.6±17.4 years [30], and 3 months to 20 years [23]. The high variation in disease duration might hamper a direct comparison between studies.
Ideally, the impact of isolated otolith dysfunction (i.e. abnormal otolith function with preserved semicircular canal function) on hippocampal and whole-brain volume should be evaluated. However, there is no consensus on defining otolith symptoms, standardized assessment of laboratory otolith function testing, and diagnostic criteria with structured definitions of isolated otolith dysfunction [12]. This often leads to mis- or underdiagnosing. Future studies should evaluate hippocampal and whole-brain volume in those participants with isolated otolith dysfunction, once a consensus regarding this pathology has been reached.
Conclusion
Neither whole-brain nor hippocampal volume differences were observed when comparing subjects with BV and healthy controls. Saccular function testing in subjects with preserved semicircular canal function resulted in no significant correlations with hippocampal volume. The association between vestibular dysfunction and an increased risk of cognitive dysfunction may only be present on the behavioral level and may not be expressed at the anatomical level.
Data-availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Financial disclosures/conflicts of interest
The authors have no relevant financial or non-financial interests to disclose. This work was supported by a Fonds voor Wetenschappelijk Onderzoek (FWO) Fundamental Research Project (Grant Number G042819N3).
